Abstract
To date, studies which utilized ultrasound (US) and optoacoustic tomography (OT) fusion (US-OT) in biochemical differentiation of malignant and benign breast conditions have relied on limited biochemical data such as oxyhaemoglobin (OH) and deoxyhaemoglobin (DH) only. There has been no data of the largest biochemical components of breast fibroglandular tissue: lipid and collagen. Here, the authors believe the ability to image collagen and lipids within the breast tissue could serve as an important milestone in breast US-OT imaging with many potential downstream clinical applications. Hence, we would like to present the first-in-human US-OT demonstration of lipid and collagen differentiation in an excised breast tissue from a 38-year-old female.
Keywords: Collagen, Lipid, Breast, Optoacoustic, Tomography
1. Introduction
Optoacoustic Tomography (OT) is an innovative optical imaging modality which is based on the “photoacoustic effect” (i.e. formation of sound waves following light absorption [1]. As different tissue components react to light energy differently, OT can provide biochemical information of the imaged tissue by allowing endogenous differentiation of tissue contents. When combined with ultrasound (US), US-OT could generate fused ultrasound images of anatomy with optoacosutic images displaying biochemical content. OT has hence been hypothesized to be an ideal complement to breast US [2], [3]., as the biochemical data could be used to reduce the high number of false positives from US, which are secondary to known inter-operator variability (image acquisition and interpretation) and inherent differences in breast heterogeneity among patients [4].
To date, studies which utilized US-OT in biochemical differentiation of malignant and benign breast conditions have relied mainly on oxygenation status of haemoglobin [5], [6]. There has been limited data on the use of OT to visualize lipids [2], [7], [8], [9] and no data on collagen, which are the two large biochemical components of the breast [10]. Collagen derived from OT has been demonstrated to be clinically feasible in superficial organs such as the skeletal muscles for monitoring of diseases such as Duchene Muscular Dystrophy [11]. This could potentially be translatable to breast tissue, and the authors believe the ability to image both lipids and collagen from OT could serve as an important milestone in breast US-OT imaging with many potential downstream clinical applications. Some of these include but not limited to differentiation of benign and malignant lesions on breast US, pre-surgical staging (particularly for non-mass lesions like ductal carcinoma in-situ), or in an intra-operative setting to evaluate tumor margins.
Herein, we present a first-in human US-OT demonstration of lipid and collagen differentiation in an excised breast tissue from a 38-year-old female and its agreement to microscopic histological examination (i.e. gold standard). The scope of the study is limited to visualizing the OT derived biochemical components such as lipid and collagen in non-neoplastic breast tissues that were surgically removed during breast conserving surgery (BCS). The authors believe these findings could be extrapolated to abnormal breast conditions and fill the gaps in clinical applications where conventional imaging is insufficient.
2. Methods
2.1. Patient characteristic
Institutional board approval and informed consent was obtained. A 38-year-old patient presented to our local hospital with a palpable left breast lump and was diagnosed with invasive ductal carcinoma after core biopsy. She underwent conservative breast surgery and was noted to have involved/positive margins on histopathological examination. A re-excision of the superior margin was performed, and the fresh tissue specimen was collected and imaged with US-OT prior to histopathological examination.
2.2. Instrument and imaging protocol
An MSOT Invision 512-echo system (iThera Medical GmbH, Munich, Germany) coupled with a customized handheld probe (2D array of 256 detector elements arranged along an arc (1250), on a 40 mm radius spherical surface, and a centre frequency of 5 MHz) was used for imaging. The excised specimen was imaged from left-to-right along x-axis on the set-up demonstrated in Fig. 1A. Detailed description of imaging setup is discussed elsewhere [2].
Fig. 1.
Equipment set-up. (A) Schematic of the handheld US-OT probe placed in heavy water tank on the specimen. The probe is then moved sequentially along the x-axis, acquiring cross-sectional images of the specimen. The axis of image acquisition is conveyed to the pathologist who then sections the specimen in a similar orientation (see (C)). (B) Normalized absorption spectra of two main endogenous chromophores in excised breast specimens from 700 to 1100 nm based on which the acquired acoustic signals were unmixed to yield distribution maps of collagen (red) and lipid (green). (C) Cut sections of tissue specimen arranged from left to right (slice 1–8) showing fibrofatty tissue with patchy areas of fibrosis. Representative slices of 3, 7 and 8 were selected for illustrations in Fig. 2, Fig. 3, Fig. 4 respectively.
A real-time image preview window generated based on the backprojection algorithm was available during data acquisition. PA images were acquired at multiple wavelengths –, 700, 730, 760, 800, 850, 920, 930, 970, 1000, 1030, 1064 and 1100 nm – 5 frames were averaged per wavelength.
