Abstract

Crystal growth from anhydrous HF solutions of M2+ (M = Ca, Sr, Ba) and [AuF6]− (molar ratio 1:2) gave [Ca(HF)2](AuF6)2, [Sr(HF)](AuF6)2, and Ba[Ba(HF)]6(AuF6)14. [Ca(HF)2](AuF6)2 exhibits a layered structure in which [Ca(HF)2]2+ cations are connected by AuF6 units, while the crystal structure of Ba[Ba(HF)]6(AuF6)14 exhibits a complex three-dimensional (3-D) network consisting of Ba2+ and [Ba(HF)2]2+ cations bridged by AuF6 groups. These results indicate that the previously reported M(AuF6)2 (M = Ca, Sr, Ba) compounds, prepared in the anhydrous HF, do not in fact correspond to this chemical formula. When the initial M2+/[AuF6]− ratio was 1:1, single crystals of [M(HF)](H3F4)(AuF6) were grown for M = Sr. The crystal structure consists of a 3-D framework formed by [Sr(HF)]2+ cations associated with [AuF6]− and [H3F4]− anions. The latter exhibits a Z-shaped conformation, which has not been observed before. Single crystals of M(BF4)(AuF6) (M = Sr, Ba) were grown when a small amount of BF3 was present during crystallization. Sr(BF4)(AuF6) crystallizes in two modifications. A high-temperature α-phase (293 K) crystallized in an orthorhombic unit cell, and a low-temperature β-phase (150 K) crystallized in a monoclinic unit cell. For Ba(BF4)(AuF6), only an orthorhombic modification was observed in the range 80–230 K. An attempt to grow crystals of Ca(BF4)(AuF6) failed. Instead, crystals of [Ca(HF)](BF4)2 were grown and the crystal structure was determined. During prolonged crystallization of [AuF]6– salts, moisture can penetrate through the walls of the crystallization vessel. This can lead to partial reduction of Au(V) to A(III) and the formation of [AuF4]− byproducts, as shown by the single-crystal growth of [Ba(HF)]4(AuF4)(AuF6)7. Its crystal structure consists of [Ba(HF)]2+ cations connected by AuF6 octahedra and square-planar AuF4 units. The crystal structure of the minor product [O2]2[Sr(HF)]5[AuF6]12·HF was also determined.
Short abstract
Crystal growth from anhydrous HF solutions of M2+ (M = Ca, Sr, Ba) and [AuF6]− (molar ratio 1:2) gave [Ca(HF)2](AuF6)2 and Ba[Ba(HF)]6(AuF6)14. These results indicate that the previously reported M(AuF6)2 (M = Ca, Sr, Ba) compounds do not correspond to this chemical formula. When the initial Sr2+/[AuF6]− ratio was 1:1, crystals of [Sr(HF)](H3F4)(AuF6) were grown. Single crystals of M(BF4)(AuF6) (M = Sr, Ba) were obtained when a small amount of BF3 was present during crystallization. The crystal structures of [Ba(HF)]4(AuF4)(AuF6)7, [O2]2[Sr(HF)]5[AuF6]12·HF, and Ca(BF4)(AuF6) byproducts were also determined.
Introduction
Although gold can occur in various oxidation states from −1 to +5, its chemistry is dominated by oxidation states (I) and (III).1,2 For most people, the only interesting oxidation state of gold is Au0, where we naturally think of the metal gold. It has attracted people for thousands of years because of its lustrous appearance. One of the goals of alchemists was to turn some other metals into gold. However, it is also interesting to scientists because of many other aspects. It is a unique element because it has very large relativistic effects, greater than any other element with Z < 100.3 This has a major effect on the chemical properties of gold and its compounds.4,5 An example is the stability of alkali or alkaline-earth metal aurides (RbAu, CsAu, CsAu·NH3, and BaAu2)5,6 and other compounds with Au– anions such as the exotic oxides M3AuO (M = K, Rb, Cs).7
Gold(I) has been used extensively in various organic gold complexes that have been shown to have physiological therapeutic value, in contrast to gold(III), whose organic complexes have been shown to be toxic.8 With the exception of AuF, all other gold(I) halides are known in the condensed state.1,9 The molecular AuF was characterized in the gas phase.10 In the solid state, it was stabilized by an N-heterocyclic carbene ligand.11 In F3As–Au+SbF6– there are strong cationic–anionic interactions through a fluorine atom.12 Therefore, the description as F3As–Au–F···SbF5 is also possible. Reports of true gold(II) compounds are sparse.13−16 A number of apparent gold(II) complexes are actually mixed gold(I)–gold(III) compounds; AuCl2 is Au2Au2IIICl8.1 Gold(III) is probably the most prominent oxidation state. AuF3, AuCl3, and AuBr3 are known in the condensed state.1,17 There are a large number of Au(III) salts containing the square-planar