BACKGROUND:
Neurophysiologic complexity has been shown to decrease during states characterized by a depressed level of consciousness, such as sleep or anesthesia. Conversely, neurophysiologic complexity is increased during exposure to serotonergic psychedelics or subanesthetic doses of dissociative anesthetics. However, the neurochemical substrates underlying changes in neurophysiologic complexity are poorly characterized. Cortical acetylcholine appears to relate to cortical activation and changes in states of consciousness, but the relationship between cortical acetylcholine and complexity has not been formally studied. We addressed this gap by analyzing simultaneous changes in cortical acetylcholine (prefrontal and parietal) and neurophysiologic complexity before, during, and after subanesthetic ketamine (10 mg/kg/h) or 50% nitrous oxide.
METHODS:
Under isoflurane anesthesia, adult Sprague Dawley rats (n = 24, 12 male and 12 female) were implanted with stainless-steel electrodes across the cortex to record monopolar electroencephalogram (0.5–175 Hz; 30 channels) and guide canulae in prefrontal and parietal cortices for local microdialysis quantification of acetylcholine levels. One subgroup of these rats was instrumented with a chronic catheter in jugular vein for ketamine infusion (n = 12, 6 male and 6 female). The electroencephalographic data were analyzed to determine subanesthetic ketamine or nitrous oxide–induced changes in Lempel-Ziv complexity and directed frontoparietal connectivity. Changes in complexity and connectivity were analyzed for correlation with concurrent changes in prefrontal and parietal acetylcholine.
RESULTS:
Subanesthetic ketamine produced sustained increases in normalized Lempel-Ziv complexity (0.5–175 Hz; P < .001) and high gamma frontoparietal connectivity (125–175 Hz; P < .001). This was accompanied by progressive increases in prefrontal (104%; P < .001) and parietal (159%; P < .001) acetylcholine levels that peaked after 50 minutes of infusion. Nitrous oxide induction produced a transient increase in complexity (P < .05) and high gamma connectivity (P < .001), which was accompanied by increases (P < .001) in prefrontal (56%) and parietal (43%) acetylcholine levels. In contrast, the final 50 minutes of nitrous oxide administration were characterized by a decrease in prefrontal (38%; P < .001) and parietal (45%; P < .001) acetylcholine levels, reduced complexity (P < .001), and comparatively weaker frontoparietal high gamma connectivity (P < .001). Cortical acetylcholine and complexity were correlated with both subanesthetic ketamine (prefrontal: cluster-weighted marginal correlation [CW r] [144] = 0.42, P < .001; parietal: CW r[144] = 0.42, P < .001) and nitrous oxide (prefrontal: CW r[156] = 0.46, P < .001; parietal: CW r[156] = 0.56, P < .001) cohorts.
CONCLUSIONS:
These data bridge changes in cortical acetylcholine with concurrent changes in neurophysiologic complexity, frontoparietal connectivity, and the level of consciousness.
KEY POINTS.
Question: What is the relationship between neurophysiologic complexity and cortical acetylcholine?
Findings: Subanesthetic ketamine- and nitrous oxide–induced changes in cortical acetylcholine correlated with concurrent changes in neurophysiologic complexity, connectivity, and behavior.
Meaning: The relationship between cortical acetylcholine and neurophysiologic complexity prompts further investigation of subcortical cholinergic nuclei in the modulation of cortical neurophysiologic complexity.
See Article, p 1114
Although the precise neural correlates of consciousness are still a matter of active debate,1,2 there is evidence that cortical neurophysiologic complexity relates to the level of consciousness.3,4 Neurophysiologic complexity has been shown to be depressed during unconscious states such as slow-wave sleep, coma, and anesthesia.5–10 Conversely, psychoactive drugs, such as classical serotonergic psychedelics or dissociative N-methyl-d-aspartate (NMDA) antagonists are known to enhance complexity with several studies establishing an association between changes in complexity, cortical connectivity, and reports of alterations to conscious contents.9,11–13 Although cortical complexity has been characterized across a broad spectrum of brain states, there is limited understanding of the relationship between measures of complexity and underlying neurochemical processes within the cortex.
The cortex receives topographically specific cholinergic projections from the basal forebrain.14 Cortical acetylcholine levels are high during states associated with high complexity and the presence of phenomenological content, such as wakefulness, rapid eye movement sleep, or after the administration of serotonergic psychedelics or subanesthetic levels of glutamatergic dissociatives.15–19 Conversely, cortical acetylcholine is suppressed during states of low complexity and reduced conscious content, such as slow-wave sleep or anesthesia.15,20,21
Despite a biologically plausible relationship that might be inferred from past investigations, there has been no study that has characterized—through concurrent measurements—the relationship between neurophysiologic complexity and cortical acetylcholine. Studies characterizing neurophysiologic complexity have often relied on temporospatial analysis of high-density electroencephalographic recordings in human subjects‚ but have not measured concurrent neurochemical changes. On the other hand, studies using animal models typically allow neurochemical analysis but have used sparse electroencephalographic recordings that prevent the application of temporospatial analyses used in human studies. To address this gap, we developed a novel approach to record high-density (30 channels) intracranial electroencephalogram (EEG) in rats while simultaneously measuring acetylcholine levels in prefrontal and parietal cortices. We then leveraged the NMDA antagonists ketamine and nitrous oxide as pharmacological tools, due to their unique dose-dependent anesthetic and psychedelic properties. Changes in acetylcholine concentration in prefrontal and parietal cortices were compared with Lempel-Ziv complexity (LZs)22 before, during, and after administration of subanesthetic ketamine or 50% nitrous oxide. To further characterize cortical dynamics, we computed normalized symbolic transfer entropy (NSTE), an information theoretic measure to estimate directed connectivity,21,23,24 within gamma bandwidths between frontal and parietal cortices. We report that changes in cortical acetylcholine levels during subanesthetic ketamine or 50% nitrous oxide exposure correlate with changes in neurophysiologic complexity and high gamma connectivity.
METHODS
Rats
The study was approved by the Institutional Animal Care and Use Committee (University of Michigan) and was performed in accordance with the Guide for the Care and Use of Laboratory Animals (8th Edition, The National Academies Press), as well as ARRIVE (Animal Research: Reporting of In Vivo Experiments) guidelines. Adult male and female Sprague Dawley rats (n = 24, 12 male:12 female, 300–350 g, Charles River Laboratories) were used. The rats were housed in a temperature- and light-controlled facility (12 hours light: 12 hours dark cycle, lights on at 8:00 am) with ad libitum access to food and water.
Surgical Procedures
The surgical procedures have been described in our recent studies21,24,25 and are provided in detail in the supplemental methods (Supplemental Digital Content http://links.lww.com/AA/D757). In brief, under surgical isoflurane anesthesia, rats were implanted with stainless-steel screw electrodes to record high-density EEG from across the cortex (Supplemental Digital Content, Figure 1, http://links.lww.com/AA/D757) and microdialysis guide tubes aimed at prefrontal cortex (prelimbic region) and parietal cortex (somatosensory barrel field region). In a subset of rats (n = 12, 6 male and 6 female), an indwelling catheter was surgically positioned into the jugular vein to allow for intravenous infusion of ketamine.
