Abstract
Parasitic nematodes of the genus Anisakis are the causative agent of anisakiosis, an important fish‐borne zoonosis. Humans are infected through consumption of raw or undercooked fish, contaminated with the parasite. Infection can result in both gastrointestinal and allergic symptoms. There are few reports of anisakiosis in Portugal, but evidence of Anisakis allergy exists, indicating that exposure is occurring in the population. The European Food Risk Assessment Fellowship Programme (EU‐FORA) work programme, entitled: ‘Food safety of fish and zoonoses: fish consumption and microbiological risk assessment and perception, from fisherman to final consumers in Portugal’ was hosted by the Interdisciplinary Centre of Marine and Environmental Research (CIIMAR), in Porto, Portugal. It aimed to gather information on risk perception and attitudes in the Portuguese population to contamination of fish with Anisakis spp. and on their knowledge of methods to prevent infection. In addition, it aimed to examine the risk of anisakiosis in the Portuguese population.
Keywords: Anisakis, fish parasite, food safety, Zoonoses
1. Introduction
1.1. European Food Risk Assessment Fellowship Programme (EU‐FORA)
The European Food Risk Assessment Fellowship Programme (EU‐FORA) is an initiative of the European Food Safety Authority (EFSA). It aims to build the European Union’s (EU) scientific assessment capacity and knowledge community, in line with the key objectives outlined in EFSA’s strategy 2020. It provides scientists working in food safety organisations across Europe with the opportunity to increase their knowledge and gain experience in food risk assessment, taking a ‘learning by doing’ approach to training. (Bronzwaer et al., 2016). The fellow was hosted by the Interdisciplinary Centre of Marine and Environmental Research (CIIMAR), in Porto, Portugal. The work programme was entitled: Food safety of fish and zoonoses: fish consumption and microbiological risk assessment and perception, from fisherman to final consumers in Portugal. The programme included a 3‐week induction training course and four 1‐week modules, each focused on different aspects of risk assessment and risk communication. These modules would usually be held in different food safety institutes across the EU; however due to the Covid‐19 pandemic, they were conducted on an online platform.
1.2. Background to Risk Assessment
Each year, almost one in 10 people fall ill from eating contaminated food. The World Health Organization has estimated that food‐borne disease resulted in 420,000 deaths in 2010, with a global burden of 33 million Disability Adjusted Life Years (DALYs) (WHO, 2015). Food‐borne zoonoses from fish and shellfish products are of significant public health concern, with high numbers of people at risk worldwide in both developed and developing countries (Lima dos Santos and Howgate, 2011). Anisakiosis is a fish‐borne parasitic zoonosis caused by nematodes of the genus Anisakis, Dujardin 1845. These parasites have an indirect life cycle, using marine mammals, usually cetaceans as their definitive host. Invertebrates such as fish or squid are intermediate or paratenic hosts and crustaceans act as first intermediate hosts (Klimpel et al., 2004; EFSA BIOHAZ Panel, 2010; Smith and Wootten, 1978). Humans are infected with Anisakis spp. through consumption of the third‐stage larval (L3) form of the parasite in raw, smoked, marinated, salted or undercooked fish or squid (Audicana et al., 2002; Caldeira et al., 2021). They are accidental hosts for the parasite, so it does not mature, but on reaching the gastrointestinal tract, the larvae can cause disease (anisakiosis) (Mattiucci et al., 2018).
Van Thiel first described gastric anisakiosis in the 1960s (Van Thiel, 1962; Van Thiel and Van Houten, 1966; Buchmann and Mehrdana, 2016), the symptoms include nausea, vomiting and abdominal pain, and are caused by the larvae penetrating the gastrointestinal tract tissues (Caramello et al., 2003; Buchmann and Mehrdana, 2016). Patients can experience both abdominal and hypersensitivity symptoms and this condition is described as gastroallergic anisakiosis (Daschner et al., 2000). The other condition associated with Anisakis infection is an allergic response to fish products that contain parasite allergens. In these cases, live parasites may not be necessary to induce an allergic reaction, although it is generally believed that an initial Anisakis infection must occur to sensitise individuals to parasite antigens. However, it has not been possible to definitively rule out the occurrence of sensitisation through exposure to antigen alone (Audicana et al., 2002; EFSA BIOHAZ Panel, 2010).
