Abstract
Purpose
Severely ill patients affected by coronavirus disease 2019 (COVID-19) develop circulatory failure. We aimed to report patterns of left and right ventricular dysfunction in the first echocardiography following admission to intensive care unit (ICU).
Methods
Retrospective, descriptive study that collected echocardiographic and clinical information from severely ill COVID-19 patients admitted to 14 ICUs in 8 countries. Patients admitted to ICU who received at least one echocardiography between 1st February 2020 and 30th June 2021 were included. Clinical and echocardiographic data were uploaded using a secured web-based electronic database (REDCap).
Results
Six hundred and seventy-seven patients were included and the first echo was performed 2 [1, 4] days after ICU admission. The median age was 65 [56, 73] years, and 71% were male. Left ventricle (LV) and/or right ventricle (RV) systolic dysfunction were found in 234 (34.5%) patients. 149 (22%) patients had LV systolic dysfunction (with or without RV dysfunction) without LV dilatation and no elevation in filling pressure. 152 (22.5%) had RV systolic dysfunction. In 517 patients with information on both paradoxical septal motion and quantitative RV size, 90 (17.4%) had acute cor pulmonale (ACP). ACP was associated with mechanical ventilation (OR > 4), pulmonary embolism (OR > 5) and increased PaCO2. Exploratory analyses showed that patients with ACP and older age were more likely to die in hospital (including ICU).
Conclusion
Almost one-third of this cohort of critically ill COVID-19 patients exhibited abnormal LV and/or RV systolic function in their first echocardiography assessment. While LV systolic dysfunction appears similar to septic cardiomyopathy, RV systolic dysfunction was related to pressure overload due to positive pressure ventilation, hypercapnia and pulmonary embolism. ACP and age seemed to be associated with mortality in this cohort.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00134-022-06685-2.
Keywords: COVID-19, Intensive care unit, Cardiac function, Echocardiography
Take-home message
| In a cohort of 677 patients affected by coronavirus disease 2019 (COVID-19) admitted in the intensive care unit (ICU), echocardiography is abnormal in one-third of cases with left and right ventricular systolic dysfunction observed in 23% and 22.5% of cases, respectively. Acute cor pulmonale is observed in 17% of cases and associated with hypercapnia, pulmonary embolism and mechanical ventilation. Acute cor pulmonale and age are associated with in-ICU mortality. |
Introduction
A large proportion of severely ill coronavirus disease 2019 (COVID-19) patients develop circulatory failure and require catecholamine infusions [1]. Myocardial injury and heart failure are associated with increased mortality [2, 3] with a large spectrum of mechanisms [4]. COVID-19 frequently induces pulmonary artery thrombosis [5]. The cytokine storm may also induce vasoplegia and distributive shock and COVID-19-related pneumonia may lead to acute respiratory distress syndrome (ARDS) [6], a condition well known to alter right ventricular (RV) function [7].
COVID-19 may then affect cardiac function by different mechanisms [8] but to the best of our knowledge, there is currently no clear information about cardiac function and echocardiographic patterns in critically ill COVID-19 patients. Echocardiographic findings was mostly reported in non-critically ill patients, frequently without any quantitative evaluation [9]. Given the mortality rate in these patients, data are urgently required to better understand this new disease and to improve hemodynamic support.
The primary aim of our study was to investigate the incidences and patterns of left and right ventricular dysfunction in the first echocardiographic examination performed after admission to the intensive care unit (ICU) in a large series of patients hospitalized for severe COVID-19. We took advantage of previous international collaborations with experts in the field of critical care echocardiography (CCE) that use echocardiography in their usual practice to manage critically ill patients [10].
Methods
This is a retrospective longitudinal observational study involving 14 intensive care units of tertiary teaching hospitals in 8 countries, registered in ClinicalTrials (April 28, 2021, number NCT 04414410). Unfortunately, we were unable to report the incidences of vasoplegia and hypovolemia due to insufficient superior vena cava (SVC) respiratory variability data available. The planned period of inclusion was extended to include the different waves of COVID-19. Consecutive patients with confirmed SARS-CoV-2 infection between 1 February 2020 and 30 June 2021, admitted to an ICU and who received at least one echocardiography, either a transthoracic (TTE) or transesophageal echocardiography (TEE), during their ICU stay were included. Patients were imaged as part of routine care, and non-identifiable patient data were captured. The study protocol was approved by ethics committee (CE SRLF 20–40). Main investigators were part of the echo working group of the European Society of Intensive Care Medicine (ESICM).
Data collection
Study data were collected and managed using REDCap, a secure web-based platform (hosted at The University of Sydney, https://redcap.sydney.edu.au). Data were collected according to recent guidelines [10] in three major domains: (1) patient characteristics, including co-morbidities, (2) clinical data, including ventilation and hemodynamic data, at the time of echocardiography, and (3) echocardiography data. Clinical and echocardiography data were collected at least once during their stay in ICU, however we only report results from the first echocardiographic examination in the present study. Length of ICU stay and outcome were also collected.
The web-based platform was opened to investigators until 30 June 2021 for uploading data. Data integrity and quality were then examined thoroughly by a methodologist (SH), and the investigators were contacted to provide details if any errors, inconsistencies or discrepancies were found. Specific data (values) were excluded if the errors were unrectifiable.
Clinical information
If available, clinical data that were relevant for the interpretation of echocardiography data were also collected, including ventilation mode and settings, arterial blood gases and hemodynamic data (vasopressor and inotrope infusions and blood pressures).
Echocardiography
Both TTE and TEE studies were included in the study. All studies were recorded and interpreted off-line by international experts in the field, all of whom have European Diploma in Advanced Critical Care Echocardiography or equivalent qualifications and experience. Common echocardiography parameters for left ventricular (LV) function, right ventricular (RV) function, and hemodynamic status were collected. These include, among others, LV ejection fraction (LVEF), RV tricuspid annular plane systolic excursion (TAPSE) and ventricular sizes. Both objective measurements and visual evaluation data were also collected for LV and RV function and sizes. Acute cor pulmonale (ACP) was defined as the presence of paradoxical septal motion (PSM) and RV end-diastolic area to LV end-diastolic area ratio (RVEDA/LVEDA) > 0.6.
