Abstract
Selumetinib is an oral, potent, and highly selective allosteric MEK1/2 inhibitor approved for the treatment of pediatric patients (aged ≥2 years) with neurofibromatosis type 1 who have symptomatic, inoperable plexiform neurofibromas. A granule formulation of selumetinib is under development to improve dosing precision for younger pediatric patients who may be unable to swallow capsules. This phase I crossover study investigated the effect of food on the pharmacokinetic (PK) properties of selumetinib capsule and granule formulations. Healthy male volunteers were randomized to receive selumetinib granules (25 mg) or capsules (50 mg [2 × 25 mg]) under fasted or fed conditions (a low‐fat meal). Plasma concentrations and PK parameters were determined less than or equal to 48 h postdose. Safety and tolerability were assessed. Across 24 volunteers, selumetinib was absorbed quickly, with a time to maximum concentration (Tmax) ranging from ~1–3 h. Geometric mean ratios (90% confidence interval [CI]) for maximum plasma concentration (Cmax) in the fed versus fasted state were 0.61 (90% CI 0.51–0.72) and 0.40 (90% CI 0.33–0.48) for the granule and capsule formulations, respectively, whereas geometric mean ratios (90% CI) for area under the plasma drug concentration‐time curve in the fed versus fasted state were 0.97 (90% CI 0.91–1.02) and 0.62 (90% CI 0.55–0.70), respectively. Levels of less than 10% conversion to the N‐desmethyl selumetinib metabolite were observed. Selumetinib was well‐tolerated, with only a few adverse events of mild intensity reported. Selumetinib administration with a low‐fat meal resulted in lower Cmax and longer Tmax for both formulations versus fasted conditions. However, area under the curve for selumetinib granules was similar under fasted and fed conditions. Overall, these findings support further development of this formulation for pediatric patients.
Study Highlights.
WHAT IS THE CURRENT KNOWLEDGE ON THE TOPIC?
Selumetinib is approved for use in pediatric patients with neurofibromatosis type 1 who have symptomatic, inoperable plexiform neurofibromas. A granule formulation is under development to improve dosing precision and swallowability for younger pediatric patients who may be unable to swallow capsules.
WHAT QUESTION DID THIS STUDY ADDRESS?
This study evaluated the effect of food on the pharmacokinetic properties of selumetinib capsule and granule formulations in healthy male volunteers to assess whether food restrictions may be needed with administration of the granule formulation.
WHAT DOES THIS STUDY ADD TO OUR KNOWLEDGE?
Absorption of selumetinib was prolonged in the fed state compared with the fasted state for both capsule and granule formulations, as determined by a lower maximum plasma concentration (Cmax) and longer Tmax). However, selumetinib granules had a similar exposure (AUC) in both fasted and fed conditions, suggesting that the rate of absorption may be affected, but not to the extent of absorption with food. Furthermore, similarities observed in AUC between the granule and capsule formulations suggest that the granule formulation can be used in a similar setting to the capsule.
HOW MIGHT THIS CHANGE CLINICAL PHARMACOLOGY OR TRANSLATIONAL SCIENCE?
These data support further clinical development of the selumetinib granule formulation for pediatric patients.
INTRODUCTION
Selumetinib (ARRY‐142886) is an oral, potent, and highly selective allosteric MEK1/2 inhibitor with a short half‐life. 1 , 2 , 3 In April 2020, selumetinib was approved by the US Food and Drug Administration (FDA) for use in pediatric patients (aged 2 years and older) with neurofibromatosis type 1 (NF1) who have symptomatic, inoperable plexiform neurofibromas, based on results from the phase II SPRINT trial (NCT01362803). 4 , 5 , 6 , 7
The commercial formulation of selumetinib is a 25 mg oral capsule formulation, a dispersion of selumetinib hydrogen‐sulphate (Hyd‐sulfate) in vitamin E polyethylene glycol succinate contained within a hypromellose shell. 6 The first‐in‐human study demonstrated significantly higher relative exposure of the 75 mg Hyd‐sulfate selumetinib capsule formulation than previously observed for the original 100 mg free‐base suspension (197%, 90% confidence interval [CI] 161–242). 2 , 8 , 9 The capsule formulation also demonstrated relatively rapid absorption, a short half‐life, and approximately dose proportional pharmacokinetics (PKs). 2
Following oral administration of [14C]‐selumetinib in healthy subjects, total recovery of radioactivity was ~93%, with most of the radioactivity (59% of the dose) eliminated in the feces. 10 Urinary elimination accounted for ~33% of the dose, most of which occurred during the first 48 h postdose. Unchanged selumetinib accounted for less than or equal to 1% of the dose in urine and between 6% and 34% of the dose in the feces. Unchanged selumetinib was identified as the major circulating drug‐related component and accounted for ~40% of the plasma radioactivity (data on file).