Experimental Design
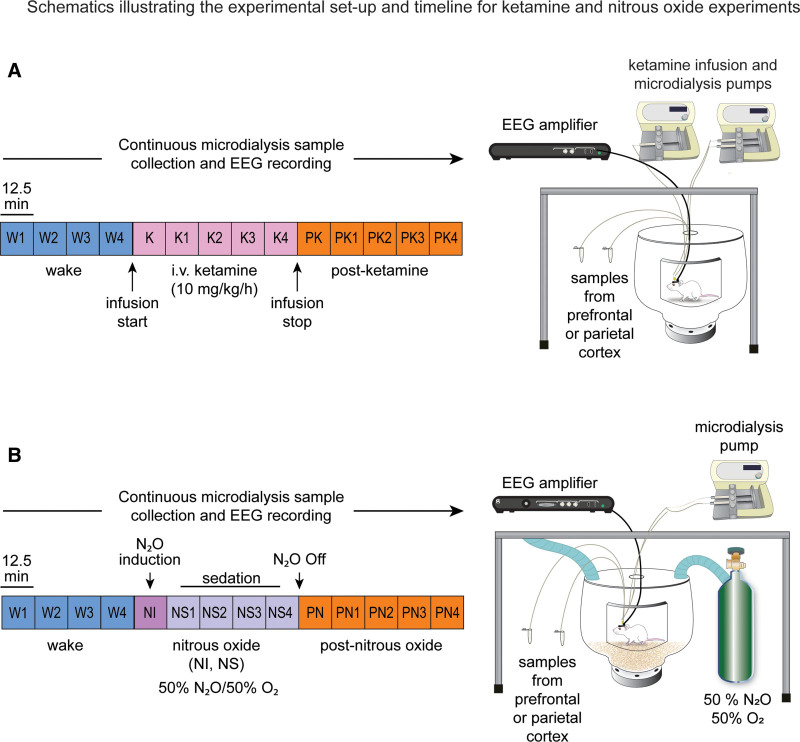
Schematics depicting the design for ketamine and nitrous oxide experiments are illustrated in Figure 1A and B, respectively. The experiments were conducted after at least 10 days of postsurgical recovery. Equal sized cohorts of rats (n = 12, 6 male and 6 female) were used for ketamine and nitrous oxide experiments. EEG data were recorded continuously throughout the experiment, while microdialysis samples were collected in 12.5-minute epochs. The EEG was recorded, and dialysis samples were collected for 50 minutes (4 microdialysis epochs) during freely moving baseline wake condition. The EEG was monitored in real time by the experimenter, and gentle tapping on the recording chamber was used to maintain a constant state of wakefulness. Following baseline recording, rats were either connected to an intravenous catheter line to allow for sustained infusion of ketamine at 10 mg/kg/h or were sealed in an airtight chamber to allow for delivery of a mixture of 50% nitrous oxide and 50% oxygen into the recording chamber (12 L/min). The EEG and dialysis samples were then collected for a period of 62.5 minutes (5 microdialysis epochs) during administration of subanesthetic ketamine or nitrous oxide. Ketamine infusion or nitrous oxide exposure was then stopped, and data were collected for another 62.5 minutes (5 microdialysis epochs) during the postdrug recovery period. At the conclusion of data collection, the sites of microdialysis were histologically verified (Supplemental Digital Content, Figure 2, http://links.lww.com/AA/D757).
Figure 1.
Schematics illustrating the experimental setup and timeline for ketamine (A) and nitrous oxide (B) experiments. The EEG data and microdialysis samples from prefrontal and parietal cortices were collected simultaneously and continuously, but the microdialysis samples were collected in 12.5-min bins. Each colored box represents 1 microdialysis epoch. Samples were collected during freely moving baseline W condition, continuous K at 10 mg/kg/h, or PK recovery period. The data collection was performed similarly for the nitrous oxide cohort, with epochs corresponding to the W state, 50% NI, 50% NS, and PN recovery period. EEG indicates electroencephalogram; K, subanesthetic ketamine infusion; NI, nitrous induction; NS, nitrous sedation; PK, postketamine; PN, post-nitrous oxide; W, wake.
EEG Data Acquisition and Quantification of Cortical Acetylcholine Levels
Monopolar EEG signals, referenced to an electrode over the nasal sinus, were acquired (0.1–500 Hz; 1-kHz sampling rate) from across the cortex. Prefrontal and parietal microdialysis samples were analyzed with high-performance liquid chromatography and electrochemical detection for quantification of acetylcholine levels. The detailed methodology is provided in the supplemental methods (Supplemental Digital Content, http://links.lww.com/AA/D757).
LZs was used to approximate temporospatial EEG complexity. LZs is a method of symbolic-sequence analysis that assesses the algorithmic complexity (ie, diversity and compressibility) of finite sequences such as EEG time series.22 LZs analysis was conducted as outlined in previous studies from our laboratory9,10,24 and others.7,8,12,13 To control for any potential bias in the resultant LZs values due to the frequency content of the EEG signal, we normalized LZs by the average of n = 50 surrogate datasets in which the spectral profile were identical to the original signal, but the phase-information was maximally randomized.7–10,12,24 This excludes the possibility that changes in complexity reflected in the normalized metric of LZs (LZsN) are attributable only to the spectral contents of the signal.
Frontoparietal Directed Connectivity: Normalized Symbolic Transfer Entropy Analysis
We used NSTE to assess the directed connectivity between frontal and parietal cortices. The analysis methods have been described in our previous studies21,23,24 and are provided in the supplemental methods (Supplemental Digital Content, http://links.lww.com/AA/D757). We focused on changes in directed connectivity between ipsilateral frontal and parietal channels and in 3 gamma bandwidths (low gamma [25–55 Hz], mid gamma [85–125 Hz], and high gamma [125–175 Hz]) because previous work from our laboratory has demonstrated directed frontoparietal connectivity in high gamma bands to be a correlate of wakefulness.21
Statistical Analyses
All statistical analyses were performed using the R software26 in consultation with the Consulting for Statistics, Computing, and Analytics Research Core at the University of Michigan.
Our primary outcome measures were changes in cortical acetylcholine, LZs, and frontoparietal connectivity. No a priori power analysis for sample size selection was performed. The sample size was informed by our previously published similar studies19,21,24,25 in which cortical acetylcholine, directed connectivity, or complexity were analyzed using identical analytic methodologies and in which we found that sample sizes of ≤11 were sufficient in detecting significant differences in these outcome measures across experimental conditions. The first ketamine infusion epoch was excluded from EEG and neurochemical analyses due to the time required for the drug to traverse dead space in the catheter and reach blood circulation. For the rest of the epochs in ketamine experiments, a within-subject design was utilized in which each rat contributed 4 wake datapoints, 4 ketamine infusion datapoints, and 4 postketamine datapoints for each measure. As opposed to intravenous administration of ketamine, which required 5 to 7 minutes to traverse the catheter, nitrous oxide exposure was quick, and the recording chamber was filled within 1 minute of the start of nitrous oxide delivery. However, the rats showed an acute response to nitrous oxide, which was primarily limited to the first 12.5 minutes of nitrous oxide delivery and differed from the subsequent 50 minutes of exposure. Therefore, we analyzed the first epoch (12.5 minutes) of nitrous oxide exposure separately as “nitrous induction” (NI), while the rest of the 4 epochs (50 minutes) were categorized as nitrous “sedation” (NS) and analyzed as a separate block. For nitrous oxide experiments, we implemented a within-subject design in which each rat contributed 4 wake datapoints, 1 nitrous oxide induction datapoint, 4 nitrous oxide sedation datapoints, and 4 post-nitrous oxide datapoints for each EEG measure and acetylcholine analysis. The statistical analyses for ketamine experiments were run on 5-minute segments of noise-free EEG from each 12.5-minute epoch, while for nitrous oxide experiments, 2-minute segments of noise-free EEG were selected. The choice to use shorter EEG epochs for nitrous oxide experiments was due to the prevalence of noise in the EEG signal driven by sporadic chewing or “bruxing” behavior. To exclude mixed states, the first postketamine and first post-nitrous oxide epochs were excluded from our analyses. Thus, each rat in the ketamine experiments contributed 12 EEG and microdialysis datapoints to the statistical analysis, while each rat in the nitrous experiments contributed to 13 data points. A linear mixed model was utilized in which “Drug State” was treated as a fixed factor. We also included “Subject Sex” as a fixed factor to account for any influences of sex on drug response. “Subject” was treated as a random intercept in the model to account for intersubject variability. An alpha threshold of P < .05 was selected, and Tukey post hoc test was used to correct for multiple pairwise comparisons (paired t test) between states. The same data analysis plan was followed to quantify the changes in spectral power. The methodology for power spectral analysis and the results are reported in Supplemental Digital Content, Figure 3, http://links.lww.com/AA/D757. To assess correlations among cortical complexity, connectivity, and acetylcholine levels, we used a Pearson correlation-based method, optimized for analysis of clustered repeated-measures data.27 The data are provided as box and whisker plots with median, interquartile range, minimum and maximum values, and the individual data points for each subject superimposed on the plots. The mean, standard deviation, and F statistics for statistical comparisons are provided in Supplemental Digital Content, Tables 1–6, http://links.lww.com/AA/D757.