Data was processed using ViewMSOT 3.8, the available commercial software present in the system. Images obtained at different wavelengths were applied a bandpass filter with cut-off frequencies 50 kHz and 6.5 Mhz and reconstructed based on a back-projection algorithm. The reconstructed images were then linearly unmixed using the absorption spectra of two endogenous contrast agents – lipid and collagen (Fig. 1B), the spectra of which were preloaded in the commercial system. Different color look up tables (LUT) were used to represent their distribution: Lipid- green; Collagen - purple. Image processing was performed on ImageJ.
3. Results
Grossly, the specimen was a flat piece of fibrofatty tissue with cut sections showing patchy areas of fibrosis (Fig. 1C). Microscopically, the excised breast tissue showed areas of fibrosis, granulation tissue, inflammatory infiltrates, fat necrosis and foreign body-type giant cell reaction, compatible with surgical site changes. No residual carcinoma was identified.
US images of the excised breast tissue showed heterogeneity of fibroglandular tissue with patchy areas of hypoechoic and hyperechoic regions (Fig. 2, Fig. 3, Fig. 4C) which can be difficult to interpret. The acquired OT images (Fig. 2, Fig. 3, Fig. 4D) accurately depict areas of collagen (i.e. fibrous tissue) and lipid (i.e. fatty tissue) within the breast tissue with near identical correlation to histological specimens (Fig. 2, Fig. 3, Fig. 4A & B).
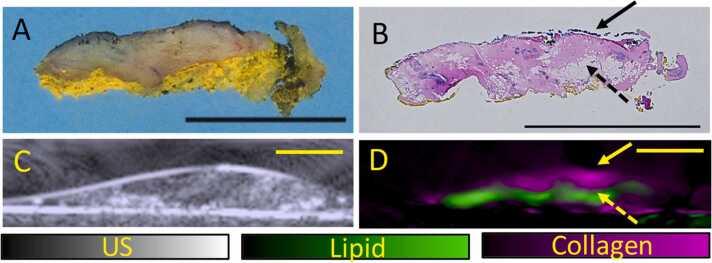
Fig. 2.
US and OT images of the excised tissue (slice 3) with histopathological correlation. (A) Gross (B) and microscopic (low power whole slide view) images of slice 3 of the re-excision specimen (black line represents 1 cm). There are dense areas of fibrosis mainly in the top portions of the specimen (bold arrow) with scattered areas of fat in the inferior portions of the specimen (dotted arrow) (C) 2D Ultrasound of the excised tissue demonstrates expected heterogeneity of the breast tissue. (D) OT images of the excised tissue shows collagen (bold arrow) and lipid (dotted arrow) distribution which correlates well with histopathology.
Fig. 3.
US and OT images of the excised tissue (slice 7) with histopathological correlation. (A) Gross (B) and microscopic (low power whole slide view) images of slice 7 of the re-excision specimen (black line represents 1 cm). There are dense areas of fibrosis in the right two-thirds of the specimen (bold arrow) with small areas of fat in the right one-third of the specimen (dotted arrow) (C) 2D Ultrasound of the excised tissue demonstrates expected heterogeneity of the breast tissue. (D) OT images of the excised tissue shows collagen (bold arrow) and lipid (dotted arrow) distribution which correlates well with histopathology.
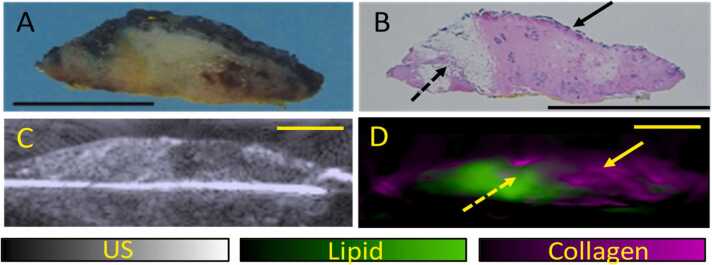
Fig. 4.
US and OT images of the excised tissue (slice 8) with histopathological correlation. (A) Gross and (B) microscopic (low power whole slide view) images of slice 8 of the re-excision specimen (black line represents 1 cm). There are patchy areas of fibrosis from the centre to the right of the specimen (bold arrow). Areas of fat can be seen in the left one-third of the specimen (dotted arrow). (C) 2D Ultrasound of the excised tissue demonstrates expected heterogeneity of the breast tissue. (D) OT images of the excised tissue shows collagen (bold arrow) and lipid (dotted arrow) distribution which correlates well with histopathology.
