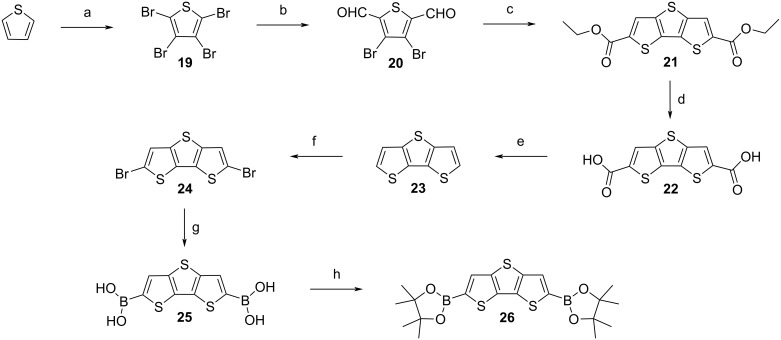
Scheme 1.
Synthetic route from thiophene to 2,6-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)dithieno[3,2-b:2’,3’-d]thiophene (26): a) Br2, CHCl3, rt, overnight, reflux, 4 h, 94% [33]; b) n-BuLi, −78 °C, 30 min, 1-formylpiperidine, anhydrous THF, −78 °C, then rt, overnight [15], 88%; c) ethyl thioglycolate, anhydrous potassium carbonate, anhydrous N,N-dimethylformamide, rt, 3 d, 83% [15]; d) 1 M aqueous lithium hydroxide, THF, 4 h, 94% [15]; e) copper powder, quinoline, 230 °C, 1 h, 81% [15]; f) N-bromosuccinimide, CHCl3/glacial acetic acid, 0 °C, 1 h, rt, 1.5 h, 94% [15] ; g) n-BuLi, −90 °C, 20 min, triisopropyl borate, −80 °C, anhydrous THF, rt, overnight, 97% [34]; h) pinacol, toluene, 115 °C, 21.5 h, 79% [35].