RESULTS
Subanesthetic Ketamine Infusion Produced a Sustained Increase in Acetylcholine Levels in Prefrontal and Parietal Cortices
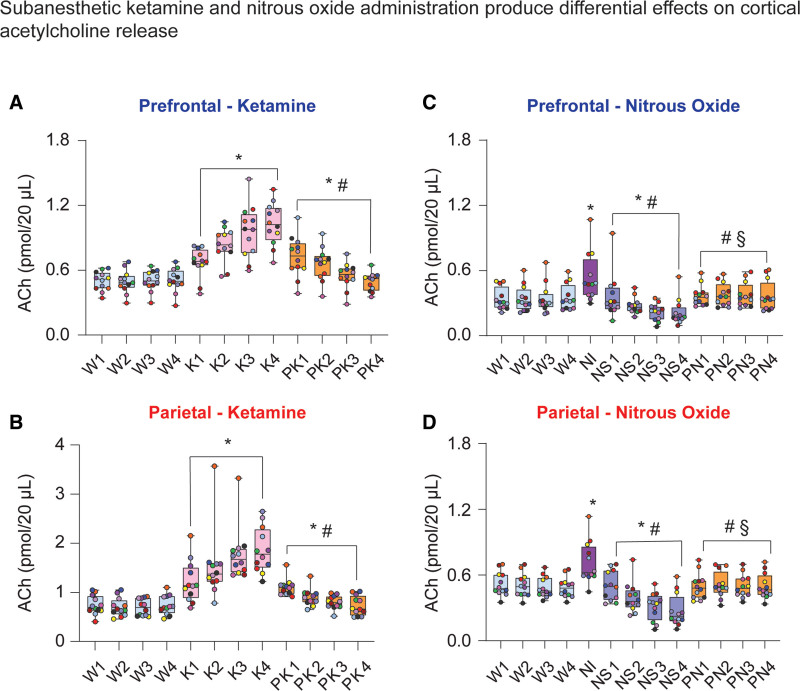
Intravenous infusion of subanesthetic ketamine resulted in a consistent increase in acetylcholine levels relative to wake state in both prefrontal (104%; t[130] = 12.49; P < .001; Figure 2A) and parietal (159%; t[130] = 13.72; P < .001; Figure 2B) cortices, lasting all 4 epochs and reaching maximum levels in the final 12.5 minutes of infusion. During subanesthetic ketamine infusion, rats displayed stereotypic head bobbing and circling behavior, interrupted by periodic bouts of ataxia. As compared to the wake state, acetylcholine levels in both prefrontal and parietal cortices remained elevated during postketamine recovery period (21%, prefrontal: t[130] = 3.54, P < .01, Figure 2A; parietal: 23%, t[130] = 2.546, P = .03, Figure 2B), with cortical acetylcholine progressively declining 12.5 minutes after the cessation of the ketamine infusion. The acetylcholine levels during the postketamine recovery period were significantly lower than that during subanesthetic ketamine infusion (prefrontal: −26%, t[130] = –8.95, P < .001, Figure 2A; −41%, parietal: t[130] = –11.17, P < .001, Figure 2B).
Figure 2.
Subanesthetic ketamine and nitrous oxide administration produced differential effects on cortical acetylcholine levels. Subanesthetic ketamine infusion produced sustained increase in prefrontal (A) and parietal (B) acetylcholine levels. During postketamine recovery, cortical acetylcholine levels remained elevated compared to wakefulness, though this effect was largely driven by the first 2 postketamine epochs (A and B). In contrast, 50% nitrous oxide treatment transiently increased prefrontal and parietal acetylcholine levels during the first 12.5 min of exposure (nitrous induction), followed by a progressive decline in acetylcholine during nitrous oxide sedation phase (C and D). Cortical acetylcholine levels during post-nitrous oxide recovery did not significantly differ from those observed during baseline wake state (C and D). A linear mixed model with a random intercept for each rat was used for statistical comparisons. Post hoc pairwise tests between states were performed with single-step correction for multiple comparisons via Tukey test. The box plots show the median (horizontal bar) and interquartile range for averaged data over all 12 subjects at each epoch. The whiskers represent the minimum and maximum values within each epoch. The data for each subject are displayed by colored dots, with each color corresponding to a single subject across all epochs. *Significant compared to wake. #Significant compared to subanesthetic ketamine infusion or nitrous oxide induction. §Significant compared to nitrous oxide sedation. The statistical comparisons are shown at P < .05. The exact P values are provided in the text in the Results section. The mean, SD, and F statistics for statistical comparisons are provided in Supplemental Digital Content, Tables 1 and 2, http://links.lww.com/AA/D757. ACh indicates acetylcholine; K, subanesthetic ketamine infusion; NI, nitrous induction; NS, nitrous sedation; PK, postketamine recovery; PN, post-nitrous oxide; SD, standard deviation; W, wake.
Nitrous Oxide Exposure Produced State-Dependent Effects on Acetylcholine Levels in Prefrontal and Parietal Cortices
Nitrous oxide exposure resulted in state-dependent effects on acetylcholine in prefrontal and parietal cortices that depended on the depth of sedation, which increased with the duration of exposure. During the first 12.5 minutes of nitrous oxide exposure, hereafter referred to as NI, rats showed active behavior such as grooming, stereotypic head bobbing, and uncoordinated locomotion. Acetylcholine levels in both prefrontal and parietal cortices were significantly elevated relative to wake (prefrontal: 56%, t[141] = 7.97, P < .001, Figure 2C; parietal: 43%, t[141] = 7.39, P < .001, Figure 2D). The following 50 minutes of nitrous oxide exposure, hereafter referred to as NS, were marked by periodically quiescent behavior interrupted by active and purposeful behaviors such as slow locomotion, grooming, or pica-like consumption of bedding. The requirement to have a sealed recording chamber precluded any behavioral manipulation to assess the level of sedation. However, despite relatively quiescent behavior, rats clearly retained righting reflex throughout nitrous oxide treatment, displayed sporadic spontaneous behavior, and were easily aroused by tapping on the wall of recording chamber. This was accompanied by a progressive decline in prefrontal (38%, t[141] = −5.01, P < .001; Figure 2C) and parietal (45%, t[141] = −7.07, P < .001; Figure 2D) acetylcholine, relative to wake state. Cortical acetylcholine showed a rapid return to prenitrous levels during postnitrous recovery and did not statistically differ from the wake state (prefrontal: t[141] = 1.86, P = .2, Figure 2C; parietal: t[141] = 0.01, P = 1, Figure 2D).
Subanesthetic Ketamine Infusion–Induced Persistent Increase in Temporospatial EEG Complexity
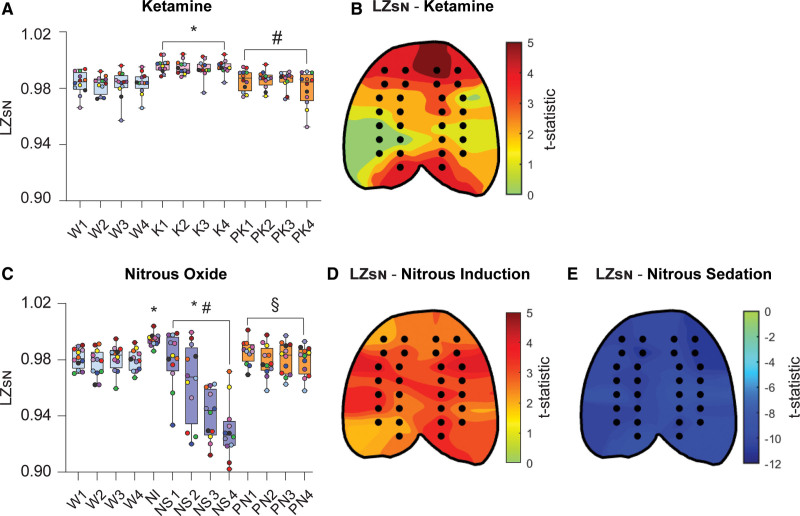
To assess the effect of subanesthetic ketamine infusion on temporospatial EEG complexity independent of the power spectrum, LZsN was computed for EEG epochs before, during, and after ketamine infusion. As compared to baseline wake state, LZsN was significantly higher during subanesthetic ketamine (t[130] = 9.77, P < .001; Figure 3A). During postketamine recovery period, LZsN values decreased relative to ketamine infusion (t[130] = −8.89, P < .001; Figure 3A), returning to values that did not significantly differ from the wake state (t[130] = 0.88, P = .7; Figure 3A). A topographic plot illustrating the degree of change in LZsN during subanesthetic ketamine relative to wake in each EEG channel (as calculated by the t statistic) is depicted in Figure 3B, with regions colored red representing the areas with the greatest magnitude of change in LZsN. EEG channels located within the frontal, posterior parietal, and occipital cortex showed the most statistically significant changes in LZsN during subanesthetic ketamine infusion, as compared to the wake state (Figure 3B).
