Abstract
The present study aimed to investigate whether prucalopride, as a 5-hydroxytryptamine 4 (5-HT4) receptor agonist, improved intestinal motility by promoting the regeneration of the enteric nervous system (ENS) in rats with diabetes mellitus (DM). A rat model of DM was established using an intraperitoneal injection of streptozotocin. The rats were randomly divided into four groups of 6 rats/group: Control, DM (DM model), DM + A (5 µg/kg prucalopride) and DM + B (10 µg/kg prucalopride). The rats in the Control group were given an equal volume of citric acid solvent. After successful model establishment, high blood glucose levels were maintained for 2 weeks before administration of prucalopride. The colonic transit time was measured using the glass bead discharge method. It was revealed that the colonic transit time of diabetic rats was the longest, and this was significantly shortened in the DM + B group. Subsequently, the colons were collected. The expression levels of Nestin, glial fibrillary acidic protein (GFAP), SOX10, RNA-binding protein human antigen D (HuD) and ubiquitin thiolesterase (PGP9.5) were determined via immunohistochemical analysis. Immunofluorescence double staining of 5-HT4 + Nestin and Ki67 + Nestin was performed. The 5-HT level was measured using ELISA. Compared with that in the control group, Nestin expression was significantly increased in the DM and DM + A groups, and it was concentrated in columnar epithelial cells and the mesenchyme. Furthermore, the expression levels of Nestin in the DM + A group were higher than those in the DM group. No difference was observed in the expression levels of Nestin between the DM + B group and the Control group. The expression levels of 5-HT protein were highest in the Control group; however, the expression levels of 5-HT protein in the DM group, DM + A group and DM + B group exhibited an increasing trend. Similar trends in the expression of 5-HT4 and Nestin were not observed; however, similar trends in the expression of Nestin and Ki67 were observed. The expression levels of GFAP, SOX10, PGP9.5 and Ki67 in the DM + A and DM + B groups were higher compared with those in the DM group. In the DM + A group, HuD expression was decreased compared with that in the Control group but it was markedly higher compared with that in the DM group. In conclusion, prucalopride may improve intestinal motility by promoting ENS regeneration in rats with DM.
Keywords: prucalopride, intestinal motility, diabetes mellitus, enteric nervous system, 5-hydroxytryptamine
Introduction
Diabetes mellitus (DM) is a widespread metabolic disease, which is accompanied by a high incidence based on statistics on individuals with diabetes in 200 countries and regions between 1980 and 2014 (1). Improper treatment will cause the body to be in a high-sugar state for a long period and can induce various complications (2). Intestinal disease is a common complication, manifested as diarrhea, constipation and fecal incontinence, which seriously affects the quality of life of patients (3,4). Constipation is the most common symptom of diabetic bowel syndrome, manifested as colonic dyskinesia and slow transportation, leading to hard stools and defecation disorders (5,6). DM can affect most parts of the gastrointestinal tract; however, to the best of our knowledge, the specific pathogenic mechanisms are yet not fully elucidated (7). Previous studies have reported that intestinal dyskinesia is closely associated with intestinal autonomic neuropathy, including enteric nervous system (ENS) disorders (8), neuron damage and reduction (9-11) and a loss of interstitial cells of Cajal (12,13).
The ENS is an endogenous nervous system, including gastrointestinal motility and sensory systems, which consists of the submucosal and intermuscular nerve plexuses (14). The ENS is distributed in the gastrointestinal tract in a network, and it receives and integrates autonomic nerve signals, releases neurotransmitters, maintains gastrointestinal movement and regulates intestinal epithelial cell secretion (15). ENS disease can cause colonic dysfunction and DM can cause pathological changes in the ENS (8,16). Therefore, patients with DM often have certain symptoms, such as constipation, diarrhea or alternation of constipation and diarrhea (17). Enteric neural stem cells in the ENS possess regeneration and repair functions (18,19). Except for the embryonic period, this regenerative function of the adult intestine occurs only when the ENS is damaged (20-22). Previous studies have revealed that enteric neural stem cells are also present in adult intestinal tissues (23-25), and these cells may be the source of ENS regeneration (26).
As the most common diabetic bowel syndrome, constipation is mainly treated with drugs; however, diet and exercise can also exert an auxiliary treatment effect (27). Osmotic and irritant laxatives are the two most commonly prescribed drugs (28,29). Although these can increase the frequency of bowel movements, the treatment effect remains unsatisfactory (30). Furthermore, 20-40% of individuals will experience adverse side effects, such as bloating (30). Initially, 5-hydroxytryptamine 4 (5-HT4) receptor agonists cisapride and tegaserod were used for the treatment of functional bowel disease; however, these were withdrawn from the market due to their damaging effects on cardiovascular function (31-33). 5-HT is a neurotransmitter expressed in the central and peripheral nervous systems, and it can regulate gastrointestinal motility and sensory functions (34). As the fourth subtype of 5-HT, 5-HT4 can regulate intestinal movement and reduce visceral sensitivity (30). Furthermore, 5-HT4 receptors serve an important role in the growth and function maintenance of enteric nerves (35,36).
As a selective and high-affinity 5-HT4 receptor agonist, prucalopride can activate the 5-HT4 receptor, enhance choline secretion, promote intestinal contraction and promote gastrointestinal motility (37-39). Prucalopride has been used to treat diabetic constipation and functional gastrointestinal diseases (37,38,40,41), and it is mainly used for the treatment of chronic constipation in women (37,38). Prucalopride was approved by the European Medicines Agency for clinical use in 2009 and was marketed in Germany and the UK in 2010 (42). Procabulide has few side effects in the treatment of chronic constipation, and exerts almost no harm to the cardiovascular system, and thus, it can also be used by elderly individuals with cardiovascular diseases (43-46). At present, most of the research examining this drug is focused on its clinical efficacy, and there are few investigations on the mechanism underlying the effects of prucalopride on improving constipation caused by DM (43-46). Understanding the mechanism via which prucalopride improves colon changes caused by DM is of great significance for optimizing treatment plans and identifying novel treatment methods.
Therefore, the present study established a rat model of DM and the rats were treated with prucalopride. The present study aimed to investigate the alterations of enteric neural stem cells and neurons by examining the expression levels of the markers of neural stem cells, neurons and glial cells, as well as measuring 5-HT4 expression, in order to determine whether prucalopride, as a 5-HT4 receptor agonist, improved intestinal motility by promoting ENS regeneration in diabetic rats.