AuF4– anion.18−21 Of the gold oxides, only the brown Au2O3 is of some importance. This Au(III) oxide is thermodynamically unstable, but its decay is kinetically inhibited up to a temperature of about 150–170 °C.7 On the other hand, there are a large number of organogold(III) compounds.1,8,22,23 The experimental preparation of gold(IV) compounds is still beyond our capabilities. The published structure of bis-benzene-1,2-dithiolato-Au(IV)24 later proved to be a compound of Au(III).25 A somewhat more recent report of Au(IV) compounds with similar ligands26 is therefore still awaiting confirmation, and the preparation of AuF4 is still a domain of theoretical chemistry.27,28 Gold(V) has only been observed in AuF5 and various [AuF6]− salts.1,2 It is the highest known oxidation state of gold.29 Since the first report of [AuF6]− salts ([Xe2F11][AuF6] and CsAuF6) in 1972,30 fewer than 40 other examples have become known (Table S1). [Xe2F11][AuF6] was synthesized by fluorination of AuF3 in the presence of excess XeF6 at elevated temperatures.29 Room-temperature syntheses require the use of strong oxidizing species such as KrF2,31 UV-irradiated F2,32 or O2F radicals.33 The reactions are usually carried out in anhydrous HF (aHF) as the solvent. Besides the crystal structure of AuF5,34 the number of known crystal structures of other Au(V) compounds is very limited. Examples with nonmetallic cations include [AuF6]− salts of [O2]+ (low and high-temperature forms),35 [KrF]+,34 and [Xe2F11]+.36 X-ray single-crystal structures have also been determined for some [AuF6]− salts with M+ and M+2 metal cations. Well-known examples include the crystal structures of KAuF6,37 M(AuF6)2 (M = Cd,36 Hg38), Mg(HF)AuF4AuF6,36 and AgFAuF6.37
Reactions between KrF2 and mixtures of MF2 and metallic Au (molar ratio 1:2) in aHF (aHF) as the solvent were assumed to lead to M(AuF6)2 (M = Ca, Sr, Ba) salts.39 These reactions were later reexamined using two different approaches, that is, KrF2 and UV-irradiated F2 in aHF.40,41 From the powder X-ray diffraction data, the Ca-salt was found to crystallize in the tetragonal unit cell and Sr and Ba salts in the cubic system.38,42 In all cases, pure M(AuF6)2 salts were assumed to be isolated. However, in one work, it was already suspected that the Ba salt was not a true Ba(AuF6)2 compound.39 The next problem was that the Raman spectra of these salts from different sources only partially agreed.38,39,41 For this reason, we made a great effort to prepare the M(AuF6)2 crystals from the corresponding solutions. We also investigated the possibility of preparing MFAuF6 (M = Ca, Sr, Ba) and M(BF4)(AuF6) salts with mixed anions. Unintentionally, single crystals of [O2]2[Sr(HF)]5[AuF6]12·HF were grown and the crystal structure was determined. The results of these experiments are described in the present work.
Results and Discussion
Crystals were grown from saturated solutions prepared in two similar ways. In the first method, “M(AuF6)2” salts were prepared by reactions between MF2/2AuF3 and KrF2 or UV-irradiated F2 in aHF.39 After yellow solutions were obtained, the volatile phases were pumped off. The solid products were recovered, characterized, and redissolved in aHF to obtain saturated solutions from which crystals were attempted to grow (Table S2). In the second method, MF2/2AuF3 mixtures were treated with UV-irradiated F2 in aHF until yellow solutions were obtained and crystallization was attempted without prior isolation (Table S3). In both cases, trace amounts of oxygen or moisture in the reaction mixture or in the crystal growth solutions were the main problem. If present during synthesis, O2AuF634 can be easily formed as a byproduct. During prolonged crystallization, moisture can penetrate through the FEP reaction vessel walls.43−46 This can lead to partial reduction of Au(V) to Au(III) and formation of [AuF4]− byproducts. For example, instead of the desired single crystals of Mg(AuF6)2, single crystals of [Mg(HF)](AuF4)(AuF6) were grown.36
The major products grown from aHF solutions of Ca2+ and [AuF6]− (molar ratio 1:2) were single crystals of [Ca(HF)2](AuF6)2. The Raman spectra, recorded on the single crystals (Figures 1 and S1), show similar features to those reported for Ca(AuF6)2.38,39,41
Figure 1.
Raman spectrum of powdered [Ca(HF)2](AuF6)240 and Raman spectrum of [Ca(HF)2](AuF6)2 recorded on single crystals checked using a diffractometer.