Selumetinib undergoes phase I metabolism, including oxidation of the side chain, N‐demethylation, and loss of the side chain to form amide and acid metabolites in vitro. 11 Cytochrome P450 3A4 is mainly responsible for oxidative metabolism of selumetinib, with other isoforms involved to a lesser extent. Glucuronidation provides a well‐established route of elimination for selumetinib phase I metabolites involving several uridine 5'‐diphospho‐glucuronosyltransferase isoforms. 11 The active metabolite N‐desmethyl selumetinib is approximately three to five‐fold more potent than selumetinib, contributing to roughly 21–35% of overall pharmacologic activity. 6 In vitro studies indicate that neither selumetinib nor N‐desmethyl selumetinib elicit clinically relevant drug interactions with cytochrome P450 or transporter substrates. 11
In ongoing clinical trials, a commercially approved selumetinib capsule formulation under the fasted state is used. 5 , 7 , 9 For children aged between greater than or equal to 1 and less than 7 years, and those who are unable to swallow capsules, a granule formulation is also under development to enable more precise dosing and improved swallowability. This selumetinib granule formulation comprises lipidic multiparticulates of selumetinib hyd‐sulfate with an enteric coating, designed for palatability. The granules are contained in a sprinkle capsule intended to be opened and the granules sprinkled on or mixed with a suitable vehicle. The final coated granules comprise 27 mg/g (free base equivalent) selumetinib, which are filled for two dosage strengths, 5 mg and 7.5 mg, but unlike the capsules do not contain vitamin E polyethylene glycol succinate.
Food effect on selumetinib when dosed as capsules was previously evaluated in other clinical trials, including clinical pharmacology food effect studies in healthy volunteers (NCT01974349) and patients with advanced solid malignancies (NCT00710515). 9 , 12 In a phase I trial investigating whether a high‐fat meal influenced the rate and extent of selumetinib capsule absorption in patients with advanced solid malignancies, selumetinib decreased maximum plasma concentration (Cmax) by 62% and diminished area under the plasma drug concentration‐time curve (AUC) by 19%. 9 In healthy volunteers, a mean reduction of both selumetinib Cmax by 50% and AUC by 16% was observed when selumetinib was administered with a high‐fat meal, although the data on the clinical significance of these reductions were not available. 12 Here, we report the effect of a low‐fat meal on the PKs of selumetinib for the granule formulation and the commercially approved capsule.
The two aforementioned clinical studies of a high‐fat meal in adult patients with cancer and adult healthy volunteers demonstrated that food reduces peak selumetinib plasma concentrations by ~50–60%, with only a minimal decrease (<20%) in overall exposure, reflecting slower absorption compared with a fasted state. 9 , 12 As selumetinib is dosed twice daily, it would be challenging to restrict access to food in children. Therefore, we conducted a clinical study in adult healthy volunteers to investigate the food effect using a low‐fat, low‐calorie meal (~400 kcal), akin to a light meal or snack versus fasted conditions. The aim was to further understand if these food restrictions may be amenable for pediatric patients and what effect, if any, a low‐fat meal has on the PK profile of selumetinib in the commercial capsule and the granule formulation.
METHODS
Participants
Healthy male volunteers aged 18–45 years (inclusive), with a body mass index of 18–30 kg/m2, body weight greater than or equal to 50 kg, and a creatinine clearance greater than 50 mL/min using Cockcroft‐Gault formula were eligible to participate. Volunteers had to be able to consume a low‐fat meal within a 30‐min period of treatment administration. Volunteers with any clinically significant condition or of an ethnicity that, in the opinion of the investigator, may put the volunteer at risk, influence the study results, or their ability to participate in the study, were excluded. Dose normalized AUC and Cmax in healthy adult subjects of Asian ethnicity were slightly higher compared with Western subjects. However, these apparent differences were lower when AUC and Cmax were corrected for body weight. Therefore, it is anticipated that the observed PK ethnic differences of selumetinib are likely not to be of clinical relevance. 13
Previous clinical pharmacology trials of selumetinib have mostly been conducted in healthy male volunteers and so this study was performed using male subjects in keeping with what had been previously done, limiting variability.
Randomization and interventions
Volunteers were randomly assigned to treatment sequence(s) by Parexel International according to the AstraZeneca randomization system (AZRand). Volunteers received selumetinib granule formulation (25 mg) or capsules (50 mg [2 × 25 mg]) as single doses to limit exposure.
As the PKs of selumetinib capsules are linear with respect to dose from 25 to 75 mg in heathy adults, a dose of 50 mg was chosen for safety reasons (a capsule dose was historically administered in healthy volunteers as a comparator with no safety issues). 6 A dose of 25 mg was chosen for the granule formulation to represent the potential doses administered to children under the age of 7 years. The 25 mg active dose is equivalent to approximately 1 g of granules, thereby allowing for a relevant assessment of palatability.
The study consisted of a screening period (maximum 28 days) followed by two treatment periods for part 1 of the study (fasted conditions) and two treatment periods for part 2 of the study (fed conditions). During these periods, volunteers were residents from the day before dosing until at least 48 h after last dosing for collection of PK samples and then for a final safety post‐study visit.
In part 1 of the study, volunteers received single doses of selumetinib and acetaminophen 500 mg, as a marker of gastric emptying, under fasted conditions. The dose was administered after an overnight fast of at least 10 h. Selumetinib and acetaminophen were administered with ~240 ml of water. No food was allowed for at least 4 h postdose, and no fluid was allowed except for water up to 1 h before and 2 h postdose. A meal was provided 4 h following drug administration.