Materials and methods
Materials
A cyanine 3 (Cy3)-conjugated goat anti-rabbit IgG antibody (cat. no. BA1032) was obtained from Wuhan Boster Biological Technology, Ltd. Cy3-conjugated goat anti-mouse IgG antibody (1:200; cat. no. A10521) and FITC-conjugated goat anti-mouse IgG antibody (1:200; cat. no. 62-6511) were purchased from Thermo Fisher Scientific, Inc. The FITC-conjugated goat anti-rabbit IgG antibody (1:100; cat. no. CW0114S) and diaminobenzidine (DAB) kit (cat. no. CW0125) were purchased from CoWin Biosciences. HRP-conjugated goat anti-rabbit IgG(H+L) antibody (1:250; cat. no. ZB-2301) was obtained from OriGene Technologies, Inc. Mouse anti-Ki-67 monoclonal antibody (1:250; cat. no. bsm-33070M) and rabbit anti-SOX10 polyclonal antibody (1:200; cat. no. bs-20563R) were purchased from BIOSS. Prucalopride was purchased from Janssen-Cilag SpA. Rabbit anti-5-HT4 polyclonal antibody (1:100; cat. no. DF3503) and rabbit anti-ubiquitin thiolesterase (PGP9.5) polyclonal antibody (1:200; cat. no. AF0243) were obtained from Affinity Biosciences. Rabbit anti-RNA-binding protein human antigen D (HuD)+ human antigen C monoclonal antibody (1:500; cat. no. ab184267) and rabbit anti-glial fibrillary acidic protein (GFAP) monoclonal antibody (1:250; cat. no. ab33922) were purchased from Abcam. Rabbit anti-Nestin polyclonal antibody (1:200; cat. no. OM264981) was obtained from Omnimabs. A rat 5-HT ELISA kit (cat. no. MM-0442R2) was obtained from Jiangsu Enzyme Industry Co., Ltd. Glass beads with a 3-mm diameter were purchased from Ziboshi Boshan Gaoqiangdu Weizhuchang.
Animals
A total of 24 male Sprague Dawley rats (6-8 weeks old; weight, 250±20 g) were provided by Hunan SJA Laboratory Animal Co., Ltd. All rats were housed in a room at 20-25°C with 55-70% relative humidity and natural light. The rats had free access to food and water. The study protocol was approved by the Ethics Committee of The First Clinical Medical College of Nanjing Medical University (Nanjing, China).
Establishment of the diabetic rat model
Sprague Dawley rats were randomly divided into four groups of 6 rats each: Control group (Control), the DM model group (DM), the 5 µg/kg prucalopride group (DM + A) and the 10 µg/kg prucalopride group (DM + B). After fasting for 12 h, the rats were given a single intraperitoneal injection of 1% streptozotocin in citrate buffer (cat. no. MB1227; Dalian Meilun Biotechnology Co., Ltd.) at a dose of 60 mg/kg. The rats in the Control group were given an equal volume of citric acid solvent via intraperitoneal injection. After 1 week, blood was collected through the tail vein and the blood glucose concentration was immediately measured using a blood glucose meter. The criterion for successful establishment of the DM model was a blood glucose level ≥16.7 mmol/l (47). After successful model establishment, high blood glucose levels were maintained for 2 weeks before administration of prucalopride. Prucalopride was dissolved in normal saline to a final concentration of 0.5 µg/ml and then administered via gavage daily for another 2 weeks. Finally, the rats were weighed.
Colonic transit time measurement
The colonic transit time was measured using the glass bead discharge method (48). After the rats were anesthetized by isoflurane inhalation at induction and maintenance doses of 5 and 2%, respectively, in an induction box, glass beads with 3-mm diameter were placed at a depth of 2 cm from the anus, so that the glass beads stayed in the colon. The rats implanted with the glass beads had free access to food and water. The time required for the rats to discharge the glass beads was recorded as the colonic transit time.
The rats were euthanized by intraperitoneal injection with 150 mg/kg pentobarbital sodium. The death of rats was determined by observing whether the rats were breathing and whether the pupils were dilated. If the breathing stopped and the pupils were dilated, it could be judged that the rats had died. The colons were collected and stored at −80°C for subsequent experiments. Both frozen and paraffin-embedded samples were prepared.
Immunohistochemical analysis
The colons were fixed in 4% paraformaldehyde for 1 h at 4°C. Then paraffin-embedded sections of colons were prepared, deparaffinated in xylene twice for 10 min each, hydrated in 100, 100, 95 and 80% ethanol and water for 5 min each, incubated in citrate buffer and heated at 115°C for 2 min in a pressure cooker. After cooling, they were washed with phosphate buffered saline, incubated in fresh 3% hydrogen peroxide at room temperature for 10 min and rinsed. Subsequently, the sections (4-µm-thick) were incubated in 5% BSA (cat. no. SW3015; Beijing Solarbio Science & Technology Co., Ltd.) at 37°C for 30 min and in primary antibody buffer at 4°C overnight. Following maintenance at room temperature for 45 min, the sections were washed and incubated in secondary antibody buffer at 37°C for 30 min. Sections were rinsed, stained in DAB at room temperature for 5-10 min, incubated in hematoxylin at room temperature for 3 min, differentiated in hydrochloric alcohol and stained blue in ammonia water at room temperature for 5-10 min. Subsequently, sections were washed with water for 1 min, dehydrated, transparentized, mounted and examined under a fluorescence microscope. Immunohistochemical images were analyzed semi-quantitatively using Image-Pro Plus software (version 6.0; Media Cybernetics, Inc.). For each sample, three visual fields were analyzed and a magnification of ×200 was used. The ratios of the integrated optical density of the selected area to the color area are presented as the semi-quantitative results.
Immunofluorescence double staining
Frozen colonic sections (-20°C; 5-µm-thick) were washed, immersed in 0.5% Triton X-100 at room temperature for 20 min and incubated in 5% BSA at 37°C for 30 min. Sections were then incubated in primary antibody buffer at 4°C overnight, washed and incubated in secondary antibody buffer at 37°C for 30 min. After washing, sections were incubated with DAPI in the dark at room temperature for 5 min, washed, mounted and examined under a fluorescence microscope (CKX53; Olympus Corporation). Immunofluorescence images were analyzed semi-quantitatively using Image-Pro Plus software (version 6.0; Media Cybernetics, Inc.). For each sample, three visual fields were analyzed and a magnification of ×200 was used. The ratios of the integrated optical density of the selected area to the color area are presented as the semi-quantitative results.
ELISA
The colons were ground and rewarmed. The levels of 5-HT were detected using a 5-HT ELISA kit according to the manufacturer's protocol. Absorbance was measured at 450 nm.
Statistical analysis
Every experiment was repeated three times. Data are presented as the mean ± SD. Statistical analysis was performed using one-way ANOVA followed by Tukey post hoc test using SPSS software (v19.0; IBM Corp.). P<0.05 was considered to indicate a statistically significant difference.
Results
Successful establishment of the DM model
The blood glucose levels of the rats at 1 and 3 weeks after model establishment are presented in Table I. The blood glucose levels of the control rats were <16.7 mmol/l and the blood glucose levels of all diabetic rats were >16.7 mmol/l, suggesting the successful establishment of the DM model.
Table I.
Blood glucose levels of the rats at 1 and 3 weeks after model establishment.
| Groups | Rat no. | Blood sugar level at 1 week, mmol/l | Blood sugar level at 3 weeks, mmol/la |
|---|---|---|---|
| Control | 1 | 7.2 | 5.4 |
| 2 | 7.9 | 6.8 | |
| 3 | 5.8 | 6.0 | |
| 4 | 6.0 | 7.9 | |
| 5 | 6.7 | 8.2 | |
| 6 | 6.4 | 5.8 | |
| DM | 7 | 25.3 | 33.2 |
| 8 | 22.7 | 33.2 | |
| 9 | 21.6 | 33.1 | |
| 10 | 22.6 | HI | |
| 11 | 23.6 | 32.3 | |
| 12 | 20.8 | HI | |
| DM + A | 13 | 20.0 | HI |
| 14 | 25.0 | HI | |
| 15 | 21.0 | HI | |
| 16 | 27.1 | HI | |
| 17 | 26.0 | 27.5 | |
| 18 | 25.6 | 29.4 | |
| DM + B | 19 | 20.7 | HI |
| 20 | 28.7 | HI | |
| 21 | 23.7 | HI | |
| 22 | 29.6 | HI | |
| 23 | 24.9 | HI | |
| 24 | 26.9 | 31.1 |
HI indicates that the level was too high and exceeded the measurement range. DM, diabetes mellitus model; DM + A, diabetic rats treated with 5 µg/kg prucalopride; DM + B, diabetic rats treated with 10 µg/kg prucalopride.
