Abstract
Stiff Person Syndrome (SPS) is an extremely rare neurological condition characterized by muscle stiffness and painful muscle spasms. The symptoms often progress slowly and can cause disability. Antibodies to glutamic acid decarboxylase (anti-GAD) have been reported in up to 80% of the classic type of SPS. Paraneoplastic syndrome comprises 5% of SPS cases. These patients present with different malignancies including lung, thymus, breast, colon, and lymph nodes. In this paper, we report a case of a 25-year-old Vietnamese female patient with SPS presenting with unusual clinical manifestations of sudden onset, rapidly progressive spinal, abdominal, and lower limb rigidity accompanied by painful spasms, autonomic disorders, and severe, multiple bone fractures. Serologic tests detected high-titer anti-GAD, combined with anti-SOX1 antibodies, suggesting paraneoplastic SPS. Intravenous immunoglobulin has been employed as the main treatment therapy, and the patient has had a complete remission.
Keywords: Stiff person syndrome, Paraneoplastic syndrome, Intravenous immunoglobulin
Introduction
Stiff person syndrome (SPS) is considered one of the extremely rare diseases involving the neurologic system. It was first described in 1956 by Moersch and Woltman [1] in a study of 14 patients who presented with progressive rigidity of the back, abdominal, and thigh muscles associated with painful spasms. The spasms can be triggered by heightened sensitivity to external stimuli [2]. SPS has been classified into three subtypes: classic SPS, paraneoplastic SPS, and variants [3]. In the classic SPS, the affected muscles are those in the lower back, abdomen, and leg. The shoulder, neck, and hip muscles may also be affected. SPS has been reported to be associated with other immune diseases such as type 1 diabetes, thyroiditis, pernicious anemia, and vitiligo [1, 3, 4]. In SPS patients, several autoantibodies have been found in serum or cerebrospinal fluid (CSF), most commonly antibodies to glutamic acid decarboxylase (anti-GAD) and anti-amphiphysin antibodies [1, 5, 6].
SPS is a paraneoplastic syndrome in 5% of cases. Lung, thymus, breast, and colon malignancies, and lymphoma have been implicated.
Anti-SOX1 is associated with small-cell lung cancer and is rarely accompanied by anti-GAD [7, 8, 9, 10, 11, 12]. Diagnosis of SPS employs the Dalakas' criteria of well-characterized clinical manifestations, electromyography (EMG) findings, and positive serology for typical autoantibodies (Table 1). Treatment for SPS is a combination of symptomatic treatment and immunomodulatory therapy. Misdiagnosis or delayed treatment can cause significant disability or life-threatening complications such as respiratory compromise and death in SPS patients. In this paper, we present a patient diagnosed with SPS who had both anti-GAD and anti-SOX1 antibodies in whom intravenous immunoglobulin (IVIG) therapy elicited a complete response.
Table 1.
Dalakas' criteria of SPS present in our patient
| Criteria | Dalakas' criteria of SPS | Our patient |
|---|---|---|
| 1 | Rigidity in limbs and trunk muscles, prominent in thoracolumbar and abdominal muscles | Fulfilled |
| 2 | Continuous contraction of agonist and antagonist muscles (clinically and on EMG) | Clinically |
| 3 | Spasms precipitated by stimuli: noise, tactile, emotion | Fulfilled |
| 4 | Absence of other neurologic disease explaining symptoms | Fulfilled |
| 5 | Presence of anti-GAD65/anti-amphiphysin antibodies in serum | Anti-GAD65 antibodies |
EMG, electromyography; GAD, glutamic acid decarboxylase.