Data analysis
Main results were of descriptive in nature. Categorical data were reported as count data, and mean ± standard deviation (SD) and/or median [Q1, Q3] were reported for continuous data. Values that were not available (not measured) were treated as missing values, and were not included in the analyses. Percentages were reported as percent of total number of patients (N) wherever possible, or as percent of available data (n) (excluding missing values) when stated. Little’s test for data missing completely at random pattern was performed. Data imputation was not carried out due to the presence of data missing not at random.
In addition to the planned investigation of the association between mechanical ventilation parameters and RV function, we also performed exploratory association analyses between outcome and global left and right heart function taking age into account. Categorical contingency tables were analyzed using Fisher’s exact test and reported as odds ratio (OR) [95% confidence interval (CI)]. Logistic regression was used in analyzing in-hospital and in-ICU mortalities. We included visual LV function, ACP and age as predictors based on the context of the study, and these are known or suspected prognosticator for ICU mortality [2, 3]. Visual assessment of RV function was not included as it showed strong association with ACP (collinearity). Nested models were compared using deviance goodness-of-fit test. All statistical tests were two-sided and were performed using the R software (version 4.1.0, R Foundation for Statistical Computing, Vienna) using tidyverse (version 1.3.1) package.
Results
Patients
A total number of 689 patients were included. Twelve patients were excluded due to missing patient information, resulting in a total of 677 patients in the analysis. This comprised of an average of 33.4% of COVID-19 patients admissions per center during this period (mean total COVID-19 admission per center = 244 ± 137 patients). Most (n = 641, 94.7%) were from European centers. Number of included patients per week, distribution according to age, gender and symptoms duration before ICU admission are reported in Supplementary Figure S1. Using 1 August 2020 as arbitrary demarcation date, the number of patients in the first and second/third waves included were 336 and 341, respectively. Patients over 60 years old constituted the majority of the admissions. Half of patients had symptoms of SARS-CoV-2 infection for 8 [6, 11] days (median [IQR]) prior to ICU admission.
Table 1 summarizes the patients’ characteristics and pre-existing medical history. The median age was 65 [56, 73]), and 71% of the patients were male. Mean body mass index (BMI) was 29.8 ± 6.4 kg m−2. Of the 675 patients with data on co-morbidities, 487 patients (72%) had at least one co-morbidity. Most frequent co-morbidities were hypertension (58%) and diabetes (30%). Only four patients had all pre-existing medical conditions and 102 (17%) had a history of cardiomyopathy. 241 patients were either taking angiotensin-converting enzyme inhibitor, angiotensin receptor blocker or both, and 146 were on beta-blockers.
Table 1.
Patient characteristics
| Characteristic | n (available/total)a# | Summary statisticsb |
|---|---|---|
| Sex | 677/677 (100%) | |
| M | 481 (71%) | |
| F | 196 (29%) | |
| Age | 581/677 (86%) | 65 (56, 73) |
| Weight | 664/677 (98%) | 85 (74, 98) |
| Height | 648/677 (96%) | 172 (165, 178) |
| BMI | 646/677 (95%) | 28.4 (25.4, 33.2) |
| Smoker | 449/677 (66%) | |
| Non-smoker | 268 (60%) | |
| Ex-smoker | 141 (31%) | |
| Current smoker | 40 (8.9%) | |
| Pre-ICU admission symptoms duration (days) | 571/677 (84%) | 8 (6, 11) |
| Cardiac failure | 600/677 (89%) | |
| Yes | 102 (17%) | |
| No | 498 (83%) | |
| Hypertension | 670/677 (99%) | |
| Yes | 388 (58%) | |
| No | 282 (42%) | |
| COPD | 670/677 (99%) | |
| Yes | 126 (19%) | |
| No | 544 (81%) | |
| Diabetes | 673/677 (99%) | |
| Yes | 204 (30%) | |
| No | 469 (70%) | |
| Chronic renal disease | 670/677 (99%) | |
| Yes | 58 (8.7%) | |
| No | 612 (91%) | |
| ACE inhibitors | 588/677 (87%) | |
| Yes | 114 (19%) | |
| No | 474 (81%) | |
| ARBs | 587/677 (87%) | |
| Yes | 127 (22%) | |
| No | 460 (78%) | |
| Beta-blockers | 587/677 (87%) | |
| Yes | 146 (25%) | |
| No | 441 (75%) |
ACE inhibitors angiotensin-converting enzymes inhibitors, ARBs angiotensin receptor blockers, CRD chronic renal disease, COPD chronic obstructive pulmonary disease
#Most data were missing at random or missing not at random. (Little’s test for missing completely at random, p < 0.0001)
aNumber of available data (%)/total number of patients (%)
bMedian (IQR) for continuous data; n (%) for categorical data
Clinical information at the time of first echocardiography
About two-thirds of the patients (69%, n = 467) were receiving mechanical ventilation, with a mean tidal volume of 435 ± 80 ml (n = 391 available data, 84% of ventilated patients) and a mean plateau pressure of 24.7 ± 4.5 cmH2O (n = 394, 84% of ventilated patients). PaO2/FiO2 (n = 634) was 134 ± 68 mmHg and the mean PaO2/FiO2 in spontaneously breathing patients and in mechanical ventilated patients were 138 ± 77 mmHg (median = 119 [88, 166]) and 133 ± 64 mmHg (median = 118 [86, 167]), respectively. The mean difference was − 4.6 mmHg [95% CI = − 16.3 to 7.1].
Two hundred and seventy-seven (41%) patients were on norepinephrine alone or in combination with another inotrope or vasopressor. Nineteen patients received extra-corporeal membrane oxygenation (ECMO) due to severe or refractory ARDS: 1 patient were on venous-arterious (VA) ECMO and 18 on venous-venous (VV) ECMO. Out of 447 patients for whom it was searched, pulmonary embolism was reported in 55 (11.5%) patients (Table 2).
Table 2.