Prucalopride shortens the colonic transit time
The colonic transit time of rats in various groups is shown in Fig. 1. Compared with that of rats in the Control group, the colonic transit time of rats in the DM group was significantly prolonged (P<0.05). Furthermore, compared with the DM group, rats treated with 5 µg/kg prucalopride (DM + A group) had a shorter colonic transit time, although the difference was not significant. However, after treatment with 10 µg/kg prucalopride (DM + B group), rats had a significantly shorter colonic transit time compared with that of rats in the DM group (P<0.05). Furthermore, the colonic transit time of rats in the DM + B group was close to that observed in the Control group, indicating that prucalopride treatment could accelerate colonic movement and shorten the colonic transit time.
Figure 1.
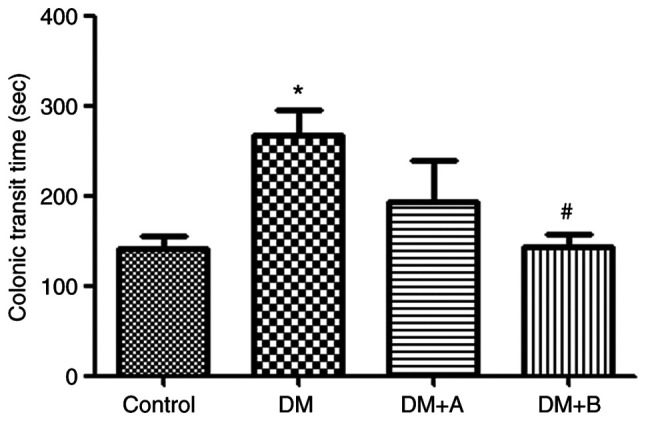
Colonic transit time of rats in the Control, DM, DM + A and DM + B groups. A rat model of DM was established using an intraperitoneal injection of streptozotocin. The rats in the Control group were given an equal volume of citric acid solvent. *P<0.05 vs. Control; #P<0.05 vs. DM. DM, diabetes mellitus model; DM + A, diabetic rats treated with 5 µg/kg prucalopride; DM + B, diabetic rats treated with 10 µg/kg prucalopride.
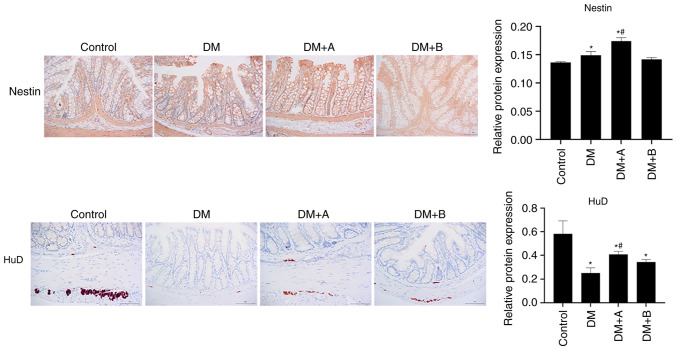
Prucalopride promotes the regeneration of colonic neural stem cells and neurons
Nestin is a marker of enteric neural stem cells in the ENS (24), whereas HuD is a marker of neurons (49). The expression levels of Nestin and HuD in various groups as determined by immunohistochemical analysis are shown in Fig. 2. Nuclei and Nestin/HuD were labeled as blue and brown, respectively. Nestin was mainly expressed in the muscularis mucosae, circular muscles and columnar epithelial cells. Compared with that in the Control group, Nestin expression was significantly increased in the DM and DM + A groups, and it was concentrated in columnar epithelial cells and the mesenchyme. The expression levels of Nestin in the DM + A group were higher than those in the DM group. There was no significant difference in the expression levels of Nestin between the DM + B group and the Control group. In the Control group, HuD was mainly expressed in the junction of the longitudinal and circular muscles of the colon, which is an area where neurons are enriched (50). Compared with the Control group, HuD expression was decreased in the longitudinal muscle and very little was observed in the submucosa in the DM group. In the DM + A group, HuD appeared in the longitudinal muscle and submucosa. Furthermore, its expression was decreased compared with that in the Control group but was markedly higher compared with that in the DM group. In the DM + B group, HuD expression was observed in the longitudinal muscle and a small amount was expressed in the submucosa. These results suggested that the ENS function of diabetic rats was impaired and that 5 µg/kg prucalopride may promote the regeneration of colonic neural stem cells and neurons.
Figure 2.
Nestin and HuD expression in the Control, DM, DM + A and DM + B groups, as determined via immunohistochemical analysis. Nuclei and Nestin/HuD were labeled as blue and brown, respectively. A rat model of DM was established using an intraperitoneal injection of streptozotocin. The rats in the Control group were given an equal volume of citric acid solvent. Magnification, ×200. *P<0.05 vs. Control; #P<0.05 vs. DM. DM, diabetes mellitus model; DM + A, diabetic rats treated with 5 µg/kg prucalopride; DM + B, diabetic rats treated with 10 µg/kg prucalopride; HuD, RNA-binding protein human antigen D.
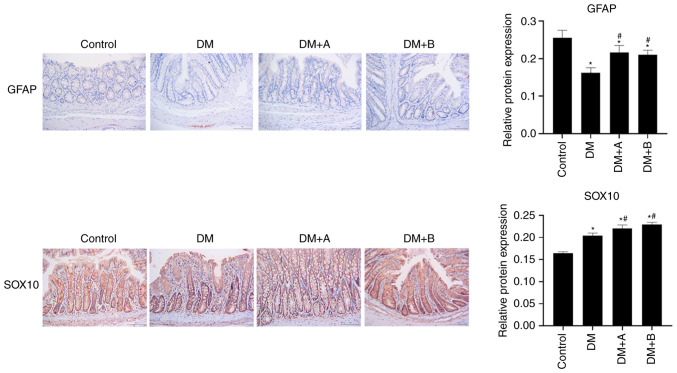
Prucalopride promotes the differentiation of colonic neural stem cells
GFAP and SOX10 are markers of glial cells, which are differentiated from neural stem cells (51,52). The expression levels of GFAP and SOX10 in various groups as determined by immunohistochemical analysis are shown in Fig. 3. Nuclei and GFAP/SOX10 were labeled as blue and brown, respectively. GFAP was lowly expressed in each group, and mainly found in the longitudinal muscle and columnar epithelial cells. Compared with the Control group, the DM, DM + A and DM + B groups exhibited decreased expression levels of GFAP. However, GFAP expression in the DM + A and DM + B groups was higher compared with that in the DM group.
Figure 3.
Expression levels of GFAP and SOX10 in the Control, DM, DM + A and DM + B groups, as determined via immunohistochemical analysis. Nuclei and GFAP/SOX10 were labeled as blue and brown, respectively. A rat model of DM was established using an intraperitoneal injection of streptozotocin. The rats in the Control group were given an equal volume of citric acid solvent. Magnification, ×200. *P<0.05 vs. Control; #P<0.05 vs. DM. DM, diabetes mellitus model; DM + A, diabetic rats treated with 5 µg/kg prucalopride; DM + B, diabetic rats treated with 10 µg/kg prucalopride; GFAP, glial fibrillary acidic protein.
