Abstract
Crinum jagus (C. jagus; J. Thomps.) Dandy (Liliaceae) is a pantropical plant known for its medicinal values and pharmacological properties. The study assessed the protective effects and changes in oxidative stress indices due to C. jagus leaf extracts on the toluene-induced liver and kidney injuries in rats. The study was conducted on 8-week-old male Wistar rats (n = 80), weighing 243.3 ± 1.42 g. Group I, 1 ml/kg distilled water for 7 days; Group II, 4.5 ml/kg toluene once, 1 ml/kg distilled water for 7 days; Group III, 4.5 ml/kg toluene once, 500 mg/kg methanolic extract for 7 days; Group IV, 4.5 ml/kg toluene once, 500 mg/kg aqueous extract for 7 days; Group V, 500 mg/kg methanolic extract for 7 days; Group VI, 500 mg/kg aqueous extract for 7 days; Group VII, 500 mg/kg of vitamin C for 7 days; Group, VIII, 4.5 ml/kg toluene once, 500 mg/kg vitamin C for 7 days, all administrations were given by oral gavage. The phytochemical contents, absolute and relative organ weights of liver and kidneys, liver and kidney function tests, antioxidant status, as well as histological tests were analyzed using standard protocols. The tannins, flavonoids, and polyphenols were in highest concentration in both extracts, content in methanol extract (57.04 ± 1.51 mgg−1, 35.43 ± 1.03 mgg−1, 28.2 ± 0.34 mgg−1 respectively) > aqueous extract (18.74 ± 1.01 mgg−1, 13.43 ± 0.47 mgg−1, 19.65 ± 0.21 mgg−1 respectively). In the negative control group (II), bodyweights significantly (P < 0.05) reduced by 22%, liver weight and kidney weight significantly (P < 0.05) increased by 42% and 83% respectively, liver-to-bodyweight and kidney-to-bodyweight ratios increased significantly (P < 0.05); serum liver function tests (LFTs) i.e., bilirubin, alkaline phosphatase (ALP), Alanine aminotransferase (ALT), Aspartate aminotransferase (AST), Gamma-glutamyl transferase (GGT), and serum kidney function tests (creatinine and urea) were significantly (P < 0.05) elevated; oxidant status (tissue malondialdehyde; MDA) was significantly (P < 0.05) elevated, antioxidant status i.e., tissue superoxide dismutase (SOD), catalase (CAT), and glutathione (GSH) levels was significantly (P < 0.05) reduced; with markedly visible renal and hepatic histopathological findings, compared to the normal control group. In C. jagus extract test groups (III and IV), the parameters were significantly (P < 0.05) alleviated and reversed to normal/near normal compared to the negative control. The LFTs, kidney function tests, and antioxidant status were significantly (P < 0.05) more improved with the methanol extract test and standard control groups compared to the aqueous extract test group; Also, the methanol extract test group showed better histological features than the aqueous extract test and standard control groups. The methanolic extract shows better antioxidant potential due to the availability of more nonenzymatic antioxidants (tannins, flavonoids, and polyphenols). The findings showed that toluene is a very aggressive xenobiotic due to the promotion of oxidative stress and peroxidation of cellular lipids, but C. jagus leaves provide significant protection through the reducing power of nonenzymatic antioxidants and their ability to induce endogenous antioxidant enzymes (SOD, CAT, and glutathione reductase or GR) causing reduced cellular lipid peroxidation and tissue damages, quickened tissue repair, and improved cell biology of liver and kidneys during toluene toxicity. The methanol leaf extract provides better protection and should be advanced for more experimental and clinical studies to confirm its efficacy in alleviating oxidative stress tissue injuries, specifically due to toluene.
Abbreviations: ALP, Alkaline phosphatase; ALT, Alanine aminotransferase; AST, Aspartate Aminotransferase; GGT, Gamma-glutamyl transferase; GSH, Glutathione; GR, glutathione reductase; LFTs, Liver function tests; MDA, malondialdehyde; SOD, Superoxide dismutase; TOL, Toluene; VC, Vitamin C
Keywords: Anti-lipid peroxidation, Antioxidants, Catalase Crinum jagus, Glutathione superoxide dismutase, Histoprotective, Malondialdehyde, Toluene toxicity
Graphical Abstract
Highlights
-
•
Toluene is toxic to body, kidneys and liver due to oxidative stress and lipid peroxidation in toluene-induced animals.
-
•
Antioxidants/ identified from aqueous and methanol Crinum jagus leaf extracts, induced the endogenous antioxidant pathway.
-
•
Improved antioxidant status reduces lipid peroxidation and necrosis in kidneys and liver of toluene-induced rodents.
-
•
Methanol leaf extract provides better anti-oxidation, necessitates further investigations in oxidative stress injuries.
1. Introduction
Toluene is a gravely toxic and fatal compound to mammals [70], [96], with the liver and kidneys being extremely sensitive to the toxic effects of xenobiotics e.g. toluene and drugs due to the presence of high detoxifying, degrading, and bioactivation enzymes and the ability of the organs to metabolize the compounds [25], [3], [45], [52], [62], [73], mostly due to the presence of cytochrome P450 enzymes (CYP450) in the liver [8], [95] and renal cortex [50], [95], which is responsible for the metabolism of the xenobiotics in the organs [50], [8], [95].
The oxidative stress induced by xenobiotics such as toluene may be one of the mechanisms responsible for numerous liver and kidney diseases [25], [31], [45], [73]. The oxidative hepatic and renal tissue damages induce protein markers of oxidative stress such as elevated levels of malondialdehyde (MDA), protein carbonyls, metallothionein, and diminished levels of superoxide dismutase (SOD), glutathione reductase (GR), and catalase (CAT) in the liver and kidney tissues; and significantly lowers antioxidant status and levels of uric acid in the blood [25], [3], [45], [52], [73], which lead to impairment of biochemical functions [2], [8] and histopathological changes such as fibrosis, apoptosis, degradation, etc. in mammalian liver and kidneys [14], [49], thus causing the burdens of drug or xenobiotic-induced kidney and liver diseases [15], [31], [59]. Renal and hepatic damage and dysfunction after exposure to xenobiotics induce in-vitro and in-vivo epigenetic transformations in mammalian tissues, predisposing to the development of acute and chronic health effects e.g. liver and kidney diseases, and in addition to their cytotoxic effects, xenobiotics have proven human carcinogenic potential [31]. For example, exposure to toluene-related compounds has been related to cancers of the prostate, lungs, and thyroid [31], [44], [72], [79], renal and hepatic cancers [1], [75], [76], and death [70], [96].
Stimulation of endogenous mammalian antioxidant systems (superoxide dismutase; SOD, catalase; CAT, and glutathione; GSH) has been proven to be useful in scavenging, binding with, and neutralizing the free oxygen radicals produced during oxidative stress conditions thereby controlling oxidative stress-induced toxicity [86], [88]. Recently, studies regarding the capability of Crinum jagus to remove toxic agents from animal tissues by the antioxidant mechanism have been conducted [32], [57], [65], [85], [90].
Crinum jagus (J. Thomps.) Dandy (Liliaceae) is a pantropical plant in Africa, Asia, and America [51], [89], it belonged to Amaryllidaceae but recently scientists have classified it under the family Liliaceae [4]. The phytochemical components of the plant include alkaloids, essential oils, tannins, reducing sugars, terpenes, steroids, and phenolics [32], [9],[34], [65] with many pharmacological properties: inhibition of acetylcholinesterase, anti-mitotic, anti-asthmatic, antihemorrhagic, antibacterial, anti-tumor, antiviral, anti-parasitic, anti-diabetic, anti-obesity, antidiarrheal, anti-toxicant, immune-stimulators, and anticonvulsant [18], [32], [38], [63], [64], [65], [82], [9].
The numerous pharmacological properties of Liliaceae spp. including C. jagus is attributable to their strong antioxidant capacity i.e., the plants induce antioxidant protective and repair mechanisms during oxidative stress injuries [58], [65], [74], [85], [86], [88], [89], [9]. Extracts from C. jagus species including Crinum L. (Amaryllidaceae J.St. Hil.) and C. jagus (J. Thomps.) Dandy (Liliaceae) have been shown to have both in vitro [32], [57], [65], [90] and in vivo [85], [90] antioxidant activities in mammalian/rodent tissues [32], [57], [65], [85], [90]. The antioxidant attributes of the plant have been exploited in testing the role of the plant in providing alleviative effects on oxidative-related injuries in several rodent tissues [23], [64], [65], [85], [90]. Therefore, the plant being tested is vital as it might be used to provide medicinal effects on liver and kidney toxicity arising from several causes, and to reduce the burdens of liver and kidney diseases.
Stimulation of antioxidant systems by antioxidants of C. jagus is one of the mechanisms by which the plant provides its protective activities in tissues subjected to oxidative stress-induced damages, here the extracts of the herb provide strong antioxidant and anti-lipid peroxidation powers [32], [9],[34], [65], [64]. Previous findings demonstrated flavonol kaempferol from Crinum jagus L. (Amaryllidaceae) leaf extract to be having strong protective powers due to its antioxidant and anti-lipid peroxidation properties in pentylenetetrazole-induced kindling of mice, and this was associated with decreased lipid peroxidation, augmented endogenous antioxidant enzymes (SOD, CAT, and GR) in the damaged brain tissues of extract-treated mice compared to the untreated negative controls [85].
The improved antioxidant status of the animals, being attributed to the nonenzymatic plant-based antioxidants causes lowered lipid peroxidation, decreased MDA levels, reduced cellular lipid peroxidation, and tissue injuries, and quickened tissue repair which protects the animal tissues from oxidative stress-induced injuries [54], [80]. These pharmacological properties are important since they could aid in the formulation of essential molecules to be used in the management of oxidative-related ailments [67]. However, to the best of our knowledge, little is known about the medicinal effects of the plant (C. jagus) in cases of liver and kidney toxicity and this formed the basis for the study.
The study aimed to elucidate the protective effects as well as changes in oxidative stress indices in hepatic and renal tissues due to C. jagus leaf extracts during toluene-induced toxicity in rats.