In part 2 of the study, volunteers received single doses of selumetinib and acetaminophen under fed conditions. Following an overnight fast of at least 10 h, volunteers could consume a low‐fat (<25% fat), low‐calorie (400–500 kcal) meal (to be eaten within 30 min or less). This meal comprised a low‐fat yogurt, apple juice, 2% milk, and a low‐fat muffin. Thirty minutes after the start of the meal, selumetinib and acetaminophen were administered with ~240 ml of water. No food was allowed for at least 4 h postdose. No fluid was allowed apart from water, which was permitted until 1 h before selumetinib and acetaminophen and then from 2 h following administration.
There was a minimum washout period of at least 5 days between selumetinib treatments. There was a follow‐up visit within 7–10 days after the last administration.
Study oversight
This study was conducted in accordance with the provisions of the Declaration of Helsinki, Good Clinical Practice guidelines (as defined by the International Conference on Harmonisation), applicable regulatory requirements, and the policy on bioethics and human biologic samples of the sponsor, AstraZeneca. The study protocol was approved by the institutional review board. All volunteers provided written informed consent prior to enrollment. The authors had full access to all data and are responsible for the accuracy of the data. Data underlying the findings described in this manuscript may be obtained in accordance with AstraZeneca’s data sharing policy described at https://astrazenecagrouptrials.pharmacm.com/ST/Submission/Disclosure.
Study objectives
This study was divided into two parts and the same volunteers participated in both parts of the study. Part 1 investigated the PK properties (relative bioavailability) of two selumetinib formulations (granule and capsule) in a fasted state (separate manuscript in development). Part 2, presented here, compared the effect of food on both capsule and granule formulations of selumetinib. The safety and tolerability of single doses of selumetinib in healthy volunteers were secondary objectives.
Study assessments
The PK analysis set included all patients who received at least one dose of selumetinib, with no major protocol deviations, and for whom at least one of the primary PK parameters (selumetinib and its metabolite N‐desmethyl selumetinib) could be calculated for at least two treatment periods. PK analyses were carried out using actual sample times recorded in the raw data, where possible. Blood samples for determination of selumetinib and N‐desmethyl selumetinib concentrations in the plasma were analyzed by Covance on behalf of Clinical Pharmacokinetic Alliance, AstraZeneca R&D. The concentrations of selumetinib, N‐desmethyl selumetinib, and acetaminophen in human plasma containing K2EDTA as an anticoagulant were determined using protein precipitation followed by analysis using high‐performance liquid chromatography, followed by tandem mass spectrometric detection (LC‐MS/MS).
Blood samples to measure selumetinib and N‐desmethyl selumetinib were taken predose and between 0.25 and 48 h postdose. PK parameters were derived using noncompartmental methods with Phoenix WinNonlin version 6.2 or higher and/or SAS version 9.2 or higher. All descriptive and inferential statistical computations were performed using SAS version 9.2 or higher. The following PK parameters were determined.
For selumetinib, AUC, Cmax, ratio of AUC in fed state to AUC in the fasted state, ratio of Cmax in fed state to Cmax in fasted state, terminal half‐life (t½λz), time to Cmax (Tmax), apparent clearance of drug from plasma (CL/F), and apparent volume of distribution during the terminal phase (Vz/F) were recorded. AUC and Cmax were also calculated for N‐desmethyl selumetinib. The terminal elimination rate constant (λz) was calculated by log‐linear regression of the terminal portion of the concentration time profile. The choice of data points used to estimate λz was made in the “lambda z ranges” dialog box using WinNonlin software. In general, the minimum data requirements were three measured concentrations spanning three half‐lives. A total of three to six observations were considered to provide a good estimation of the elimination constant. Use of λz values obtained from data spanning less than three half‐lives was permissible based on expert judgment provided that the resulting extrapolated area was not more than 20% of the total AUC determined. The AUC was calculated using the trapezoidal rule. The standard calculation method of “linear up/log down” is recommended for use in WinNonlin. From single dosing, the AUC from zero to infinity was calculated by extrapolation to infinity of the AUC up to the time of the last quantifiable plasma concentration using λz. The minimum requirement for the calculation of AUC will be the inclusion of at least three consecutive plasma concentrations above the lower limit of quantification (LLOQ), with at least one of these concentrations following Cmax.
In addition, any adverse events (AEs) were monitored at assigned timepoints throughout the study and were graded using the Common Terminology Criteria for Adverse Events Version 4.0.
Statistical methods
Briefly, a sample size of 24 healthy volunteers was planned to gain adequate information while exposing as few healthy volunteers as possible to the study procedures. It was expected that at least 20 evaluable volunteers would complete the study (allowing for an approximate 20% dropout rate). The following analysis populations were utilized: randomized set (all volunteers randomized into the study), safety analysis set (SAS; all volunteers who received at least one dose of selumetinib and for whom any safety postdose data were available), and PK analysis set. The SAS was used for all demographic and disposition data as well as safety analyses.
Plasma concentrations and PK parameters were summarized by treatment using appropriate descriptive statistics.
RESULTS
Demographics
A total of 24 male volunteers were screened and enrolled in this study. Demographics are reported in Table 1. All randomized volunteers completed the study (N = 24) with no premature withdrawals.
TABLE 1.