Clinical data during the first echocardiography
| Clinical parameters | n (available/total)a# | Summary statisticsb |
|---|---|---|
| Ventilation mode | 677/677 (100%) | |
| Spontaneously breathing | 210 (31%) | |
| Mechanically ventilated | 467 (69%) | |
| FiO2 (%) | 661/677 (98%) | 65 (50, 90) |
| High flow O2 | 199/677 (29%) | |
| Yes | 149 (75%) | |
| No | 50 (25%) | |
| Respiratory rate (per min) | 597/677 (88%) | 24 (20, 28) |
| Tidal volume (ml)c | 391/467 (84%) | 440 (400, 480) |
| PEEP (cmH2O)c | 294/467 (63%) | 10 (8, 12) |
| Total PEEP (cmH2O)c | 359/467 (77%) | 11 (10, 14) |
| Plateau pressure (cmH2O)c | 394/467 (84%) | 25 (22, 28) |
| PaO2 (mmHg) | 649/677 (96%) | 75 (64, 90) |
| PaO2/FiO2 | 634/677 (94%) | 118 (86, 167) |
| SaO2 | 503/677 (74%) | 94 (91, 96.2) |
| PaCO2 (mmHg) | 597/677 (88%) | 42 (35, 50) |
| pH | 597/677 (88%) | 7.40 (7.32, 7.46) |
| Base deficit (mmol/L) | 403/677 (60%) | 0.7 (− 2, 2.8) |
| Lactate (mmol/L) | 510/677 (75%) | 1.50 (1.10, 2) |
| Heart rate (bpm) | 639/677 (94%) | 87 (74, 103) |
| Systolic arterial pressure (mmHg) | 634/677 (94%) | 125 (110, 140) |
| Diastolic arterial pressure (mmHg) | 634/677 (94%) | 65 (55, 77) |
| Mean arterial pressure (mmHg) | 627/677 (93%) | 83 (72, 95) |
| Central venous pressure (cmH2O) | 183/677 (27%) | 9 (7, 11) |
| Pulmonary embolism | 447/677 (66%) | |
| Yes | 55 (12%) | |
| No | 392 (88%) | |
| ECMO | 509/677 (75%) | |
| VA ECMO | 1 (0.2%) | |
| VV ECMO | 18 (3.5%) | |
| No | 490 (96%) | |
| Vasopressors/inotropes | 677/677 (100%) | |
| Dobutamine | 31 (4.6%) | |
| Dopamine | 1 (0.1%) | |
| Epinephrine | 4 (0.6%) | |
| Norepinephrine | 277 (40.9%) | |
| Not administered | 364 (53.8%) |
PEEP positive end-expiratory pressure, VA and VV ECMO veno-arterial and veno-venous extra-corporeal membrane oxygenation
aNumber of available data/Total number of patients (%)
bMedian (IQR) for continuous data; n (%) for categorical data
cTotal number of patients receiving mechanical ventilation = 467
#Most data were missing at random or missing not at random. (Little’s test for missing completely at random pattern, p < 0.0001)
Echocardiography
In half of the patients, the first echocardiography was performed on the first two days of ICU admission (median = 2 [1, 4]) and 80% were performed on or before day 5 (Table 3). Most patients (81.4%, n = 551) received TTE of which 342 were mechanically ventilated, and 126 (18.6%) patients received TEE where all except one were mechanically ventilated. Most of the patients were in sinus rhythm (91.8%).
Table 3.
Echocardiography findings
| Echo parameters | n (available/total)a# | Summary statisticsb |
|---|---|---|
| Echo types | 677/677 (100%) | |
| TTE | 551 (81%) | |
| TEE | 126 (19%) | |
| Day in ICU had echo | 677/677 (100%) | 2 (1, 4) |
| Cardiac rhythm | 660/677 (97%) | |
| Sinus | 606 (92%) | |
| Atrial fibrillation / flutter | 54 (8.2%) | |
| LV function (visual) | 676/677 (99.1%) | |
| Hyperkinetic | 113 (17%) | |
| Normal | 414 (61%) | |
| Moderately depressed | 105 (16%) | |
| Severely depressed | 44 (6.5%) | |
| LVEF (%) | 598/677 (88%) | 60 (50, 65) |
| SWMA | 563/677 (83%) | |
| Yes | 77 (14%) | |
| No | 486 (86%) | |
| LV size (visual) | 675/677 (99.7%) | |
| Reduced | 49 (7.3%) | |
| Not dilated | 555 (82%) | |
| Moderately dilated | 54 (8%) | |
| Severely dilated | 17 (2.5%) | |
| LV end-diastolic diameter (mm) | 345/677 (51%) | 46 (42, 50) |
| LV end-diastolic volume (ml) | 433/677 (64%) | 90 (70, 118) |
| LVEDV indexed (per BSA) (ml/mb) | 409/677 (60%) | 46 (37, 57) |
| RV function (visual) | 675/677 (99.7%) | |
| Hyperkinetic | 58 (8.6%) | |
| Normal | 465 (69%) | |
| Moderately depressed | 108 (16%) | |
| Severely depressed | 44 (6.5%) | |
| TAPSE (mm) | 537/677 (79%) | 20.9 (17, 24) |
| FAC (%) | 333/677 (49%) | 36 (28, 45) |
| RV size (visual) | 668/677 (99%) | |
| Reduced | 28 (4.2%) | |
| Not dilated | 363 (54%) | |
| Moderately dilated | 217 (32%) | |