Figure 3.
Changes in temporospatial EEG complexity during subanesthetic ketamine infusion and nitrous oxide exposure mirror concomitant changes in cortical acetylcholine levels. LZsN was significantly increased during subanesthetic ketamine infusion, returning to wake levels after the infusion was stopped (A). Changes in LZsN during subanesthetic ketamine infusion were also quantified at the level of single channels (black dots in the topographic plots), demonstrating that the most pronounced changes in temporal complexity, relative to wake, occurred in frontal, posterior parietal, and occipital channel clusters (B). LZsN significantly increased during the first 12.5 min of nitrous oxide exposure (induction phase) as compared to baseline wake state (C). During the subsequent nitrous oxide sedation phase, the LZsN declined to levels significantly lower than that observed during baseline wake. LZsN returned to levels comparable to baseline wakefulness during post-nitrous oxide recovery (C). The most significant increases in temporal complexity during nitrous oxide induction, as compared to baseline wake state, were located in frontotemporal, parietal, and occipital regions (D). Nitrous oxide sedation was characterized by a significant decrease in temporal complexity across the entire cortical surface (E). A linear mixed model with a random intercept for each rat was used for statistical comparisons. Post hoc pairwise tests between states were performed with single-step correction for multiple comparisons via Tukey test. The box plots show the median (horizontal bar) and interquartile range for averaged data over all 12 subjects at each epoch. The whiskers represent the minimum and maximum values within each epoch. The data for each subject are displayed by colored dots, with each color corresponding to a single subject across all epochs. *Significant compared to wake. #Significant compared to subanesthetic ketamine infusion or nitrous oxide induction. §Significant compared to nitrous oxide sedation. The statistical comparisons are shown at P < .05. The exact P values are provided in the text in the Results section. The mean, standard deviation, and F statistics for statistical comparisons are provided in Supplemental Digital Content, Tables 3 and 4, http://links.lww.com/AA/D757. EEG indicates electroencephalogram; K, subanesthetic ketamine infusion; LZsN, normalized Lempel-Ziv complexity; NS, nitrous sedation; PK, postketamine recovery; PN, post-nitrous oxide; W, wake.
Nitrous Oxide Exposure Caused State-Dependent Changes in Temporospatial EEG Complexity
Mirroring its effects on cortical acetylcholine, nitrous oxide exposure produced changes in EEG complexity that largely depended on the duration of exposure. The LZsN during the first 12.5 minutes of nitrous oxide exposure (ie, the induction phase) was found to be significantly higher than that observed during baseline wake state (t[141] = 2.64, P = .04; Figure 3C). However, in the following 50 minutes of nitrous oxide exposure, the animals showed behavioral signs of sedation (ie, reduced movement, slow stereotypic head bobbing, and sporadic grooming) accompanied with progressive decline in LZsN values, which were significantly lower than that observed during both wakefulness (t[141] = −7.34, P < .001; Figure 3C) and the nitrous oxide induction phase (t[141] = −7.28, P < .001; Figure 3C). During the postnitrous recovery period, LZsN values returned to levels that were significantly greater than nitrous oxide sedation (t[141] = 7.8, P < .001; Figure 3C) but did not significantly differ from levels observed during baseline wake state (t[141] = 0.45, P = 1; Figure 3C). Figure 3D and E illustrates topographic maps of the t statistic representing changes in LZsN relative to wake during nitrous oxide treatment within each EEG channel, with red regions representing channels with the most significant increases in LZsN and the darkest blue regions representing the most significant decreases in LZsN. Nitrous oxide induction caused a broad increase in neurophysiologic complexity across the EEG montage, with channels within the temporal, parietal, and occipital regions showing the most significant increases in LZsN, as compared to the wake state (Figure 3D). In contrast, nitrous oxide sedation in the following 50 minutes caused global suppression of LZsN relative to the wake state, with significant decreases in EEG complexity spanning the entirety of the cortex (Figure 3E).
Prefrontal and Parietal Acetylcholine Levels Correlate With the Changes in Cortical Complexity
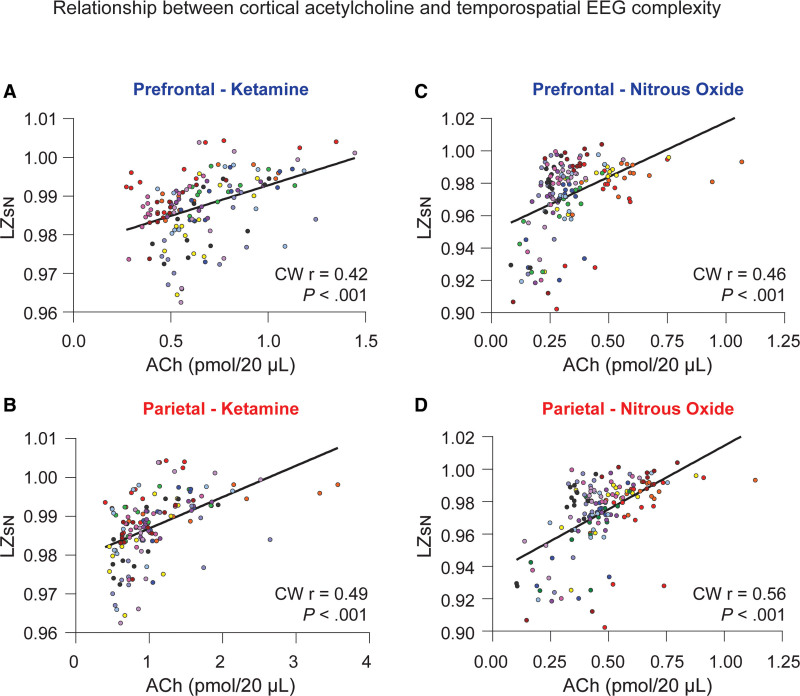
To assess the correlation between changes in prefrontal and parietal acetylcholine levels and temporospatial EEG complexity, the cluster-weighted marginal correlation (CW r) was computed for both ketamine and nitrous oxide groups. Prefrontal and parietal acetylcholine levels for each microdialysis epoch were compared with the concomitant changes in LZsN measured across all 144 individual datapoints in ketamine experiments or all 156 datapoints in nitrous oxide experiments, with data dependencies based on individual rats factored into the analysis. Acetylcholine levels in the prefrontal and parietal cortices showed significant positive correlations with LZsN both in the ketamine group (prefrontal: CW r[144] = 0.42, P < .001, Figure 4A; parietal: CW r[144] = 0.49, P < .001, Figure 4B) and the nitrous oxide group (prefrontal: CW r[156] = 0.46, P < .001, Figure 4C; parietal: CW r[156] = 0.56, P < .001, Figure 4D).
Figure 4.