Case Presentation
A 25-year-old female patient with a previously unremarkable medical history presented with the sudden onset of severe vomiting, neck pain, and high fever. A few days later, she noted limb tremors lasting for 20–30 s occurring while she was awake. After the episodes, she felt tired and noted weakness and pain in her leg muscles sufficient to prevent her from being able to sit up unaided. There were many similar attacks during the day. Routine biochemistry and a full blood count were normal, as were sequential electroencephalograms. Magnetic resonance imaging (MRI) of the brain and cervical spine was normal. Her CSF results revealed white blood cells of 4/µL; protein of 0.2 g/L; and a negative Pandy's test. CSF multiplex PCR for E. coli, H. influenzae, Listeria monocytogenes, Neisseria meningitidis, Streptococcus agalactiae, Streptococcus pneumoniae, Enterovirus, adenovirus, cytomegalovirus, mumps virus, parvovirus B19, Epstein-Barr virus, herpes simplex virus 1, 2, human herpesvirus 6, 7, human parechovirus, and vesicular stomatitis virus) were negative. Serology tests for autoimmune encephalitis and antinuclear antibodies, screened by ELISA, were negative. She was transferred to our hospital after 10 days of treatment with antibiotics, and supportive therapy failed to result in any improvement.
During admission, she continued to have multiple daily episodes of limb tremors which lasted a few seconds to 2 min and were accompanied by sweating, tachypnea, and tachycardia (120 beats/min). It was also noted that she was anxious and easily startled by sounds (please refer to the video).
Clinical examination revealed an alert individual with no cranial nerve palsies or limb paralysis. Assessment of the patient's lower extremities, deep tendon reflexes, and Babinski sign was impossible due to her hypersensitivity to touch. Muscle tone was normal. Fever spikes in the vicinity of 38°C were recorded. Her white blood cell count was elevated up to 15 g/L. Blood and urine cultures grew no organisms. The patient's serum was screened for antinuclear antibodies using an indirect immunofluorescence assay (Hep-2 cells; Euroimmun Medizinische Labordiagnostika AG, Lübeck, Germany), anti-phospholipid antibodies (anti-phospholipid, anti-beta2 glycoprotein I) using ELISA and lupus anticoagulant. Results were negative. Complement 3 was at 157.9 mg/dL (normal range [NR] 90–180); C4 was elevated at 81.6 mg/dL [NR 10–40 mg/dL]); ferritin was elevated at 328 μg/L (NR 13–150 for female); and erythrocyte sedimentation rate was slightly elevated at 80 mm/h (NR 0–29 mm/h) after 2 h. The patient was diagnosed with suspected conversion disorder and treated symptomatically with diazepam, valproate, and mirtazapine. These medications reduced the startle responses and the shaking of her extremities; however, these signs did not resolve completely. The patient could sit up and walk. After that, she was discharged and referred to a psychologist.
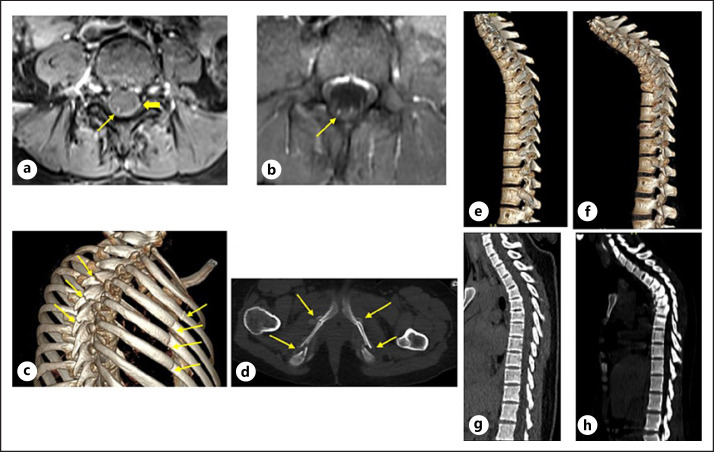
Three weeks later, the patient returned with urinary retention and numbness on both sides of the buttocks radiating down the right leg. Urinary catheterization was performed with a Foley catheter. The spasms of the whole body and limbs had not improved. However, after the spasms, we found abdominal, thigh, and neck rigidity. Spinal MRI with gadolinium contrast media was performed and demonstrated lumbar spinal dural sac and nerve root enhancement (shown in Fig. 1a, b). We also found edema of multiple pelvic muscles, bilateral paraspinal muscles with diffuse hyperintense STIR images (shown in Fig. 2a, b), and multiple thoracic vertebral bodies collapsed from T4-T7 (shown in Fig. 1e-h). Brain MRI was normal. Nerve conduction studies showed normal motor and sensory conduction velocities. Compound muscle action potentials and sensory nerve action potentials