There has been an increase in notifications of anisakiosis over the past few decades. This is probably due to increased inspection measures for fishery products, higher detection rates of contaminated seafood and of human infections due to improved detection and diagnostic techniques, along with the increasing popularity of raw or lightly cooked fish products (McCarthy and Moore, 2000; Chai et al., 2005; Audicana and Kennedy, 2008; Caldeira et al., 2021). The economic consequences of this parasite can be substantial, caused by increased rejection rates of contaminated products and decreased consumer confidence due to the negative aesthetic effects of visible nematode larvae in seafood products (D’amico et al., 2014; Llarena‐Reino et al., 2015). Anisakis was ranked fourth of 14 food‐borne parasites within the category of international trade importance in a report from the FAO/WHO (2014).
There are very few reported cases of anisakiosis in Portugal (Santos et al., 2022). The first case was reported in 2017 by Carmo et al. (2017). A case–control study by Falcão et al. (2008) found that 7% of the control group in the study population were sensitised to Anisakis simplex, while 22.5% of the patients in the case group, suffering from acute, relapsing urticaria, were sensitised. An earlier study by Nunes and Ladeira (2003) demonstrated that 8% of a population tested in a coastal region of Portugal were allergic to Anisakis spp. indicating that exposure to the parasite is occurring in the population. Portugal does not have a tradition of eating raw fish, but this may change with the global trend towards consumption of raw or lightly processed foods (McCarthy and Moore, 2000; Käferstein, 2003). Also, grilling is a common method of cooking fish and this does not always result in sufficiently high temperatures throughout the fish to inactivate larvae. Furthermore, dead Anisakis larvae may be allergenic to previously sensitised individuals. Hake gonads are a popular Portuguese dish and this area of the fish is frequently parasitised by Anisakis (Santos et al., 2022). The factors outlined above indicate that the Portuguese population is at risk of exposure to this parasite.
2. Description of work programme
2.1. Aims
The work programme had three main aims:
Use survey data to characterise the situation in Portugal with regard to the zoonotic fish parasite Anisakis. The survey gathered information on general fish consumption, raw fish consumption and on consumer risk perception and attitudes to contamination of fish with Anisakis spp. Also, on consumer knowledge of methods of preventing infection.
Examine the risk of anisakiosis in the Portuguese population from consumption of raw or undercooked fish.
Determine the prevalence of Anisakis spp. in European hake (Merluccius merluccius), captured in North‐East Atlantic Portuguese waters.