2. Materials and methods
2.1. Plant materials
Freshly harvested plant material (Crinum jagus leaves) were collected in Ishaka-Bushenyi municipality, western Uganda (GPS coordinates; Latitude: −0.53828 S 0°32'17.79583" Longitude: 30.14546 E 30°8′43.64405"), and identified as Crinum jagus (J. Thomps.) Dandy (Liliaceae/ Amaryllidaceae) by a botanist at Mbarara University of Science and Technology (code No. SHO 1000). The methanol and aqueous extracts were prepared using standard methods [32], [81]. Briefly, “dry leaves were ground using a manual metallic mortar and pestle into fine powders that were kept in air-tight glass containers. The methanol and aqueous extracts of the plant were prepared by soaking 200 g of the powdered sample in 1 L of respective solvent (absolute methanol or distilled water) in an Erlenmeyer conical flask (Corning® CLS431145, Darmstadt, Germany). This was placed on an orbital shaker incubator (Amerex®, Gyromax 727, NY, USA) to allow complete mixing (24 h, 100 rpm, 30 oC) after which the debris was removed from the extracts by centrifugation (5000 rpm, 15 min). The solvents from the methanol and aqueous extracts were filtered using Whatman number 1 filter papers (Whatman® qualitative filter paper, Grade 1, WHA1001325, Darmstadt, Germany) and were concentrated to dryness using a rotary evaporator (Büchi® Rotavapor® R-210, Büchi 23011V010, Darmstadt, Germany) at 53 °C to evaporate off the methanol and water respectively. The remaining dry concentrated extracts were weighed and the yield was stored in universal bottles put in a refrigerator at 5 °C (Samsung RF22N9781SR, UPC: 887276259277, Brooklyn, NY, USA) till used. When needed for use, the dry concentrates were dissolved in known volumes of respective solvents to make the required dose for phytochemical and in vivo antioxidant activities” [32], [81].
2.2. Animals
The study was performed on male Wistar rats of similar age (8 weeks of age) and weight range (243.3 ± 1.42 g) at the time of study initiation following previous studies [21], [64], [8], [85], and male rats were used as previous laboratory studies have shown no differences between female and male rats [19], [20]. The rats were obtained and contained in separate stainless-steel wire mesh cages (60 cm × 50 cm x 30 cm) on Paper Pellet Bedding (So Phresh® Paper Pellet Small Animal Litter, ASIN: B013GL52Y8, Amazon.com Inc., USA), at the laboratory animal facility in the school of Pharmacy, Kampala International University, western campus, Bushenyi, Uganda. Standard commercial diet (Nuvilab Cr-1 ®, Nuvital Nutrientes, S/A) and drinking water were available ad libitum to all the animals, and were maintained in well-regulated environmental conditions; continuous monitoring, good husbandry practices, the humidity of 55 ± 5%, room temperature (25–30 °C), and under 12:12-hour light: dark cycles, in line with ARRIVE guidelines [29], [6], [61].
2.3. Experimental design
The study was conducted for one week based on previous studies [21], [64], [8], [85]. The sample size was based on previous studies [26], [37]. After two weeks of adaptation 80 rats were randomly allocated to 8 experimental groups as follows: Group, I received 1 ml/kg of distilled water by oral gavage (per os., i.e., p.o) for 7 successive days (normal control, n = 10); Group, II received 4.5 ml/kg of toluene once, p.o, plus 1 ml/kg of distilled per os. for 7 successive days (negative control, n = 10); Group, III received 4.5 ml/kg of toluene once p.o, and 500 mg/kg of methanol extract p.o for 7 successive days (methanol extract test group, n = 10); Group, IV received 4.5 ml/kg of toluene once p.o, and 500 mg/kg of the aqueous extract p.o for 7 successive days (aqueous extract test group, n = 10); Group, V received 500 mg/kg of methanol extract p.o for 7 successive days (methanol extract normal control group, n = 10) while group VI, received 500 mg/kg of aqueous extract p.o for 7 successive days (aqueous extract normal control group, n = 10); Group VII received 500 mg/kg of vitamin C dissolved in distilled water p.o for 7 successive days (standard normal control group, n = 10), while Group, VIII received 4.5 ml/kg of toluene once p.o, and 500 mg/kg vitamin C dissolved in distilled water p.o for 7 successive days (standard control group, n = 10).
Toluene was administered once throughout the whole study (single oral dose exposure) per os. as based on previous studies [21], [8], whereas the plant extracts were administered once daily for seven days per os (multiple oral dose exposures) using therapeutic doses of C. jagus from previous studies [64], [85], vitamin C dose (500 mg/kg) used in our study had been previously shown to provide significant antioxidant potential on oxidative stress conditions in mice and rats [33]. Analytical grade liquid toluene- 99.5% pure (Santa Cruz Biotechnology, Germany; 99.5%) was used to induce toxicity in the study animals, the toluene dose used in the currently (4.5 ml/kg) was 75% of the benchmark dose (BD) described previously [21], [8], BD is the maximum dose that is associated with significant neurotoxicity without causing rodent death. Toluene is non-soluble in water, corrosive, and was therefore administered by oral gavage without dilution based on previous methods [21], [8].
At the termination of the studies (on the morning of sacrifice), the rats were weighted using a large plastic beaker (Kent Scientific®, Torrington, USA), with a digital animal weighing scale (Kent Scientific®, SCL-1053, Torrington, USA) using standard methods [28], [36], [7], sacrificed using thiopental sodium (25 mg/kg body weight, Santa Cruz Biotechnology, Germany) based a method described previously [42], and immediately blood samples were collected by cardiac puncture and placed in red top plain vacutainer tubes for laboratory analysis of serum liver enzyme activities (alanine aminotransferase-ALT, alkaline phosphatase- ALP, aspartate Aminotransferase- AST, and gamma-glutamyl transferase- GGT), serum bilirubin and serum kidney function tests (creatine and urea) using previous methods [28], [29]. After getting the blood specimen, the kidneys and liver were harvested via linea alba incision, dissected free of adhering tissue, and weighted (absolute organ weight) using a large plastic beaker (Kent Scientific®, Torrington, USA) on an electronic weighing scale (Kent Scientific®, SCL-1053, Torrington, USA) using previous methods [28], [41]. Immediately after the weights of the organs were taken, the tissues were quickly cut and washed in ice-chilled normal saline, pieces of organ specimens were stored in 10% phosphate-buffered saline (PBS) and 10% neutral buffered formalin (NBF) fixative at room temperature (22–300 C) for biochemical assays (SOD, CAT, GSH, and MDA) in tissue homogenates and histopathological examination of tissues respectively [13], [42], [47], [84].
2.4. Analytical procedures
2.4.1. Phytochemical analysis of C. jagus leaf extracts
Phytochemical analyses of methanol and aqueous leaf extracts of the herb were performed in triplicates in the Ethnobotany Laboratory at the Department of Biological Sciences, Makerere University in Uganda. Qualitative analysis for the main active groups of bioactive components such as alkaloids, saponins, tannins, cardiac glycosides, hydroxyanthraquinones, phlobatannins, terpenoids, flavonoids, phenols, among others was carried out using standard qualitative analytical methods as described by Trease and Evans, and Oke-Altuntas et al. Oke-Altuntas et al. [66], [87]. Quantitative analyses of the majorly compounds identified from the above qualitative analysis of the two crude extracts of the herb were then carried out in the same Laboratory using standard spectrophotometric analytical methods (PerkinElmer’s Lambda 35 UV/Vis, USA) described previously [22], [55], [56], [71], [77], [78], [93].
2.4.1.1. Polyphenol analysis
The polyphenol contents were assessed using the Folin-Ciocalteu method described previously [22], [71], at a spectrophotometer absorbance (O.D) of 730 nm, and the result was expressed as milligrams of phenol per gram of dry extract (g/100 g).
2.4.1.2. Flavonoid analysis
The flavonoid levels were analyzed using the aluminum chloride test using a method described previously [71], [93], at O.D = 420 nm expressed as milligrams of flavonoid per gram of dry extract.
2.4.1.3. Tannin analysis
The tannin levels were determined using the vanillin-hydrochloric acid test by employing a method by Morrison et al. Morrison et al. [55] as suggested by Piana [71], at O.D = 500 nm expressed. The data were expressed as milligrams of tannin per gram of dry extract.
2.4.1.4. Saponin analysis
The amounts of saponins were determined by the vanillin–sulfuric acid method described by Shiau et al. Shiau et al., [78], at O.D 535 nm, expressed as milligrams of saponin per gram of dry extract.
2.4.1.5. Cardiac glycoside analysis
The amounts of glycosides in the powdered sample were determined using the Baljet’s test described previously [56], and the absorbance of the colored complex was determined spectrophotometrically at 495 nm, expressed as milligrams of glycoside per gram of dry extract.
2.4.1.6. Alkaloid analysis
The contents of alkaloids were analyzed using the Dragendorff’s test described previously [71], [83], at O.D = 435 nm expressed. The data were expressed as milligrams of alkaloids per gram of each fraction.
2.4.1.7. Terpenoid analysis
The terpenoid content of the extracts was measured using the spectrophotometric analytical method (PerkinElmer’s Lambda 35 UV/Vis, USA) following the method described previously [48], at O.D = 471 nm. The data were expressed as milligrams of terpenoids per gram of each fraction.
2.4.2. In vivo antioxidant and protective effects of C. jagus leaf extracts extract
2.4.2.1. Determination of the bodyweight, absolute, and relative organ weights of liver and kidney
The rats were weighed using a large plastic beaker (BrandTech®) on an electronic weighing scale (Kent Scientific®) using methods described previously [28], [36], [7]. The kidneys and liver were weighted to get the absolute organ weights using a large plastic beaker (BrandTech®) on an electronic weighing scale (Kent Scientific®) using previous methods [28], [41]. Body and absolute organ weights were expressed in grams, and the relative organ weight was obtained as a percentage of body weight based on the organ-to-bodyweight ratio (OBR) in the rats; (OBR % = organ weight/bodyweight x 100%) using previous methods [12], [53], and OBRs in combination with absolute organ weight data were used as accurate indicators of organ toxicity [12], [53].
2.4.2.2. Assay of the serum liver and kidney function biochemical markers
This was done by measuring the activity of serum liver biochemical enzymes, and levels of serum creatinine and urea respectively. The laboratory analysis of serum liver enzyme activities (ALT, ALP, AST, GGT), serum bilirubin, and serum kidney function tests (creatine and urea) were done using previous methods [29], [8]. Briefly, the collected samples of blood were left to stand for an hour to clot and serum samples were separated from the clots by centrifugation at 3000 rpm for 10 min (CYAN CL008N, USA). Biochemical parameters in serum (ALT, ALP, AST, GGT, creatinine, urea, and bilirubin) were measured using an automated biochemical analyzer (Roche Diagnostics, Roche Cobas Integra 800, Mannheim, Germany) using standard kits supplied by Santa Cruz Biotechnology, Germany [29], [8]. ALT, ALP, AST, and GGT values were expressed in units per liter (U/L), whereas creatinine, urea, and bilirubin were expressed in mg/dL.
2.4.2.3. Determination of tissue biochemical markers in animal tissues
Kidney and liver tissue extracts were prepared using previous methods, as described in brief, pieces of kidney and liver (1 g) were dipped in 9% w/v of ice-cold buffered sucrose (0.25 M) with HEPES (1 mM, pH = 7.4). The tissues were cut into many pieces and spun in the buffer to remove blood. The kidney and liver pieces were then crushed into fine components and transferred to the ice-cold homogenizing vessel to be homogenized (6 strokes of pestle, full speed) to make homogenates of 10% w/v using sucrose buffer solution, and the homogenate was stored at − 80°C. The homogenates (5 ml) were centrifuged (CYAN CL008N, USA; 4 °C, 9000 rpm, 10, minutes) to get supernatants for the analysis of GSH, MDA, and antioxidant enzymes (SOD, CAT) [37], [94].