were also within NRs. It was impossible to measure the H-reflex and F wave because of muscle spasms caused by stimulation. For the same reason, lumbar puncture and EMG were not able to be performed. Paraneoplastic autoantibodies were tested, using the EUROLINE Paraneoplastic Neurologic Syndromes 12 Ag (IgG). Pulse therapy of corticosteroids at a dose of 1 mg/kg was commenced. Diazepam, gabapentin, and baclofen were administered for symptom relief, and slight improvement was observed. When anti-GAD antibodies with intensity of 91 (+++) and anti-SOX1 antibodies with intensity of 38 (++) were detected, screening for systemic cancer was initiated. 18F-FDG PET/CT showed multiple fractures of the thoracic vertebral spinous process, ribs, sacrum, and pelvic bones (shown in Fig. 1c, d). Avid FDG metabolism was observed in multiple paraspinal and erector spinal muscles in the cervical, thoracic, and lumbar regions. No malignant lesion was detected. Further full blood counts, fasting glucose, and renal and liver function tests were normal. Thyroid hormones, parathyroid hormones, and calcium were within normal limits, except for a high titer of anti-TPO (23.7 IU/mL; NR <5.61 IU/mL) antibody. Protein electrophoresis was unremarkable. The patient refused a Dual Energy X-ray Absorptiometry (DEXA) scan. A multidisciplinary review by a rheumatologist, oncologist, immunologist, endocrinologist, and neurologist determined that there was no evidence of cancer or bone disease.
Fig. 1.
a, b MRI lumbar spine with gadolinium-DTPA demonstrates caudal equine (thin arrow) and dural sac enhancement (thick arrow in A) on axial postcontrast T1W images. c Multiple rib and thoracic vertebral spinous process fractures (arrows) on 3D-volume rendering MSCT images. d Bilateral multiple fractures at the pubic bone (arrows) on axial MSCT bone window. e, f Progressive thoracic spine scoliosis on 3D-volume rendering MSCT images: (e) October 2020 and (f) November 2020. g, h Progressive thoracic spine scoliosis on MSCT MPR sagittal images: (g) October 2020 and (h) November 2020.
Fig. 2.
a, b MRI shows multiple muscular edema pre-IVIG treatment at bilateral obturator internus muscles on axial STIR images (arrows in a) and at bilateral paraspinal muscles on coronal STIR images (arrows in b). c, d Muscular edema markedly resolved following by IVIG treatment (arrows in c, d).
Based on clinical manifestations and laboratory findings, the patient was diagnosed with SPS with a poor response to symptomatic treatment. Thus, we decided to give a 5-day course of IVIG (0.4 g/kg/day), followed by rehabilitation. After 3 weeks of treatment, the symptoms gradually improved. Her urinary catheter was able to be removed. Abdominal and thigh muscles were soft without noticeable spasms. The modified Rankin score at discharge was 2 (modified Rankin score of 5 at admission).
After 1 month, the dosage of IVIG was reduced to 0.2 g/kg/day. A DEXA scan showed osteopenia with Z score values of −2.5 at the lumbar vertebrae and femoral necks. The spinal MRI showed an increase in kyphosis and vertebral body collapse but markedly reduced lumbar-pelvic muscle edema (shown in Fig. 2c, d). Treatment with zoledronic acid was recommended and consideration to spinal surgery.
Discussion
SPS is a rare neurological autoimmune condition with an incidence of 1 per million [2, 3]. Main clinical symptoms include muscle rigidity and muscle spasms, which predominantly affect muscles of the trunk and lower limbs. Here, we present a case of a female patient diagnosed with SPS. Her main symptoms were simultaneous activation of both agonist and antagonist muscles. The spasms were provoked by the unexpected noise, light touch, or sudden movement. Muscle rigidity affecting paraspinal, abdominal, and lower limb muscles was also noted. These symptoms are typical presenting symptoms for SPS (shown in Table 1).