2.2. Activities/Methods
2.2.1. Assessing raw fish consumption trends and sociodemographic and health characteristics of raw fish consumers
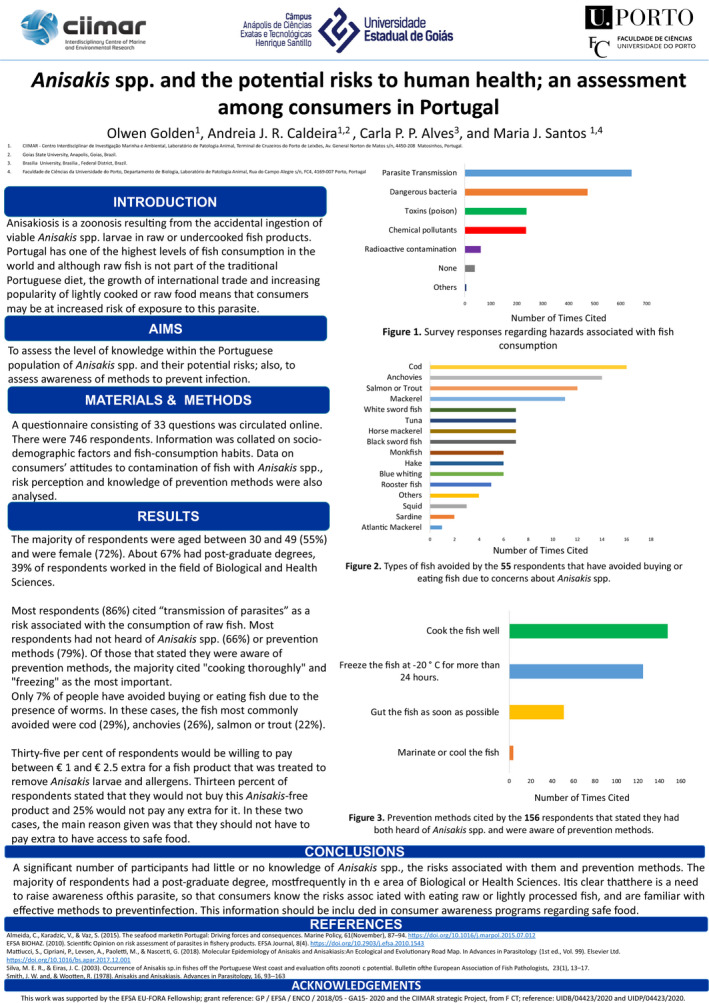
A survey had been carried out prior to the start of the work programme consisting of 33 questions that gathered information on demographics, general health, fish consumption habits for both cooked and raw/undercooked products and knowledge of Anisakis. Data from questions regarding raw fish consumption were analysed in conjunction with data from sociodemographic and health‐based questions, including those relating to allergy. This allowed analysis of the subset of respondents that consumed raw fish in relation to the type and volume of raw fish consumed, their sociodemographic profile, perceived health status and frequency of allergies.
2.2.2. Assessing consumer risk perception, awareness of Anisakis and ‘willingness to pay’ for Anisakis‐free fish
Survey respondents were asked to describe what hazards they associated with fish consumption, to indicate if they had any prior knowledge of Anisakis and if this had affected their purchasing habits in the past. They were also asked if they were aware of methods to prevent infection with this parasite. In the final section of the survey, respondents were provided with information on Anisakis and also presented with a hypothetical scenario regarding a treatment that could remove all parasites and parasite antigens from the fish without affecting the quality. They were then asked if they would be willing to pay extra for such a product and given options of price ranges to choose from, for those that were willing to pay extra. This scenario and question were included to facilitate a contingent valuation study as an additional analysis of the survey data. Contingent valuation (CV) studies are used to assess the monetary trade‐offs that consumers would be willing to make for a good or service (Carson, 2012). This approach was used by Bao et al. (2018) to ascertain the value that Spanish consumers would place on Anisakis‐free fish.
2.2.3. Examine the risk of anisakiosis in the Portuguese population from consumption of raw or undercooked fish
The initial plan for this part of the project had been to carry out a quantitative risk assessment regarding the risk of anisakiosis posed to the Portuguese population from consumption of a given species of fish, raw or undercooked. One of the objectives of assessing raw fish consumption trends (see Section 2.2.1) was to identify a suitable hazard vehicle for this analysis. However, following analysis of these data, no suitable hazard vehicle was identified. It was decided to focus on the five species most frequently eaten raw or undercooked in Portugal, identified by our survey, and examine the risks that they might pose to the population.