In the Control group, SOX10 was abundantly expressed in the columnar epithelial cells, the nuclei of lamina propria cells and enteraden, and it was slightly expressed in the nucleus of the submucosa of the circular muscle. In the DM group, SOX10 was highly expressed in the columnar epithelial cells and its expression was significantly increased compared with that in the Control group. In the DM + A group, SOX10 expression was significantly higher compared with that in the DM group, and it was mainly expressed in the columnar epithelial nuclei and enteraden. In the DM + B group, SOX10 was expressed in small amounts in the circular muscle; however, it was highly expressed in the columnar epithelial cells, the nuclei of lamina propria cells and enteraden. Furthermore, SOX10 expression in the DM + B group was higher compared with that in the DM group. The results of SOX10 and GFAP expression analysis indicated that prucalopride could promote the differentiation of colonic neural stem cells, activate the expression of glial proteins and promote the recovery of neuronal injury to a certain extent.
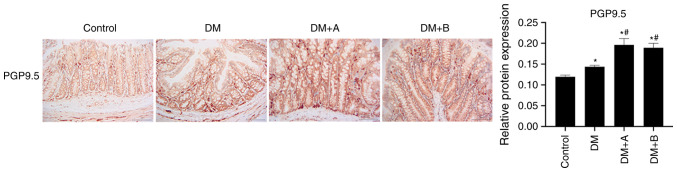
Prucalopride promotes the differentiation of the colonic nerve node
PGP9.5 is mainly expressed in differentiated nerve nodes, and the faster the differentiation, the higher the expression levels of PGP9.5 (53,54). Therefore, PGP9.5 is a marker of colonic nerve node differentiation (55). The expression levels of PGP9.5 in various groups as determined using immunohistochemical analysis are presented in Fig. 4. Nuclei and PGP9.5 were labeled as blue and brown, respectively. PGP9.5 was generally expressed in columnar epithelial cells, lamina propria, enteraden and circular muscles, and a small amount was expressed in the submucosa. The expression levels of PGP9.5 in the DM group were markedly higher compared with those in the Control group. In both the DM + A group and the DM + B group, the expression levels of PGP9.5 were significantly increased compared with those in the Control and DM groups. The results of PGP9.5 expression analysis demonstrated that prucalopride was beneficial for the differentiation of the colonic nerve node.
Figure 4.
PGP9.5 expression in the Control, DM, DM + A and DM + B groups, as determined via immunohistochemical analysis. Nuclei and PGP9.5 were labeled as blue and brown, respectively. A rat model of DM was established using an intraperitoneal injection of streptozotocin. The rats in the Control group were given an equal volume of citric acid solvent. Magnification, ×200. *P<0.05 vs. Control; #P<0.05 vs. DM. DM, diabetes mellitus model; DM + A, diabetic rats treated with 5 µg/kg prucalopride; DM + B, diabetic rats treated with 10 µg/kg prucalopride; PGP9.5, ubiquitin thiolesterase.
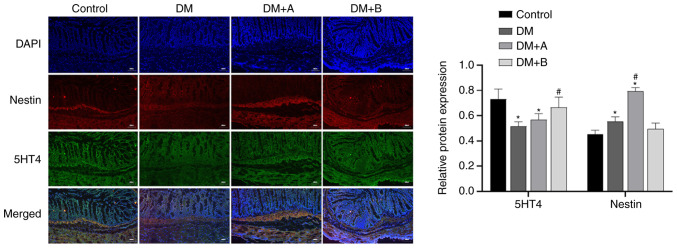
Prucalopride increases the secretion of 5-HT4 in the colon
Immunofluorescence double staining images of Nestin and 5-HT4 in the Control, DM, DM + A and DM + B groups are shown in Fig. 5. DAPI-stained nuclei appeared blue, while Nestin and 5-HT4 are indicated by red fluorescence and green fluorescence, respectively. Nestin was mainly distributed in the cytoplasm of muscularis mucosae and circular muscles, while 5-HT4 was expressed in interstitium, muscularis mucosae, mucosal layers and circular muscles. The main areas in which Nestin and 5-HT4 were co-expressed included the muscularis mucosae and circular muscles. Small amounts of these proteins were expressed in the intercellular substance. Compared with those in the Control group, the expression levels of 5-HT4 in the DM and DM + A groups were significantly decreased, and the expression levels of Nestin in the DM and DM + A groups were significantly increased. Compared with those in the DM group, the expression levels of Nestin in the DM + A group were significantly increased, and the expression levels of 5-HT4 in the DM + B group were significantly increased. Similar trends in the expression of 5-HT4 and Nestin were not observed. These results suggested that prucalopride promoted the secretion of 5-HT4 in the colon.
Figure 5.
Immunofluorescence double staining images of Nestin and 5-HT4 in the Control, DM, DM + A and DM + B groups. DAPI-stained nuclei appeared blue. Nestin and 5-HT4 are indicated by red fluorescence and green fluorescence, respectively. A rat model of DM was established using an intraperitoneal injection of streptozotocin. The rats in the Control group were given an equal volume of citric acid solvent. Magnification, ×100. *P<0.05 vs. Control; #P<0.05 vs. DM. DM, diabetes mellitus model; DM + A, diabetic rats treated with 5 µg/kg prucalopride; DM + B, diabetic rats treated with 10 µg/kg prucalopride; 5-HT4, 5-hydroxytryptamine 4.
Prucalopride enhances the regeneration of colonic neural stem cells
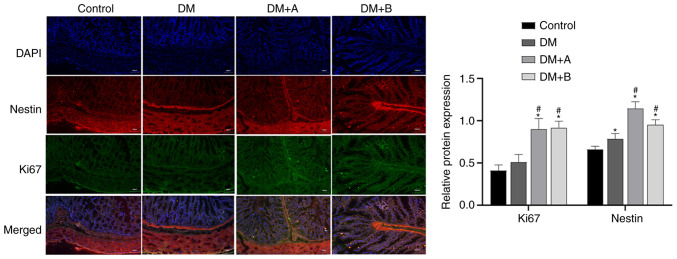
Ki67 is a marker of the cell proliferation rate, and the faster the cell proliferation rate, the higher its expression levels (56). Immunofluorescence double staining images of Nestin and Ki67 in the Control, DM, DM + A and DM + B groups are shown in Fig. 6. DAPI-stained nuclei appeared blue. Nestin and Ki67 are indicated by red fluorescence and green fluorescence, respectively. In the Control group, Nestin was mainly distributed in the cytoplasm of muscularis mucosae and circular muscles, and a small amount was expressed in the intercellular substance. Ki67 was mainly expressed in the intercellular substance, muscularis mucosae and circular muscles. The main areas in which Nestin and Ki67 were co-expressed included the muscularis mucosae and circular muscles. Compared with those in the Control group, the expression levels of Nestin in the DM group were significantly increased. The expression levels of Ki67 and Nestin in the DM + A and DM + B groups were markedly higher compared with those in the DM group. Furthermore, similar trends in the expression of Ki67 and Nestin were observed. These results indicated that prucalopride could promote the regeneration of colonic neural stem cells.
Figure 6.