Demographic characteristics (all volunteers)
| Category | All volunteers (N = 24) |
|---|---|
| Age, years | |
| Mean (SD) | 33.2 (5.8) |
| Median | 33.0 |
| Min–max | 23–44 |
| Sex, n (%) | |
| Female | 0 |
| Male | 24 |
| Race, n (%) | |
| White | 3 (12.5) |
| Black or African American | 20 (83.3) |
| Other | 1 (4.2) |
| Ethnicity, n (%) | |
| Hispanic or Latino | 4 (16.7) |
| Not Hispanic or Latino | 20 (83.3) |
| Height, cm | |
| Mean (SD) | 177.6 (8.0) |
| Median | 175.5 |
| Min–max | 162–190 |
| Weight, kg | |
| Mean (SD) | 78.4 (10.8) |
| Median | 80.5 |
| Min–max | 53.9–94.5 |
| BMI, kg/m2 | |
| Mean (SD) | 24.9 (3.5) |
| Median | 25.1 |
| Min–max | 18.7–29.8 |
Abbreviations: BMI, body mass index; Max, maximum recorded values; Min, minimum recorded values; n, number of volunteers in the given category; N, number of volunteers randomized; SD, standard deviation.
PK and food effect
Selumetinib granule and capsule formulations plasma PK profiles in both fasted and fed conditions are presented in Figure 1a,b. Absorption of selumetinib generally occurred quickly under fasted conditions; however, absorption was prolonged in the fed state by ~1.3 h for the granule formulation and ~0.9 h for the capsule formulation. The Tmax (median [range]) for selumetinib granule and capsule formulations were 1.73 h (1.20–5.00) and 1.14 h (0.75–2.25), respectively, in the fasted stated and 3.03 h (1.73–5.00) and 2.00 h (0.98–3.53), respectively, in the fed state. Absorption of acetaminophen when co‐administered with selumetinib was also prolonged by ~1.2 h in the fed state compared with the fasted state for both the granule and capsule formulations. The Tmax (median [range]) for acetaminophen granule and capsule formulations were 0.75 h (0.47–1.97) and 0.76 h (0.48–2.50), respectively, in the fasted state and 1.98 h (0.23–3.95) and 2.00 h (0.50–4.00), respectively, in the fed state (Table 2).
FIGURE 1.
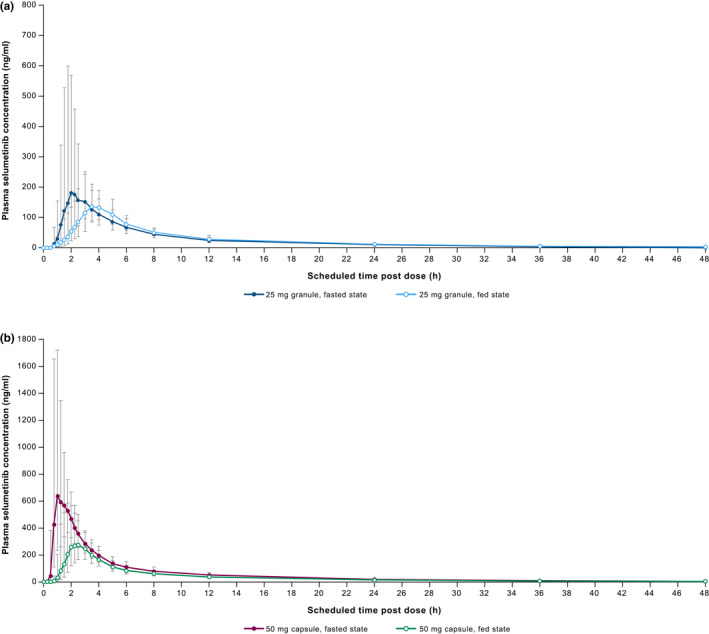
Geometric mean (±gSD) plasma concentration (ng/ml) of selumetinib granule (a) and capsule (b) formulations versus time (pharmacokinetic analysis set). gSD, geometric standard deviation
TABLE 2.