| Severely dilated | 60 (9%) | |
| RV hypertrophy | 489 / 677 (72%) | |
| Yes | 82 (17%) | |
| No | 407 (83%) | |
| RVEDA/LVEDA ratio | 521/677 (77%) | 0.61 (0.5, 0.8) |
| PSM | 670/677 (99%) | |
| Yes | 130 (19%) | |
| No | 540 (81%) | |
| Acute cor pulmonale | 519/677 (77%) | |
| Yes | 90 (17%) | |
| No | 429 (83%) | |
| TRmax velocity (cm/s) | 278/677 (41%) | 2.7 (2.15, 3.1) |
| PAAT | 133/677 (20%) | 87 (66, 105) |
| IVC diameter (TTE, n = 551) | 433/677 (64%) | |
| Small | 52 (12%) | |
| Normal | 198 (46%) | |
| Dilated | 183 (42%) | |
| Mitral E/A ratio | 567/677 (84%) | 0.9 (0.75, 1.17) |
| Aorta VTI (cm) | 570/677 (84%) | 19.2 (16.7, 22.6) |
| Aortic regurgitation | 582/677 (86%) | |
| None | 482 (83%) | |
| Mild | 90 (15%) | |
| Moderate | 10 (1.7%) | |
| Severe | 0 (0%) | |
| Mitral regurgitation | 592/677 (87%) | |
| None | 381 (64%) | |
| Mild | 175 (30%) | |
| Moderate | 35 (5.9%) | |
| Severe | 1 (0.2%) | |
| Pericardial effusion | 605/677 (89%) | |
| Yes | 74 (12%) | |
| No | 531 (88%) | |
| PFO | 256/677 (38%) | |
| Yes | 14 (5.5%) | |
| No | 242 (95%) |
TTE transthoracic echocardiography, TEE transesophageal echocardiography, SWMA segmental wall motion abnormality, LVEDV left ventricular end-diastolic volume, BSA body surface area, TAPSE tricuspid annular plane systolic excursion, FAC fractional area change, EDA end-diastolic area, PSM paradoxical septal motion, ACP acute cor pulmonale, TR tricuspid regurgitation, PAAT pulmonary artery acceleration time, IVC inferior vena cava, VTI velocity time integral
aNumber of available data/total number of patients (%)
bMedian (IQR) for continuous data; n (%) for categorical data
#Most data were missing at random or missing not at random. (Little’s test for missing completely at random pattern, p < 0.0001)
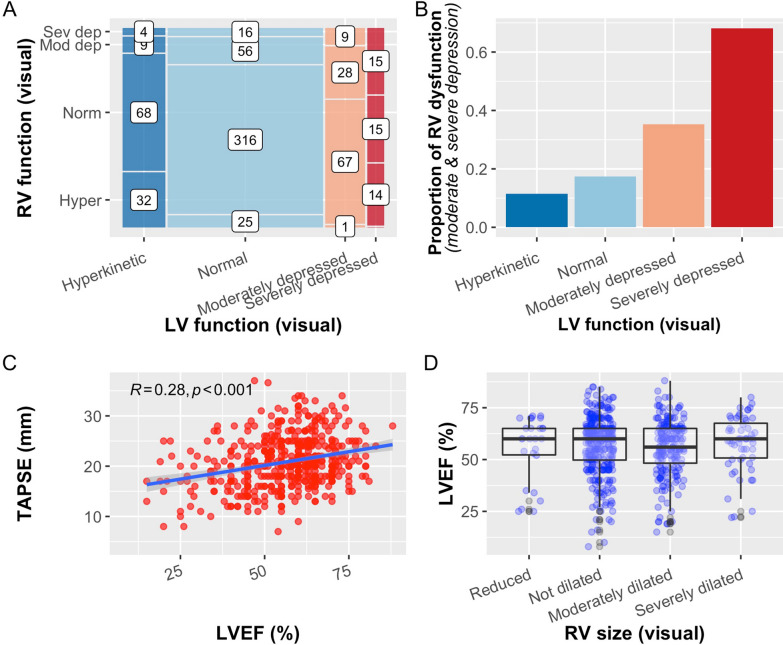
Four hundred and sixty-five patients had one or more echocardiographic identifiable pathologies and the presentations were shown in Supplementary Figure S2. A total of 234 (34.5%) patients were reported to have LV and/or RV systolic dysfunction visually: 67 (9.9%) patients had biventricular dysfunction, 85 (12.5%) had isolated RV dysfunction, 82 (12.1%) had isolated LV dysfunction, and 441 (65.1%) patients displayed normal LV and RV function (Fig. 1A).
Fig. 1.
A Mosaic plot for number of patients with different combinations of RV and LV functions (Hyper hyperkinetic, Norm normal, Mod dep moderately depressed, Sev dep severely depressed). Number denotes number of patients for each category. B Proportion of patients with depressed RV function with different LV function, both assessed visually. C Relationship between LVEF and TAPSE. The blue line shows the fitted linear regression line. Pearson correlation (R) is shown. D LV ejection fraction (LVEF) vs RV size
Characteristics of LV systolic dysfunction
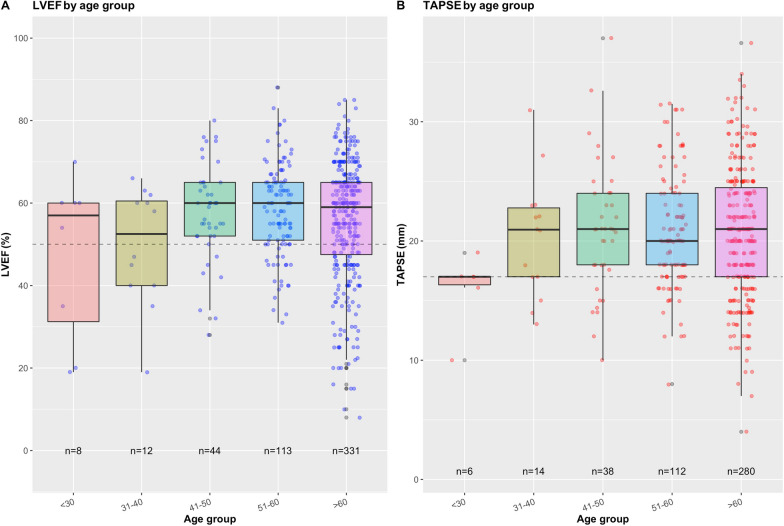
LV systolic dysfunction (moderate or severe) was observed in 149 (22%) patients, among them 45 (30.2%) had previous history of cardiomyopathy, 50 previously received beta-blockers and only 15 were under dobutamine. The visual assessment of LV systolic function was consistent with measured LVEF (Supplementary Figure S3A). There were no discernible differences in the means of LVEF between the age groups, although a larger proportion of patients (27.5%, n = 91) in the > 60 age group had LVEF < 50% (Fig. 2A). Only a relatively small proportion of patients (10.5%) displayed visually dilated LV. LV end-diastolic volume (LVEDV) indexed to body surface area was available in 409 patients (Table 3), and its relationship with visual assessment of LV size is shown in Supplementary Figure S3B. Out of 563 patients with available information on segmental wall motion abnormality (SWMA), 77 (13.7%) were found to have SWMA, and 46 of these patients were reported to have visually impaired LV function (Supplementary Figure S2). The mean LV E/A ratio was 1 ± 0.37 (n = 567).
Fig. 2.