2.4.2.4. Determination of SOD activity in animal tissues
Assay for superoxide dismutase (SOD) activity was done by spectrophotometric methods previously described [13], [84], this involved suppression of nitroblue tetrazolium reduction, and xanthine-xanthine oxidase was employed as the superoxide generator. The assays were done using tissue samples in a reaction with 0.1 M sodium phosphate buffer Ph 7.8, 0.3 mM hypoxanthine, 7.5 mM cytochrome c. The reaction was initiated by combining 5 U of xanthine oxidase in 1 ml of sample. Cytochrome c reduction was observed for 60 s at 550 nm in a microplate spectrophotometer (PerkinElmer’s Lambda 35 UV/Vis, USA) [13], [84]. The values were expressed as units of SOD per milligram of protein or tissue (Units/mg protein).
2.4.2.5. Determination of CAT activity in animal tissues
Assay of catalase (CAT) activity was done using a standard technique by Beer & Seizer. In brief, “295 µL of H2O2 solution was added to 5 µL of samples and the absorbance, OD, was measured (240 nm, 5 min at 30-second intervals). The quantity of catalase was quantified as a difference in OD x 1000/43.6 x mg of protein per ml of the reaction mixture” [80]. The activity was expressed as units of CAT per milligram of protein or tissue (Units/mg protein).
2.4.2.6. Determination of glutathione concentration in animal tissues
The determination of glutathione (GSH) was done by the dithiobis (2-nitrobenzoic acid); DTNB spectrophotometric method as previously described [47]. “Briefly, specimens were diluted in DTNB buffer to the required concentrations. 50 µL of specimen or GSH standard was measured on a microplate and 100 µL of DTNB reaction buffer was added. Absorption was monitored (405 nm, 3 min) in association with TNB formation with a Scientific microplate spectrophotometer (PerkinElmer’s Lambda 35 UV/Vis, USA). The absorption difference between 2 and 0 min was used for the quantification of GSH. The values were expressed as micromoles of GSH per milligram of protein or tissue (μMol/mg protein)” [47].
2.4.2.7. Determination of malondialdehyde concentration in animal tissues
The determination of malondialdehyde (MDA) was done by the nonspecific spectrophotometric thiobarbituric acid reactive substances (TBARS) measurement as previously described [42]. “In particular, the concentration was determined by using the equation;.
Concentration = Absorbance/(Molar extinction co-efficient x 1 L) x 106 nm where an extinction coefficient of 156 was used. The values were expressed as micromoles of MDA per milligram of protein or tissue (μMol/mg protein)” [42].
2.4.2.8. Histopathological examination of animal tissues
Histopathological tests were performed using standard histological methods as previously described [30], [37]. Briefly, portions of liver and kidney tissues were fixed in 10% NBF fixative solution for 48 h, after which tissues were processed using routine histology techniques by dehydrating with graded alcohols, clearing with xylene, then embedding into paraffin wax, and finally, every organ was randomly sectioned into six, 5 µm-thick histological sections. The sections were then placed on slides and stained with hematoxylin and eosin (H &E) stain [30], [37]. The stained sections were mounted in mounting media and qualitative histological examination of the sections was done and photographed with a light microscope (Nikon Eclipse Ci-L Upright Microscope, New York, USA), at a magnification of 250x or 400x, digital camera (Nikon DS-Fi1c Digital Camera, New York, USA), and imaging software (Nikon NIS- NIS-Elements F Ver4.60.00 Imaging software), for image analysis and documentation. A qualitative examination of the tissues was done under the light microscope using previously described methods [37], [46], [94].
2.5. Statistical analysis
The data was recorded and then entered in Graph Pad Prism Version 6 for statistical analysis. Data were tested for normalcy and parametric tests were conducted using Graph Pad Prism. Data were subjected to one-way analysis of variance (ANOVA) and Tukey’s multiple comparison test was used to determine sources of variation and significant differences (P < 0.05) at 95% confidence interval were indicated with different superscript letters (a, b, c, and d). The information was expressed as mean ± SD and presented on graphs and tables.
3. Results
3.1. Phytochemical composition of C. jagus leaf extracts
Phytochemical analysis of both the aqueous and methanol C. jagus leaf extracts revealed the presence of alkaloids, flavonoids, tannins, terpenoids, cardiac glycosides, polyphenols, and saponins. The standard spectrophotometric analytical methods showed the content of phytochemicals in methanol extract as being in the order tannins > flavonoids > polyphenols > alkaloids > terpenoids > cardiac glycosides > saponins, while that of the aqueous extract was in the order polyphenols > tannins > flavonoids > alkaloids > terpenoids > cardiac glycosides > saponins. Considering both extracts, the tannins, polyphenols, and flavonoids were in the highest concentration (Table 1).
Table 1.
Total phytochemical content in methanol and aqueous leaf extracts of C. jagus.
| Phytochemical compound | Methanol (mgg−1 Extract) | Aqueous (mgg−1 Extract) |
|---|---|---|
| Tannins | 57.04 ± 1.51 | 18.74 ± 1.01 |
| Flavonoids | 35.43 ± 1.03 | 13.43 ± 0.47 |
| Polyphenols | 28.2 ± 0.34 | 19.65 ± 0.21 |
| Alkaloids | 6.64 ± 0.42 | 4.56 ± 0.03 |
| Terpenoids | 3.20 ± 0.11 | 2.78 ± 0.23 |
| Cardiac glycosides | 3.06 ± 0.13 | 1.13 ± 0.67 |
| Saponins | 1.34 ± 0.02 | 0.87 ± 0.04 |
Values represent means ± standard deviations (SD) for triplicate experiments.
3.2. C. jagus leaf extracts alleviated the bodyweight, and the absolute and relative liver weight and kidney weight of male Wistar rats under toluene toxicity
The negative control group showed significantly (P < 0.05) reduced bodyweights by 22%, and significantly (P < 0.05) increased absolute liver weight and kidney weight by 42% and 83% respectively compared to the normal control group. Administration of aqueous and methanol C. jagus extracts in the two extract test groups (group III and IV) led to significantly (P < 0.05) increased bodyweights, and significantly (P < 0.05) reduced absolute liver weight and kidney weight compared to the negative control group, with no significant (P > 0.05) difference on the body and organ weights between the methanol extract test group and aqueous extract test group. Additionally, there was no significant (P > 0.05) difference in the bodyweights and absolute organ weights of the extract test groups (group III and IV) compared to the normal control, standard control, standard normal control as well as to the extract normal control groups (group V and VI) (Fig. 1 A, C & supplementary file 1:Table 1).
Fig. 1.
Effect of methanol and aqueous C. jagus leaf extracts on the absolute and relative organ weights of toluene-exposed male Wistar rats. A. Absolute liver weight. B. Relative liver weight. C. Absolute kidney weight. D. Relative kidney weight. Each value is expressed as mean ± SD. Different letters (a, and b) meant significant differences among the groups, (p < 0.05). Abbreviations: TOL = toluene, ME = methanol extract, AE = aqueous extract, VC = vitamin C.
Furthermore, the negative control group showed significantly (P < 0.05) higher liver and kidney-to-body weight ratios by 80% and 140% respectively compared to the normal control; aqueous and methanol extracts test groups showed significantly (P < 0.05) reduced organ-to-bodyweight ratios of liver and kidney compared to the negative control group, with no significant (P > 0.05) difference on the organ-to-bodyweight ratios of methanol extract test group and aqueous extract test animals. Also, there was no significant (P > 0.05) difference in the organ-to-bodyweight ratios of liver and kidney of extract-treated test groups compared to the normal control, standard control, standard normal control, as well as to the extract-treated normal control rats (Fig. 1 B, D).
3.3. C. jagus leaf extracts alleviated the serum liver and kidney function biochemical markers of male Wistar rats under toluene toxicity
Animals in the negative control group showed significantly (P < 0.05) higher serum levels of liver and kidney function biochemical parameters (ALT, ALP, AST, GGT, bilirubin, creatinine, and urea) compared to normal control; extract-treated test group revealed significantly (P < 0.05) lower levels of liver and kidney function biochemical markers compared to the negative control group, with significant (P < 0.05) difference on the parameters between methanol extract test group and the aqueous extract test group. Additionally, there was no significant (P > 0.05) difference in serum levels of liver and kidney function test parameters of the methanol extract test group compared to the normal control, standard control, standard normal control, and the extract normal control groups. However, there was a significant (P < 0.05) difference in the levels of parameters of the aqueous extract test group compared to the normal control, standard control, standard normal control, methanol extract test group, and extract normal control groups (Fig. 2).
Fig. 2.
Effect of methanol and aqueous C. jagus leaf extracts on serum liver and kidney function tests of toluene-exposed male Wistar rats. A. Alanine aminotransferase. B. Alkaline phosphatase. C. Aspartate Aminotransferase. D. Gamma-glutamyl transferase. E. Bilirubin. F. Creatinine. G. Urea. Each value is expressed as mean ± SD. Different letters (a, b, and c) meant significant differences among the groups, (p < 0.05). Abbreviations: TOL = toluene, ME = methanol extract, AE = aqueous extract. ALT = Alanine aminotransferase, ALP = Alkaline phosphatase, AST = Aspartate Aminotransferase, GGT = Gamma-glutamyl transferase, VC = vitamin C.
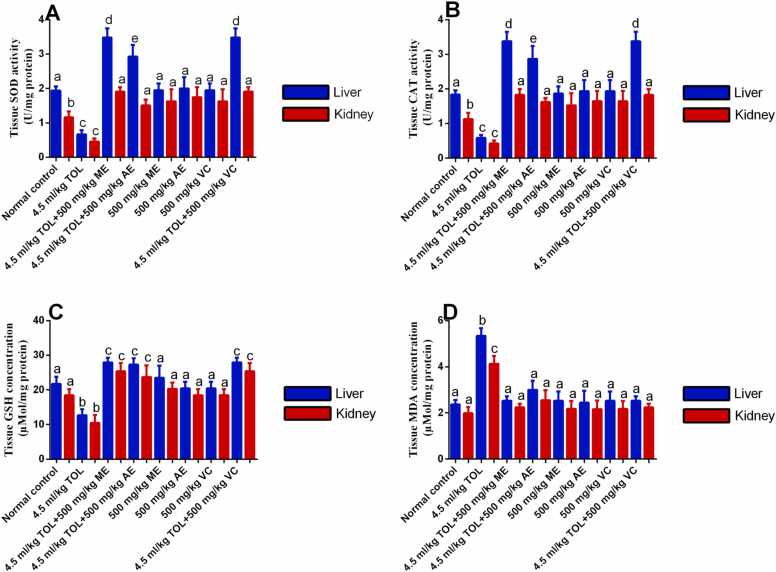
3.4. C. jagus leaf extracts alleviated the activities of SOD and CAT, and concentrations of GSH and MDA in liver and kidney tissues of male Wistar rats under toluene toxicity
Animal tissues of the negative control group showed significantly (P < 0.05) reduced SOD and CAT activities, and GSH levels, and significantly (P < 0.05) increased MDA levels in liver and kidney compared to normal control; extract test group tissues showed significantly (P < 0.05) higher SOD and CAT activities, and GSH levels, and significantly (P < 0.05) lower MDA levels in liver and kidney compared to the negative control group, with no significant (P > 0.05) difference on the GSH, and MDA levels but with significant (P < 0.05) difference in SOD and CAT activities of methanol extract test group compared to the aqueous extract test group. The SOD and CAT activities and GSH levels were significantly (P < 0.05) higher in extract test groups compared to the normal control and extract normal control groups. However, there was no significant (P > 0.05) difference in the MDA level between the extract test groups compared to the normal control and extract normal controls. There were no significant (P > 0.05) differences in the SOD, CAT, GSH, and MDA levels between the methanol extract test group compared to the standard control group. Furthermore, significantly (P < 0.05) higher levels of SOD, CAT, and MDA were found in the liver than kidney as follows: in the normal control and extract test groups there was significantly (P < 0.05) higher SOD and CAT activity in the liver compared to the kidney; negative control group showed significantly (P < 0.05) higher MDA levels in liver compared to kidney (Fig. 3).
