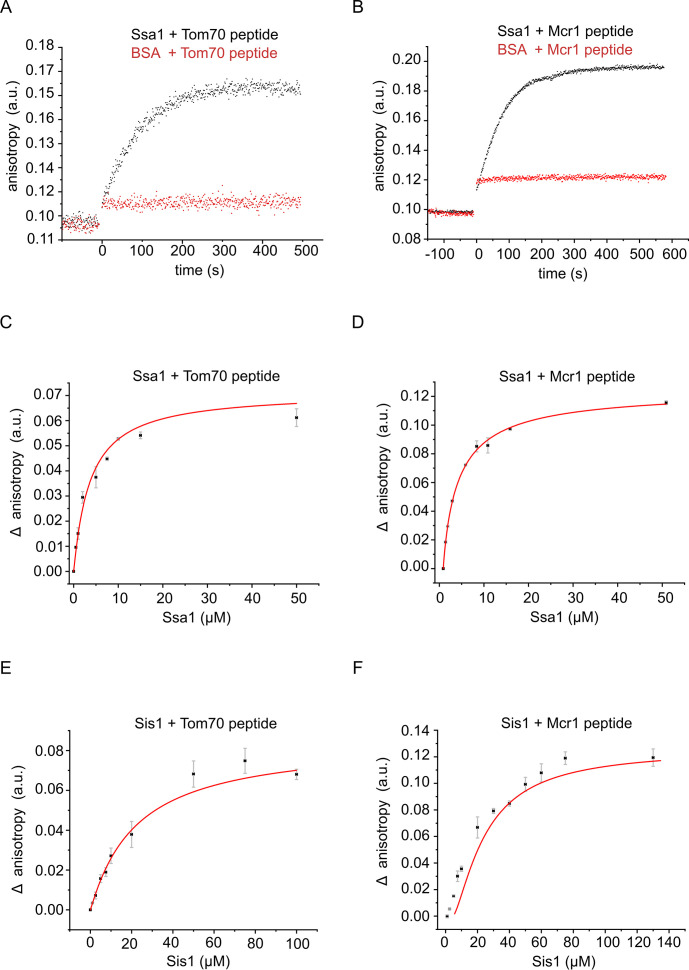
Figure 6. The hydrophobic segment of the signal-anchored proteins interacts with the Hsp70 chaperone and its co-chaperone Sis1.
(A and B) The fluorescence anisotropy of TAMRA-labeled peptides corresponding to the TMSs of either Tom70 (A) or Mcr1 (B) was measured in the presence of 10 µM of the Hsp70 Ssa1 (black circles) or 30 µM BSA, as a control (red circles). (C–F) For affinity determinations, the TMS-labelled peptides of either Tom70 (C and E) or Mcr1 (D and F) were mixed with the indicated concentrations of either Ssa1 (C and D) or Sis1 (E and F), and the difference in anisotropy (Δ anisotropy) between the bound and free peptide was plotted against the (co)chaperone concentrations.