Summary of key plasma pharmacokinetic parameters of selumetinib and acetaminophen (pharmacokinetic analysis set)
| Pharmacokinetic parameters of selumetinib | |||||
|---|---|---|---|---|---|
| Parameter | Statistic | Fasted | Fed | ||
| 25 mg granule (n = 24) | 50 mg capsule (n = 24) | 25 mg granule (n = 24) | 50 mg capsule (n = 24) | ||
| Cmax, ng/ml | Geomean (gCV%) | 329.7 (39.95) | 1009 (41.48) | 199.6 (32.32) | 404.2 (52.14) |
| AUC, h*ng/ml | Geomean (gCV%) | 1299 (26.03) | 3004 (25.29) | 1253 a (23.70) | 1871 (38.66) |
| Tmax, h | Median (range) | 1.73 (1.20–5.00) | 1.14 (0.75–2.25) | 3.03 (1.73–5.00) | 2.00 (0.98–3.53) |
| t½λz b , h | Arithmean (SD) | 8.764 (2.349) | 9.733 (2.810) | 11.79 a (3.853) | 11.91 (2.654) |
| CL/F, L/h | Arithmean (SD) | 19.86 (5.201) | 17.15 (4.377) | 20.47 a (4.751) | 28.50 (10.19) |
| Vz/F, L | Arithmean (SD) | 245.7 (80.52) | 243.6 (99.53) | 348.5 a (159) | 493.2 (215.8) |
| Pharmacokinetic parameters of acetaminophen | |||||
|---|---|---|---|---|---|
| Parameter | Statistic | Fasted | Fed | ||
| 25 mg granule (n = 24) | 50 mg capsule (n = 24) | 25 mg granule (n = 24) | 50 mg capsule (n = 24) | ||
| Cmax, ng/ml | Geomean (gCV%) | 6189 (31.28) | 5811 (36.14) | 4970 (29.41) | 5028 (35.20) |
| AUC, h*ng/ml | Geomean (gCV%) | 21190 (22.46) | 21690 a (22.50) | 22750 (20.63) | 22600 (20.36) |
| Tmax, h | Median (range) | 0.75 (0.47–1.97) | 0.76 (0.48–2.50) | 1.98 (0.23–3.95) | 2.00 (0.50–4.00) |
| t½λz b , h | Arithmean (SD) | 3.789 (1.081) | 4.081 a (1.176) | 4.374 (0.8479) | 4.267 (0.7217) |
| CL/F, L/h | Arithmean (SD) | 24.17 (5.628) | 23.61 (5.355) | 22.43 (4.887) | 22.57 (4.830) |
| Vz/F, L | Arithmean (SD) | 126.3 (27.09) | 135.4 (39.69) | 137.8 (25.39) | 136.2 (24.41) |
Abbreviations: Arithmean, arithmetic mean; AUC, area under the plasma drug concentration‐time curve; CL/F, apparent clearance for parent drug estimated as dose divided by AUC; Cmax, observed maximum plasma concentration; gCV%, geometric coefficient of variation; Geomean, geometric mean; n, number of subjects in the given category; SD, standard deviation; Tmax, time to reach maximum plasma concentration; t½λz, terminal half‐life; Vz/F, apparent volume of distribution during the terminal phase (extravascular administration).
One subject with regression fit with the R 2 adjusted value <0.8.
The t½λz calculated over a period that was less than three times the resultant half‐life for several subjects in each treatment.
Mean t½λz and CL/F for selumetinib were similar in the fed and fasted states for the granule formulation. For the capsule formulation, mean t½λz was similar between the fed and fasted states, whereas mean CL/F appeared to be lower in the fasted state compared with the fed state (17.15 L/h vs. 28.50 L/h). In addition, high variability in the fed state was observed for the capsule formulation (Table 2). In the fasted versus fed states, mean Vz/F for the granule formulation was 245.7 (±80.5 L) versus 348.5 (±159.0 L), and for the capsule formulation was 243.6 (±99.5 L) versus 493.2 (±215.8 L; Table 2). Geometric means for AUC in the fed versus fasted states were 1253 h*ng/ml (geometric coefficient [gCV%] 23.70) versus 1299 h*ng/ml (gCV% 26.03) for the granule, and 1871 h*ng/ml (gCV% 38.66) versus 3004 h*ng/ml (gCV% 25.29) for the capsule formulations. Geometric means for Cmax in the fed versus fasted states were 199.6 ng/ml (gCV% 32.32) versus 329.7 ng/ml (gCV% 39.95) for the granule and 404.2 ng/ml (gCV% 52.14) versus 1009 ng/ml (gCV% 41.48) for the capsule formulations (Table 2).
Fed to fasted ratios for Cmax and AUC for the granule and capsule formulations are presented in Figure 2. Geometric mean ratios (90% CI) for Cmax in the fed versus fasted states were 0.61 (0.51–0.72) and 0.40 (0.33–0.48) for the granule and capsule formulations, respectively. Geometric mean ratios (90% CI) for AUC in the fed versus fasted states were 0.97 (0.91–1.02) and 0.62 (0.55–0.70) for the granule and capsule formulations, respectively. As expected, lower Cmax was observed when the granule formulation was administered in the fed state compared with the fasted state. For the capsule formulation, lower Cmax and AUC were observed in the majority of subjects in the fed state compared with the fasted state. The geometric mean for the fraction of selumetinib granule dose systemically available (relative to the capsule dose) was 1.34 (90% CI 1.17–1.53) and 0.87 (90% CI 0.81–0.92) in the fed and fasted states, respectively (Figure 2).
FIGURE 2.

Food effect ratios for selumetinib pharmacokinetic parameters (pharmacokinetic analysis set). AUC, area under the plasma drug concentration‐time curve; CI, confidence interval; Cmax, observed maximum plasma concentration; PK, pharmacokinetic
Pharmacokinetics of N‐desmethyl selumetinib
For both capsule and granule formulations, conversion of selumetinib to N‐desmethyl selumetinib was low and similar in the fasted and fed states (Figure 3). The geometric mean metabolite to parent ratios ranged from 6.7 to 7.2% for Cmax and 5.9 to 8.3% for AUC. Due to the low doses of selumetinib administered in this study, only limited levels of N‐desmethyl selumetinib were detected.
FIGURE 3.