There are three possibilities: (a) both salts have very similar spectra; (b) the single crystals of [Ca(HF)2](AuF6)2 were partially decomposed at the surface and covered with Ca(AuF6)2; (c) the Raman spectra really belong to [Ca(HF)2](AuF6)2. We believe that the third option is correct. This is in agreement with the results obtained in other MF2/XF5 (M = Ca, Sr, Ba; X = As, Sb, Ta, Ru) systems, where the crystallization products from the HF saturated solution always contained HF coordinated to Ca, Sr, or Ba atoms. Some examples are [Ca(HF)n](AsF6)2 (n = 1, 6),47 [Ca(HF)2](SbF6)2,48 [Sr(HF)3](TaF6)2,49 [Ba(HF)](RuF6)2,48 and [Ba(HF)](AsF6)2.50 To date, no crystals of M(XF6)2 (M = Ca, Sr, Ba; X = As, Sb, Ta, Ru) have been prepared so far. In some of the Ca2+/[AuF6]− crystallizations, crystals containing [AuF4]− anions were observed (Table S2). The resulting compound has the formula Ca(AuF4)(AuF6). Some Au5+ was reduced upon contact with moisture that can penetrate through the FEP reaction vessel walls. An attempt to grow single crystals of the Ca-salt with a lower content of [AuF6]− [i.e., CaFAuF6 or Ca[HnFn+1](AuF6)] failed. At a lower content of [AuF6]− (i.e., initial molar ratio Ca2+/[AuF6]− = 1:1), only [Ca(HF)2](AuF6)2 and [Ca(HF)](AuF4)(AuF6) were detected in the crystallization products.
In the case of strontium, crystals of [Sr(HF)2](AuF6)2 and [Sr(HF)]2(AuF4)(AuF6)3 were grown from solutions in which the Sr2+/[AuF6]− molar ratio was equal to 1:2 (Table S2). At a molar ratio of 1:1, Sr(H2F3)(AuF6) and [Sr(HF)](H3F4)(AuF6) were observed. In some experiments, single crystals of [O2]2[Sr(HF)]5[AuF6]12·HF were grown. This was due to the contamination of the reaction mixture with oxygen during the photochemical preparation of the crystallization solution by UV-irradiated F2 in aHF.
Crystallizations from Ba2+/[AuF6]− hydrogen fluoride solutions (molar ratios 1:1 and 1:2) resulted in crystal growth of Ba[Ba(HF)]6(AuF6)14 and [Ba(HF)]4(AuF4)(AuF6)7 (Figure S2). When a 1:1 ratio was used, single crystals of Ba(H3F4)251 were also observed.
In an attempt to prepare M(BF4)(AuF6) salts with mixed anions, a small amount of BF3 was added to the crystallization mixture. Single crystals of M(BF4)(AuF6) (M = Sr, Ba) were successfully grown (Figures S3 and S4), while in the case of calcium, single crystals of [Ca(HF)](BF4)2 (Figure S5) and a yellow crystalline product of unknown composition (Figure S6) were detected.
Crystal Structures
The corresponding crystal data and refinement results for [Ca(HF)2](AuF6)2, Ba[Ba(HF)]6(AuF6)14, [Sr(HF)](H3F4)(AuF6), [Ba(HF)]4(AuF4)(AuF6)7, M(BF4)(AuF6) (M = Sr, Ba), [Ca(HF)](BF4)2, and [O2]2[Sr(HF)]5[AuF6]12·HF are summarized in Table 1.
Table 1. Summary of Crystal Data and Refinement Results of [Ca(HF)2](AuF6)2, Ba[Ba(HF)]6(AuF6)14, [Sr(HF)](H3F4)(AuF6), [Ba(HF)]4(AuF4)(AuF6)7, M(BF4)(AuF6) (M = Sr, Ba), [Ca(HF)](BF4)2, and [O2]2[Sr(HF)]5[AuF6]12·HF Compounds.