Immunofluorescence double staining images of Nestin and Ki67 in the Control, DM, DM + A and DM + B groups. DAPI-stained nuclei appeared blue. Nestin and Ki67 are indicated by red fluorescence and green fluorescence, respectively. A rat model of DM was established using an intraperitoneal injection of streptozotocin. The rats in the Control group were given an equal volume of citric acid solvent. Magnification, ×100. *P<0.05 vs. Control; #P<0.05 vs. DM. DM, diabetes mellitus model; DM + A, diabetic rats treated with 5 µg/kg prucalopride; DM + B, diabetic rats treated with 10 µg/kg prucalopride.
Prucalopride promotes 5-HT secretion in the colon
The levels of 5-HT in the colon, which were measured via ELISA, are presented in Fig. 7. Compared with those in the Control group, the levels of 5-HT in the DM group were significantly reduced. Furthermore, the DM + A group and the DM + B group exhibited significantly higher levels of 5-HT compared with the DM group, indicating that both 5 and 10 µg/kg prucalopride promoted 5-HT secretion in the colon.
Figure 7.
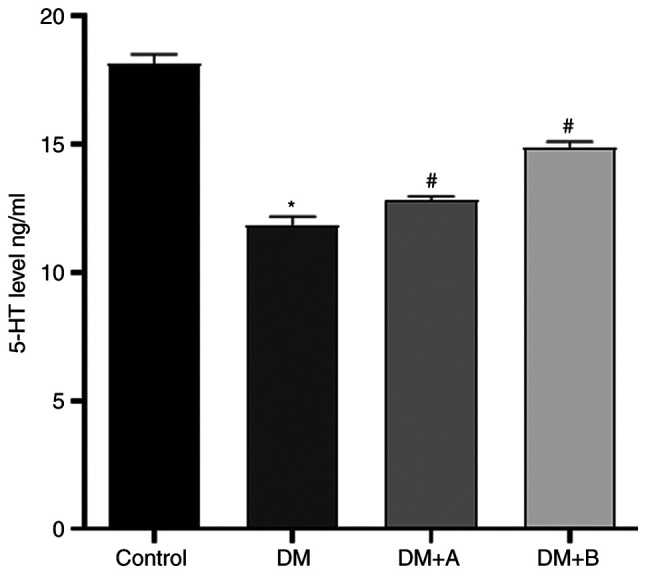
Levels of 5-HT in the colon samples of the Control, DM, DM + A and DM + B groups, as detected using an ELISA. A rat model of DM was established using an intraperitoneal injection of streptozotocin. The rats in the Control group were given an equal volume of citric acid solvent. *P<0.05 vs. Control; #P<0.05 vs. DM. DM, diabetes mellitus model; DM + A, diabetic rats treated with 5 µg/kg prucalopride; DM + B, diabetic rats treated with 10 µg/kg prucalopride; 5-HT, 5-hydroxytryptamine.
Discussion
In animal experiments, the numbers of enteric neural stem cells and intestinal neurons expressing Nestin are markedly increased in normal adult mice after treatment with the 5-HT4 receptor agonists mosapride and tegaserod (39). However, the use of 5-HT4 receptor agonists in 5-HT4 receptor-knockout mice does not increase the number of colon neurons (39). Furthermore, an in vitro study has demonstrated that 5-HT4 receptor agonists can promote neuron survival and reduce apoptosis (39). Therefore, as a 5-HT4 receptor agonist, prucalopride may have an effect on enteric neural stem cells.
Nestin is a protein specific to the central nervous system, which is specifically expressed in undifferentiated neural pluripotent stem cells (57,58). Neural stem cells expressing Nestin in the ENS can differentiate into enteric neurons or glial cells (35). When neural stem cells differentiate into neurons and glial cells, Nestin is no longer expressed (57). Therefore, Nestin is considered to be a marker of enteric neural stem cells in the ENS (24). In the present study, the results of Nestin immunohistochemical analysis identified that colonic stem cells of diabetic rats possessed a regenerative function and exhibited high protein expression levels of Nestin. Furthermore, compared with those of the Control rats and DM rats, the expression levels of Nestin in the DM + A group were significantly increased.
HuD can promote the mitosis of neural stem cells, and it is an important protein involved in the differentiation and maturation of neurons (59). HuD is abundantly expressed in the healthy ENS and its expression is decreased in diseased tissues with loss of intestinal function (49). Furthermore, the loss or reduction of HuD protein may impair embryonic development of the ENS, which causes the absence or decrease of enteric ganglion cells (60). The present study demonstrated that, compared with the control group, HuD expression was significantly decreased in the DM, DM + A and DM + B groups, indicating that colon neurons were damaged in rats with DM. Compared with those in the DM group, the expression levels of HuD in the DM + A group were increased. These results suggested that prucalopride could promote the regeneration of colonic neural stem cells and neurons.
Neural stem cells will differentiate into neurons and glial cells. PGP9.5 is a neuron-specific protein and it is mainly expressed in endocrine cells of the central and peripheral nervous systems (61). PGP9.5 regulates the cell cycle, differentiation, proliferation and apoptosis via a non-lysosomal degradation pathway (62). Furthermore, PGP9.5 is expressed during the differentiation of neural nodules, and it is a cancer biomarker due to its high expression levels in various cancer types, such as esophagus cancer and colorectal cancer (53,54). In the present study, compared with those in the Control group, the expression levels of PGP9.5 in the DM, DM + A and DM + B groups were significantly increased, and its expression levels in the DM + A and DM + B groups were significantly higher compared with those in the DM group. These results suggested that prucalopride could promote the differentiation of the colonic nerve node.
GFAP is expressed in astrocytes of the central nervous system and in enteroglial cells (63). SOX10 is a transcription factor expressed in the neural crest and peripheral nervous system, and it is highly expressed in multiple healthy tissues, such as the esophagus, stomach and colorectum (51,52). SOX10 serves an important role in promoting the survival and proliferation of enteric neural crest cells, as well as glial cell formation (62). In the present study, compared with those in the Control group, the expression levels of GFAP in the DM, DM + A and DM + B groups were significantly decreased, and its expression levels in the DM + A and DM + B groups were significantly higher compared with those in the DM group. These results demonstrated that DM caused damage to intestinal glial cells and decreased the numbers of glial cells, and that prucalopride could promote the regeneration of glial cells. As a marker of glial cells, SOX10 is abundantly expressed in healthy tissues, but is negatively expressed in cancerous tissues (64). Compared with those in the DM group, the expression levels of SOX10 in the DM + A and DM + B groups were significantly increased, indicating that after colonic glial cell injury, prucalopride could promote glial cell regeneration.
The aforementioned results regarding the expression levels of Nestin, HuD, PGP9.5, GFAP and SOX10 suggested that colon cells of diabetic rats had the ability to regenerate after damage. Furthermore, these results indicated that prucalopride may promote the proliferation of neural stem cells and enhance the differentiation of neural stem cells into neurons and glial cells. Regeneration of neurons expressing PGP9.5 and HuD, and glial cells expressing GFAP and SOX10, could repair the damaged ENS and restore gastrointestinal motility.
Since prucalopride, as an agonist of the 5-HT4 receptor, could promote the regeneration of colonic nerve cells, it was suggested that 5-HT4 expression may also promote the regeneration and recovery of colonic nerve cells. In the present study, the ELISA results indicated that the levels of 5-HT protein were highest in the Control group, although the levels of 5-HT protein in the DM, DM + A and DM + B groups showed an increasing trend, indicating that prucalopride could promote the secretion of 5-HT protein in the colon. The results of immunofluorescence double-labeling of 5-HT4 were consistent with the ELISA results, suggesting that prucalopride may upregulate the protein expression levels of 5-HT4. However, similar trends in the expression of 5-HT4 and Nestin were not observed.