Geometric mean (±gSD) plasma concentration (ng/ml) of N‐desmethyl selumetinib granule (a) and capsule (b) formulations vs. time (pharmacokinetic analysis set). gSD, geometric standard deviation
Safety
AEs were reported across all treatment groups and although the frequency was low, more AEs were reported after the administration of either selumetinib formulation in the fasted state compared with the fed state (6 [25%] each in the fasted group vs. 3 [13%] each in the fed group; Table 3). The most common AEs reported by system organ class were dry eye (fasted: 3 [13%] with granule; fed: one [4%] each with granule and capsule) and headache (fasted: one [4%] each with granule and capsule; fed: one [4%] with granule). Two events of dry eye were reported in two volunteers (8%) in the fasted, granule group, which were considered to be drug related by the investigator. All AEs reported were mild in intensity and resolved by the end of the study, with no dose modifications. There were no serious AEs, discontinuations, or deaths during the study. No clinically significant changes in laboratory values and vital signs were reported in the study.
TABLE 3.
Volunteers who experienced at least one AE, by preferred term, arranged by system organ class (safety analysis set)
| System organ class/ preferred term | Fasted | Fed | ||
|---|---|---|---|---|
|
25 mg granule (N = 24) n (%) |
50 mg capsule (N = 24) n (%) |
25 mg granule (N = 24) n (%) |
50 mg capsule (N = 24) n (%) |
|
| Subjects with any AE | 6 (25.0) | 6 (25.0) | 3 (12.5) | 3 (12.5) |
| Eye disorders | 4 (16.7) | 0 (0.0) | 1 (4.2) | 1 (4.2) |
| Blepharitis | 1 (4.2) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Dry eye | 3 (12.5) | 0 (0.0) | 1 (4.2) | 1 (4.2) |
| Infections and infestations | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (4.2) |
| Hordeolum | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (4.2) |
| Injury, poisoning, and procedural complications | 1 (4.2) | 1 (4.2) | 0 (0.0) | 0 (0.0) |
| Skin injury | 1 (4.2) | 1 (4.2) | 0 (0.0) | 0 (0.0) |
| Nervous system disorders | 1 (4.2) | 1 (4.2) | 1 (4.2) | 0 (0.0) |
| Headache | 1 (4.2) | 1 (4.2) | 1 (4.2) | 0 (0.0) |
| Respiratory, thoracic, and mediastinal disorders | 1 (4.2) | 1 (4.2) | 2 (8.3) | 0 (0.0) |
| Nasal congestion | 1 (4.2) | 1 (4.2) | 0 (0.0) | 0 (0.0) |
| Rhinorrhea | 0 (0.0) | 0 (0.0) | 2 (8.3) | 0 (0.0) |
| Skin and subcutaneous tissue disorders | 1 (4.2) | 3 (12.5) | 0 (0.0) | 1 (4.2) |
| Dermatitis contact | 1 (4.2) | 0 (0.0) | 0 (0.0) | 0 (0.0) |
| Rash | 0 (0.0) | 1 (4.2) | 0 (0.0) | 0 (0.0) |
| Skin irritation | 0 (0.0) | 1 (4.2) | 0 (0.0) | 0 (0.0) |
| Swelling face | 0 (0.0) | 1 (4.2) | 0 (0.0) | 1 (4.2) |
Abbreviations: AE, adverse event; n, number of subjects with the event; N, number of subjects in the safety analysis.
DISCUSSION
In this phase I, open‐label study in 24 healthy volunteers, the effect of a low‐fat meal on the PK properties of selumetinib capsule and granule formulations were investigated. For the granule formulation, the absorption of selumetinib (Tmax) observed in the fed state was prolonged compared with the fasted state by ~1.3 h. [Correction added on 23 March 2022, after first online publication: Difference in Tmax values for fed vs fasted state for granule formulation has been corrected to ~1.3 h]. However, the administration of food did not affect selumetinib elimination in the granule formulation, as t½λz, CL/F, and metabolite to parent ratios for Cmax and AUC were similar in the fed and fasted states. Selumetinib Cmax was reduced by ~40% in the fed state compared with the fasted state with the granule formulation but no significant effect on overall exposure (AUC) was observed.
For the capsule formulation, the absorption of selumetinib (Tmax) was also prolonged in the fed state versus the fasted state by ~0.9 h. A reduction (60%) in selumetinib Cmax was observed with the capsule formulation in the fed state versus the fasted state, which is similar to that observed in previous studies. 9 , 12 [Correction added on 23 March 2022, after first online publication: Difference in Tmax values for fed vs fasted state for capsule formulation has been corrected to ~0.9 h]. Furthermore, a 38% reduction in AUC occurred with the capsule formulation in the fed versus fasted state. The overall effect is consistent with observations in previous studies. 9 , 12
Data from clinical studies identified the presence of selumetinib N‐desmethyl in human subjects at about 3% of the total selumetinib plasma radioactivity. Parent to N‐desmethyl metabolite ratio was low, with geometric mean metabolite to parent ranging from 6.7% to 7.2% for Cmax and 5.9% to 8.3% for AUC. Together with the low doses of selumetinib administered in this study, this resulted in limited detectable concentrations of the metabolite. 10 However, the derived PK parameters were considered insufficiently precise to draw robust conclusions.