A LVEF in different age groups. Dashed line denotes LVEF = 50%. B TAPSE in different age groups. Dashed line denotes TAPSE = 17 mm
Characteristics of RV systolic dysfunction
RV systolic dysfunction (moderate or severe dysfunction) was observed in 152 (22.5%) patients. The visual assessment of RV function was consistent with measured TAPSE (Supplementary Figure S3C). There were no discernible differences in the means of TAPSE between the age groups, although a slightly larger proportion of patients (22.1%, n = 62) in the > 60 age group had TAPSE ≤ 16 mm (Fig. 2B). About 41% (n = 277, Navailable = 675) of the patients were reported to have either moderately or severely dilated RV visually, and higher RVEDA/LVEDA (Supplementary Figure S3D). The mean RVEDA/LVEDA, when available (Navailable = 521), was 0.67 ± 0.25 and 262 (50%) patients had a dilated RV (RVEDA/LVEDA > 0.6). The proportion of RV systolic dysfunction increases with the severity of LV dysfunction (p < 0.001) (Fig. 1B). TAPSE was also positively correlated with LVEF [r = 0.28 (95% CI 0.19, 0.36)] (Fig. 1C). The mean LVEF was similar in different RV size groups (Fig. 1D).
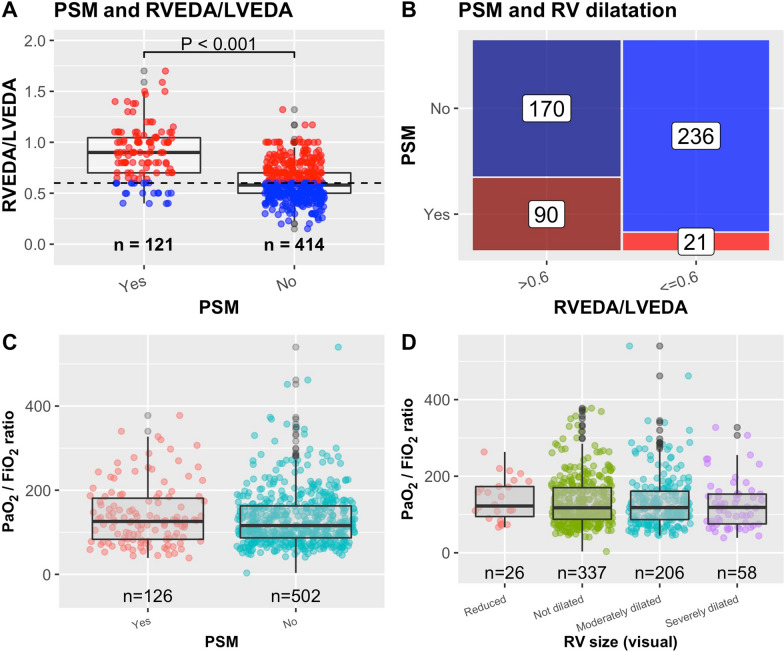
PSM was found in 130 out of 670 patients (19.4%) with an RVEDA/LVEDA ratio higher than in patients without PSM [difference = 0.28 (95% CI 0.23 to 0.33)] (Fig. 3A). 90 (17.4%, Navailable = 517) patients displayed PSM and RVEDA/LVEDA > 0.6 indicating ACP (Fig. 3B). The PaO2/FiO2 between those with and without PSM were similar, 140 ± 72 vs 133 ± 67 mmHg, respectively [difference = 7.5 mmHg (95% CI − 5.8 to 20.7)] (Fig. 3C) and also similar in patients with and without ACP (138 ± 68 mmHg vs 131 ± 67 mmHg) [difference = 6.5 mmHg (95% CI − 9 to 21.9)]. There was also no difference in PaO2/FiO2 in different RV size (Fig. 3D). However, patients with PSM or ACP was associated with higher incidences of pulmonary embolism (OR > 5) or mechanical ventilation (OR > 4) (Supplementary Table S1 and Supplementary Figure S4). They also displayed higher PaCO2, while plateau pressure did not differ. Tidal volume was slightly lower in PSM (Supplementary Table S1). On the other hand, a significant proportion of patients with RV dysfunction (57%) did not experienced ACP, and there was no association between visual RV function and pulmonary embolism [OR = 0.614 (95% CI 0.319 to 1.223)].
Fig. 3.
A Relationship between paradoxical septal motion (PSM) and right to left ventricular end-diastolic areas ratio (RVEDA/LVEDA). Dashed line denotes RVEDA/LVEDA = 0.6. B Mosaic plot showing the number of patients with different combinations of PSM and RV dilatation using RVEDA/LVEDA > 0.6 as cutoff. 90 patients had acute cor pulmonale by definition (presence of PSM and RVEDA/LVEDA > 0.6). C PaO2/FiO2 ratio in patients with vs without PSM. D PaO2/FiO2 ratio vs RV size assessed visually
Other echo findings
Valvular (aortic and mitral) regurgitations were absent in most patients, and only a few had severe valvular regurgitations (Table 3). Among the 256 patients in whom it was actively sought, patent foramen ovale (PFO) was detected in 14 patients. 74/605 (12.2%) patients had a pericardial effusion, with no case of tamponade.
Patient outcomes
The median ICU length of stay was 14 [8, 26] days. The in-ICU mortality rate was 32.1% and in-hospital mortality rate 34.3%. Exploratory analyses in this cohort showed that both ACP and age were significantly associated with in-hospital and in-ICU mortalities. Patients with ACP were twice as likely to die when compared to those without (OR = 2.02 [1.11, 3.66] for in-hospital mortality, and OR = 2.01 [1.09, 3.69] for in-ICU mortality). The odds for dying increases by 6% for every year increase in age (OR = 1.06 [1.04, 1.08]) for both in-hospital and in-ICU mortalities) (Supplementary Figure S5). Visual LV systolic dysfunction was associated with in-hospital mortality (OR = 1.52 [1.04, 2.23]), but this association disappeared after adjusting for age. There is no significant association between visual RV dysfunction and mortality (results not shown). Sensitivity analyses excluding patients (n = 19) receiving ECMO yielded similar results and conclusions.
Discussion
We report here cardiac function evaluated by CCE in 677 severe COVID-19 patients admitted in 14 different ICUs, mostly in Europe. Cardiac function at the time of the first echocardiography after ICU admission was abnormal in 34% of the patients, 149 (22%) exhibiting LV systolic dysfunction and 152 (22.5%) RV systolic dysfunction. It is known that COVID-19 may affect the heart by different mechanisms [8]. Clear data regarding cardiac function is very limited in critically ill patients. In a large international survey, Dweck et al. reported LV and RV abnormalities in 39% and 33% respectively [9]. However, this survey only included 54% of critically ill patients and no quantitative evaluation of cardiac function was done. In another study of 43 consecutive patients admitted in 2 ICUs for COVID-19, Doyen et al. found that 30% of patients with myocardial injury had concurrent LV wall motion abnormalities, 47% had RV systolic dysfunction and 30% had ACP [11]. Patients’ characteristics in our cohort were very similar to previous publications in terms of age, gender, BMI, delay between symptoms onset and ICU admission and co-morbidities, as well as outcome [12, 13]. We included a similar number of patients between the first wave and the second/third wave. Many patients required catecholamine infusions at the time of CCE.
