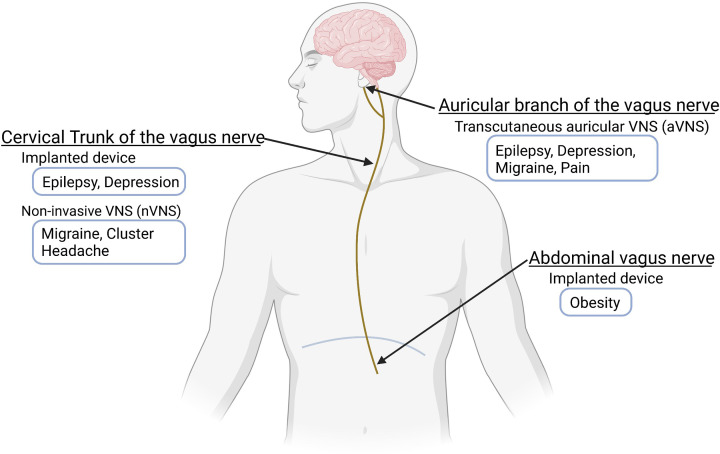
Figure 1. FDA-approved clinical uses of VNS.
The FDA approved an implanted cervical VNS device consisting of the pulse generator, lead wire, and external remote controls for epilepsy and depression, and non-invasive VNS device (applied to the neck) for migraine and cluster headache. In transcutaneous aVNS, the auricular branch of VN is stimulated with electrodes placed on the skin of the external ear in a non-invasive manner for epilepsy, depression, migraine, and pain. The FDA also approved the Maestro Rechargeable System, with implanted electrode wires at the subdiaphragmatic level, for the treatment of obesity.