Ki67 is a marker of cell proliferation rate, and the faster the cell proliferation rate, the higher its expression levels (65). The results of immunofluorescence double-labeling of Ki67 demonstrated that the Control group had the lowest expression levels of Ki67, and its expression levels in the DM + A group and DM + B group were significantly higher compared with those in the DM group. Furthermore, similar trends in the expression of Nestin and Ki67 were observed. These findings indicated that DM caused colon damage and initiated the regeneration of colon cells, and that prucalopride could increase the rate of cell proliferation and accelerate the proliferation of colon stem cells. The aforementioned results also suggested that prucalopride could promote the secretion of 5-HT4 in the colon, enhance 5-HT4 expression and promote the regeneration of colon stem cells.
It was subsequently examined whether prucalopride exerted an effect on intestinal motility in diabetic rats, and whether it promoted bowel movement. The results of colonic transit time analysis demonstrated that the colonic transit time of diabetic rats (DM group) was significantly longer compared with that of the Control group. In the DM+ A group, the time for removing the glass beads appeared to be shorter compared with that in the DM group. Furthermore, the colonic transit time in the DM + B group was significantly shorter compared with that in the DM group, and it was similar to that observed in the Control group. The treatment of the DM + B group appeared to have the best efficacy to improve intestinal motility. Although the difference was not significant, the treatment of DM + B group appeared to be superior compared with that of the DM + A group in terms of improving intestinal motility. These results suggested that prucalopride improved intestinal motility and reduced bowel movement time.
The results of immunohistochemical analysis identified that the promoting effect of 5 µg/kg prucalopride (DM + A) on colon cell regeneration was more favorable compared with that of 10 µg/kg Prucalopride (DM + B). However, the results of 5-HT4 expression, Ki67 expression and colonic transit time analyses demonstrated that 10 µg/kg prucalopride had more favorable effects on promoting 5-HT4 expression and enhancing the intestinal motility. It was suggested that, compared with the normal dose of prucalopride, an increase in its dose may inhibit the rate of cell proliferation and slow cell repair. However, the specific regulatory mechanism remains to be studied in the future.
In conclusion, DM could cause ENS damage, which was manifested by a decrease in neurons and glial cells, leading to bowel movement disorders and prolonged bowel movement time. As a 5-HT4 receptor agonist, prucalopride could promote the secretion of 5-HT4 in colon cells, enhance 5-HT4 expression and improve bowel movement function. Prucalopride could also repair colon damage by promoting the proliferation of enteric neural stem cells, as well as the differentiation of enteric neurons and glial cells. Furthermore, prucalopride may improve intestinal motility by promoting ENS regeneration in diabetic rats. These results suggest a novel role of prucalopride in the management of diabetic neuropathy, and this should be further validated in clinical trials.
Acknowledgments
Not applicable.
Funding Statement
The present study was supported by the National Natural Science Foundation of China (grant no. 81600430).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
YW and LL designed the study, analyzed the data, wrote the paper and confirmed the authenticity of all the raw data. YW, XX and LL performed the experiments and collected data. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The study protocol was approved by the Ethics Committee of The First Clinical Medical College of Nanjing Medical University (Nanjing, China).
Patient consent for publication
Not applicable.
Competing interests
The authors declare they have no competing interests.
References
- 1.NCD Risk Factor Collaboration (NCD -RisC): Worldwide trends in diabetes since 1980: A pooled analysis of 751 population-based studies with 4.4 million participants. Lancet. 2016;387:1513–1530. doi: 10.1016/S0140-6736(16)00618-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Yoo H, Choo E, Lee S. Study of hospitalization and mortality in Korean diabetic patients using the diabetes complications severity index. BMC Endocr Disord. 2020;20:122. doi: 10.1186/s12902-020-00605-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Horowitz M, Samsom M. Gastrointestinal function in diabetes mellitus. Chichester: John Wiley & Sons, Ltd; 2004. pp. 1–337. [Google Scholar]
- 4.Kalra S, Zargar AH, Jain SM, Sethi B, Chowdhury S, Singh AK, Thomas N, Unnikrishnan AG, Thakkar PB, Malve H. Diabetes insipidus: The other diabetes. Indian J Endocrinol Metab. 2016;20:9–21. doi: 10.4103/2230-8210.172273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Krishnan B, Babu S, Walker J, Walker AB, Pappachan JM. Gastrointestinal complications of diabetes mellitus. World J Diabetes. 2013;4:51–63. doi: 10.4239/wjd.v4.i3.51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Azpiroz F, Malagelada C. Diabetic neuropathy in the gut: Pathogenesis and diagnosis. Diabetologia. 2016;59:404–408. doi: 10.1007/s00125-015-3831-1. [DOI] [PubMed] [Google Scholar]
- 7.Ordög T, Hayashi Y, Gibbons SJ. Cellular pathogenesis of diabetic gastroenteropathy. Minerva Gastroenterol Dietol. 2009;55:315–343. [PMC free article] [PubMed] [Google Scholar]
- 8.Yarandi SS, Srinivasan S. Diabetic gastrointestinal motility disorders and the role of enteric nervous system: Current status and future directions. Neurogastroenterol Motil. 2014;26:611–624. doi: 10.1111/nmo.12330. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Domènech A, Pasquinelli G, De Giorgio R, Gori A, Bosch F, Pumarola M, Jiménez M. Morphofunctional changes underlying intestinal dysmotility in diabetic RIP-I/hIFNβ transgenic mice. Int J Exp Pathol. 2011;92:400–412. doi: 10.1111/j.1365-2613.2011.00789.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Du F, Wang L, Qian W, Liu S. Loss of enteric neurons accompanied by decreased expression of GDNF and PI3K/Akt pathway in diabetic rats. Neurogastroenterol Motil. 2009;21:1229–e114. doi: 10.1111/j.1365-2982.2009.01379.x. [DOI] [PubMed] [Google Scholar]
- 11.Furlan MM, Molinari SL, Miranda Neto MH. Morphoquantitative effects of acute diabetes on the myenteric neurons of the proximal colon of adult rats. Arq Neuropsiquiatr. 2002;60:576–581. doi: 10.1590/S0004-282X2002000400012. [DOI] [PubMed] [Google Scholar]
- 12.Ordög T. Interstitial cells of Cajal in diabetic gastroenteropathy. Neurogastroenterol Motil. 2008;20:8–18. doi: 10.1111/j.1365-2982.2007.01056.x. [DOI] [PubMed] [Google Scholar]