As expected, CCE was mainly performed by a transthoracic route, even when patients were mechanically ventilated. While LV and RV systolic function were only evaluated visually in most patients, our results show that CCE was reliably interpreted by experts in the field as visual and quantitative data, when available, did not show significant discrepancies. CCE was also performed early after ICU admission (half were within 2 days) and so reports the initial cardiac function before potential complications. As recommended by the PRICES consensus statement [10], we were also able to report important clinical data at the time of CCE, helping us to adequately interpret our findings.
A larger proportion of patients with age > 60 years had LV and RV dysfunction (LVEF < 50% or TAPSE ≤ 16 mm), while the means of LVEF and TAPSE did not differ across the age. Depressed LV systolic function, which was observed in 22%, was usually global and in most patients, the LV was non-dilated, suggesting an acute injury. In the majority of patients who did not have previous history of cardiomyopathy, the pattern of LV systolic dysfunction is similar to that previously described in septic cardiomyopathy [14], and is likely mediated by systemic inflammation [15, 16]. This was reinforced by a non-elevated E/A ratio, suggesting the absence of LV filling pressure elevation [17]. However, the presence of potential LV ischemia cannot be formally ruled out as a minority of patients had segmental wall motion abnormalities, and we did not collect data on myocardial injury as well as types I and II myocardial infarction. Interestingly, only 12 patients had the association of LV systolic dysfunction with segmental wall motion abnormalities and LV dilatation which frequently occur in case of chronic cardiomyopathy. In 156 patients with COVID-19 in shock, Hollenberg et al. reported a 15% incidence of LV systolic dysfunction [18].
RV systolic dysfunction was found in 22.5%. The RV was frequently dilated with an incidence of 41% from visual assessment alone, and 50% when quantitative measurements were available. Paradoxical septal motion, a marker of RV pressure overload, was reported in 19% of cases. In the 517 patients who had required data [19], ACP was observed in 17% of cases. This remains slightly lower than what was reported in moderate to severe non-COVID-19 ARDS [7], but similar than what was reported in a previous small series of 18 COVID-19 patients [20]. In a recent retrospective study including 510 selected patients, of which 68% were admitted to ICU, RV dilatation was observed in 35% of cases but RV dysfunction, based on decreased TAPSE and tissue Doppler imaging velocity, was much less frequent (15%) [21]. Interestingly, Bleakley et al. reported in 90 critically ill patients, 42% of them under VV ECMO, that RV systolic dysfunction was frequent but that COVID-19 led to a specific phenotype of radial impairment with sparing of longitudinal function [22]. As a matter of fact, TAPSE and tissue Doppler imaging velocity were less sensitive than fractional area change to detect RV abnormalities [22]. They also suggested that strain approach was less sensitive which has to be confirmed in the future. Three mechanisms partly responsible for ACP may be supported by our results: (1) the effect of positive pressure ventilation (at least 4 times the risk of spontaneously breathing patients), despite the application of a lung protective ventilation, (2) the effect of hypercapnia as PaCO2 was slightly but significantly higher in case of ACP, and (3) the consequence of pulmonary embolism (more than 5 times the risk of patients without pulmonary embolism), all being intricate with strong pathophysiological rationale. Although history of lung disease, as COPD, is also a potential cause of RV failure, we were unable to draw any conclusion due to the very small number of patients (n = 9) who had concomitant ACP and history of COPD, of which 3 of them had pulmonary embolism. Hypercapnia and mechanical ventilation, by increasing RV afterload, are already known risk factors for ACP in non-COVID ARDS [7]. Pulmonary embolism, when severe, is associated with RV failure in general, and has been reported as a risk for ACP in COVID-19 related ARDS in particular [23]. On the other hand, it is noteworthy that alterations in the pulmonary circulation in patients with COVID-19 through angiogenesis development [24] and limited adaptive hypoxic vasoconstriction [25, 26] compared to more usual ARDS could also protect the right ventricle by limiting the increase in pulmonary pressure. As a matter of fact, we did not find any association between PaO2/FiO2 and ACP or RV size while it was previously reported in non-COVID-19 ARDS [7].
We found that ACP and age, but not LV dysfunction, are associated with mortality. In 2 studies performed in non-critically ill COVID-19 patients, RV dilatation was independently associated with death or subsequent ICU transfer [27] in one and echo parameters of LV and RV dysfunction were independently associated with poor prognosis in the other [28].
Our study suffered from the usual limitations seen in retrospective studies. The impact of pandemic on the burden of healthcare workers, and the ICU surge with its unforeseeable and unpredictable COVID-19 infection rate (and trajectory) at the time precluded us from conducting a prospective study. The main limitations of this retrospective observational study were: first, echocardiography studies were only performed as per clinical necessity and not on every patients (only 33.4% of all COVID-19 patients per center), and these resulted in selection bias and limits its generalization to other COVID-19 patients. However, the large number of recruitment centers in different geographical locations provided a meaningful representative of COVID-19 patients with characteristics similar to our cohort. Second, the echocardiography procedures were not designed for a proper echocardiography research study, hence the procedure were not standardized and missing data were common. Most missing data were not missing completely at random, but were missing at random (e.g., some centers may not perform certain measurements in their practice) or not at random (e.g., LVEF was missing due to poor image quality, or PFO or thrombus was not entered because it was not observed). While we were unable to impute missing data due to large number of the latter, our main results were based on visual assessments which were available in most patients. Third, we were unable to report any definitive causality between ACP and mortality but only a statistical association. We also could not evaluate the potential impact of VV ECMO on this association as the number of patients with such a technique was too small, while we found similar results after excluding the 19 patients who received ECMO. Finally, another (minor) limitation was the lack of severity index (Acute Physiology and Chronic Health Disease Classification System (APACHE), Simplified Acute Physiology Score (SAPS) and Sequential Organ Failure Assessment (SOFA) scores), although all patients were admitted to ICU due to severe COVID-19. On the other hand, there were several strengths in our study. First, the visual assessments were performed by experts in CCE, and we find the relationship between visual and quantitative assessment was consistent and reliable. Second, previous studies in hospitalized COVID-19 patients have shown that myocardial injury (as reflected by increased troponin level) is common and is associated with death [2, 3]. However, in these studies, cardiac biomarkers were used as the sole marker of cardiac injury [2, 3]. Although we did not report cardiac biomarkers in our study, we used echocardiography to assess cardiac function which has better specificity in identifying clinically important cardiac dysfunction, the types of dysfunction and other salient features not detected using cardiac biomarkers. While this retrospective study showed some limitations, the strength of the study is that it is one of the largest critical care echocardiography study in ICU COVID-19 patients and has provided important clinical and echocardiography information in this special population. This study was not designed to provide explanation of pathophysiology nor treatment options in COVID-19 patients.