Fig. 3.
Effect of methanol and aqueous C. jagus leaf extracts on the levels of antioxidants and oxidant enzymes in liver and kidney tissues of toluene-exposed male Wistar rats. A. Superoxide dismutase. B. Catalase. C. Glutathione. D. Malondialdehyde. Each value is expressed as mean ± SD. Different letters (a, b, and c) meant significant differences among the groups, (p < 0.05). Abbreviations: TOL = toluene, ME = methanol extract, AE = aqueous extract. SOD = superoxide dismutase, CAT = catalase, GSH = glutathione, MDA = malondialdehyde, VC = vitamin C.
3.5. C. jagus leaf extracts alleviated the histopathological changes in liver and kidney tissues of male Wistar rats under toluene toxicity
Kidney sections from the negative control group showed dilatation of Bowman’s space with glomerular atrophy in the renal cortex compared to the normal control and extract normal controls groups whose sections showed normal structural integrity of Bowman’s space, glomeruli, tubules, and renal cortex. In the extract test groups, histological appearances of Bowman’s space and glomeruli were markedly improved, with the methanol extract test group similar to the standard control group, providing a better (normal) histological appearance of the structures compared to the aqueous extract test group (Fig. 4).
Fig. 4.
Effect of methanol and aqueous C. jagus leaf extracts on the histopathological changes in kidney tissue of toluene-induced male Wistar rats. a. Normal control. b. Methanol extract normal control. c. Aqueous extract normal control. d. Negative control. e. Aqueous extract test. f. Standard control. g. Methanol extract test. Glomerulus (G), inflammatory cell infiltration (black arrow), dilation of bowman space (red arrow). H&E; 250x).
Liver sections from the negative control group showed histopathological changes marked by the extensive presence of fatty droplets, pyknotic cells, and inflammatory cell infiltration compared to normal control and extract normal controls groups whose sections showed normal morphology of the hepatocytes with a regular arrangement, and visibly clear stained nucleus and cytoplasm. In the extract test groups, inflammatory cell infiltration, pyknotic cells, and fatty droplets were markedly reduced and histological appearance and arrangement of hepatocytes were normal/near normal, with the methanol extract test group and standard control group providing a better (normal) histological appearance of the structures compared to the aqueous extract test group (Fig. 5).
Fig. 5.
Effect of methanol and aqueous C. jagus leaf extracts on the histopathological changes in liver tissue of toluene-induced male Wistar rats. a. Normal control. b. Methanol extract normal control. c. Aqueous extract normal control. d. Negative control. e. Aqueous extract test. f. Standard control. g. Methanol extract test. Hepatocyte (H), central vein (CV), pyknotic cell (black arrow), and fatty droplet (red arrow). (H&E; 250x).
4. Discussion
At the time of the study, little was known regarding the antioxidant and protective effects of Crinum jagus (C. jagus); only three studies were found in the National Center for Biotechnology Information (NCBI) library using the keywords ‘Crinum jagus antioxidant’ [17], [85], [89], also, data involving the antioxidant or protective attributes of C. jagus on toluene-induced damages in liver and kidneys of rodents were not found during NCBI search with the key words ‘Crinum jagus toluene toxicity’, which formed the basis for this study. Accordingly, the current findings showed that the major phytochemical components of the methanolic Crinum jagus leaf extract (more than the aqueous extract) are associated with significant antioxidant and protective effects against toluene-induced oxidative stress injuries in the liver and kidneys of rats due to the activation of the endogenous antioxidant enzymes (SOD, CAT, and GR) with a resultant increased antioxidant status, inhibition of lipid peroxidation pathways and lowering of cellular lipid peroxidation in the two organs.
The isolated phytochemicals of the aqueous and methanol Crinum jagus (J. Thomps.) Dandy (Liliaceae) leaf extracts (alkaloids, flavonoids, tannins, terpenoids, cardiac glycosides, polyphenols, and saponins) are comparable to those previously isolated from the aqueous and methanol leaf extracts of Crinum jagus (Amaryllidaceae J.St. Hil.) i.e. higher levels of phytochemicals were isolated from the methanolic than aqueous extract, and the tannins, polyphenols, and flavonoids were in the highest concentration in the two extracts of methanolic and water [32]. However, results from the present study found three extra phytochemicals (terpenoids, cardiac glycosides, and saponins) and about thrice less phytochemical concentration in the current C. jagus leaf extracts compared to [32]. The disparities in phytochemical content between the current herb and others might be due to environmental or seasonal changes where the herb was collected, changes during extraction and/or storage, uniqueness of individual species of the plant that may alter the available plant components [34]. The phytochemicals found in the current C. jagus leaf extracts are in tandem with previous findings where phytochemical analysis of the aqueous and ethanolic extracts of C. jagus (Amaryllidaceae/Lilaceae J.St. Hil.) bulb found the presence of alkaloids, flavonoids, tannins, terpenoids, phenolics, saponins, and glycosides [9,34], and an abundant concentration of flavonoids, phenolics, and tannins [9,34].
Most studies attribute the protective and antioxidant scavenging capacities from herbs against the reactive oxygen species (ROS) like H2O2, nitric oxide, and hydroxyl peroxide radicals generated during oxidative stress-induced pathological processes, to be due to the presence of the numerous bioactive compounds in the medicinal plants [27], [50], [69]. There is a positive correlation between the total phytochemical content of plant extracts, and their antioxidant potential and lipid peroxidation inhibition activities [43], [50]. Thus, since tannins, flavonoids, and polyphenols were in the highest concentration in the current C. jagus extracts, it follows that the antioxidant and lipid inhibition protective capacity of the secondary metabolites against toluene toxicity is highest with tannins, flavonoids, and polyphenols. The presence of significant levels of tannins, flavonoids, and polyphenols in C. jagus provides strong antioxidant and anti-lipid peroxidation activities in vitro and in vivo [32], [9],[34], [65], [64], agrees with data from other plant extracts that attribute the antioxidant, anti-lipid peroxidation, and the protective capacities of herbs to be mainly due to the presence of phenolics, tannins, polyphenols, and flavonoids (nonenzymatic antioxidants) [10], [16], [39], [50].
During the acute one-week period of the study, administration of a single oral dose of toluene, 4.5 ml/kg (3900 mg/kg) per gavage was non-lethal to our rats but caused significant toxic effects to the body tissues, these findings concur with previous studies where similar doses of toluene (3–6 ml/kg) were associated with acute tissue toxicity in rats [21], [5], [8], agree with data that stipulates that the xenobiotic is generally toxic to mammals including humans and rodents [70], [96]. The toluene toxicity caused significant bodily and organ toxicity (significant decrease in body weight, elevated absolute and relative organ weights kidneys and liver) of the animals in the negative control group compared to the normal control group, similar findings are observed with acute-exposure oral doses of toluene between 1500 and 5200 ppm [12], [21], [53], [91], [92].
Again, toluene toxicity in our animals was associated with significantly decreased antioxidant status (SOD and CAT activities and GSH levels), and increased oxidant status (amounts of MDA) in the liver and kidneys of the negative control group compared to normal control. Additionally, there was a significant distortion of renal and hepatic histology evidenced by a marked increase in inflammation and apoptosis/necrosis in the kidney and liver tissues, and significant impairment of liver and kidney functions i.e. significantly raised liver function tests-LFTs (serum ALT, ALP, AST, GGT, and total bilirubin) and raised kidney function tests (serum creatinine and urea) in the negative control group compared to the normal control. These concur with previous data where toluene caused renal and hepatic damage through increased oxidative stress reactions leading to a reduction in antioxidant status, levels of uric acid in the blood, and antioxidant potential in tissues [11], [25], [45], [73]. It is this excessive oxidative stress status in the body that results in increased amounts of protein markers of oxidative stress (diminished levels of SOD, CAT, and GSH), and attenuated lipid peroxidase pathway mechanisms (increased cellular lipid peroxidation or elevated levels of MDA in the liver and kidney tissues) [25], [3], [45], [52], [73]. As observed in the current research, these reactions trigger progressive hepatic injury and hepatocyte degradation, apoptosis, and impairment of liver biochemical functions in agreement with previous findings [8], defects of which arise hugely from hepatic mitochondrial dysfunction [8]. Toluene is mainly excreted from mammals via the kidneys [24], [8], and the resultant oxidative nephrotoxicity in rodents and humans [3], [60], cause serious renal cell damage, increased lactate dehydrogenase activity and renal impairment indicated by increased levels of serum creatinine, urea, and blood urea nitrogen which are plasma biomarkers of adverse renal toxicity [2], [50], [8], [95], findings of which are consistent with our findings.
Extract exposure significantly increased the antioxidant status (SOD and CAT activities, GSH levels) and reduced the oxidant status (MDA levels) in the extract test animals, as well as the standard control animals treated with VC compared to the negative control group, the values of SOD and CAT activities and GSH levels in the extract test and standard control groups significantly surpassed values of the normal control indicating that our C. jagus extracts induce significant antioxidant and anti-lipid peroxidation activities (induce surges of endogenous activities of SODs, CATs, GRs-production of GSH, and inhibit MDA production) in the injured liver and kidney tissues under toluene toxicity, with a similar antioxidant capacity to the standard control drug of our study (VC). Similar to our findings, VC (500 mg/kg) has been found to provide significant antioxidant potential in oxidative stress conditions in mice and rats [33]. Antioxidants correct oxidative stress by preventing or delaying the progression of oxidative chain reactions; nonenzymatic plant-based antioxidants such as tannins, polyphenols, and flavonoids identified in our plant can donate their free electrons which neutralize exogenous and endogenous free radicals, can scavenge free radicals resulting in amelioration of tissue damages and a reduction in the risk of diseases [35]. However, nonenzymatic plant-based antioxidants mainly work by stimulation of endogenous antioxidant enzymes (SOD, CAT, and GR), as was observed in the current paper, causing increased release of protons and free electrons which reduce the free radicals and oxidized intermediates of lipid peroxidation in liver and kidney tissues during oxidative stress [50], [85], [97]. The improved antioxidant status causes a lowering of the lipid peroxidation to bring about a decrease in MDA levels, reduced cellular lipid peroxidation, reduced tissue damage, and quickened tissue repair which protects tissues from oxidative stress-induced injuries [54], [80].