Although our patient exhibited typical signs and symptoms of classical SPS, she had an unusually sudden onset and additional presenting symptoms of fever, vomiting, and neck stiffness, suggesting infection or a diagnosis of progressive encephalomyelitis with rigidity and myoclonus; however, brainstem symptoms did not appear in this patient. In addition, anti-SOX1 was detected in the serum, indicating the patient should be having neoplasia excluded, to rule out paraneoplastic SPS.
Sudden death and paroxysmal autonomic dysfunction are uncommon neurological symptoms in patients with SPS. Mitsumoto et al. [13] have reported sudden death in two women with typical SPS who developed increasingly frequent attacks of muscle spasms accompanied by severe paroxysmal autonomic dysfunction such as transient hyperpyrexia, diaphoresis, tachypnea, tachycardia, pupillary dilation, and arterial hypertension [14]. Anti-GAD antibodies were identified in the serum of both patients and in the CSF of one. Misra et al. [15] reported a patient who initially suffered from hiccups and vomiting that responded to diazepam. Our patient also had frequent muscle spasms with severe paroxysmal autonomic dysfunction which responded to diazepam, suggesting an underlying autonomic neurological disorder.
Some reports have described myositis in patients with SPS [2, 14, 16]. Dalakas et al. [2] reported 5 of 20 SPS patients with mild creatine kinase elevations. Maramattom [17] and No and colleagues [14] reported marked hyperintensities in the muscles of SPS patients. These authors speculated that muscle edematous change (rhabdomyolysis) may be related to a sustained contraction episode [17, 18]. Similarly, the spinal MRI and PET/CT of our patient showed muscle edema most likely related to fluctuating muscle rigidity.
Fractures and dislocations are the main complications of SPS [19]. There have been several reports of fractures in SPS patients [18, 20, 21]. Podobinski et al., and Dubow [22] and Jamil et al. [21] reported cases with bilateral hip, scaphoid, or femoral neck fracture. Our patient presented with multiple fractures of ribs, vertebrae, sacrum, and pelvic girdle. The thoracic spine MRI showed vertebral collapse from T4-T7. Osteopenia was seen on the DEXA scan (after 1 month of corticosteroids) with a Z score of −2.5 to −2.6. Thus, the multiple fractures in this patient were likely due to a combination of prolonged, frequent muscle contractions and osteopenia, as CT and PET-CT imaging showed no evidence of specific bone pathology. In addition, thyroid and parathyroid hormones and renal function were normal.
Dumitrascu et al. [23] and Barker et al. [24] reported some SPS patients presenting with urinary retention, but normal lumbar MRI. Our patient also had urinary retention; however, the MRI with contrast showed spinal dura and nerve root enhancement in the lumbar and sacral regions. Therefore, in this case, urinary retention is most likely due to nerve root damage. According to the literature, patients with anti-SOX1 antibodies can have concomitant neurological disorders, including neuropathy, in about 8.2% [25, 26, 27]. However, no case of SPS has been reported with nerve root and dural lesions [27]. Sun et al. [25] in their review of paraneoplastic neurological syndrome found that most patients had anti-SOX1 antibodies identified in their serum, while some patients showed positive anti-SOX1 antibody reactivity in the CSF. Although we were unable to perform a spinal fluid tap or the H-reflex, based on MRI scans and clinical manifestations, we hypothesized there may be anti-SOX1/anti-GAD antibodies in the patient's CSF responsible for the inflammation of the nerve roots and dura.