2.2.4. Determining the prevalence and distribution of Anisakis spp. larvae in a sample of European hake
2.2.4.1. UV‐Press analysis
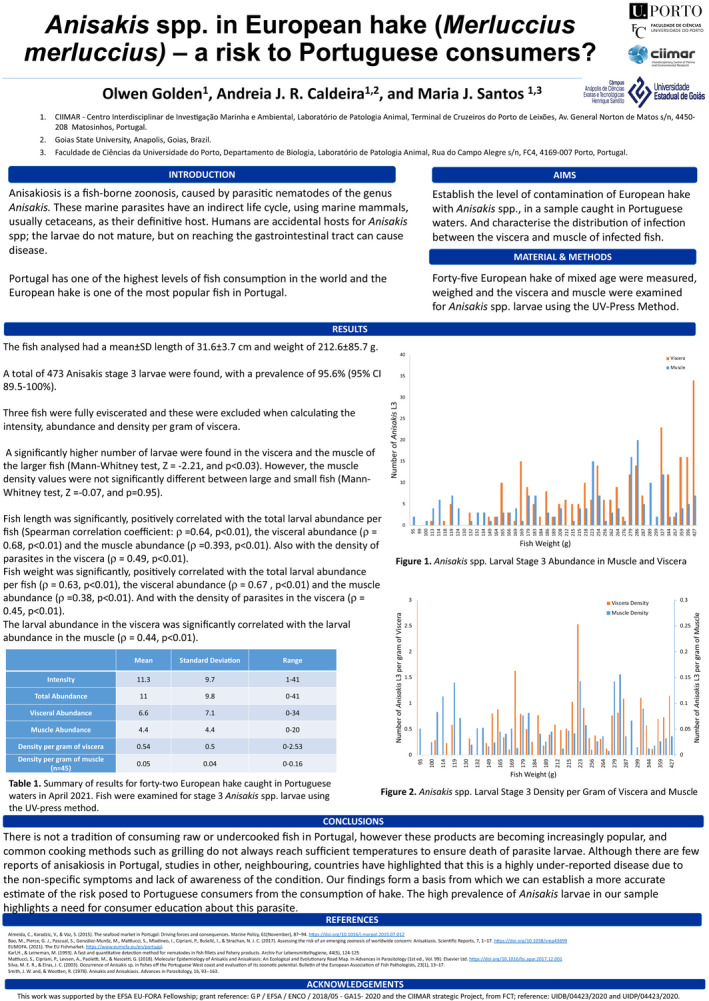
Forty‐five European hake were examined for Anisakis L3. The UV‐Press method was used to estimate the number of parasites in the muscle and viscera of the fish. This method was first described by Karl and Leinemann (1993). Anisakis larvae fluoresce under UV light after freezing, allowing them to be identified and their location marked on the plastic bag. The larvae were removed from the samples, washed in 0.9% saline solution and examined using a stereoscope to distinguish between Type I and Type II Anisakis L3.
This was the first time that the UV‐Press method had been used in the laboratory, so some work was required to optimise the method such as establishing the most suitable light source to obtain the best visualisation of the larvae.
2.2.4.2. Statistical analysis of UV‐Press results
From the findings of the UV‐Press analysis, the intensity and abundance of infection per fish were calculated, as defined by Bush et al. (1997), as well as the intensity and abundance of infection in the muscle and viscera of each fish. The density of infection in the muscle and viscera was also calculated (Bush et al., 1997). The Mann–Whitney U test was used to establish if measurements such as infection abundance differed significantly between heavier and lighter fish, and Spearman’s rank‐order correlation was used to evaluate some host–parasite correlations, such as fish weight and length with intensity and density of infection in the muscle and viscera. SPSS version 27 was used for this analysis.
2.2.5. Molecular analysis of Anisakis L3 obtained from a range of fish species
Anisakis L3 isolated from a sample of 16 fish of the subclass Elasmobranchii and 14 gurnards were selected for identification using conventional PCR. DNA was extracted and amplified by PCR, using primers for Internal Transcribed Spacer rDNA (ITS) (Gasser et al., 1993; Gasser and Hoste, 1995), Mitochondrial‐encoded cytochrome c oxidase subunit 2 (COX‐2) (Nadler and Hudspeth, 2000) and elongation factor 1 alpha 1 (Mattiucci et al., 2016). PCR products were analysed by electrophoresis on a 1% agarose gel and submitted for forward and reverse Sanger sequencing with an external company. The sequences obtained were analysed using MEGA software and compared with sequences deposited on GenBank.
3. Conclusions
The survey analysis provided the fellow with hands‐on training in data analysis and an understanding of how contingent valuation studies are conducted. It resulted in a data set with useful insights into the fish consumption habits of the Portuguese population, their knowledge and perception of risks associated with the parasite Anisakis and their awareness of methods of preventing infection.