4. Discussion
Herein, the authors have successfully demonstrated the use of OT in differentiating biochemical components - collagen and lipid in a non-neoplastic breast tissue. It is noteworthy that OH and DH are not investigated in this study as there is no on-going blood blow. It is also noteworthy that other chromophores abundant in breast, namely elastin and water are also not included in this study. The former has similar absorption profile like collagen and is closely intertwined with collagen as supporting structures of the breast, making it seemingly impossible to differentiate between the two at the resolution offered by the current set-up (Fig. S1). As collagen is the dominant supporting structure in breast [10], the authors believe that OT-derived collagen can be a true representation of the collagen within the breast. Water is in abundance in all our body organs and the breast is not an exception. Collagen has a tight water-binding capacity and shows high uniform signal intensity in the mammary zone of the breast as demonstrated by Diot et. al [7]. This uniformity is exacerbated in our study (Fig. S1) by the abundance of water in the surrounding ultrasound gel as a coupling agent during image acquisition. Given its uniformity, water may not be a useful biomarker/chromophore in the ex-vivo setting but will likely play an important role in future in-vivo studies where cystic lesions of the breast are involved.
We have also demonstrated that OT derived collagen and lipid are in qualitative agreement with histopathology. While the correlation is accurate, it may not be perfect. This is because the tissue specimen was collected fresh and imaged prior to histological fixation. This was performed to ensure the integrity of the imaged specimen and to investigate feasibility of future clinical translation (e.g. evaluation of intra-operative tumor margins). Post fixation changes coupled with variability in sectioning of specimen would account for minor discrepancies in Fig. 2, Fig. 3, Fig. 4.
From a scientific perspective, the ability to visualize OT-derived lipid and collagen in breast tissue is a milestone for future radiology-pathology correlation. The combination of collagen, lipids and blood chromophores would intensify one’s understanding of biochemical patterns in different breast conditions on US in future in-vivo settings. For example, sclerosing adenosis or stromal fibrosis (benign fibrotic conditions) of the breast are localized area of fibrosis within the breast characterized by the presence of dense collagen bundles [10]. These benign conditions are unfortunately common mimickers of breast malignancy on US, resulting in unnecessary invasive procedures such as breast biopsies [4], [12]. In our representative case, the US-OT derived collagen was in agreement with the histopathology analysis showing the presence of dense collagen in areas of fibrosis within the normal excised breast tissue. From a clinical perspective, these findings could potentially assist in differentiating benign mimickers from true breast malignancies. Collagen also plays an important role in tumor microenvironment as demonstrated by Gole et. al [13], as increased stromal collagen has been demonstrated to facilitate breast cancer metastasis. The use of US-OT to offer biochemical information may hence be useful in pre-surgical staging for breast malignancies or even be translatable to the intra-operative setting for evaluation of tumor margins.
While results of this single case study may be promising, the current findings remain limited to an ex-vivo setting. As it is important to obtain full pathological correlation with US-OT images, excised tissues had to be obtained. Validation of these findings with larger samples would have to be performed to ensure reproducibility of results across breast tissues of different ages, different breast conditions (i.e. benign and malignant lesions) as well as tissues with different thickness. As the penetration depth of OT can be limited as compared to conventional US, significant improvements in devices as well as unmixing algorithms would have to made before direct translation into clinical in-vivo imaging can be achieved.
5. Conclusion
We have demonstrated using a commercial US-OT system, the successful visualization of lipid and collagen in a non-neoplastic breast tissue in the near infrared wavelength region. Distribution of lipid and collagen as visualised by US-OT is qualitatively in agreement with the gold standard microscopic histological examination. Extension of this clinical study to investigate more BCS specimens across several tumor types will not only establish the robustness of the technique but open a wide range of avenues to be explored for future applications.
Declaration of Competing Interest
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests:Goh Yonggeng has patent #IBB-016 issued to Licensee.
Acknowledgements
This work was supported by (1) Agency of Science, Technology and Research (A*STAR), under its BMRC Central Research Fund (UIBR) 2021 and (2) NMRC clinician-scientist individual research grant new investigator grant (CS-IRG NIG).
Footnotes
Supplementary data associated with this article can be found in the online version at doi:10.1016/j.pacs.2022.100377.
Contributor Information
Malini Olivo, Email: Malini_Olivo@sbic.a-star.edu.sg.
Swee Tian Quek, Email: swee_tian_quek@nuhs.edu.sg.
Appendix A. Supplementary material
Supplementary material Fig. S1 US and OT images of excised tissue (slice 7) with elastin and water distribution. (i) Gross (ii) and microscopic (low power whole slide view) images of slice 7 of the re-excision specimen (black line represents 1 cm), (iii) 2D Ultrasound of the excised tissue demonstrates expected heterogeneity of the breast tissue. (iv) OT images of the excised tissue showing distribution of collagen (magenta) and lipid (green), (v) OT images of the excised tissue showing distribution of elastin (red) and lipid (green) distribution when spectrum of collagen was replaced by elastin while unmixing and (vi) OT images of the excised specimen showing distribution of water when unmixed for water in addition to lipid and collagen.