Anti-SOX1 antibodies are classified as onco-neuronal autoantibodies. Cancers were identified in 93.5% of 520 patients with anti-SOX1 antibodies [25], but 6.5% of those patients developed neurological symptoms without an underlying tumor, even after years of follow-up [25]. 245 patients (47.1%) showed the coexistence of multiple onco-neuronal and cell surface autoantibodies. However, only 4 patients (1.9%) had anti-GAD antibodies [25]. Zhang et al. reported a patient with both anti-GAD antibodies and anti-SOX1 antibodies in serum, but the clinical presentation was as Lambert-Eaton myasthenic syndrome [28, 29]. Hardy-Werbi et al. [11] reported 2 cases with both anti-GAD and anti-SOX1 antibodies in serum presenting with small-cell lung cancer, but no neurological presentations. Our patient with clinical SPS also had serum anti-GAD and anti-SOX1 but had no tumor detected.
The treatment of SPS includes GABAergic drugs and IVIG. Our patient did not respond well to first-line treatment but had remarkable improvement in her symptoms after IVIG.
Conclusion
We have described a case of a young, female patient with the sudden onset of SPS and rapid progression of lumbar nerve root damage, multiple fractures, and accompanying autonomic neurological disorders, which responded well to IVIG. The coexistence of anti-GAD and anti-SOX1 antibodies in our patient is very rare, especially when presenting with clinical neurological symptoms of SPS. Considering our patient's response, IVIG may be an appropriate option when treating other SPS patients with various coexisting autoantibodies.
Statement of Ethics
The patient and their family have given their written informed consent to publish the details of her medical case and any accompanying images and video. Information revealing the subject's identity is to be avoided. This study was reviewed and approved by Vinmec Healthcare System-VinUniversity institutional ethical review board for biomedical research: No. 166.QD/VMEC.
Conflict of Interest Statement
The authors have no conflicts of interest to declare.
Funding Sources
No funding sources have been funded for this study.
Author Contributions
All authors have been satisfactory to the ICMJE Criteria for Authorship. Phuong Minh Nguyen and Dinh Van Nguyen: examination, treatment and following up, data analysis/clinical findings, and manuscript writing; Dung Duy Vu and Kien Dung Vu: examination, treatment and following up, and data analysis/clinical findings; Hai Thanh Nguyen: data analysis/clinical findings and manuscript writing.
Data Availability Statement
All data have been made available and presented in the case report.
Supplementary Material
Supplemental Video
Acknowledgments
We are grateful to our patient and her family who supported us in reporting her journey. The authors also wish to thank Clinical Professor Sheryl van Nunen, Faculty of Medicine, Macquarie University, and Faculty of Medicine and Health, The University of Sydney, for reviewing this paper.
References
- 1.Moersch FP, Woltman HW. Progressive fluctuating muscular rigidity and spasm (“stiff-man” syndrome); report of a case and some observations in 13 other cases. Proc Staff Meet Mayo Clin. 1956 Jul 25;31((15)):421–7. [PubMed] [Google Scholar]
- 2.Dalakas MC, Fujii M, Li M, McElroy B. The clinical spectrum of anti-GAD antibody-positive patients with stiff-person syndrome. Neurology. 2000 Nov 28;55((10)):1531–5. doi: 10.1212/wnl.55.10.1531. [DOI] [PubMed] [Google Scholar]