Through working on the UV press method, the fellow also gained experience in introducing a new method to the host institute laboratory and this allowed collection of data regarding prevalence and distribution of Anisakis larvae in European hake from Portuguese waters. In addition, the fellow received training in molecular techniques to allow speciation of Anisakis larvae.
Preparation for the quantitative risk assessment that was planned, allowed the fellow to go through the process of gathering and organising data for such an assessment. When it became apparent from analysis of survey data, that no suitable hazard vehicles could be identified, the fellow worked on examining the risks of anisakiosis posed to the Portuguese population from the species of fish most commonly consumed raw in Portugal. This consolidated the knowledge of the topic already gained over the course of the programme, and provided an opportunity to acquire a broader understanding of this food safety issue, through a comprehensive review of the relevant literature.
The fellow also had the opportunity to attend regular on‐line seminars at the host institute, learning about a wide variety of research projects conducted there. The EU‐FORA modules provided training in a broad range of topics related to risk assessment, along with the opportunity to network with other scientists in this field.
Results of the fellow’s project were presented in two posters at the international conference of the European Association of Fish Pathologists 2021 (Annex A and B), and will be published in four articles in peer‐reviewed journals.
Abbreviations
- CIIMAR
Interdisciplinary Centre of Marine and Environmental Research
- CV
Contingent Valuation
- DALY
Disability Adjusted Life Years
- FAO
Food and Agriculture Organization
- L3
Stage‐three parasite larva
- PCR
Polymerase Chain Reaction
- QMRA
Quantitative Microbiological Risk Assessment
- UV
Ultraviolet
- WHO
World Health Organisation
Annex A – Poster on UV‐Press method results presented at the European Association of Fish Pathologists 20th international conference (virtual) on diseases of fish and shellfish 20–23 September 2021
Annex B – Poster on survey results presented at the European Association of Fish Pathologists 20th international conference (virtual) on diseases of fish and shellfish 20–23 September 2021
Suggested citation: Golden O, Caldeira AJR, Rangel LF and Santos MJ, 2022. Seafood safety and food‐borne zoonoses from fish. EFSA Journal 2022;20(S1):e200409, 10 pp. 10.2903/j.efsa.2022.e200409
Declarations of interest: The declarations of interest of all scientific experts active in EFSA’s work are available at https://ess.efsa.europa.eu/doi/doiweb/doisearch.
Acknowledgements: This report is funded by EFSA as part of the EU‐FORA programme. This research was supported by national funds through FCT – Foundation for Science and Technology within the scope of UIDB/04423/2020 and UIDP/04423/2020. This work was also funded by the project Ocean3R (NORTE‐01‐0145‐FEDER‐000064), supported by the North Portugal Regional Operational Programme (NORTE2020), under the Portugal 2020 Partnership Agreement and through the European Regional Development Fund (ERDF).
Approved: 31 January 2022
References
- Audicana MT, Ansotegui IJ, De Corres LF and Kennedy MW, 2002. Anisakis simplex: dangerous ‐ dead and alive? Trends in Parasitology, 18, 20–25. 10.1016/S1471-4922(01)02152-3 [DOI] [PubMed] [Google Scholar]