In this study, acetaminophen was administered as a gastric emptying marker together with selumetinib. It was observed that median Tmax of acetaminophen was delayed by ~1.2 h under fed conditions compared with the fasted state. This indicates a delay in gastric emptying in the fed state, which would contribute to a delay in selumetinib absorption. In other food effect selumetinib studies using a 75 mg (3 × 25 mg) capsule dose, median Tmax was prolonged in the fed versus fasted state by ~1.5 h in healthy volunteers. 12
A phase I healthy volunteer study on the relative bioavailability and food effect of capsule and granule formulations of selumetinib showed that exposure with respect to AUC, was broadly similar for the capsule and granule formulations. 14 The Cmax was lower for selumetinib granules than selumetinib capsules. A physiologically based biopharmaceutics model (PBBM) was developed using in vitro dissolution profiles as input data and validated against fasted and fed state clinical data. This model was used to investigate the mechanism of food effect. This model will be published separately.
The observed food effect on selumetinib capsule and granule PK involves several mechanisms. A reduction in Cmax is expected due to the delayed gastric emptying and a reduction in first pass liver extraction is expected due to a higher blood flow in the fed state compared to the fasted state. 15 For capsules, the PBBM indicated a reduction in absorption due to a higher precipitation upon stomach emptying and an increase in first pass gut extraction, due to a higher dilution of the drug in the stomach chyme, combined with a slower gastric emptying in the fed state (data on file). The granule shows no impact of prandial state on overall exposure (AUC). For the granule formulation, the PBBM indicates that dissolution starts after stomach emptying and is slower for granule in vivo, compared with capsules (data on file). The slower gastric emptying in the fed state also induces a delay to the granule’s dissolution, which is reflected in the absorption pattern. The slower dissolution prevents precipitation and even if the gut extraction is higher in the fed compared to fasted state for the granules, it is almost entirely compensated by a reduced first pass liver extraction (data on file).
The PKs of selumetinib is comparable between healthy adult subjects and pediatric patients at the investigated dose range. Therefore, the observations and conclusions from the clinical pharmacology studies performed in healthy adult subjects are considered of direct relevance for pediatric patients. In the pediatric population, at the investigated dose range, no clear exposure–response relationship was found between selumetinib exposure and efficacy or safety. 13 In addition, the population PK analysis of 461 subjects (women, 15%) indicated gender had minimal impact on selumetinib PKs. Hence, the exposure of selumetinib was similar between male and female subjects. 11
Considering the results from this study, other food effect studies, 9 , 12 , 14 and the exposure response analysis in pediatric patients, selumetinib capsules (at the approved dose of 25 mg/m2) is recommended to be taken twice daily on an empty stomach as per label. 6 No food should be consumed 2 h prior to each dose or for one hour following each dose. As a follow‐up, a phase I study assessing the effect of food on the gastrointestinal toxicity and PKs of selumetinib in adolescents with NF1‐related plexiform neurofibromas is also in progress. 16 The study aims to evaluate the steady‐state systemic exposure and optimal dosage of selumetinib when taken with a low‐fat, low‐calorie meal, and to assess the effect of food on gastrointestinal toxicity.
Moreover, in view of the similar relative bioavailability between the capsule formulation and the granule formulation, with only 14% decrease in AUC with the granule formulation in the fasted state, 14 and the flat exposure response in pediatric patients, this is unlikely to alter efficacy or safety in pediatric patients and thereby the benefit:risk of selumetinib as noted with the capsule formulation. 17
In conclusion, results of this food effect study show that absorption of selumetinib (Tmax) in the fed state was prolonged compared with the fasted state for both capsule and granule formulations. Although Cmax values were lower in the fed state for both formulations, the selumetinib granule formulation had similar exposure (AUC) in both fasted and fed conditions, indicating that the rate of absorption may be affected with food, but not the extent of absorption. Overall, selumetinib was well‐tolerated when administered in both a fasted state and following a low‐fat meal in both formulations; however, more AEs were reported in the fasted state compared with the fed state. All reported AEs were mild in intensity and no new safety concerns were identified in this study.
CONFLICT OF INTEREST
S.C.R. reports employment at AstraZeneca. A.M. reports ownership of stocks/shares and employment at AstraZeneca. K.S. reports ownership of stocks/shares, stock options, and employment at AstraZeneca. N.W. has no disclosures to declare. R.G. reports employment at Parexel International, which received funding from AstraZeneca for this study.
AUTHOR CONTRIBUTIONS
S.C.R., A.M., and K.S. wrote the manuscript. S.C.R., A.M., and K.S. designed the research. S.C.R. and R.G. performed the research. S.C.R., K.S., and N.W. analyzed the data. All authors participated in the drafting, review, and approval of the manuscript and in the decision to submit for publication. All authors agree to be accountable for all aspects of the work, which includes ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
ACKNOWLEDGEMENTS
The authors would like to thank all of the study volunteers. The authors would like to acknowledge David Carlile (former AstraZeneca employee) and Eileen McBride (AstraZeneca employee) for their contributions to the development of the study design and data interpretation. The authors would also like to acknowledge Rachel Gater, PhD, of Ashfield MedComms, an Ashfield Health company part of UDG Healthcare plc, and Robyn Fowler, PhD, of OPEN Health Communications, London, UK, for medical writing support that was funded by AstraZeneca and Merck Sharp & Dohme Corp, a subsidiary of Merck & Co, Inc, in accordance with Good Publications Practice (GPP3) guidelines (http://www.ismpp.org/gpp3).