- 3.Buechner S, Florio I, Capone L. Stiff person syndrome: a rare neurological disorder, heterogeneous in clinical presentation and not easy to treat. Case Rep Neurol Med. 2015;2015:278065–5. doi: 10.1155/2015/278065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Smith SR, Fu JB. Paraneoplastic stiff person syndrome: inpatient rehabilitation outcomes of a rare disease from two cancer rehabilitation programmes. J Rehabil Med. 2016;48((7)):639–42. doi: 10.2340/16501977-2089. [DOI] [PubMed] [Google Scholar]
- 5.Walikonis JE, Lennon VA. Radioimmunoassay for glutamic acid decarboxylase (GAD65) autoantibodies as a diagnostic aid for stiff-man syndrome and a correlate of susceptibility to type 1 diabetes mellitus. Mayo Clin Proc. 1998 Dec;73((12)):1161–6. doi: 10.4065/73.12.1161. [DOI] [PubMed] [Google Scholar]
- 6.McKeon A, Robinson MT, McEvoy KM, Matsumoto JY, Lennon VA, Ahlskog JE, et al. Stiff-man syndrome and variants: clinical course, treatments, and outcomes. Arch Neurol. 2012 Feb 1;69((2)):230. doi: 10.1001/archneurol.2011.991. [DOI] [PubMed] [Google Scholar]
- 7.Berger B, Dersch R, Ruthardt E, Rasiah C, Rauer S, Stich O. Prevalence of anti-SOX1 reactivity in various neurological disorders. J Neurol Sci. 2016 Oct;369:342–6. doi: 10.1016/j.jns.2016.09.002. [DOI] [PubMed] [Google Scholar]
- 8.Tschernatsch M, Singh P, Gross O, Gerriets T, Kneifel N, Probst C, et al. Anti-SOX1 antibodies in patients with paraneoplastic and non-paraneoplastic neuropathy. J Neuroimmunol. 2010 Sep 14;226((1–2)):177–80. doi: 10.1016/j.jneuroim.2010.07.005. [DOI] [PubMed] [Google Scholar]
- 9.Sabater L, Titulaer M, Saiz A, Verschuuren J, Güre AO, Graus F. SOX1 antibodies are markers of paraneoplastic Lambert-Eaton myasthenic syndrome. Neurology. 2008 Mar 18;70((12)):924–8. doi: 10.1212/01.wnl.0000281663.81079.24. [DOI] [PubMed] [Google Scholar]
- 10.Titulaer MJ, Klooster R, Potman M, Sabater L, Graus F, Hegeman IM, et al. SOX antibodies in small-cell lung cancer and Lambert-Eaton myasthenic syndrome: frequency and relation with survival. J Clin Oncol. 2009 Sep 10;27((26)):4260–7. doi: 10.1200/JCO.2008.20.6169. [DOI] [PubMed] [Google Scholar]
- 11.Hardy-Werbin M, Arpí O, Taus A, Rocha P, Joseph-Pietras D, Nolan L, et al. Assessment of neuronal autoantibodies in patients with small cell lung cancer treated with chemotherapy with or without ipilimumab. Oncoimmunology. 2018 Feb;7((2)):e1395125. doi: 10.1080/2162402X.2017.1395125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Sabater L, Höftberger R, Boronat A, Saiz A, Dalmau J, Graus F. Antibody repertoire in paraneoplastic cerebellar degeneration and small cell lung cancer. PLoS One. 2013 Mar 25;8((3)):e60438. doi: 10.1371/journal.pone.0060438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Mitsumoto H, Schwartzman MJ, Estes ML, Chou SM, La Franchise EF, De Camilli P, et al. Sudden death and paroxysmal autonomic dysfunction in stiff-man syndrome. J Neurol. 1991;238((2)):91–6. doi: 10.1007/BF00315688. [DOI] [PubMed] [Google Scholar]
- 14.No SW, Im IK, Kim DH. Stiff person syndrome with evidence of nonspecific focal myositis secondary to sustained muscle contraction: a case report. PM R. 2018 Dec;10((12)):1426–30. doi: 10.1016/j.pmrj.2018.04.007. [DOI] [PubMed] [Google Scholar]
- 15.Misra UK, Maurya PK, Kalita J, Gupta RK. Stiff limb syndrome: end of spectrum or a separate entity? Pain Med. 2009 Apr;10((3)):594–7. doi: 10.1111/j.1526-4637.2009.00578.x. [DOI] [PubMed] [Google Scholar]