- Audicana MT and Kennedy MW, 2008. Anisakis simplex: from obscure infectious worm to inducer of immune hypersensitivity. Clinical Microbiology Reviews, 21, 360–379. 10.1128/CMR.00012-07 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bao M, Pierce GJ, Strachan NJC, Martínez C, Fernández R and Theodossiou I, 2018. Consumers’ attitudes and willingness to pay for Anisakis‐free fish in Spain. Fisheries Research, 202, 149–160. 10.1016/j.fishres.2017.06.018 [DOI]
- Bronzwaer S, Le Gourierec N and Koulouris S, 2016. Editorial: The European Food Risk Assessment Fellowship Programme (EU‐FORA). EFSA Journal 2016;14(11):e14111, 2 pp. 10.2903/j.efsa.2016.e14111 [DOI] [Google Scholar]
- Buchmann K and Mehrdana F, 2016. Effects of anisakid nematodes Anisakis simplex (s.l.), Pseudoterranova decipiens (s.l.) and Contracaecum osculatum (s.l.) on fish and consumer health. Food and Waterborne Parasitology, 4, 13–22. 10.1016/j.fawpar.2016.07.003 [DOI] [Google Scholar]
- Bush A, Lafferty K, Lotz J and Shostak A, 1997. Parasitology meets ecology on its own terms: Margolis et al revisited. Journal of Parasitology, 83, 575–583. Available online: https://www.ncbi.nlm.nih.gov/pubmed/9267395 [PubMed] [Google Scholar]
- Caldeira AJR, Alves CPP and Santos MJ, 2021. Anisakis notification in fish: an assessment of the cases reported in the European Union Rapid Alert System for Food and Feed (RASFF) database. Food Control, 124, 107913. 10.1016/j.foodcont.2021.107913 [DOI] [Google Scholar]
- Caramello P, Vitali A, Canta F, Caldana A, Santi F, Caputo A, Lipani F and Balbiano R, 2003. Intestinal localization of anisakiasis manifested as acute abdomen. Clinical Microbiology and Infection, 9, 734–737. 10.1046/j.1469-0691.2003.00660.x [DOI] [PubMed] [Google Scholar]
- Carmo J, Marques S, Bispo M and Serra D, 2017. Anisakiasis: a growing cause of abdominal pain! BMJ Case Reports, 2017, 2016–2017. 10.1136/bcr-2016-218857 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carson RT, 2012. Contingent valuation: a practical alternative when prices aren’t available. Journal of Economic Perspectives, 26, 27–42. 10.1257/jep.26.4.27 [DOI] [Google Scholar]
- Chai JY, Murrell KD and Lymbery AJ, 2005. Fish‐borne parasitic zoonoses: status and issues. International Journal for Parasitology, 35, 1233–1254. 10.1016/j.ijpara.2005.07.013 [DOI] [PubMed] [Google Scholar]
- D’amico P, Malandra R, Costanzo F, Castigliego L, Guidi A, Gianfaldoni D and Armani A, 2014. Evolution of the Anisakis risk management in the European and Italian context. Food Research International, 64, 348–362. 10.1016/j.foodres.2014.06.038 [DOI] [PubMed] [Google Scholar]
- Daschner A, Alonso‐Gómez A, Cabañas R, Suarez‐de‐Parga JM and López‐Serrano MC, 2000. Gastroallergic anisakiasis: borderline between food allergy and parasitic disease ‐ Clinical and allergologic evaluation of 20 patients with confirmed acute parasitism by Anisakis simplex. Journal of Allergy and Clinical Immunology, 105, 176–181. 10.1016/s0091-6749(00)90194-5 [DOI] [PubMed] [Google Scholar]
- EFSA BIOHAZ Panel , 2010. Scientific Opinion on risk assessment of parasites in fishery products. EFSA Journal 2010;8(4):1543, 45 pp. 10.2903/j.efsa.2010.1543 [DOI] [Google Scholar]
- Falcão H, Lunet N, Neves E, Iglésias I and Barros H, 2008. Anisakis simplex as a risk factor for relapsing acute urticaria: a case‐control study. Journal of Epidemiology and Community Health, 62, 634–637. 10.1136/jech.2007.061572 [DOI] [PubMed] [Google Scholar]
- FAO/WHO , 2014. Multicriteria‐based ranking for risk management of food‐borne parasites. In W. H. Organization (Ed.), Microbiological Risk Assessment Series, Food and Agriculture Organization of the United Nations/World Health Organization (Issue No.23). World Health Organization. Available online: https://www.who.int/iris/handle/10665/112672