In conclusion, cardiac function evaluated during the first echocardiography performed after ICU admission was abnormal in 34% in this cohort of critically ill COVID-19 patients. While LV systolic dysfunction seemed similar to septic cardiomyopathy, RV dysfunction was related to pressure overload due to positive pressure ventilation, hypercapnia and pulmonary embolism, while not to severity of hypoxemia which appears different from non-COVID-19 ARDS. No other significant cardiac abnormalities were observed. Our data should help intensivists to better understand hemodynamic impairment in these patients to guide therapy.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
Collaborators of the ECHO-COVID study are listed as follows: Anne-Marie Welsh (Nepean Hospital, Sydney, Australia), H Didriksson (Linköping University hospital, Sweden), Yoann Zerbib (University hospital of Amiens, Amiens, France), Clément Brault (University hospital of Amiens, Amiens, France), Laetitia Bodénes (CHU La Cavale Blanche, Brest, France), Nicolas Ferrière (CHU La Cavale Blanche, Brest, France), S Clavier (Hôpital Européen Georges Pompidou, AP-HP and Université de Paris, Paris, France), I Ma (Hôpital Européen Georges Pompidou, AP-HP and Université de Paris, Paris, France), Luigi La Via (Department of Anesthesia and Intensive Care, Policlinico-Vittorio Emanuele University Hospital, Catania, Italy), Veronica Dezio (Department of Anesthesia and Intensive Care, Policlinico-Vittorio Emanuele University Hospital, Catania, Italy), JB Rius (Vall d’Hebron University hospital, Barcelona, Spain), JR Palomares (Vall d’Hebron University hospital, Barcelona, Spain), Fernando Piscioneri (Department of Clinical Internal, Anesthesiological and Cardiovascular Sciences, University of Rome, "La Sapienza", Policlinico Umberto Primo), S Giglioli (CHIREC Hospitals, Université Libre de Bruxelles, Brussels, Belgium), S Banauch-Mayer (The University hospital for University of Massachusetts, Worcester, MA, USA), Bruno François (Medical-surgical ICU, Dupuytren Teaching Hospital, Limoges, France), Anne-Laure Fedoux (Medical-surgical ICU, Dupuytren Teaching Hospital, Limoges, France) and Thomas Daix (Medical-surgical ICU, Dupuytren Teaching Hospital, Limoges, France).
Author contributions
All the authors contribute to read, interpret and enter patients in the secure web-based platform. SH did the statistical analysis. AV-B interpreted the results. SH and AV-B wrote the manuscript. All the authors confirm they have full access to all of data in the study and accept responsibility to submit for publication. They all agree with the manuscript.
Funding
None.
Declarations
Conflicts of interest
AVB declares research grant from GSK and Air Liquid companies. DDB declares honoraria from Philips company for lecture and honoraria for consulting from Edwards Lifesciences, Pfizer and Baxter companies. The other authors declare that they have no conflict of interest.
Ethical approval
The study protocol was approved by ethics committee (CE SRLF 20–40).
Footnotes
The members of the ECHO-COVID research group are listed in the Acknowledgment section at the end of the manuscript.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Antoine Vieillard-Baron, Email: antoine.vieillard-baron@aphp.fr.
the ECHO-COVID research group:
Anne-Marie Welsh, H. Didriksson, Yoann Zerbib, Clément Brault, Laetitia Bodénes, Nicolas Ferrière, S. Clavier, I. Ma, Luigi La Via, Veronica Dezio, J. B. Rius, J. R. Palomares, Fernando Piscioneri, S. Giglioli, S. Banauch-Mayer, Bruno François, Anne-Laure Fedoux, and Thomas Daix
References
- 1.Michard F, Vieillard-Baron A. Critically-ill patients with COVID-19: are they hemodynamically unstable and do we know why? Intensive Care Med. 2021;47(2):254–255. doi: 10.1007/s00134-020-06238-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhou F, Yu T, Fan G, Liu Z, Xiang J, Wang Y, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Shi S, Qin M, Shen B, Cai Y, Liu T, Yang F, et al. Association of cardiac injury with mortality in hospitalized patients with COVID-19 in Wuhan. China JAMA Cardiol. 2020;5(7):802–810. doi: 10.1001/jamacardio.2020.0950. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hendren N, Drazner M, Bozkurt B, Cooper L. Description and proposed management of the acute COVID-19 cardiovascular syndrome. Circulation. 2020;141:1903–1914. doi: 10.1161/CIRCULATIONAHA.120.047349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wichmann D, Sperhake JP, Lütgehetmann M, Steurer S, Edler C, Heinemann A, et al. Autopsy findings and venous thromboembolism in patients with COVID-19. A prospective cohort study. Ann Intern Med. 2020;173(4):268–277. doi: 10.7326/M20-2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gattinoni L, Chiumello D, Caironi P, Busana M, Rominiti F, Brazzi L, et al. COVID-19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med. 2020;46(6):1099–1102. doi: 10.1007/s00134-020-06033-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mekontso-Dessap A, Boissier F, Charron C, Bégot E, Repessé X, Legras A, et al. Acute cor pulmonale during protective ventilation for acute respiratory distress syndrome: prevalence, predictors, and clinical impact. Intensive Care Med. 2016;42:862–870. doi: 10.1007/s00134-015-4141-2. [DOI] [PubMed] [Google Scholar]
- 8.Topol EJ. COVID-19 can affect the heart. Science. 2020;370(6515):408–409. doi: 10.1126/science.abe2813. [DOI] [PubMed] [Google Scholar]