The current findings concur with previous in vitro data where Crinum jagus L. (Amaryllidaceae J.St. Hil.) leaf extracts and Crinum jagus (J. Thomps.) Dandy (Liliaceae) bulb extracts showed strong antioxidant potential owing to the presence of significant amounts of tannins, phenolics, and flavonoids [32], [57], [65]. Additionally, the current data are in tandem with H. Zailani et al.’s study where the aqueous and ethanolic bulb extracts of Crinum jagus (J. Thomps.) Dandy (Amaryllidaceae) showed anti-snake venom in rat tissues by an antioxidant mechanism owed to the presence of alkaloids, tannins, phenolics, terpenoids, steroids, and flavonoids [34]. In addition, the current data are in agreement with a previous in vivo study in which flavonol kaempferol from Crinum jagus L. (Amaryllidaceae) leaf extract showed strong antioxidant and anti-lipid peroxidation properties in pentylenetetrazole-induced kindling of mice, similarly, they also found decreased lipid peroxidation (MDA), augmented endogenous antioxidant enzymes (SOD, CAT, and GR) in damaged brain tissues of extract-treated animals compared to the untreated negative controls; the antioxidation capacity of the extract was coupled with significant protective (anticonvulsant) activity in the mice [85]. When endogenous antioxidant (SOD, CAT, and GSH) levels increase, we see reduced cellular lipid peroxidation, reduced tissue damage, and quickened tissue repair [68], [85]. Lipid peroxidation is facilitated by the lipid peroxidase pathway whose activities cause the production of MDA in tissues causing structural impairment and loss of function of cell membranes [68], [85], yet this was able to be checked by our plant extracts.
In any case, the two extracts significantly reduced bodily injury and organ toxicity as evidenced by the significantly improved bodyweight, reduced absolute and relative organ weights of liver and kidneys to normal levels, and alleviated the histopathological features of liver and kidneys to normal; coupled with improved LFTs, and kidney function tests to normal and near-normal values with the methanol and aqueous extracts respectively in the extract-treated animals compared to the untreated negative control group. This indicates that the extracts have anti-toxicant protective potentials on toluene-induced oxidative stress damages in rats (pretreatment with C. jagus ameliorates the injuries by protecting against the development of oxidative stress in the animals). This is in concurrence with previous data that found the extracts of C. jagus as possessing protective activities against oxidative stress-induced injuries in tissues due to the presence of abundant plant-based antioxidants mainly tannins, flavonoids, and polyphenols that show strong antioxidant and anti-lipid peroxidation capacities [32], [9],[34], [65], [64]. The improved antioxidant status in the animals causes lowered lipid peroxidation, decreased MDA levels, reduced cellular lipid peroxidation, and tissue injuries, and quickened tissue repair which protects the tissues from oxidative stress-induced injuries [54], [80].
Other mechanisms by which C. jagus extracts might be offering protection on the xenobiotically-injured tissues could be by the provision of secondary pharmacological properties (antimicrobial, antihemorrhagic, anti-inflammatory, anticarcinogenic, and anti-toxicant), [58], [65], [74], [85], [88], [9], such properties have been identified with Aloe vera. (L.) Burm. f. (Liliaceae) and facilitate the repair and regeneration of tissues following oxidative stress injuries thereby resulting in improved mammalian cell biology and function [58], [74], [86], [88], [89], however, these need more exploration.
Current data shows no differences in the levels/magnitudes of all our parameters (body weights, absolute and relative organ weights, SOD, CAT, GSH, MDA, LFTs, kidney function tests, and liver and kidney histology) in the extract normal control groups compared to the normal controls. This is in line with previous findings and indicates that when used in established therapeutic doses [64], [85], [89], C. jagus extracts are not toxic and maintain the morphological, biochemical, and physiological status of the animals [64], [85], [89]. There was more antioxidant status (higher tissue levels of SOD, CAT, and GSH) in the liver compared to kidney tissues (in normal control and extract test groups), and more oxidant status (higher MDA levels) in the liver compared to kidneys (in the negative control group). This is because the liver is the main organ of occurrence for the lipid peroxidase pathway and antioxidant enzymes activities in agreement with Jurczuk et al. who suggested that cellular lipid peroxidation produces higher levels of MDA in the liver following toluene toxicity [40], our extracts induce the liver to respond to the high MDA activity by causing increases of SOD and CAT activities to neutralize the excessive oxidative stress and lipid peroxidation (xenobiotic exposure triggers higher lipid peroxidase pathway activities in liver than kidney leading to higher levels of MDA) [40]. Antioxidant exposure (normal control and extract test groups) triggers higher antioxidant (SOD, CAT, and GSH) activity in the liver than kidneys in agreement with previous data [13], [40], [42].
The LFTs, kidney function tests, SOD and CAT activities, as well as liver and kidney histology, were more improved with methanol compared to the aqueous extract indicating that in general, the methanol extract of our plant showed better antioxidant protective potential than its aqueous counterpart, agrees with previous studies where the antioxidant potentials of C. jagus leaf and bulb extracts were highest in the order ethanol > methanol > aqueous attributed to the high content of phytochemicals mainly tannins, flavonoids, phenolics, alkaloids, and proanthocyan in the alcohol extracts compared to the aqueous one [32], [57], [65]. This corresponds to the phytochemical contents of our extracts, i.e., we obtained significantly higher concentrations of plant-based antioxidants (phenols, tannins, flavonoids) and correspondingly higher antioxidant, anti-lipid peroxidation, and protective effects with the methanol extract compared to its aqueous counterpart.
Altogether, the current study has established that the methanol leaf extract of C. jagus (J. Thomps.) Dandy (Liliaceae) provides more antioxidant and ameliorative potentials against toluene-induced oxidative stress damages in the liver and kidneys than its aqueous counterpart due to higher levels of plant-derived antioxidants (tannins, polyphenols, and flavonoids) in the former extract. Thus, the antioxidant protection is achieved largely by the activation of endogenous antioxidant enzymes (SOD, CAT, GR) by the plant-based non-enzymatic antioxidants (mainly polyphenols and flavonoids) and provision of strong reducing powers against free radicals and oxidized intermediates of lipid peroxidation by the phytochemicals which cause inhibition of the lipid peroxidation pathways in liver and kidneys of rats. Consequently, there is a need for the advancement of the methanol leaf extraction rather than the aqueous extraction of the herbal phytochemicals as a protective antioxidant and anti-lipid peroxidation medicinal molecules for toluene toxicity.
Some of the limitations of the current study are that it did not emphasize the evaluation of the antioxidant pathway via the CYP450 enzymes and Nrf2 (nuclear factor-erythroid factor 2-related factor 2) signaling mechanisms; prospective studies will be done to assess the practical attributes of CYP450 and Nrf2-associated pathways in L. inermis-related ameliorative effects during oxidative stress damages in animal tissues.
5. Conclusion
The aqueous and methanol C. jagus leaf extracts of C. jagus exert protective properties against toluene-induced damages by improving antioxidant mechanisms with improved antioxidant status and lowered lipid peroxidation in the liver and kidney of toluene-exposed rats thereby improving the biological processes of the organs. On the other hand, the methanol leaf extract of C. jagus shows better antioxidant protection than its aqueous counterpart due to higher amounts of plant-based antioxidants mainly flavonoids, tannins, and polyphenols in the former compared to the latter extract. Thus, the major herb-based antioxidants of the extract (polyphenols and flavonoids), protect the toluene-induced oxidative stress injuries of the animals by stimulation of endogenous antioxidant enzymes (SOD, CAT, and GR), but also due to the radical scavenging and reducing powers of the plant-based antioxidants. For this reason, these processes result in improved antioxidant status, reduced cellular lipid peroxidation, decreased tissue damages, quickened tissue repair, and improved histology in the liver and kidneys of toluene-induced rats.
Therefore, the methanolic extract contains high amounts of nonenzymatic plant-based antioxidants reflecting huge antioxidative and protective powers of C. jagus leaf extracts in cases of xenobiotic (toluene)-induced liver and kidney toxicity of the animals. Thus, we recommend more experimental and clinical studies to confirm the efficacy of this extract in treating xenobiotic-induced toxicity due to oxidative stress damages.
Declarations
Funding
Not applicable.
Ethical approval
The study was conducted after being given formal approval by the Institutional Review Ethics Committee (IREC) of Kampala International University, Western Campus, Uganda (UG-REC-0100/2021100). Upon approval of the experimental protocol by IREC, animals obtained from Experimental Animals Breeding and Research Facility, School of Pharmacy, Kampala International University, western campus, Uganda, were cared for based on the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
CRediT authorship contribution statement
Mariama Salihu: Conceptualization, Methodology, Formal analysis, Visualization, Writing – original draft, Project administration. Gaber El-Saber Batiha: Methodology, Visualization, Writing – original draft, Project administration. Keneth Iceland Kasozi: Methodology, Visualization, Investigation, Data curation, Writing – review & editing. George D. Zouganelis: Methodology, Formal analysis, Visualization, Data curation, Writing – review & editing. Souty M.Z. Sharkawi: Supervision, Data curation, Formal analysis, Writing – original draft. Eman Ibrahim Ahmed: Methodology, Visualization, Investigation, Data curation, Writing – review & editing. Ibe Micheal Usman: Methodology, Visualization, Investigation, Data curation, Writing – review & editing. Halima Nalugo: Investigation, Supervision, Data curation, Writing – review & editing. Juma J. Ochieng: Investigation, Supervision, Data curation, Formal analysis, Writing – review & editing. Ibrahim Ssengendo: Investigation, Supervision, Data curation, Writing – review & editing. Olatayo Segun Okeniran: Investigation, Supervision, Data curation, Writing – review & editing. Theophilus Pius: Methodology, Visualization, Investigation, Data curation, Writing – review & editing. Kyobe Ronald Kimanje: Investigation, Methodology, Visualization, Writing – original draft, Project administration. Eric Simidi Kegoye: Investigation, Supervision, Data curation, Writing – review & editing. Ritah Kenganzi: Methodology, Visualization, Investigation, Data curation, Writing – review & editing. Fred Ssempijja: Conceptualization, Methodology, Data curation, Formal analysis, Writing – original draft, Project administration.
Declaration of Competing Interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgments
Not applicable.
Handling Editor: Dr. Lawrence Lash
Footnotes
Supplementary data associated with this article can be found in the online version at doi:10.1016/j.toxrep.2022.03.026.
Contributor Information
George D. Zouganelis, Email: G.Zouganelis@derby.ac.uk.
Fred Ssempijja, Email: kalanzifr@yahoo.com.
