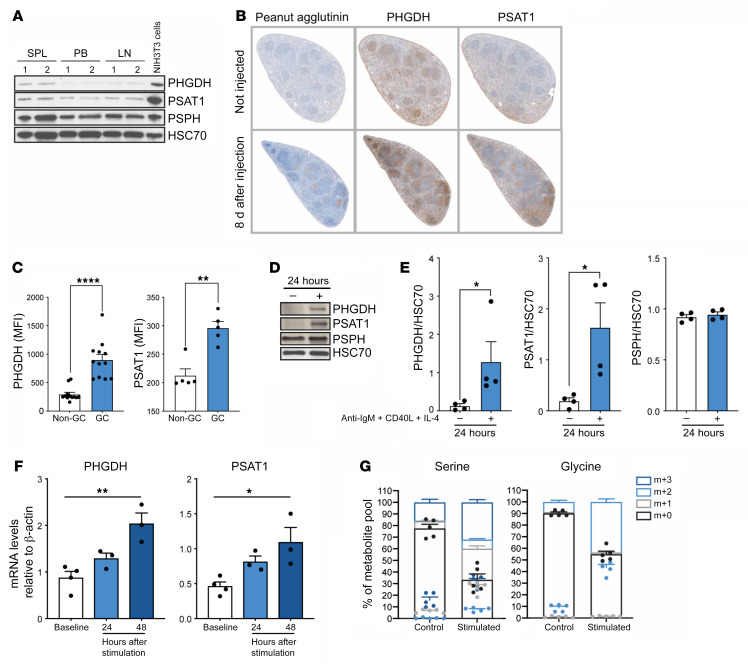
Figure 2. Characterization of the SSP in WT mice after activation in vivo.
(A) Analysis of PHGDH, PSAT1, and PSPH protein levels in resting B cells isolated from mouse spleen (SPL), peripheral blood (PB), and lymph nodes (LN). NIH3T3 murine cells were used as control for high expression of SSP-related enzymes. (B) Representative IHC staining for PNA as GC marker, PHGDH, and PSAT1 on consecutive spleen sections derived from mouse spleens 8 days after sheep RBC immunization (×5 magnification). (C) Expression of PHGDH and PSAT1 in GC B cells and non-GC B cells harvested from mouse spleen 8 days after immunization with sheep RBC. (D) Representative immunoblots of PHGDH, PSAT1, and PSPH proteins levels in murine resting and activated B cells. (E) Mouse B cells were isolated from spleen and left unstimulated (–) or stimulated (+) with anti-IgM/G antibody, CD40L, and IL-4 for 24 hours before protein extraction and quantification of protein levels normalized to HSC70. Individual samples (dots) and means (bars) values are plotted (n = 4). (F) Relative mRNA expression of SSP enzyme genes in resting and activated mouse B cells as determined by qPCR. Isolated mouse B cells were left unstimulated or stimulated with anti-IgM/G antibody, CD40L, and IL-4 for 24 and 48 hours before mRNA extraction. Specific transcript levels were determined relative to β-actin mRNA levels (n = 4). (G) Mass isotopologue distribution of U-[13C6]-glucose–derived serine and glycine from murine resting and activated murine B cells. Cells were left unstimulated or stimulated with anti-IgM/G antibody, CD40L, and IL-4 for 48 hours. Cells were then cultured for 2 hours in serine/glycine-deplete media containing U-[13C]-glucose. 13C isotopologue distribution in serine and glycine was determined by LC-MS. Data are shown as the mean ± SEM. *P < 0.05, **P < 0.01, and ****P < 0.0001, by Mann-Whitney U test (C, E) or by 1-way ANOVA (F).