Relationship between cortical acetylcholine and temporospatial EEG complexity. Changes in prefrontal and parietal acetylcholine levels were significantly correlated with changes in temporospatial EEG complexity in the subanesthetic ketamine infusion (A and B) and nitrous oxide exposure (C and D) cohorts. The data for each subject are displayed by colored dots, with each color corresponding to a single subject across all epochs. To account for clustering of the data within each rat, we calculated the cluster-weighted marginal correlation. The line represents points of best fit. ACh indicates acetylcholine; CW r, cluster-weighted marginal correlation; EEG, electroencephalogram; LZsN, normalized Lempel-Ziv complexity.
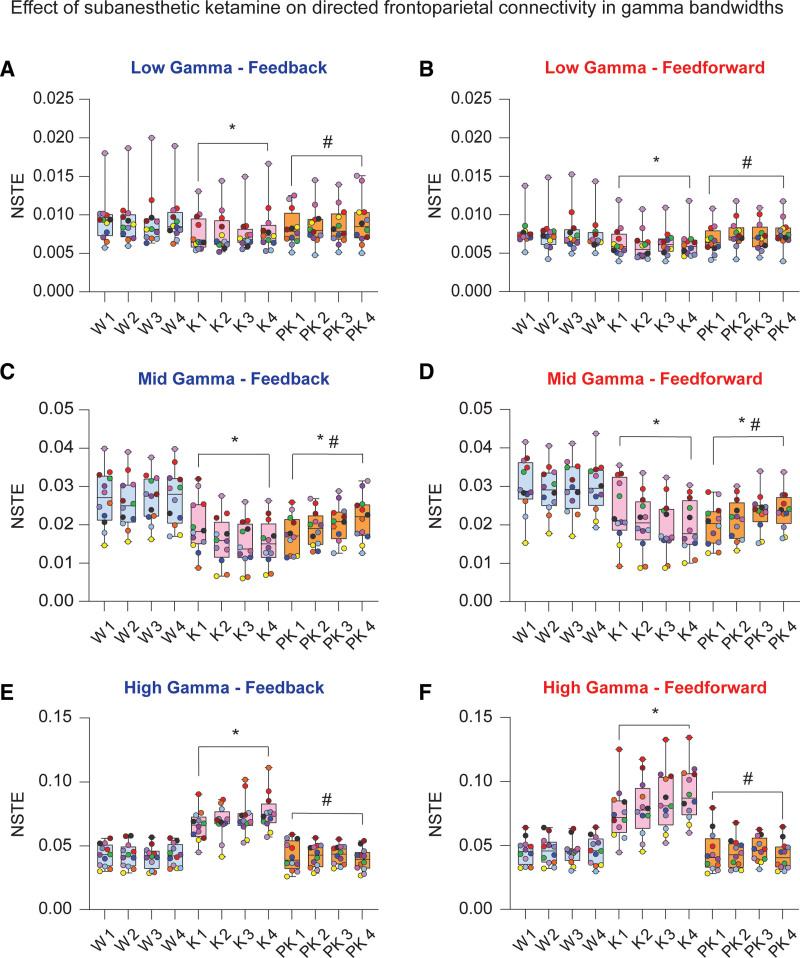
Effects of Subanesthetic Ketamine on Frontoparietal Connectivity in Gamma Bandwidths
NSTE was computed across low (25–55 Hz), mid (85–125 Hz), and high gamma (125–175 Hz) bandwidths between ipsilateral frontal and parietal channels as an estimate of directed feedback (frontal to parietal) and feedforward (parietal to frontal) connectivities. Subanesthetic ketamine suppressed low and mid gamma frontoparietal connectivities in both feedback (low: t[130] = −6.89, P < .001, Figure 5A; mid: t[130] = −15.12, P < .001, Figure 5C) and feedforward directions (low: t[130] = −8.20, P < .001, Figure 5B; mid: t[130] = −14.90, P < .001, Figure 5D). Conversely, high gamma frontoparietal connectivity was significantly increased during subanesthetic ketamine in both feedback (t[130] = 17.59, P < .001; Figure 5E) and feedforward (t[130] = 17.24, P < .001; Figure 5F) directions. As compared to wake state, postketamine recovery was characterized by reduced frontoparietal connectivity in the mid gamma band (feedback: t[130] = −10.54, P < .001, Figure 5C; feedforward: t[130] = −12.79, P < .001, Figure 5D), while the low and high gamma connectivities returned to levels that did not significantly differ from that observed during the baseline wake state. In the low gamma band, feedback and feedforward connectivities were elevated relative to subanesthetic ketamine (feedback: t[130] = 3.85, P < .001, Figure 5A; feedforward: t[130] = 6.24, P < .001, Figure 5B). High gamma connectivity during postketamine recovery remained attenuated as compared to subanesthetic ketamine (feedback: t[130] = −18.02, P < .001, Figure 5E; feedforward: t[130] = −17.89, P < .001, Figure 5F). The correlations between frontoparietal connectivity in gamma bandwidths and acetylcholine levels in prefrontal and parietal cortices are reported in Supplemental Digital Content, Table 7, http://links.lww.com/AA/D757.
Figure 5.
Effect of subanesthetic ketamine infusion on directed frontoparietal connectivity in gamma bandwidths. Subanesthetic ketamine infusion significantly decreased frontoparietal connectivity in the low gamma bandwidth (25–55 Hz) in both feedback and feedforward directions relative to baseline wake state (A and B). Frontoparietal connectivity in the mid gamma bandwidth (85–125 Hz) decreased in feedback and feedforward directions during subanesthetic ketamine and remained depressed relative to baseline wakefulness during postketamine recovery (C and D). Feedback and feedforward connectivities between frontal and parietal cortices were significantly increased in the high gamma bandwidth (125–175 Hz) during subanesthetic ketamine treatment, returning to levels comparable to baseline wakefulness during postketamine recovery (E and F). A linear mixed model with a random intercept for each rat was used for statistical comparisons. Post hoc pairwise tests between states were performed with single-step correction for multiple comparisons via Tukey test. The box plots show the median (horizontal bar) and interquartile range for averaged data over all 12 subjects at each epoch. The whiskers represent the minimum and maximum values within each epoch. The data for each subject are displayed by colored dots, with each color corresponding to a single subject across all epochs. *Significant compared to wake. #Significant compared to subanesthetic ketamine infusion. The statistical comparisons are shown at P < .05. The exact P values are provided in the text in the Results section. The mean, SD, and F statistics for statistical comparisons are provided in Supplemental Digital Content, Table 5, http://links.lww.com/AA/D757. K indicates subanesthetic ketamine infusion; NSTE, normalized symbolic transfer entropy; PK, postketamine recovery; SD, standard deviation; W, wake.