Appendix A. Supplementary material
Supplementary material
.
Data availability
Data files used in the study can be found at https://figshare.com/s/8b47bcac1da1cd91b56b.
References
- 1.Abouee-Mehrizi A., Rasoulzadeh Y., Mehdipour A., Alihemmati A., Rahimi E. Hepatotoxic effects caused by simultaneous exposure to noise and toluene in New Zealand white rabbits: a biochemical and histopathological study. Ecotoxicology. 2021;30:154–163. doi: 10.1007/s10646-020-02288-z. [DOI] [PubMed] [Google Scholar]
- 2.Abouee-Mehrizi A., Rasoulzadeh Y., Mesgari-Abbasi M., Mehdipour A., Ebrahimi-Kalan A. Nephrotoxic effects caused by co-exposure to noise and toluene in New Zealand white rabbits: a biochemical and histopathological study. Life Sci. 2020;259 doi: 10.1016/j.lfs.2020.118254. [DOI] [PubMed] [Google Scholar]
- 3.Afravy M., Angali K., Khodadadi A., Ahmadizadeh M. The protective effect of Buffalo’s milk against toluene induced-nephrotoxicity in rats. J. Nephropathol. 2016;6:174–179. doi: 10.15171/jnp.2017.30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Afroz S., Rahman M.O., Hassan M.A. Taxonomic revision of the genus Crinum L. (Liliaceae) of Bangladesh. Bangladesh J. Plant Taxon. 2018;25:257–271. doi: 10.3329/bjpt.v25i2.39531. [DOI] [Google Scholar]
- 5.Armenta-Reséndiz M., Ríos-Leal E., Rivera-García M.T., López-Rubalcava C., Cruz S.L. Structure-activity study of acute neurobehavioral effects of cyclohexane, benzene, m-xylene, and toluene in rats. Toxicol. Appl. Pharmacol. 2019;376:38–45. doi: 10.1016/j.taap.2019.05.016. [DOI] [PubMed] [Google Scholar]
- 6.ARRIVE Guidelines, 2021. Housing and husbandry [WWW Document]. arriveguidelines.org. 〈https://arriveguidelines.org/arrive-guidelines/housing-and-husbandry〉. (Accessed 12.23.21).
- 7.A.O. Atilade, O. Victoria, D.B. Joseph, E.A. Segun, Aqueous leaf extract of alafia barteri maintained renal integrity in diabetic wistar rats 2 (2018 )1–8.
- 8.Ayan M., Tas U., Sogut E., Kuloglu T., Cayli S., Kocaman N., Karaca Z.I., Sahin M. The apoptotic effect of a high dose of toluene on liver tissue during the acute phase: an experimental study. Toxicol. Ind. Health. 2013;29:728–736. doi: 10.1177/0748233712442731. [DOI] [PubMed] [Google Scholar]
- 9.Azikiwe C., Siminialayi I., Brambaifa N., Amazu L., Enye J., Ezeani M. Anticonvulsant activity of the fractionated extract of Crinum jagus bulbs in experimental animals. Asian Pacific J. Trop. Dis. 2012;2:S446–S452. doi: 10.1016/S2222-1808(12)60201-1. [DOI] [Google Scholar]
- 10.Badoni Semwal R., Semwal D.K., Combrinck S., Cartwright-Jones C., Viljoen A. Lawsonia inermis L. (henna): ethnobotanical, phytochemical and pharmacological aspects. J. Ethnopharmacol. 2014;155:80–103. doi: 10.1016/j.jep.2014.05.042. [DOI] [PubMed] [Google Scholar]
- 11.Baelum J., Molhave L., Honore Hansen S., Dossing M. Hepatic metabolism of toluene after gastrointestinal uptake in humans. Scand. J. Work. Environ. Health. 1993;19:55–62. doi: 10.5271/sjweh.1503. [DOI] [PubMed] [Google Scholar]
- 12.Bailey S.A., Zidell R.H., Perry R.W. Relationships between organ weight and body/brain weight in the rat: what is the best analytical endpoint? Toxicol. Pathol. 2004;32:448–466. doi: 10.1080/01926230490465874. [DOI] [PubMed] [Google Scholar]
- 13.Barroso A.S., Quissell D.O., Colepicolo P. Modulation of SOD activity in rat submandibular glands. Arch. Oral Biol. 2003;48:133–139. doi: 10.1016/S0003-9969(02)00169-3. [DOI] [PubMed] [Google Scholar]
- 14.Ben Hamed S., Guardiola F., Cuesta A., Martínez S., Martínez-Sánchez M.J., Pérez-Sirvent C., Esteban M.Á. Head kidney, liver and skin histopathology and gene expression in gilthead seabream ( Sparus aurata L.) exposed to highly polluted marine sediments from Portman Bay (Spain) Chemosphere. 2017;174:563–571. doi: 10.1016/j.chemosphere.2017.02.009. [DOI] [PubMed] [Google Scholar]
- 15.Boniche C., Rossi S.A., Kischkel B., Vieira Barbalho F., Nogueira D′Aurea Moura Á., Nosanchuk J.D., Travassos L.R., Pelleschi Taborda C. Immunotherapy against systemic fungal infections based on monoclonal antibodies. J. Fungi. 2020;6(31) doi: 10.3390/jof6010031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Boubaya A., Marzougui N., Yahia L., Ben, Ferchichi A. Chemical diversity analysis of Tunisian Lawsonia inermis L. populations. African J. Biotechnol. 2011;10:4980–4987. doi: 10.5897/AJB10.2527. [DOI] [Google Scholar]
- 17.Calderón A.I., Cubilla M., Espinosa A., Gupta M.P. Screening of plants of Amaryllidaceae and related families from Panama as sources of acetylcholinesterase inhibitors. Pharm. Biol. 2010;48:988–993. doi: 10.3109/13880200903418514. [DOI] [PubMed] [Google Scholar]
- 18.A. Cca, A. Lu, The potential organo-toxicity safety of Morpholine and Crinum jagus in rats 10 (2015) 113–120.
- 19.Cervantes-Durán C., Ortega-Varela L.F., Godínez-Hernández D., Granados-Soto V., Gauthereau-Torres M.Y. Toluene exposure enhances acute and chronic formalin-induced nociception in rats: participation of 5-HT 3 receptors. Neurotoxicology. 2017;63:97–105. doi: 10.1016/j.neuro.2017.09.010. [DOI] [PubMed] [Google Scholar]
- 20.Cervantes-Durán C., Vidal-Cantú G.C., Godínez-Chaparro B., Granados-Soto V. Role of spinal 5-HT 2 receptors subtypes in formalin-induced long-lasting hypersensitivity. Pharmacol. Rep. 2016;68:434–442. doi: 10.1016/j.pharep.2015.11.009. [DOI] [PubMed] [Google Scholar]
- 21.Chander S Mehta, Pei Nan Sun A. Acute toxicity of toluene in male and female rats:a single oral dose exposure 2-week study. Toxic Subst. Mech. 1998;17:43–56. doi: 10.1080/107691898229440. [DOI] [Google Scholar]
- 22.Chandra S., Gonzalez de Mejia E. Polyphenolic compounds, antioxidant capacity, and quinone reductase activity of an aqueous extract of ardisia compressa in comparison to mate ( Ilex paraguariensis) and green ( Camellia sinensis) Teas. J. Agric. Food Chem. 2004;52:3583–3589. doi: 10.1021/jf0352632. [DOI] [PubMed] [Google Scholar]
- 23.Cortes N., Posada-Duque R.A., Alvarez R., Alzate F., Berkov S., Cardona-Gómez G.P., Osorio E. Neuroprotective activity and acetylcholinesterase inhibition of five Amaryllidaceae species: a comparative study. Life Sci. 2015;122:42–50. doi: 10.1016/j.lfs.2014.12.011. [DOI] [PubMed] [Google Scholar]
- 24.Cosnier F., Nunge H., Bonfanti É., Grossmann S., Lambert-Xollin A.-M., Muller S., Sébillaud S., Thomas A., Gaté L., Campo P. Toluene and methylethylketone: effect of combined exposure on their metabolism in rat. Xenobiotica. 2018;48:684–694. doi: 10.1080/00498254.2017.1362604. [DOI] [PubMed] [Google Scholar]
- 25.Dar M.A., Khan A.M., Raina R., Verma P.K., Sultana M. Effect of repeated oral administration of bifenthrin on lipid peroxidation and anti-oxidant parameters in Wistar rats. Bull. Environ. Contam. Toxicol. 2013;91:125–128. doi: 10.1007/s00128-013-1022-7. [DOI] [PubMed] [Google Scholar]
- 26.El-Dessouki A.M., El Fattah M.A., Awad A.S., Zaki H.F. Zafirlukast and vincamine ameliorate tamoxifen-induced oxidative stress and inflammation: role of the JNK/ERK pathway. Life Sci. 2018;202:78–88. doi: 10.1016/j.lfs.2018.04.002. [DOI] [PubMed] [Google Scholar]
- 27.Elansary H.O., Szopa A., Kubica P., Ekiert H., Al-Mana A., Al-Yafrsi, M.A F. Antioxidant and biological activities of Acacia saligna and Lawsonia inermis natural populations. Plants. 2020;9:908. doi: 10.3390/plants9070908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Eteng M.U., Ibekwe H.A., Abolaji A.O., Okoi A.I., Onwuka F.C., Osuchukwu N.C. Effect of Rauwolfia vomitoria Afzel (Apocynaceae) extract on serum amino transferase and alkaline phosphatase activities and selected indices of liver and kidney functions. Afr. J. Biotechnol. 2009;8:4604–4607. doi: 10.5897/AJB2009.000-9422. [DOI] [Google Scholar]
- 29.Eze E.D., Afodun A.M., Kasolo J., Kasozi K.I. Lycopene improves on basic hematological and immunological parameters in diabetes mellitus. BMC Res. Notes. 2019;12:805. doi: 10.1186/s13104-019-4841-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Fischer A.H., Jacobson K.A., Rose J., Zeller R. Hematoxylin and eosin staining of tissue and cell sections. Cold Spring Harb. Protoc. 2008:2008. doi: 10.1101/pdb.prot4986. pdb.prot4986-pdb.prot4986. [DOI] [PubMed] [Google Scholar]
- 31.Genchi G., Sinicropi M.S., Lauria G., Carocci A., Catalano A. The effects of cadmium toxicity. Int. J. Environ. Res. Public Health. 2020;17:3782. doi: 10.3390/ijerph17113782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ghane S.G., Attar U.A., Yadav P.B., Lekhak M.M. Antioxidant, anti-diabetic, acetylcholinesterase inhibitory potential and estimation of alkaloids (lycorine and galanthamine) from Crinum species: an important source of anticancer and anti-Alzheimer drug. Ind. Crop. Prod. 2018;125:168–177. doi: 10.1016/j.indcrop.2018.08.087. [DOI] [Google Scholar]
- 33.Gomez-Cabrera M.-C., Domenech E., Romagnoli M., Arduini A., Borras C., Pallardo F.V., Sastre J., Viña J. Oral administration of vitamin C decreases muscle mitochondrial biogenesis and hampers training-induced adaptations in endurance performance. Am. J. Clin. Nutr. 2008;87:142–149. doi: 10.1093/ajcn/87.1.142. [DOI] [PubMed] [Google Scholar]