.
Data Availability
Data will be made available on request.
References
- 1.Bell A.G. The production of sound by radiant energy. Science. 1881;2(49):242–253. doi: 10.1126/science.os-2.49.242. [DOI] [PubMed] [Google Scholar]
- 2.Balasundaram G., Goh Y., Moothanchery M., et al. Optoacoustic characterization of breast conserving surgery specimens - a pilot study. Photoacoustics. 2020;19 doi: 10.1016/j.pacs.2020.100164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Menezes G.L.G., Pijnappel R.M., Meeuwis C., et al. Downgrading of breast masses suspicious for cancer by using optoacoustic breast imaging. Radiology. 2018;288(2):355–365. doi: 10.1148/radiol.2018170500. [DOI] [PubMed] [Google Scholar]
- 4.Brem R.F., Lenihan M.J., Lieberman J., Torrente J. Screening breast ultrasound: past, present. Future AJR Am. J. Roentgenol. 2015;204(2):234–240. doi: 10.2214/AJR.13.12072. [DOI] [PubMed] [Google Scholar]
- 5.Becker A., Masthoff M., Claussen J., et al. Multispectral optoacoustic tomography of the human breast: characterisation of healthy tissue and malignant lesions using a hybrid ultrasound-optoacoustic approach. Eur. Radio. 2018;28(2):602–609. doi: 10.1007/s00330-017-5002-x. [DOI] [PubMed] [Google Scholar]
- 6.Butler R., Lavin P.T., Tucker F.L., et al. Optoacoustic breast imaging: imaging-pathology correlation of optoacoustic features in benign and malignant breast masses. AJR Am. J. Roentgenol. 2018;211(5):1155–1170. doi: 10.2214/AJR.17.18435. [DOI] [PubMed] [Google Scholar]
- 7.Diot G., Metz S., Noske A., et al. Multispectral optoacoustic tomography (MSOT) of human breast cancer. Clin. Cancer Res. 2017;23(22):6912–6922. doi: 10.1158/1078-0432.CCR-16-3200. [DOI] [PubMed] [Google Scholar]
- 8.Goh Y., Balasundaram G., Moothanchery M., et al. Ultrasound guided optoacoustic tomography in assessment of tumor margins for lumpectomies. Transl. Oncol. 2020;13(2):254–261. doi: 10.1016/j.tranon.2019.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kosik I., Brackstone M., Kornecki A., Chamson-Reig A., Wong P., Carson J.J.L. Lipid-weighted intraoperative photoacoustic tomography of breast tumors: volumetric comparison to preoperative MRI. Photoacoustics. 2020;18 doi: 10.1016/j.pacs.2020.100165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Martinez-Hernandez A., Francis D.J., Silverberg S.G. Elastosis and other stromal reactions in benign and malignant breast tissue: an ultrastructural study. Cancer. 1977;40(2):600–606. doi: 10.1002/1097-0142(197708)40:2<700::aid-cncr2820400217>3.0.co;2-w. [DOI] [PubMed] [Google Scholar]
- 11.Regensburger A.P., Fonteyne L.M., Jungert J., et al. Detection of collagens by multispectral optoacoustic tomography as an imaging biomarker for Duchenne muscular dystrophy. Nat. Med. 2019;25(12):1905–1915. doi: 10.1038/s41591-019-0669-y. [DOI] [PubMed] [Google Scholar]
- 12.Guirguis M.S., Adrada B., Santiago L., Candelaria R., Arribas E. Mimickers of breast malignancy: imaging findings, pathologic concordance and clinical management. Insights Imaging. 2021;12(1):53. doi: 10.1186/s13244-021-00991-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Gole L., Yeong J., Lim J.C.T., et al. Quantitative stain-free imaging and digital profiling of collagen structure reveal diverse survival of triple negative breast cancer patients. Breast Cancer Res. 2020;22(1):42. doi: 10.1186/s13058-020-01282-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary material Fig. S1 US and OT images of excised tissue (slice 7) with elastin and water distribution. (i) Gross (ii) and microscopic (low power whole slide view) images of slice 7 of the re-excision specimen (black line represents 1 cm), (iii) 2D Ultrasound of the excised tissue demonstrates expected heterogeneity of the breast tissue. (iv) OT images of the excised tissue showing distribution of collagen (magenta) and lipid (green), (v) OT images of the excised tissue showing distribution of elastin (red) and lipid (green) distribution when spectrum of collagen was replaced by elastin while unmixing and (vi) OT images of the excised specimen showing distribution of water when unmixed for water in addition to lipid and collagen.
Data Availability Statement
Data will be made available on request.