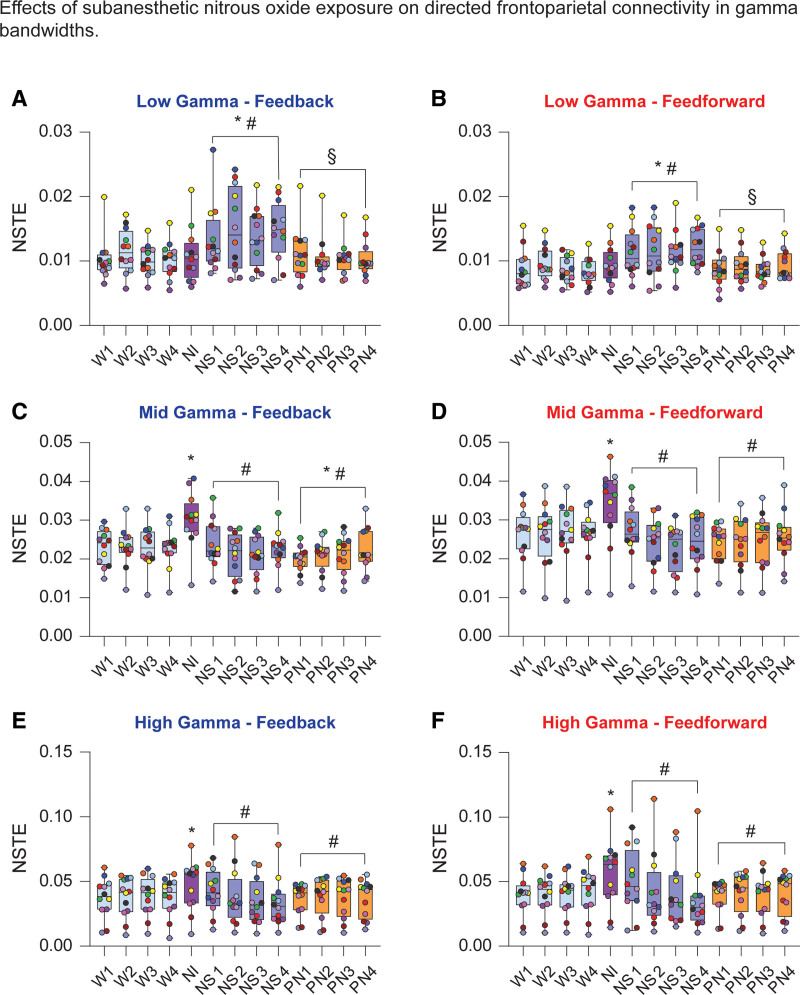
Effects of Nitrous Oxide Exposure on Frontoparietal Connectivity in Gamma Bandwidths
Nitrous oxide exposure produced no significant effect on low gamma frontoparietal connectivity in the first 12.5-minute induction period (feedback: t[141] = 0.44, P = 1, Figure 6A; feedforward: t[141] = 1, P = .7, Figure 6B). However, the following 50 minutes of nitrous oxide sedation significantly increased low gamma frontoparietal connectivity in both feedforward and feedback directions, as compared to the wake state (feedback: t[141] = 6.41, P < .001, Figure 6A; feedforward: t[141] = 6.84, P < .001, Figure 6B). As compared to nitrous oxide sedation, the postnitrous recovery was characterized by significant attenuation of both feedback and feedforward low gamma frontoparietal connectivities (feedback: t[141] = −6.18, P < .001, Figure 6A; feedforward: t[141] = 3.33, P = .005, Figure 6B), with no significant difference as compared to the baseline wake levels (low gamma feedback: t[141] = 0.24, P = 1, Figure 6A; low gamma feedforward: t[141] = 0.5, P = .9, Figure 6B).
Figure 6.
Effects of nitrous oxide exposure on directed frontoparietal connectivity in gamma bandwidths. During nitrous oxide induction, frontoparietal connectivity in the low gamma bandwidth (25–55 Hz) did not differ from wake (A and B). However, feedback and feedforward connectivities in low gamma frequencies were significantly increased during the following 50-min nitrous oxide sedation (A and B). Frontoparietal connectivity in feedback and feedforward directions were increased during nitrous oxide induction in the mid gamma bandwidth (85–125 Hz) (C and D), whereas nitrous oxide sedation was characterized by a decrease in connectivity as compared to wakefulness. During post-nitrous oxide recovery period, connectivity in the feedback direction showed a modest increase relative to baseline wakefulness, while feedforward connectivity did not statistically differ from wake. Nitrous oxide induction transiently increased the strength of connectivity between frontal and parietal cortices in the high gamma bandwidth (125–175 Hz) in feedback and feedforward directions (E and F), whereas the nitrous oxide sedation phase was characterized by decrease in both feedback and feedforward connectivities to wake levels. Frontoparietal connectivity did not differ between baseline wakefulness and post-nitrous oxide recovery. A linear mixed model with a random intercept for each rat was used for statistical comparisons. Post hoc pairwise tests between states were performed with single-step correction for multiple comparisons via Tukey test. The box plots show the median (horizontal bar) and interquartile range for averaged data over all 12 subjects at each epoch. The whiskers represent the minimum and maximum values within each epoch. The data for each subject are displayed by colored dots, with each color corresponding to a single subject across all epochs. *Significant compared to wake. #Significant compared to nitrous oxide induction, §Significant compared to nitrous oxide sedation. The statistical comparisons are shown at P < .05. The exact P values are provided in the text in the Results section. The mean, SD, and F statistics for statistical comparisons are provided in Supplemental Digital Content, Table 6, http://links.lww.com/AA/D757. NI indicates nitrous induction; NS, nitrous sedation; NSTE, normalized symbolic transfer entropy; PN, post-nitrous oxide; SD, standard deviation; W, wake.
In the mid gamma bandwidth, nitrous oxide induction was characterized by increase in bidirectional frontoparietal connectivity (feedback: t[141] = 6.43, P < .001, Figure 6C; feedforward: t[141] = 6.09, P < .001, Figure 6D). During NS, mid gamma connectivity did not significantly differ from wake but was significantly lower than NI (feedback: t[141] = −7.15, P < .001, Figure 6C; feedforward: t[141] = −7.72, P < .001, Figure 6D). As compared to the wake state, the postnitrous period was characterized by a significant attenuation of mid gamma feedback connectivity (t[141] = −2.69, P = .04, Figure 6C), while there was no significant effect on feedforward connectivity (t[141] = −2.50, P = .06, Figure 6D).
High gamma frontoparietal connectivity during nitrous oxide induction increased in feedback (t[141] = 3.90, P < .001, Figure 6E) and feedforward (t[141] = 4.52, P < .001, Figure 6F) directions. During nitrous oxide sedation, high gamma connectivity returned to the baseline wake state but was significantly attenuated as compared to that observed during nitrous oxide induction (feedback: t[141] = −4.19, P < .001, Figure 6E; feedforward: t[141] = −4.02, P < .001, Figure 6F). Frontoparietal connectivity during post-nitrous oxide recovery wakefulness did not statistically differ from the baseline wake state (feedback: t[141] = −0.35, P = 1, Figure 6E; feedforward: t[141] = −0.24, P = 1, Figure 6F) and was significantly attenuated when compared with nitrous oxide induction (feedback: t[141] = −4.126, P < .001, Figure 6E; feedforward: t[141] = −4.673, P < .001, Figure 6F). Correlations between frontoparietal connectivity in gamma bandwidths and acetylcholine levels in prefrontal and parietal cortices are reported in Supplemental Digital Content, Table 8, http://links.lww.com/AA/D757.
DISCUSSION
The principal findings in our study demonstrate that changes in temporospatial EEG complexity during subanesthetic ketamine or 50% nitrous oxide exposure correlate with concomitant changes in prefrontal and parietal acetylcholine levels. Of note, the changes observed in EEG complexity showed distinct drug-dependent temporal and spatial profiles. Subanesthetic ketamine produced increases in signal complexity and cortical acetylcholine that lasted throughout the ketamine infusion (62.5 minutes), with the largest complexity changes occurring in frontal, posterior parietal, and occipital channel clusters. Conversely, nitrous oxide increased EEG complexity and cortical acetylcholine only during the first 12.5 minutes of nitrous administration (induction), with the largest magnitude of change in frontotemporal and parietal regions. The next 50 minutes of nitrous oxide exposure (sedation) were marked by periodically quiescent behavior, gradual decline in prefrontal and parietal acetylcholine levels, and global suppression of EEG complexity that was most pronounced in frontal channel clusters. The topographic distribution of changes in EEG complexity during subanesthetic ketamine and nitrous oxide sedation in our study broadly mirrored recent magnetoencephalographic and EEG data published during ketamine and nitrous oxide administration in humans,12,28 suggesting a comparable dynamic landscape during the administration of dissociative anesthetics in rodents.