- 13.Lin HZ, Sarosiek I, Forster J, Damjanov I, Hou Q, McCallum RW. Association of the status of interstitial cells of Cajal and electrogastrogram parameters, gastric emptying and symptoms in patients with gastroparesis. Neurogastroenterol Motility. 2010;22:56–61. doi: 10.1111/j.1365-2982.2009.01365.x. [DOI] [PubMed] [Google Scholar]
- 14.Rao M, Gershon MD. The bowel and beyond: The enteric nervous system in neurological disorders. Nat Rev Gastroenterol Hepatol. 2016;13:517–528. doi: 10.1038/nrgastro.2016.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lake JI, Heuckeroth RO. Enteric nervous system development: Migration, differentiation, and disease. Am J Physiol Gastrointest Liver Physiol. 2013;305:G1–G24. doi: 10.1152/ajpgi.00452.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Farrugia G. Histologic changes in diabetic gastroparesis. Gastroenterol Clin North Am. 2015;44:31–38. doi: 10.1016/j.gtc.2014.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Ihana-Sugiyama N, Nagata N, Yamamoto-Honda R, Izawa E, Kajio H, Shimbo T, Kakei M, Uemura N, Akiyama J, Noda M. Constipation, hard stools, fecal urgency, and incomplete evacuation, but not diarrhea is associated with diabetes and its related factors. World J Gastroenterol. 2016;22:3252–3260. doi: 10.3748/wjg.v22.i11.3252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Becker L, Kulkarni S, Tiwari G, Micci MA, Pasricha PJ. Divergent fate and origin of neurosphere-like bodies from different layers of the gut. Am J Physiol Gastrointest Liver Physiol. 2012;302:G958–G965. doi: 10.1152/ajpgi.00511.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Burns AJ, Pachnis V. Development of the enteric nervous system: Bringing together cells, signals and genes. Neurogastroenterol Motil. 2009;21:100–102. doi: 10.1111/j.1365-2982.2008.01255.x. [DOI] [PubMed] [Google Scholar]
- 20.Laranjeira C, Sandgren K, Kessaris N, Richardson W, Potocnik A, Vanden Berghe P, Pachnis V. Glial cells in the mouse enteric nervous system can undergo neurogenesis in response to injury. J Clin Invest. 2011;121:3412–3424. doi: 10.1172/JCI58200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Joseph NM, He S, Quintana E, Kim YG, Núñez G, Morrison SJ. Enteric glia are multipotent in culture but primarily form glia in the adult rodent gut. J Clin Invest. 2011;121:3398–3411. doi: 10.1172/JCI58186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Matsuyoshi H, Kuniyasu H, Okumura M, Misawa H, Katsui R, Zhang GX, Obata K, Takaki M. A 5-HT(4)-receptor activation-induced neural plasticity enhances in vivo reconstructs of enteric nerve circuit insult. Neurogastroenterol Motil. 2010;22:806–813.e226. doi: 10.1111/j.1365-2982.2010.01474.x. [DOI] [PubMed] [Google Scholar]
- 23.Bixby S, Kruger GM, Mosher JT, Joseph NM, Morrison SJ. Cell-intrinsic differences between stem cells from different regions of the peripheral nervous system regulate the generation of neural diversity. Neuron. 2002;35:643–656. doi: 10.1016/S0896-6273(02)00825-5. [DOI] [PubMed] [Google Scholar]
- 24.Kruger GM, Mosher JT, Bixby S, Joseph N, Iwashita T, Morrison SJ. Neural crest stem cells persist in the adult gut but undergo changes in self-renewal, neuronal subtype potential, and factor responsiveness. Neuron. 2002;35:657–669. doi: 10.1016/S0896-6273(02)00827-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Heanue TA, Pachnis V. Enteric nervous system development and Hirschsprung's disease: Advances in genetic and stem cell studies. Nat Rev Neurosci. 2007;8:466–479. doi: 10.1038/nrn2137. [DOI] [PubMed] [Google Scholar]
- 26.Burns AJ, Goldstein AM, Newgreen DF, Stamp L, Schäfer KH, Metzger M, Hotta R, Young HM, Andrews PW, Thapar N, et al. White paper on guidelines concerning enteric nervous system stem cell therapy for enteric neuropathies. Dev Biol. 2016;417:229–251. doi: 10.1016/j.ydbio.2016.04.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ibrahim A, Ali RAR, Manaf MRA, Ahmad N, Tajurruddin FW, Qin WZ, Desa SHM, Ibrahim NM. Multi-strain probiotics (Hexbio) containing MCP BCMC strains improved constipation and gut motility in Parkinson's disease: A randomised controlled trial. PLoS One. 2020;15:e0244680. doi: 10.1371/journal.pone.0244680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Shafe AC, Lee S, Dalrymple JS, Whorwell PJ. The LUCK study: Laxative usage in patients with GP-diagnosed constipation in the UK, within the general population and in pregnancy. An epidemiological study using the general practice research database (GPRD) Therap Adv Gastroenterol. 2011;4:343–363. doi: 10.1177/1756283X11417483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Menees SB, Guentner A, Chey SW, Saad R, Chey WD. How Do US gastroenterologists use over-the-counter and prescription medications in patients with gastroesophageal reflux and chronic constipation? Am J Gastroenterol. 2015;110:1516–1525. doi: 10.1038/ajg.2015.156. [DOI] [PubMed] [Google Scholar]
- 30.Gonzalez-Martinez MA, Ortiz-Olvera NX, Mendez-Navarro J. Novel pharmacological therapies for management of chronic constipation. J Clin Gastroenterol. 2014;48:21–28. doi: 10.1097/01.mcg.0000436440.05887.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hennessy S, Leonard CE, Newcomb C, Kimmel SE, Bilker WB. Cisapride and ventricular arrhythmia. Br J Clin Pharmacol. 2008;66:375–385. doi: 10.1111/j.1365-2125.2008.03249.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Wysowski DK, Corken A, Gallo-Torres H, Talarico L, Rodriguez EM. Postmarketing reports of QT prolongation and ventricular arrhythmia in association with cisapride and Food and Drug Administration regulatory actions. Am J Gastroenterol. 2001;96:1698–1703. doi: 10.1111/j.1572-0241.2001.03927.x. [DOI] [PubMed] [Google Scholar]
- 33.Thompson CA. Novartis suspends tegaserod sales at FDA's request. Am J Health Syst Pharm. 2007;64:1020. doi: 10.2146/news070044. [DOI] [PubMed] [Google Scholar]
- 34.Pan H, Gershon MD. Activation of intrinsic afferent pathways in submucosal ganglia of the guinea pig small intestine. J Neurosci. 2000;20:3295–3309. doi: 10.1523/JNEUROSCI.20-09-03295.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Belkind-Gerson J, Carreon-Rodriguez A, Benedict LA, Steiger C, Pieretti A, Nagy N, Dietrich J, Goldstein AM. Nestin-expressing cells in the gut give rise to enteric neurons and glial cells. Neurogastroenterol Motil. 2013;25:61–69.e7. doi: 10.1111/nmo.12015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu MT, Kuan YH, Wang J, Hen R, Gershon MD. 5-HT4 receptor-mediated neuroprotection and neurogenesis in the enteric nervous system of adult mice. J Neurosci. 2009;29:9683–9699. doi: 10.1523/JNEUROSCI.1145-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bouras EP, Michael C, Burton DD, Thomforde G, McKinzie S, Zinsmeister AR. Prucalopride accelerates gastrointestinal and colonic transit in patients with constipation without a rectal evacuation disorder. Gastroenterology. 2001;120:354–360. doi: 10.1053/gast.2001.21166. [DOI] [PubMed] [Google Scholar]
