Cascade oxidation/Knoevenagel condensation of different benzyl alcohols 2a–f catalyzed by the Cu@EDTAD-PMO (1) under optimized conditionsa.

| ||||
|---|---|---|---|---|
| Entry | Substrate 2 | Product | Time (h) | Yield (%) |
| 1 |

|

|
6 | 95 |
| 2 |

|

|
10 | 90 |
| 3 |

|

|
16 | 86 |
| 4 |

|

|
13 | 78 |
| 5 |

|

|
10 | 85 |
| 6 |
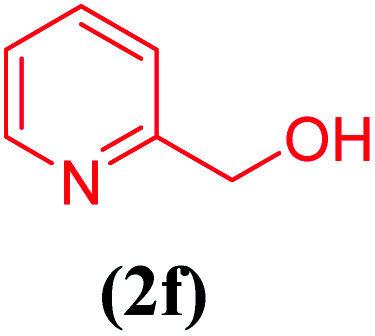
|

|
15 | 88 |
| 7 |

|

|
8 | 91 |
Reaction conditions: benzyl alcohol (2, 1 mmol), TBHP (1 mmol), and Cu@EDTAD-PMO (1, 4.2 mol%) were added into CH3CN (2 mL) and stirred at 50 °C. Then, malononitrile (3, 1.1 mmol) was added to the reaction mixture.