We have shown earlier that ketamine, in the absence of any behavioral arousal, can increase prefrontal acetylcholine.19 A previous study quantifying the effect of nitrous oxide on cortical acetylcholine showed similar effects on acetylcholine as reported by us in the current study, that is, a sharp increase after nitrous administration followed by a progressive decrease.20 Cortical acetylcholine is also known to correlate with behavioral arousal.17 Therefore, changes in cortical acetylcholine can result from a direct drug effect or changes in level of behavioral arousal. In the current study, it is difficult to ascribe the increase in cortical acetylcholine only to drug (ketamine/nitrous oxide) administration or only behavioral effects; it is likely that changes in cortical acetylcholine are a product of interaction between drug and behavioral effects. In contrast, increase in complexity is likely due to increased cortical acetylcholine or EEG activation, rather than behavioral arousal. This conclusion is supported by our recent studies in which we showed that a pharmacologically induced increase in prefrontal acetylcholine in sevoflurane-anesthetized rats was accompanied with EEG activation and an increase in complexity to baseline wake levels, with or without concomitant behavioral arousal.24,25
In addition to the relationship between neurophysiologic complexity and cortical acetylcholine, our study characterized changes in directed frontoparietal connectivity in low (25–55 Hz), mid (85–125 Hz), and high (125–175 Hz) gamma bandwidths. A previous study from our laboratory demonstrated that frontoparietal connectivity in high gamma bandwidth is a correlate of wakefulness, being reduced in conjunction with suppressed cortical levels of acetylcholine during general anesthesia.21 Our data extend these findings to dynamics characteristic of psychedelic states, demonstrating that periods of elevated cortical acetylcholine and neurophysiologic complexity during subanesthetic ketamine or nitrous oxide induction were correlated with increased high gamma frontoparietal connectivity. Conversely, low gamma frontoparietal connectivity was reduced when cortical acetylcholine levels were high and increased when acetylcholine levels were suppressed. Our data relating to cortical complexity and connectivity are broadly supportive of studies demonstrating hyperfrontality during classical psychedelic and dissociative drug administration, wherein excessive glutamatergic or cholinergic tone in frontal cortex accompanies aberrant cortical dynamics and alterations to the contents of consciousness.29–31
Reductions in measures of neurophysiologic complexity have been demonstrated across a broad range of pharmacologic and nonpharmacologic models of unconsciousness,6–10,24,32,33 while an increase in neurophysiologic complexity during psychedelic states has been shown to correlate with subjective intensity of altered conscious contents.12,13 Our finding of concomitant changes in cortical acetylcholine levels, neurophysiologic complexity, and high gamma frontoparietal connectivity during elevated and depressed states of consciousness is consistent with the purported role of acetylcholine in supporting cortical activation and arousal, expanding this relationship to dynamic neurophysiologic signatures related to the level of consciousness.3–5 Heterogeneous cell populations within the basal forebrain drive acetylcholine release at topographically specific targets across the cortex,14,34 with cortical cholinergic transmission shaping the activity of pyramidal and GABAergic (gamma aminobutyric acid) interneuron populations implicated in cortical activation and wakefulness.35–37 Acetylcholine release within the cortex is thought to create conditions that are permissive of high levels of neurophysiologic complexity, such as the reduction of slow cortical oscillations,38 thalamocortical desynchronization,39 and the promotion of high-frequency cortical gamma oscillations.19,35,40 Thus, while further testing of this hypothesis is required, cortical cholinergic transmission is a plausible neuromodulatory mechanism by which dynamics such as complexity can be shaped within cortical networks.
Although an increase in cortical acetylcholine levels during subanesthetic ketamine and nitrous oxide in rats has been reported, there are key differences as compared to our study. First, to our knowledge, none of the previous studies recorded simultaneous changes in EEG, which in the current study has allowed us to bridge neurochemical events in the cortex with concurrent changes in neurophysiological complexity and corticocortical connectivity. Second, while previous studies in rodents have reported changes in complexity across altered states of arousal,6,10,24 our use of high-density EEG allowed the application of temporospatial measures of complexity that more readily translate to those used in human studies.7,8,12 Third, because of our carefully titrated and timed drug administration, we were able to clearly characterize neurochemical and neurophysiological changes before, during, and after drug administration. Of note, although there are known sex differences in sensitivity and response to anesthetics, we did not find any overt behavioral differences between male and female rats. The lack of overt behavioral differences could be due to the graded nature of subanesthetic state that encompasses a wide range of behaviors, unlike anesthetic induction or emergence, which have binary end points (ie, awake or anesthetized).
There are limitations to our study. Given the low temporal resolution of microdialysis, our results should only be interpreted as they relate to sustained tonic acetylcholine release. Alternative approaches such as cholinergic biosensors may be better suited to investigate phasic cholinergic dynamics as they relate to neurophysiologic complexity at finer scales such as spikes or event-related potentials. Although cortical acetylcholine appears to vary with the level of consciousness, neurophysiologic complexity is likely to be governed by a complex neurochemical milieu, and thus, further research investigating the relationship between complexity and other neurotransmitter systems within the cortex is warranted. We did not study complexity and cortical acetylcholine at a higher ketamine concentration sufficient to produce sedation or anesthesia, which could have provided insightful comparisons with the changes in these outcome measures during the sedation phase produced by nitrous oxide. However, in a previous study, we did demonstrate that neurophysiologic complexity is reduced during the state of anesthesia induced by intraperitoneal ketamine, demonstrating a relationship between state and complexity.10 Interestingly, while subanesthetic ketamine in human volunteers increases neurophysiologic complexity,9,12 anesthetic doses of ketamine have been shown to produce an alternating pattern of high- and low-complexity states.9 Finally, our data are correlative, and thus, further studies establishing a causal relationship between cholinergic neurotransmission and complexity are necessary.
In conclusion, we demonstrate that changes in cortical acetylcholine are correlated with measures of EEG complexity and frontoparietal connectivity in high gamma bandwidths during subanesthetic ketamine and nitrous oxide. These findings expand our understanding of the relationship between neurochemical and neurophysiologic signatures purported to correlate with consciousness.
ACKNOWLEDGMENTS
We thank Chris Andrews PhD (CSCAR, the University of Michigan) for consultation and assistance with statistical analysis.
DICLOSURES
Name: Michael A. Brito, PhD.
Contribution: This author helped design the study, conduct the experiments, analyze and interpret the data, and write the manuscript.
Name: Duan Li, PhD.
Contribution: This author helped design the study, interpret the data, and write the manuscript.
Name: Christopher W. Fields, BS.
Contribution: This author helped conduct the experiments, analyze the data, and write the manuscript.
Name: Chloe Rybicki-Kler, BS.
Contribution: This author helped conduct the experiments, analyze the data, and write the manuscript.
Name: Jon G. Dean, PhD.
Contribution: This author helped conduct the experiments and write the manuscript.
Name: Tiecheng Liu, MD.
Contribution: This author helped conduct the experiments and write the manuscript.
Name: George A. Mashour, MD, PhD.
Contribution: This author helped design the study, analyze and interpret the data, and write the manuscript.
Name: Dinesh Pal, PhD.
Contribution: This author helped design the study, analyze and interpret the data, and write the manuscript.
This manuscript was handled by: Oluwaseun Johnson-Akeju, MD, MMSc.
Supplementary Material
FOOTNOTES
GLOSSARY
- ACh
- acetylcholine
- ARRIVE
- Animal Research: Reporting of In Vivo Experiments
- CW r
- cluster-weighted marginal correlation
- EEG
- electroencephalogram
- GABAergic
- gamma aminobutyric acid
- K
- subanesthetic ketamine infusion
- K
- subanesthetic ketamine infusion
- LZs
- Lempel-Ziv complexity
- LZsN
- normalized Lempel-Ziv complexity
- NI
- nitrous induction
- NMDA
- N-methyl-d-aspartate
- NS
- nitrous sedation
- NSTE
- normalized symbolic transfer entropy
- PK
- postketamine
- PN
- post-nitrous oxide recovery
- SD
- standard deviation
- W
- wake
Published ahead of print December 20, 2021.
Funding: This study was supported by the National Institutes of Health (grant no. R01GM111293), the National Science Foundation Graduate Research Fellowship (grant no. DGE 1256260), the Center for Consciousness Science, and the Department of Anesthesiology, University of Michigan.
The authors declare no conflicts of interest.
Supplemental digital content is available for this article. Direct URL citations appear in the printed text and are provided in the HTML and PDF versions of this article on the journal’s website (www.anesthesia-analgesia.org).
Reprints will not be available from the authors.