- 38.Karin S, Ken W. Chapter 35-gastrointestinal drugs. Side Eff Drugs Annu. 2015;37:433–459. doi: 10.1016/bs.seda.2015.05.008. [DOI] [Google Scholar]
- 39.Bharucha AE, Wouters MM, Tack J. Existing and emerging therapies for managing constipation and diarrhea. Curr Opin Pharmacol. 2017;37:158–166. doi: 10.1016/j.coph.2017.10.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ueno N, Inui A, Satoh Y. The effect of mosapride citrate on constipation in patients with diabetes. Diabetes Res Clin Pract. 2010;87:27–32. doi: 10.1016/j.diabres.2009.09.024. [DOI] [PubMed] [Google Scholar]
- 41.Gershon MD, Liu MT. Serotonin and neuroprotection in functional bowel disorders. Neurogastroenterol Motil. 2007;19(Suppl 2):S19–S24. doi: 10.1111/j.1365-2982.2007.00962.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Bassotti G, Gambaccini D, Bellini M. Prucalopride succinate for the treatment of constipation: An update. Expert Rev Gastroenterol Hepatol. 2016;10:291–300. doi: 10.1586/17474124.2016.1129897. [DOI] [PubMed] [Google Scholar]
- 43.Omer A, Quigley EMM. An update on prucalopride in the treatment of chronic constipation. Therap Adv Gastroenterol. 2017;10:877–887. doi: 10.1177/1756283X17734809. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bassotti G, Usai Satta P, Bellini M. Prucalopride for the treatment of constipation: A view from 2015 and beyond. Expert Rev Gastroenterol Hepatol. 2019;13:257–262. doi: 10.1080/17474124.2019.1568238. [DOI] [PubMed] [Google Scholar]
- 45.Garnock-Jones KP. Prucalopride: A review in chronic idiopathic constipation. Drugs. 2016;76:99–110. doi: 10.1007/s40265-015-0518-3. [DOI] [PubMed] [Google Scholar]
- 46.Daniali M, Nikfar S, Abdollahi M. An overview of the efficacy and safety of prucalopride for the treatment of chronic idiopathic constipation. Expert Opin Pharmacother. 2019;20:2073–2080. doi: 10.1080/14656566.2019.1668927. [DOI] [PubMed] [Google Scholar]
- 47.Xie F, Lei J, Ran M, Li Y, Deng L, Feng J, Zhong Y, Li J. Attenuation of diabetic nephropathy in diabetic mice by fasudil through regulation of macrophage polarization. J Diabetes Res. 2020;2020:4126913. doi: 10.1155/2020/4126913. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.McClain J, Grubišić V, Fried D, Gomez-Suarez RA, Leinninger GM, Sévigny J, Parpura V, Gulbransen BD. Ca2+ responses in enteric glia are mediated by connexin-43 hemichannels and modulate colonic transit in mice. Gastroenterology. 2014;146:497–507.e1. doi: 10.1053/j.gastro.2013.10.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Butler Tjaden NE, Trainor PA. The developmental etiology and pathogenesis of Hirschsprung disease. Transl Res. 2013;162:1–15. doi: 10.1016/j.trsl.2013.03.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Nagy N, Goldstein AM. Enteric nervous system development: A crest cell's journey from neural tube to colon. Semin Cell Dev Biol. 2017;66:94–106. doi: 10.1016/j.semcdb.2017.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Kiefer JC. Back to basics: Sox genes. Dev Dyn. 2007;236:2356–2366. doi: 10.1002/dvdy.21218. [DOI] [PubMed] [Google Scholar]
- 52.Sarkar A, Hochedlinger K. The sox family of transcription factors: Versatile regulators of stem and progenitor cell fate. Cell Stem Cell. 2013;12:15–30. doi: 10.1016/j.stem.2012.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hurst-Kennedy J, Chin LS, Li L. Ubiquitin C-terminal hydrolase l1 in tumorigenesis. Biochem Res Int. 2012;2012:123706. doi: 10.1155/2012/123706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Burger AM, Seth AK. The ubiquitin-mediated protein degradation pathway in cancer: Therapeutic implications. Eur J Cancer. 2004;40:2217–2229. doi: 10.1016/j.ejca.2004.07.006. [DOI] [PubMed] [Google Scholar]
- 55.Uesaka T, Nagashimada M, Enomoto H. Neuronal differentiation in schwann cell lineage underlies postnatal neurogenesis in the enteric nervous system. J Neurosci. 2015;35:9879–9888. doi: 10.1523/JNEUROSCI.1239-15.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Gerdes J. Ki-67 and other proliferation markers useful for immunohistological diagnostic and prognostic evaluations in human malignancies. Semin Cancer Biol. 1990;1:199–206. [PubMed] [Google Scholar]
- 57.Lendahl U, Zimmerman LB, Mckay RD. CNS stem cells express a new class of intermediate filament protein. Cell. 1990;60:585–595. doi: 10.1016/0092-8674(90)90662-X. [DOI] [PubMed] [Google Scholar]
- 58.Dahlstrand J, Lardelli M, Lendahl U. Nestin mRNA expression correlates with the central nervous system progenitor cell state in many, but not all, regions of developing central nervous system. Brain Res Dev Brain Res. 1995;84:109–129. doi: 10.1016/0165-3806(94)00162-S. [DOI] [PubMed] [Google Scholar]
- 59.Zheng Z, Chen B, Jin Z, Gao M, Tang C, Mao Y, Qu Y, Liu Y. Downregulation of P2Y2 and HuD during the development of the enteric nervous system in fetal rats with anorectal malformations. Mol Med Rep. 2019;20:1297–1305. doi: 10.3892/mmr.2019.10356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Giorgio RD, Volta U, Stanghellini V, Cogliandro RF, Barbara G, Corinaldesi R, Towns R, Guo C, Hong S, Wiley JW. Neurogenic chronic intestinal pseudo-obstruction: Antineuronal antibody-mediated activation of autophagy via Fas. Gastroenterology. 2008;135:601–609. doi: 10.1053/j.gastro.2008.05.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Akishima-Fukasawa Y, Ino Y, Nakanishi Y, Miura A, Moriya Y, Kondo T, Kanai Y, Hirohashi S. Significance of PGP9.5 expression in cancer-associated fibroblasts for prognosis of colorectal carcinoma. Am J Clin Pathol. 2010;134:71–79. doi: 10.1309/AJCPRJP39MIDSGBH. [DOI] [PubMed] [Google Scholar]
- 62.Kostouros A, Koliarakis I, Natsis K, Spandidos D, Tsatsakis A, Tsiaoussis J. Large intestine embryogenesis: Molecular pathways and related disorders (Review) Int J Mol Med. 2020;46:27–57. doi: 10.3892/ijmm.2020.4583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Gomes FC, Paulin D, Moura Neto V. Glial fibrillary acidic protein (GFAP): Modulation by growth factors and its implication in astrocyte differentiation. Braz J Med Biol Res. 1999;32:619–631. doi: 10.1590/S0100-879X1999000500016. [DOI] [PubMed] [Google Scholar]
- 64.Tong X, Li L, Li X, Heng L, Zhong L, Su X, Rong R, Hu S, Liu W, Jia B, et al. SOX10, a novel HMG-box-containing tumor suppressor, inhibits growth and metastasis of digestive cancers by suppressing the Wnt/β-catenin pathway. Oncotarget. 2014;5:10571–10583. doi: 10.18632/oncotarget.2512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Miller I, Min M, Yang C, Tian C, Gookin S, Carter D, Spencer SL. Ki67 is a graded rather than a binary marker of proliferation versus quiescence. Cell Rep. 2018;24:1105–1112.e5. doi: 10.1016/j.celrep.2018.06.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.