- Gasser RB, Chilton NB, Hoste H and Beveridge I, 1993. Rapid sequencing of rDNA from single worms and eggs of parasitic helminths. Nucleic Acids Research, 21, 2525–2526. 10.1093/nar/21.10.2525 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gasser RB and Hoste H, 1995. Genetic markers for closely‐related parasitic nematodes. Molecular and Cellular Probes, 9, 315–319. 10.1016/S0890-8508(95)91588-5 [DOI] [PubMed] [Google Scholar]
- Käferstein FK, 2003. Actions to reverse the upward curve of foodborne illness. Food Control, 14, 101–109. 10.1016/S0956-7135(02)00017-8 [DOI] [Google Scholar]
- Karl H and Leineman M, 1993. A fast and quantitative detection method for nematodes in fish fillets and fishery products. Archiv Fur Lebensmittelhygiene, 44, 124–125. [Google Scholar]
- Klimpel S, Palm HW, Rückert S and Piatkowski U, 2004. The life cycle of Anisakis simplex in the Norwegian Deep (northern North Sea). Parasitology Research, 94, 1–9. 10.1007/s00436-004-1154-0 [DOI] [PubMed] [Google Scholar]
- Lima dos Santos CAM and Howgate P, 2011. Fishborne zoonotic parasites and aquaculture: a review. Aquaculture, 318, 253–261. 10.1016/j.aquaculture.2011.05.046 [DOI] [Google Scholar]
- Llarena‐Reino M, Abollo E, Regueira M, Rodríguez H and Pascual S, 2015. Horizon scanning for management of emerging parasitic infections in fishery products. Food Control, 49, 49–58. [Google Scholar]
- Mattiucci S, Acerra V, Paoletti M, Cipriani P, Levsen A, Webb SC, Canestrelli D and Nascetti G, 2016. No more time to stay ‘single’ in the detection of Anisakis pegreffii, A. simplex (s. s.) and hybridization events between them: a multi‐marker nuclear genotyping approach. Parasitology, 143, 998–1011. 10.1017/S0031182016000330 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mattiucci S, Cipriani P, Levsen A, Paoletti M and Nascetti G, 2018. Molecular Epidemiology of Anisakis and Anisakiasis: An Ecological and Evolutionary Road Map. In Advances in Parasitology (1st ed., Vol. 99). Elsevier Ltd. 10.1016/bs.apar.2017.12.001 [DOI] [PubMed] [Google Scholar]
- McCarthy J and Moore TA, 2000. Emerging helminth zoonoses. International Journal for Parasitology, 30, 1351–1359. 10.1016/S0020-7519(00)00122-3 [DOI] [PubMed] [Google Scholar]
- Nadler SA and Hudspeth DSS, 2000. Phylogeny of the ascaridoidea (Nematoda: Ascaridida) based on three genes and morphology: hypotheses of structural and sequence evolution. Journal of Parasitology, 86, 380–393. 10.1645/0022-3395(2000)086[;0380:POTANA];2.0.CO;2 [DOI] [PubMed] [Google Scholar]
- Nunes C and Ladeira S, 2003. Allergy to Anisakis simplex in the Portuguese Population. Journal of Allergy and Clinical Immunology, 111, 255. [Google Scholar]
- Santos MJ, Matos M, Guardone L, Golden O, Armani A, Caldeira AJR and Vieira‐pinto M, 2022. Preliminary Data on the Occurrence of Anisakis spp. in European Hake (Merluccius merluccius) Caught off the Portuguese Coast and on Reports of Human Anisakiosis in Portugal. Microorganisms, 10, 331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith JW and Wootten R, 1978. Anisakis and Anisakiasis. Advances in Parasitology, 16, 93–163. [DOI] [PubMed] [Google Scholar]
- Van Thiel PH, 1962. Anisakiasis. [Abstract]. Parasitology, 52, 16P–17P. [Google Scholar]
- Van Thiel PH and Van Houten H, 1966. The Herring Worm Anisakis marina as a Human Parasite outside the Wall of the Gastro‐Intestinal Tract. [Abstract]. Nederlands Tijdschrift Voor Geneeskunde, 110, 1524–1528. Available online: https://www.cabdirect.org/cabdirect/abstract/19672900175 [PubMed] [Google Scholar]
- WHO , 2015. WHO estimates of the global burden of foodborne diseases. In World Health Organisation. 10.1007/978-3-642-27769-6_3884-1 [DOI] [Google Scholar]