REFERENCES
- 1.Boly M, Massimini M, Tsuchiya N, Postle BR, Koch C, Tononi G. Are the neural correlates of consciousness in the front or in the back of the cerebral cortex? Clinical and neuroimaging evidence. J Neurosci. 2017;37:9603–9613. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Odegaard B, Knight RT, Lau H. Should a few null findings falsify prefrontal theories of conscious perception? J Neurosci. 2017;37:9593–9602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Tononi G, Boly M, Massimini M, Koch C. Integrated information theory: from consciousness to its physical substrate. Nat Rev Neurosci. 2016;17:450–461. [DOI] [PubMed] [Google Scholar]
- 4.Carhart-Harris RL. The entropic brain - revisited. Neuropharmacology. 2018;142:167–178. [DOI] [PubMed] [Google Scholar]
- 5.Casali AG, Gosseries O, Rosanova M, et al. A theoretically based index of consciousness independent of sensory processing and behavior. Sci Transl Med. 2013;5:198ra105. [DOI] [PubMed] [Google Scholar]
- 6.Abásolo D, Simons S, Morgado da Silva R, Tononi G, Vyazovskiy VV. Lempel-Ziv complexity of cortical activity during sleep and waking in rats. J Neurophysiol. 2015;113:2742–2752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Schartner M, Seth A, Noirhomme Q, et al. Complexity of multi-dimensional spontaneous EEG decreases during propofol induced general anaesthesia. PLoS One. 2015;10:e0133532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schartner MM, Pigorini A, Gibbs SA, et al. Global and local complexity of intracranial EEG decreases during NREM sleep. Neurosci Conscious. 2017;2017:niw022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Li D, Mashour GA. Cortical dynamics during psychedelic and anesthetized states induced by ketamine. Neuroimage. 2019;196:32–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Brito MA, Li D, Mashour GA, Pal D. State-dependent and bandwidth-specific effects of ketamine and propofol on electroencephalographic complexity in rats. Front Syst Neurosci. 2020;14:50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tagliazucchi E, Carhart-Harris R, Leech R, Nutt D, Chialvo DR. Enhanced repertoire of brain dynamical states during the psychedelic experience: enhanced repertoire of brain dynamical states. Hum Brain Mapp. 2014;35:5442–5456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Schartner MM, Carhart-Harris RL, Barrett AB, Seth AK, Muthukumaraswamy SD. Increased spontaneous MEG signal diversity for psychoactive doses of ketamine, LSD and psilocybin. Sci Rep. 2017;7:46421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Timmermann C, Roseman L, Schartner M, et al. Neural correlates of the DMT experience assessed with multivariate EEG. Sci Rep. 2019;9:16324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zaborszky L, Csordas A, Mosca K, et al. Neurons in the basal forebrain project to the cortex in a complex topographic organization that reflects corticocortical connectivity patterns: an experimental study based on retrograde tracing and 3D reconstruction. Cereb Cortex. 2015;25:118–137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Marrosu F, Portas C, Mascia MS, et al. Microdialysis measurement of cortical and hippocampal acetylcholine release during sleep-wake cycle in freely moving cats. Brain Res. 1995;671:329–332. [DOI] [PubMed] [Google Scholar]
- 16.Nelson CL, Burk JA, Bruno JP, Sarter M. Effects of acute and repeated systemic administration of ketamine on prefrontal acetylcholine release and sustained attention performance in rats. Psychopharmacology (Berl). 2002;161:168–179. [DOI] [PubMed] [Google Scholar]
- 17.Lydic R, Baghdoyan HA. Sleep, anesthesiology, and the neurobiology of arousal state control. Anesthesiology. 2005;103:1268–1295. [DOI] [PubMed] [Google Scholar]
- 18.Nair SG, Gudelsky GA. Activation of 5-HT2 receptors enhances the release of acetylcholine in the prefrontal cortex and hippocampus of the rat. Synapse. 2004;53:202–207. [DOI] [PubMed] [Google Scholar]
- 19.Pal D, Hambrecht-Wiedbusch VS, Silverstein BH, Mashour GA. Electroencephalographic coherence and cortical acetylcholine during ketamine-induced unconsciousness. Br J Anaesth. 2015;114:979–989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Shichino T, Murakawa M, Adachi T, Arai T, Miyazaki Y, Mori K. Effects of inhalation anaesthetics on the release of acetylcholine in the rat cerebral cortex in vivo. Br J Anaesth. 1998;80:365–370. [DOI] [PubMed] [Google Scholar]
- 21.Pal D, Silverstein BH, Lee H, Mashour GA. Neural correlates of wakefulness, sleep, and general anesthesia: an experimental study in rat. Anesthesiology. 2016;125:929–942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Lempel A, Ziv J. On the complexity of finite sequences. IEEE Trans Inf Theory. 1976;22:75–81. [Google Scholar]
- 23.Lee U, Ku S, Noh G, Baek S, Choi B, Mashour GA. Disruption of frontal-parietal communication by ketamine, propofol, and sevoflurane. Anesthesiology. 2013;118:1264–1275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Pal D, Li D, Dean JG, et al. Level of consciousness is dissociable from electroencephalographic measures of cortical connectivity, slow oscillations, and complexity. J Neurosci. 2020;40:605–618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Pal D, Dean JG, Liu T, et al. Differential role of prefrontal and parietal cortices in controlling level of consciousness. Curr Biol. 2018;28:2145–2152.e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.R Core Team. R: A Language and Environment for Statistical Computing. R Foundation for Statistical Computing; 2016. [Google Scholar]
- 27.Lorenz DJ, Datta S, Harkema SJ. Marginal association measures for clustered data. Stat Med. 2011;30:3181–3191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Vrijdag XCE, van Waart H, Mitchell SJ, Sleigh JW. An electroencephalogram metric of temporal complexity tracks psychometric impairment caused by low-dose nitrous oxide. Anesthesiology. 2021;134:202–218. [DOI] [PubMed] [Google Scholar]
- 29.Vollenweider FX, Leenders KL, Scharfetter C, et al. Metabolic hyperfrontality and psychopathology in the ketamine model of psychosis using positron emission tomography (PET) and [18F]fluorodeoxyglucose (FDG). Eur Neuropsychopharmacol. 1997;7:9–24. [DOI] [PubMed] [Google Scholar]
- 30.Abdallah CG, De Feyter HM, Averill LA, et al. The effects of ketamine on prefrontal glutamate neurotransmission in healthy and depressed subjects. Neuropsychopharmacology. 2018;43:2154–2160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mason NL, Kuypers KPC, Müller F, et al. Me, myself, bye: regional alterations in glutamate and the experience of ego dissolution with psilocybin. Neuropsychopharmacology. 2020;45:2003–2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hudetz AG, Liu X, Pillay S, Boly M, Tononi G. Propofol anesthesia reduces Lempel-Ziv complexity of spontaneous brain activity in rats. Neurosci Lett. 2016;628:132–135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bodart O, Gosseries O, Wannez S, et al. Measures of metabolism and complexity in the brain of patients with disorders of consciousness. Neuroimage Clin. 2017;14:354–362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Benarroch EE. Acetylcholine in the cerebral cortex: effects and clinical implications. Neurology. 2010;75:659–665. [DOI] [PubMed] [Google Scholar]
- 35.Liljenström H, Hasselmo ME. Cholinergic modulation of cortical oscillatory dynamics. J Neurophysiol. 1995;74:288–297. [DOI] [PubMed] [Google Scholar]
- 36.Poulet JFA, Crochet S. The cortical states of wakefulness. Front Syst Neurosci. 2019;12:64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yi F, Ball J, Stoll KE, et al. Direct excitation of parvalbumin-positive interneurons by M1 muscarinic acetylcholine receptors: roles in cellular excitability, inhibitory transmission and cognition. J Physiol. 2014;592:3463–3494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen L, Yin D, Wang TX, et al. Basal forebrain cholinergic neurons primarily contribute to inhibition of electroencephalogram delta activity, rather than inducing behavioral wakefulness in mice. Neuropsychopharmacology. 2016;41:2133–2146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Colangelo C, Shichkova P, Keller D, Markram H, Ramaswamy S. Cellular, synaptic and network effects of acetylcholine in the neocortex. Front Neural Circuits. 2019;13:24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Howe WM, Gritton HJ, Lusk NA, et al. Acetylcholine release in prefrontal cortex promotes gamma oscillations and theta–gamma coupling during cue detection. J Neurosci. 2017;37:3215–3230. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.