- 34.Zailani H., Magaji A., K., Sarkiyayi P., Wurochekke, A S., U. Anti-snake venom activity of aqueous and ethanolic extracts of Crinum jagus bulb. Asian J. Res. Biochem. 2018;2:1–9. doi: 10.9734/ajrb/2018/v2i2455. [DOI] [Google Scholar]
- 35.Haida Z., Hakiman M. A comprehensive review on the determination of enzymatic assay and nonenzymatic antioxidant activities. Food Sci. Nutr. 2019;7:1555–1563. doi: 10.1002/fsn3.1012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.C. Hill, A. Flyvbjerg, R. Rasch, M. Bak, A. Logan, Transforming growth factor- 2 antibody attenuates fibrosis in the experimental diabetic rat kidney (2001) 647–651. [DOI] [PubMed]
- 37.Hismiogullari A.A., Hismiogullari S.E., Karaca O., Sunay F.B., Paksoy S., Can M., Kus I., Seyrek K., Yavuz O. The protective effect of curcumin administration on carbon tetrachloride (CCl4)-induced nephrotoxicity in rats. Pharmacol. Rep. 2015;67:410–416. doi: 10.1016/j.pharep.2014.10.021. [DOI] [PubMed] [Google Scholar]
- 38.Houghton P.J., Agbedahunsi J.M., Adegbulugbe A. Choline esterase inhibitory properties of alkaloids from two Nigerian Crinum species. Phytochemistry. 2004;65:2893–2896. doi: 10.1016/j.phytochem.2004.08.052. [DOI] [PubMed] [Google Scholar]
- 39.Jamshed H., Siddiqi H.S., Gilani A., Arslan J., Qasim M., Gul B. Studies on antioxidant, hepatoprotective, and vasculoprotective potential of <scp> Viola odorata </scp> and Wrightia tinctoria. Phyther. Res. 2019;33:2310–2318. doi: 10.1002/ptr.6411. [DOI] [PubMed] [Google Scholar]
- 40.Jurczuk M., Brzóska M., Moniuszko-Jakoniuk M., Gałażyn-Sidorczuk J., Kulikowska-Karpińska, E M. Antioxidant enzymes activity and lipid peroxidation in liver and kidney of rats exposed to cadmium and ethanol. Food Chem. Toxicol. 2004;42:429–438. doi: 10.1016/j.fct.2003.10.005. [DOI] [PubMed] [Google Scholar]
- 41.Kalange M., Nansunga M., Kasozi K.I., Kasolo J., Namulema J., Atusiimirwe J.K., Ayikobua E.T., Ssempijja F., Munanura E.I., Matama K., Semuyaba I., Zirintunda G., Okpanachi A.O. Antimalarial combination therapies increase gastric ulcers through an imbalance of basic antioxidative-oxidative enzymes in male Wistar rats. BMC Res. Notes. 2020;13:230. doi: 10.1186/s13104-020-05073-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kasozi K.I., Namubiru S., Safiriyu A.A., Ninsiima H.I., Nakimbugwe D., Namayanja M., Valladares M.B. Grain Amaranth is associated with improved hepatic and renal calcium metabolism in type 2 diabetes mellitus of male Wistar rats. Evid. Based Complement. Altern. Med. 2018;2018:1–10. doi: 10.1155/2018/4098942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kaur C., Kapoor H.C. Anti-oxidant activity and total phenolic content of some Asian vegetables. Int. J. Food Sci. Technol. 2002;37:153–161. doi: 10.1046/j.1365-2621.2002.00552.x. [DOI] [Google Scholar]
- 44.Kim S., Park E., Song S.-H., Lee C.-W., Kwon J.-T., Park E.Y., Kim B. Toluene concentrations in the blood and risk of thyroid cancer among residents living near national industrial complexes in South Korea: a population-based cohort study. Environ. Int. 2021;146 doi: 10.1016/j.envint.2020.106304. [DOI] [PubMed] [Google Scholar]
- 45.Lafi B., Chaâbane M., Elwej A., Grati M., Jamoussi K., Mnif H., Boudawara T., Ketata Bouaziz H., Zeghal N. Effects of co-exposure to imidacloprid and gibberellic acid on redox status, kidney variables and histopathology in adult rats. Arch. Physiol. Biochem. 2018;124:175–184. doi: 10.1080/13813455.2017.1371195. [DOI] [PubMed] [Google Scholar]
- 46.Liu J., Man S., Li J., Zhang Y., Meng X., Gao W. Inhibition of diethylnitrosamine-induced liver cancer in rats by Rhizoma paridis saponin. Environ. Toxicol. Pharmacol. 2016;46:103–109. doi: 10.1016/j.etap.2016.07.004. [DOI] [PubMed] [Google Scholar]
- 47.Lőrincz T., Szarka A. The determination of hepatic glutathione at tissue and subcellular level. J. Pharmacol. Toxicol. Methods. 2017;88:32–39. doi: 10.1016/j.vascn.2017.05.004. [DOI] [PubMed] [Google Scholar]
- 48.Luterotti S., Marković K., Franko M., Bicanic D., Madžgalj A., Kljak K. Comparison of spectrophotometric and HPLC methods for determination of carotenoids in foods. Food Chem. 2013;140:390–397. doi: 10.1016/j.foodchem.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 49.Ma X., Luo Q., Zhu H., Liu X., Dong Z., Zhang K., Zou Y., Wu J., Ge J., Sun A. Aldehyde dehydrogenase 2 activation ameliorates CCl 4 -induced chronic liver fibrosis in mice by up-regulating Nrf2/HO-1 antioxidant pathway. J. Cell. Mol. Med. 2018;22:3965–3978. doi: 10.1111/jcmm.13677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mbarki S., Alimi H., Bouzenna H., Elfeki A., Hfaiedh N. Phytochemical study and protective effect of Trigonella foenum graecum (Fenugreek seeds) against carbon tetrachloride-induced toxicity in liver and kidney of male rat. Biomed. Pharmacother. 2017;88:19–26. doi: 10.1016/j.biopha.2016.12.078. [DOI] [PubMed] [Google Scholar]
- 51.Meerow A.W., Lehmiller D.J., Clayton J.R. Phylogeny and biogeography of Crinum L. (Amaryllidaceae) inferred from nuclear and limited plastid non-coding DNA sequences. Bot. J. Linn. Soc. 2003;141:349–363. doi: 10.1046/j.1095-8339.2003.00142.x. [DOI] [Google Scholar]
- 52.Meydan S., Esrefoglu M., Selek S., Akbas Tosunoglu E., Ozturk O., Kurbetli N., Bayındır N., Bulut H., Meral I. Protective effects of caffeic acid phenethyl ester and thymoquinone on toluene induced liver toxicity. Biotech. Histochem. 2019;94:277–282. doi: 10.1080/10520295.2018.1554825. [DOI] [PubMed] [Google Scholar]
- 53.Michael B., Yano B., Sellers R.S., Perry R., Morton D., Roome N., Johnson J.K., Schafer K. Evaluation of organ weights for rodent and non-rodent toxicity studies: a review of regulatory guidelines and a survey of current practices. Toxicol. Pathol. 2007;35:742–750. doi: 10.1080/01926230701595292. [DOI] [PubMed] [Google Scholar]
- 54.Mir N.T., Saleem U., Anwar F., Ahmad B., Ullah I., Hira S., Ismail T., Ali T., Ayaz M. Lawsonia inermis markedly improves cognitive functions in animal models and modulate oxidative stress markers in the brain. Medicina. 2019;55:192. doi: 10.3390/medicina55050192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Morrison I.M., Asiedu E.A., Stuchbury T., Powell A.A. Determination of lignin and tannin contents of cowpea seed coats. Ann. Bot. 1995;76:287–290. doi: 10.1006/anbo.1995.1097. [DOI] [Google Scholar]
- 56.Morsy N. Phytochemical analysis of biologically active constituents of medicinal plants. Main Gr. Chem. 2014;13:7–21. doi: 10.3233/MGC-130117. [DOI] [Google Scholar]
- 57.Mvongo C., Noubissi P.A., Kamgang R., Sara C., Minka M., Mfopa A., Essame Oyono J.-L. Phytochemical studies and in vitro antioxidant potential of two different extracts of Crinum Jagus. Int. J. Pharm. Sci. Res. IJPSR. 2015;6:2354–2359. doi: 10.13040/IJPSR.0975-8232.6(6).2354-59. [DOI] [Google Scholar]
- 58.Nahar T., Uddin B., Hossain S., Sikder A.M., Ahmed S. Aloe vera gel protects liver from oxidative stress-induced damage in experimental rat model. J. Complement. Integr. Med. 2013;10:1–7. doi: 10.1515/jcim-2012-0020. [DOI] [PubMed] [Google Scholar]
- 59.Nakano M., Lockhart C.M., Kelly E.J., Rettie A.E. Ocular cytochrome P450s and transporters: roles in disease and endobiotic and xenobiotic disposition. Drug Metab. Rev. 2014;46:247–260. doi: 10.3109/03602532.2014.921190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Neghab M., Nourozi M.A., Shahtaheri S.J., Mansoori Y., Bazzaz J.T., Nedjat S. Effects of genetic polymorphism on susceptibility to nephrotoxic properties of BTEXs compounds. J. Occup. Environ. Med. 2018;60:e377–e382. doi: 10.1097/JOM.0000000000001364. [DOI] [PubMed] [Google Scholar]
- 61.Nevalainen T. Animal husbandry and experimental design. ILAR J. 2014;55:392–398. doi: 10.1093/ilar/ilu035. [DOI] [PubMed] [Google Scholar]
- 62.Nigam A.K., Ojha A.A., Li J.G., Shi D., Bhatnagar V., Nigam K.B., Abagyan R., Nigam S.K. Molecular properties of drugs handled by kidney OATs and liver OATPs revealed by chemoinformatics and machine learning: implications for kidney and liver disease. Pharmaceutics. 2021;13:1720. doi: 10.3390/pharmaceutics13101720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Nwakanma N.M.C., Okoli B.E. Cytological effects of the root extracts of Boerhaavia diffusa on root tips of Crinum jagus. EurAsian J. Biosci. 2010;111:105–111. doi: 10.5053/ejobios.2010.4.0.13. [DOI] [Google Scholar]
- 64.Ode O.J., Asuzu I.U. The anti-snake venom activities of the methanolic extract of the bulb of Crinum jagus (Amaryllidaceae) Toxicon. 2006;48:331–342. doi: 10.1016/j.toxicon.2006.06.003. [DOI] [PubMed] [Google Scholar]
- 65.Ode O.J., Nwaehujor C.O., Onakpa M.M. Evaluation of antihaemorrhagic and antioxidant potentials of Crinum jagus BULB Department of Veterinary Pharmacology and Toxicology, University of Abuja, PMB Department of Veterinary Physiology and Pharmacology, University of Nigeria, Nsukka. Departm. Pharm. Technol. 2010:1330–1336. [Google Scholar]