Cohen‐Rabbie S, Mattinson A, So K, Wang N, Goldwater R. Effect of food on capsule and granule formulations of selumetinib. Clin Transl Sci. 2022;15:878‐888. doi: 10.1111/cts.13209
Funding information
This study (NCT03649165) was funded by AstraZeneca, Cambridge, UK and Merck Sharp & Dohme Corp, a subsidiary of Merck & Co, Inc, Kenilworth, NJ, USA.
REFERENCES
- 1. Yeh TC, Marsh V, Bernat BA, et al. Biological characterization of ARRY‐142886 (AZD6244), a potent, highly selective mitogen‐activated protein kinase kinase 1/2 inhibitor. Clin Cancer Res. 2007;13:1576‐1583. [DOI] [PubMed] [Google Scholar]
- 2. Banerji U, Camidge DR, Verheul HMW, et al. The first‐in‐human study of the hydrogen sulfate (Hyd‐sulfate) capsule of the MEK1/2 inhibitor AZD6244 (ARRY‐142886): a phase I open‐label multicenter trial in patients with advanced cancer. Clin Cancer Res. 2010;16:1613‐1623. [DOI] [PubMed] [Google Scholar]
- 3. Denton CL, Gustafson DL. Pharmacokinetics and pharmacodynamics of AZD6244 (ARRY‐142886) in tumor‐bearing nude mice. Cancer Chemother Pharmacol. 2011;67:349‐360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Gross AM, Wolters PL, Dombi E, et al. Selumetinib in children with inoperable plexiform neurofibromas. N Engl J Med. 2020;382:1430‐1442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Dombi E, Baldwin A, Marcus LJ, et al. Activity of selumetinib in neurofibromatosis type 1‐related plexiform neurofibromas. N Engl J Med. 2016;375:2550‐2560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. AstraZeneca Pharmaceuticals LP . KOSELUGO (selumetinib). U.S. Food and Drug Administration. Prescribing Information. 2020. [Google Scholar]
- 7. Gross AM, Wolters P, Baldwin A, et al. SPRINT: phase II study of the MEK 1/2 inhibitor selumetinib (AZD6244, ARRY‐142886) in children with neurofibromatosis type 1 (NF1) and inoperable plexiform neurofibromas (PN). J Clin Oncol. 2018;36(15 suppl):10503. [Google Scholar]
- 8. Adjei AA, Cohen RB, Franklin W, et al. Phase I pharmacokinetic and pharmacodynamic study of the oral, small‐molecule mitogen‐activated protein kinase kinase 1/2 inhibitor AZD6244 (ARRY‐142886) in patients with advanced cancers. J Clin Oncol. 2008;26:2139‐2146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Leijen S, Soetekouw PMMB, Evans JTR , et al. A phase I, open‐label, randomized crossover study to assess the effect of dosing of the MEK 1/2 inhibitor selumetinib (AZD6244; ARRY‐142866) in the presence and absence of food in patients with advanced solid tumors. Cancer Chemother Pharmacol. 2011;68:1619‐1628. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Dymond AW, Howes C, Pattison C, et al. Metabolism, excretion, and pharmacokinetics of selumetinib, an MEK1/2 inhibitor, in healthy adult male subjects. Clin Ther. 2016;38:2447‐2458. [DOI] [PubMed] [Google Scholar]
- 11. Cohen‐Rabbie S, Zhou L, Vishwanathan K, et al. Physiologically based pharmacokinetic modeling for selumetinib to evaluate drug‐drug interactions and pediatric dose regimens. J Clin Pharmacol. 2021;61:1493‐1504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Tomkinson H, McBride E, Martin P, et al. Comparison of the pharmacokinetics of the phase II and phase III capsule formulations of selumetinib and the effects of food on exposure: results from two randomized crossover trials in healthy male subjects. Clin Ther. 2017;39:2260‐2275. [DOI] [PubMed] [Google Scholar]
- 13. AstraZeneca Pharmaceuticals LP . Koselugo (selumetinib). European Medicines Agency. Assessment Report. 2021. [Google Scholar]
- 14. Cohen‐Rabbie S, Mattinson A, So K, et al. A phase I healthy volunteer study on the relative bioavailability and food effect of capsule and granule formulations of selumetinib. Neuro‐oncology. 2020;22(Suppl 2):ii43.: Poster presented at: SNO Virtual Congress 2020 (CTNI‐2009). [Google Scholar]
- 15. Emami Riedmaier A, DeMent K, Huckle J, et al. Use of physiologically based pharmacokinetic (PBPK) modeling for predicting drug‐food interactions: an industry perspective. AAPS J. 2020;22:1‐15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Viskochil D, Burgess A, Oliver S, Collins T, Wang N, Karen S. A phase 1 study to assess the effect of food on the gastrointestinal toxicity and pharmacokinetics of selumetinib in adolescents with neurofibromatosis type 1‐related plexiform neurofibromas: trial in progress. Poster presented at: Children's Tumor Foundation NF Virtual Conference, 14–16 June 2021.
- 17. Schalkwijk S, Zhou L, Cohen‐Rabbie S, et al. Population pharmacokinetics and exposure‐response of selumetinib and its N‐desmethyl metabolite in pediatric patients with neurofibromatosis type 1 and inoperable plexiform neurofibromas. Cancer Chemother Pharmacol. 2021;88:189‐202. [DOI] [PubMed] [Google Scholar]