- 16.Pakeerappa PN, Birthi P, Salles S. Botulinum toxin A injection to facial and cervical paraspinal muscles in a patient with stiff person syndrome: a case report. PM R. 2015 Mar;7((3)):326–8. doi: 10.1016/j.pmrj.2014.10.013. [DOI] [PubMed] [Google Scholar]
- 17.Maramattom BV. Status spasticus and psoas muscle edema due to anti-GAD antibody associated stiff-man syndrome. Indian J Crit Care Med. 2015;19((8)):493. doi: 10.4103/0972-5229.162474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Castelnovo G, Renard D, Bouly S, Labauge P. Isolated hypertrophy of the tibialis anterior muscle in the stiff leg syndrome. Muscle Nerve. 2011 Aug;44((2)):306. doi: 10.1002/mus.22152. [DOI] [PubMed] [Google Scholar]
- 19.Baizabal-Carvallo JF, Jankovic J. Stiff-person syndrome: insights into a complex autoimmune disorder. J Neurol Neurosurg Psychiatry. 2015 Aug;86((8)):840–8. doi: 10.1136/jnnp-2014-309201. [DOI] [PubMed] [Google Scholar]
- 20.Sullivan CW, Desai K, Arain AR, Alley RM. A proximal humerus fracture dislocation in a patient with stiff person syndrome. Case Rep Orthop Res. 2019 Jul 9;2((1–3)):28–32. [Google Scholar]
- 21.Jamil M, Saleem M, Habib A, Khan S, Qahir M. Femur neck fracture in a patient with stiff person syndrome: a case report. JBJS Case Connect. 2019;9((3)):e0049e0049. doi: 10.2106/JBJS.CC.18.00049. [DOI] [PubMed] [Google Scholar]
- 22.Sullivan CW, Desai K, Arain AR, Alley RM. A proximal humerus fracture dislocation in a patient with stiff person syndrome. Case Rep Orthop Res. 2019;2((1–3)):28–32. [Google Scholar]
- 23.Dumitrascu OM, Tsimerinov EI, Lewis RA. Gastrointestinal and urologic sphincter dysfunction in stiff person syndrome. J Clin Neuromuscul Dis. 2016 Dec;18((2)):92–5. doi: 10.1097/CND.0000000000000143. [DOI] [PubMed] [Google Scholar]
- 24.Barker RA, Revesz T, Thom M, Marsden CD, Brown P. Review of 23 patients affected by the stiff man syndrome: clinical subdivision into stiff trunk (man) syndrome, stiff limb syndrome, and progressive encephalomyelitis with rigidity. J Neurol Neurosurg Psychiatry. 1998 Nov 1;65((5)):633–40. doi: 10.1136/jnnp.65.5.633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sun X, Tan J, Sun H, Liu Y, Guan W, Jia J, et al. Anti-SOX1 antibodies in paraneoplastic neurological syndrome. J Clin Neurol. 2020;16((4)):530. doi: 10.3988/jcn.2020.16.4.530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Graus F, Vincent A, Pozo-Rosich P, Sabater L, Saiz A, Lang B, et al. Anti-glial nuclear antibody: marker of lung cancer-related paraneoplastic neurological syndromes. J Neuroimmunol. 2005 Aug;165((1–2)):166–71. doi: 10.1016/j.jneuroim.2005.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Saltık S, Türkeş M, Tüzün E, Cakır A, Ulusoy C. Peripheral neuropathy associated with antiglutamic acid decarboxylase antibodies. Pediatr Neurol. 2013 May;48((5)):403–6. doi: 10.1016/j.pediatrneurol.2012.12.031. [DOI] [PubMed] [Google Scholar]
- 28.Li C, Wang X, Sun L, Deng H, Han Y, Zheng W. Anti‐SOX1 antibody‐positive paraneoplastic neurological syndrome presenting with Lambert‐Eaton myasthenic syndrome and small cell lung cancer: a case report. Thorac Cancer. 2020 Feb;11((2)):465–9. doi: 10.1111/1759-7714.13290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang RR, Han T, Guo F, Liu ZZ, Han YL, Chen WC, et al. Lambert-Eaton myasthenic syndrome in a patient with small-cell lung cancer: a case report. Oncol Lett. 2015 Sep;10((3)):1339–42. doi: 10.3892/ol.2015.3473. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental Video
Data Availability Statement
All data have been made available and presented in the case report.