- 66.Oke-Altuntas F., Demirci M.A., Demirtas I., Yaglioglu A.S., Behcet L. Phytochemical screening, antiproliferative and antioxidant properties of various extracts from endemic Origanum acutidens. Comb. Chem. High Throughput Screen. 2018;21:281–291. doi: 10.2174/1386207321666180416154404. [DOI] [PubMed] [Google Scholar]
- 67.Oladimeji A.O. Vol. 12. 2018. Variation in pre-extraction processes influences the differences in chemical constituent, quantity and biochemical activities of volatile oils from Crinum jagus (Th.) D.: Gas Chromatography-Mass Spectrometry analysis; pp. 748–753. (J. Taibah Univ. Sci.). [DOI] [Google Scholar]
- 68.Panneerselvam S.R., Govindasamy S. Effect of sodium molybdate on the status of lipids, lipid peroxidation and antioxidant systems in alloxan-induced diabetic rats. Clin. Chim. Acta. 2004;345:93–98. doi: 10.1016/j.cccn.2004.03.005. [DOI] [PubMed] [Google Scholar]
- 69.Pasandi Pour A., Farahbakhsh H. Lawsonia inermis L. leaves aqueous extract as a natural antioxidant and antibacterial product. Nat. Prod. Res. 2019;0:1–5. doi: 10.1080/14786419.2019.1569006. [DOI] [PubMed] [Google Scholar]
- 70.Pelletti G., Rossi F., Garagnani M., Barone R., Roffi R., Pelotti S. Medico-legal implications of toluene abuse and toxicity. Review of cases along with blood concentrations. Leg. Med. 2018;34:48–57. doi: 10.1016/j.legalmed.2018.08.008. [DOI] [PubMed] [Google Scholar]
- 71.Piana M., Boligon A.A., Brum T.F., De, Zadra M., Belke B.V., Froeder A.L.F., Frohlich J.K., NUNES L.T., PAPPIS L., BOLIGON A.A., ATHAYDE M.L. Phytochemical analysis and antioxidant capacity ofTabernaemontana catharinensis A. DC. Fruits and branches. An. Acad. Bras. Cienc. 2014;86:881–888. doi: 10.1590/0001-3765201420120020. [DOI] [PubMed] [Google Scholar]
- 72.Prueitt R.L., Lynch H.N., Zu K., Shi L., Goodman J.E. Dermal exposure to toluene diisocyanate and respiratory cancer risk. Environ. Int. 2017;109:181–192. doi: 10.1016/j.envint.2017.09.017. [DOI] [PubMed] [Google Scholar]
- 73.Raina R., Baba N.A., Verma P.K., Sultana M., Singh M. Hepatotoxicity induced by subchronic exposure of fluoride and chlorpyrifos in Wistar rats: mitigating effect of ascorbic acid. Biol. Trace Elem. Res. 2015;166:157–162. doi: 10.1007/s12011-015-0263-1. [DOI] [PubMed] [Google Scholar]
- 74.Sahin H., Yener A.U., Karaboga I., Sehitoglu M.H., Dogu T., Altinisik H.B., Altinisik U., Simsek T. Protective effect of gel form of gastric gavage applicated Aloe vera on ischemia reperfusion injury in renal and lung tissue. Cell. Mol. Biol. 2017;63:34. doi: 10.14715/cmb/2017.63.12.9. [DOI] [PubMed] [Google Scholar]
- 75.Seidler A., Brüning T., Taeger D., Möhner M., Gawrych K., Bergmann A., Haerting J., Bolt H.M., Straif K., Harth V. Cancer incidence among workers occupationally exposed to dinitrotoluene in the copper mining industry. Int. Arch. Occup. Environ. Health. 2014;87:117–124. doi: 10.1007/s00420-012-0842-9. [DOI] [PubMed] [Google Scholar]
- 76.Seidler A., Harth V., Taeger D., Möhner M., Gawrych K., Bergmann A., Haerting J., Kahmann H.-J., Bolt H.M., Straif K., Brüning T. Dinitrotoluene exposure in the copper mining industry and renal cancer: a case-cohort study. Occup. Environ. Med. 2014;71:259–265. doi: 10.1136/oemed-2013-101850. [DOI] [PubMed] [Google Scholar]
- 77.Shaik S., Singh N., Nicholas A. Comparison of the selected secondary metabolite content present in the cancer-bush Lessertia (Sutherlandia) frutescens L. extracts. Afr. J. Tradit. Complement. Altern. Med. 2011;8:429–434. doi: 10.4314/ajtcam.v8i4.14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Shiau I.L., Shih T.L., Wang Y.N., Chen H.T., Lan H.F., Lin H.C., Yang B.Y., Ko C.H., Murase Y. Quantification for saponin from a soapberry (Sapindus mukorossi Gaertn) in cleaning products by a chromatographic and two colorimetric assays. J. Fac. Agric. Kyushu Univ. 2009;54:215–221. [Google Scholar]
- 79.Shipley W.U., Seiferheld W., Lukka H.R., Major P.P., Heney N.M., Grignon D.J., Sartor O., Patel M.P., Bahary J.-P., Zietman A.L., Pisansky T.M., Zeitzer K.L., Lawton C.A.F., Feng F.Y., Lovett R.D., Balogh A.G., Souhami L., Rosenthal S.A., Kerlin K.J., Dignam J.J., Pugh S.L., Sandler H.M. Radiation with or without antiandrogen therapy in recurrent prostate cancer. N. Engl. J. Med. 2017;376:417–428. doi: 10.1056/NEJMoa1607529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Singh D.K., Cheema H.S., Saxena A., Jyotshana, Singh S., Darokar M.P., Bawankule D.U., Shanker K., Luqman S. Fraxetin and ethyl acetate extract from Lawsonia inermis L. ameliorate oxidative stress in P. berghei infected mice by augmenting antioxidant defence system. Phytomedicine. 2017;36:262–272. doi: 10.1016/j.phymed.2017.09.012. [DOI] [PubMed] [Google Scholar]
- 81.Solanki R., Nagori B.P. New method for extracting phytoconstituents from plants. Int. J. Biomed. Adv. Res. 2012;3:770–774. doi: 10.7439/ijbar.v3i10.779. [DOI] [Google Scholar]
- 82.Sonibare M., Gbile Z. Ethnobotanical survey of anti-asthmatic plants in South Western Nigeria. Afr. J. Tradit. Complement. Altern. Med. 2008;5:340. doi: 10.4314/ajtcam.v5i4.31288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Sreevidya N., Mehrotra S. Spectrophotometric method for estimation of alkaloids precipitable with Dragendorff’s reagent in plant materials. J. AOAC Int. 2003;86:1124–1127. doi: 10.1093/jaoac/86.6.1124. [DOI] [PubMed] [Google Scholar]
- 84.Sun Y., Oberley L.W., Li Y. A simple method for clinical assay of superoxide dismutase. Clin. Chem. 1988;34:497–500. doi: 10.1093/clinchem/34.3.497. [DOI] [PubMed] [Google Scholar]
- 85.Taiwe G.S., Tchoya T.B., Menanga J.R., Dabole B., De Waard M. Anticonvulsant activity of an active fraction extracted from Crinum jagus L. (Amaryllidaceae), and its possible effects on fully kindled seizures, depression-like behaviour and oxidative stress in experimental rodent models. J. Ethnopharmacol. 2016;194:421–433. doi: 10.1016/j.jep.2016.10.023. [DOI] [PubMed] [Google Scholar]
- 86.Tran Doan, Nguyen Nguyen, Wang . Vol. 11. 2019. Anti-oxidant and anti-diabetes potential of water-soluble chitosan–glucose derivatives produced by Maillard reaction; p. 1714. (Polymers). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Trease G.E., Evans W.C. Textbook of Pharmacognosy. 12th ed. Balliers Tindall; 1983. [Google Scholar]
- 88.Tu W., Wang H., Li S., Liu Q., Sha H. The anti-inflammatory and anti-oxidant mechanisms of the Keap1/Nrf2/ARE signaling pathway in chronic diseases. Aging Dis. 2019;10:637. doi: 10.14336/AD.2018.0513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Udegbunam S., Kene R., Anika S., Udegbunam R., Nnaji T., Anyanwu M. Evaluation of wound healing potential of methanolic Crinum jagus bulb extract. J. Intercult. Ethnopharmacol. 2015;4:194. doi: 10.5455/jice.20150405064050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Udegbunam S., Udegbunam R., Nnaji T., Anyanwu M., Kene R., Anika S. Antimicrobial and antioxidant effect of methanolic Crinum jagus bulb extract in wound healing. J. Intercult. Ethnopharmacol. 2015;4:239. doi: 10.5455/jice.20150511022858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.US Department of Health and Human Services . ATSDR’s Toxicological Profiles. CRC Press; 2002. Toxicological profile for toluene. [DOI] [Google Scholar]
- 92.US Department of Health and Human Services Toxicology and carcinogenesis studies of in F344 / N rats. Natl. Toxicol. Program Tech. Rep. Ser. 1992;409:1–171. [PubMed] [Google Scholar]
- 93.Woisky R.G., Salatino A. Analysis of propolis: some parameters and procedures for chemical quality control. J. Apic. Res. 1998;37:99–105. doi: 10.1080/00218839.1998.11100961. [DOI] [Google Scholar]
- 94.Wu T., Shen M., Liu S., Yu Q., Chen Y., Xie J. Ameliorative effect of Cyclocarya paliurus polysaccharides against carbon tetrachloride induced oxidative stress in liver and kidney of mice. Food Chem. Toxicol. 2020;135 doi: 10.1016/j.fct.2019.111014. [DOI] [PubMed] [Google Scholar]
- 95.Wu X., Chen Y., Wang X., Wei W., Liang Y. Origin of site selectivity in toluene hydroxylation by cytochrome P450 enzymes. J. Org. Chem. 2021;86:13768–13773. doi: 10.1021/acs.joc.1c01295. [DOI] [PubMed] [Google Scholar]
- 96.Yavari F., van Thriel C., Nitsche M.A., Kuo M.-F. Effect of acute exposure to toluene on cortical excitability, neuroplasticity, and motor learning in healthy humans. Arch. Toxicol. 2018;92:3149–3162. doi: 10.1007/s00204-018-2277-x. [DOI] [PubMed] [Google Scholar]
- 97.Zhong X.-K., Jin X., Lai F.-Y., Lin Q.-S., Jiang J.-G. Chemical analysis and antioxidant activities in vitro of polysaccharide extracted from Opuntia ficus indica Mill. cultivated in China. Carbohydr. Polym. 2010;82:722–727. doi: 10.1016/j.carbpol.2010.05.042. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material
Data Availability Statement
Data files used in the study can be found at https://figshare.com/s/8b47bcac1da1cd91b56b.