- 9.Dweck M, Bularga A, Hahn R, Bing R, Lee KK, Chapman AR, et al. Global evaluation of echocardiography in patients with COVID-19. Eur Heart J Cardiovasc Imaging. 2020;21(9):949–958. doi: 10.1093/ehjci/jeaa178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sanfilippo F, Huang S, Herpain A, Balik M, Chew M, Clau-Terré F, et al. The PRICES statement: an ESICM expert consensus on methodology for conducting and reporting critical care echocardiography research studies. Intensive Care Med. 2021;47(1):1–13. doi: 10.1007/s00134-020-06262-5. [DOI] [PubMed] [Google Scholar]
- 11.Doyen D, Dupland P, Morand L, Fourrier E, Saccheri C, Buscot M, et al. Characteristics of cardiac injury in critically ill patients with coronavirus disease 2019. Chest. 2021;159(5):1974–1985. doi: 10.1016/j.chest.2020.10.056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Grasselli G, Zangrillo A, Zanella A, Antonelli M, Cabrini L, Castelli A, et al. Baseline characteristics and outcomes of 1591 patients infected with SARS-COV-2 admitted to ICUs of the Lombardy region. Italy JAMA. 2020;323(16):1574–1581. doi: 10.1001/jama.2020.5394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.COVID-ICU group on behalf of the REVA network and the COVID-ICU investigators Clinical characteristics and day-90 outcomes of 4244 critically ill adults with COVID-19: a prospective cohort study. Intensive Care Med. 2021;47(1):60–73. doi: 10.1007/s00134-020-06294-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Vieillard-Baron A, Prin S, Chergui K, Dubourg O, Jardin F. Hemodynamic instability in sepsis: bedside assessment by Doppler echocardiography. Am J Respir Crit Care Med. 2003;168(11):1270–1276. doi: 10.1164/rccm.200306-816CC. [DOI] [PubMed] [Google Scholar]
- 15.Kox M, Waalders N, Kooistra E, Gerretsen J, Pickkers P. Cytokine levels in critically ill patients with COVID-19 and other conditions. JAMA. 2020;324(15):1565–1567. doi: 10.1001/jama.2020.17052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Del Valle D, Kim-Schulze S, Huang H, Beckmann N, Nirenberg S, Wang B, et al. An inflammatory cytokine signature predicts COVID-19 severity and survival. Nat Med. 2020;26(10):1636–1643. doi: 10.1038/s41591-020-1051-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Nagueh SF, Smiseth OA, Appleton CP, Byrd B, Dokainish H, Edvardsen T, et al. Recommendations for the evaluation of left ventricular diastolic function by echocardiography: an update from the American society of echocardiography and the European association of cardiovascular imaging. Eur Heart J Cardiovasc Imaging. 2016;17(12):1321–1360. doi: 10.1093/ehjci/jew082. [DOI] [PubMed] [Google Scholar]
- 18.Hollenberg S, Safi L, Parrillo J, Fata M, Klinkhammer B, Gayed N, et al. Hemodynamic profiles of shock in patients with COVID-19. Am J Cardiol. 2021;153:135–139. doi: 10.1016/j.amjcard.2021.05.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Vieillard-Baron A, Prin S, Chergui K, Dubourg O, Jardin F. Echo-Doppler demonstration of acute cor pulmonale at the bedside in the medical intensive care unit. Am J Respir Crit Care Med. 2002;166(10):1310–1319. doi: 10.1164/rccm.200202-146CC. [DOI] [PubMed] [Google Scholar]
- 20.Evrard B, Goudelin M, Montmagnon N, Fedou A, Lafon T, Vignon P. Cardiovascular phenotypes in ventilated patients with COVID-19 acute respiratory distress syndrome. Crit Care. 2020;24(1):236. doi: 10.1186/s13054-020-02958-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Kim J, Volodarskiy A, Sultan R, Pollie MP, Yum B, Nambiar L, et al. Prognostic utility of right ventricular remodeling over conventional risk stratification in patients with COVID-19. J Am Coll Cardiol. 2020;76(17):1965–1977. doi: 10.1016/j.jacc.2020.08.066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Bleakley C, Singh S, Garfield B, Morosin M, Surkova E, Mandalia S, Dias B, Androulakis E, Proce LC, McCabe C, Wort SJ, West C, Li W, Khattar R, Senior R, Patel BV, Price S. Right ventricular dysfunction in critically illCOVID-19 ARDS. Int J Cardiol. 2021;327:251–258. doi: 10.1016/j.ijcard.2020.11.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cavaleiro P, Masi P, Bagate F, d’Humières T, Mekontso-Dessap A. Acute cor pulmonale in COVID-19 related acute respiratory distress syndrome. Crit Care. 2021;25(1):346. doi: 10.1186/s13054-021-03756-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ackermann M, Verleden S, Kuehnel M, Haverich A, Welte T, Laenger F, et al. Pulmonary vascular endothelialitis thrombosis and angiogenesis in COVID-19. N Engl J Med. 2020;383(2):120–128. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Archer S, Sharp W, Weir E. Differentiating COVID-19 pneumonia from acute respiratory distress syndrome and high altitude pulmonary edema: therapeutic implications. Circulation. 2020;142(2):101–104. doi: 10.1161/CIRCULATIONAHA.120.047915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Caravita S, Baratto C, Di Marco F, Calabrese A, Balestrieri G, Russo F, et al. Haemodynamic characteristics of COVID-19 patients with acute respiratory distress syndrome requiring mechanical ventilation. An invasive assessment using right heart catheterization. Eur J Heart Fail. 2020;22(12):228–2237. doi: 10.1002/ejhf.2058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Soulat-Dufour L, Fauvel C, Weizman O, Barbe T, Pezel T, Mika D, et al. Prognostic value of right ventricular dilatation in patients with COVID-19: a multicenter study. Eur Heart J Cardiovasc Imaging. 2021 doi: 10.1093/ehjci/jeab067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Silverio A, Di Maio M, Scudiero F, Russo V, Esposito L, Attena E, et al. Clinical conditions and echocardiographic parameters associated with mortality in COVID-19. Eur J Clin Invest. 2021;51:e13638. doi: 10.1111/eci.13638. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.