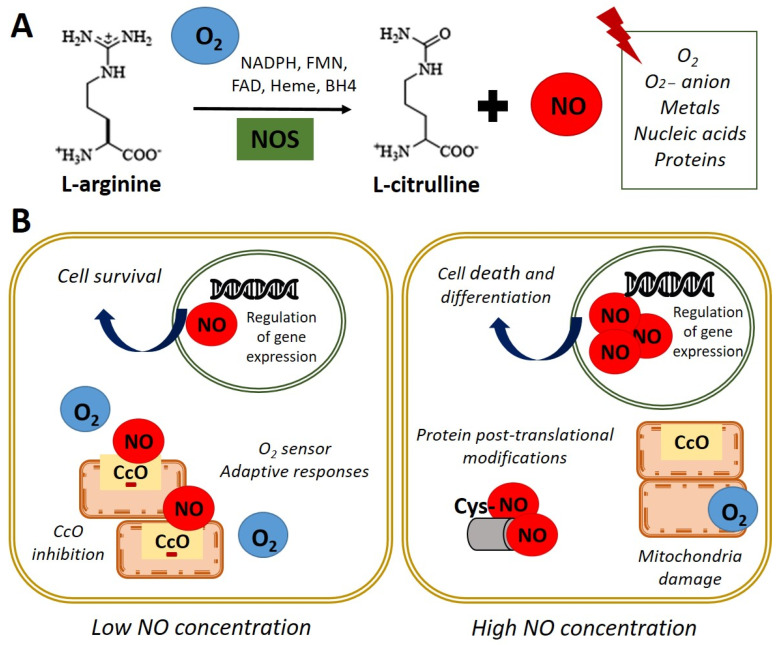
Figure 1.
Nitric oxide synthesis and biological functions. (A) In normoxia conditions (21% O2), nitric oxide synthase (NOS) catalyzes the oxidation of the terminal guanidinyl nitrogen of the amino acid L-arginine to form L-citrulline and nitric oxide (NO) in presence of NADPH and cofactors such as flavin mononucleotide (FMN), flavin adenine dinucleotide (FAD), heme, and tetrahydrobiopterin (BH4) [3]. Once produced, NO readily interacts with O2, O2− anion, metals, nucleic acids, and proteins. (B) Left panel. NO at low concentration inhibits cytochrome c oxidase (CcO) activity by competing with O2. Adaptive responses to O2 concentration and cell survival genes are activated. Right panel. High concentrations of NO induce damage in all mitochondrial complexes, nitrosylation, or oxidation of protein thiol groups and induce cell death and differentiation.