| chem. formula | [Ca(HF)2]–(AuF6)2 | Ba[Ba(HF)]6–(AuF6)14 | [Sr(HF)]–(H3F4)(AuF6) | [Ba(HF)]4–(AuF4)(AuF6) | [Ca(HF)]–(BF4)2 |
|---|---|---|---|---|---|
| crystal system | triclinic | rhombohedral | monoclinic | tetragonal | triclinic |
| space group | P1̅ | R3̅ | P21/m | I41/a | P1̅ |
| a (Å) | 5.6598(3) | 19.8309(6) | 5.8708(2) | 11.1380(3) | 5.1827(4) |
| b (Å) | 8.8838(7) | 19.8309(6) | 8.1261(3) | 11.1380(3) | 6.5414(6) |
| c (Å) | 10.1017(5) | 14.2361(4) | 8.6531(3) | 32.0799(11) | 9.7870(7) |
| α (deg) | 93.884(5) | 90 | 90 | 90 | 108.679(7) |
| β (deg) | 91.773(4) | 90 | 92.399(4) | 90 | 91.057(6) |
| γ (deg) | 97.742(5) | 120 | 90 | 90 | 93.386(7) |
| V (Å3) | 501.72(5) | 4848.5(3) | 412.45(3) | 3979.7(3) | 313.53(5) |
| Z | 2 | 3 | 2 | 4 | 2 |
| T (K) | 150 | 150 | 150 | 150 | 150 |
| R1a | 0.0398 | 0.0410 | 0.0225 | 0.0263 | 0.0535 |
| wR2b | 0.1051 | 0.1204 | 0.0539 | 0.0601 | 0.1404 |
| chem. formula | α-Sr(BF4)–(AuF6) | β-Sr(BF4)–(AuF6) | Ba(BF4)–(AuF6)c | [O2]2[Sr(HF)]5–[AuF6]12·HF |
|---|---|---|---|---|
| crystal system | monoclinic | orthorhombic | orthorhombic | monoclinic |
| space group | P21/c | Pnma | Pnma | P21/n |
| a (Å) | 6.1193(3) | 10.2969(5) | 10.4846(3) | 21.8471(10) |
| b (Å) | 11.4512(6) | 6.1506(4) | 6.4416(2) | 10.5392(4) |
| c (Å) | 10.2122(5) | 11.4529(6) | 11.7209(4) | 27.0291(12) |
| α (deg) | 90 | 90 | 90 | 90 |
| β (deg) | 90.106(4) | 90 | 90 | 104.565(5) |
| γ (deg) | 90 | 90 | 90 | 90 |
| V (Å3) | 715.60(6) | 725.34(7) | 791.60(4) | 6023.5(5) |
| Z | 1 | 4 | 4 | 4 |
| T (K) | 150 | 293 | 150 | 150 |
| R1a | 0.0245 | 0.0298 | 0.0275 | 0.0409 |
| wR2b | 0.0470 | 0.0731 | 0.0646 | 0.0911 |
R1 = Σ∥Fo| – |Fc∥/Σ|Fo| for I > 2σ(I).
wR2 = [Σ[w(Fo2 – Fc2)2]/Σw(Fo2)2]1/2.
The crystal structures determined at 80 and 296 K are the same as at 150 K.
Crystal Structure of [Ca(HF)2](AuF6)2
[Ca(HF)2](AuF6)2 crystallizes in the triclinic space group (P1̅, Z = 2 at 150 K) and is not isotypic with [Ca(HF)2](SbF6)2 (monoclinic, P21/n at 200 K).47 [Ca(HF)2](AuF6)2 has a layered structure in which Ca atoms are linked by AuF6 units (Figure 2).
Figure 2.

Packing of slabs in the crystal structure of [Ca(HF)2](AuF6)2 (yellow octahedra: AuF6; red circles: Ca; green circles: F; small colorless circles: H).
Eight fluorine atoms provided by six AuF6 units and two HF molecules coordinate each of the crystallographically distinct Ca atoms (Figure 3). The Ca–F(−H) distances are 2.371(6) and 2.415(7) Å, and the Ca–F(−AuF5) bond lengths range from 2.302(6) to 2.443(6) Å. The corresponding bond lengths in [Ca(HF)2](SbF6)2, where Ca is also eight-coordinated, are 2.304(3)/2.315(3) Å for Ca–F(−H) and 2.328(3)–2.430(3) Å for Ca–F(−SbF5).47 There are two crystallographically nonequivalent Au atoms (Figure S7). The Au–Fb bond lengths between gold and the bridging fluorine atoms (Au–Fb–Ca) are elongated (1.913(6)–1.929(5) Å) compared to the Au–Ft bonds between gold and the terminal fluorine atoms (Ft: 1.871(6)–1.887(6) Å). Two of the latter are slightly elongated (1.883(5) and 1.887(6) Å), which is due to hydrogen bonding with the HF molecule. There are intralayer and interlayer F–H···F–AuF5 hydrogen bonds (the F···F distances for the former are 2.676(9) and 2.713(8) Å for the latter).
Figure 3.

Coordination of Ca2+ atoms in the crystal structure of [Ca(HF)2](AuF6)2.
Crystal Structure of Ba[Ba(HF)]6(AuF6)14
The crystal structure of Ba[Ba(HF)]6(AuF6)14 shows a complex three-dimensional network (Figure 4) consisting of two crystallographically distinct Ba atoms (Figures 5 and 6, and S8), three crystallographically independent AuF6 groups (Figure S9), and HF molecules bonded to Ba atoms via their F atom.
Figure 4.

Rhombohedral unit cell and packing of cations, anions, and HF molecules in the crystal structure of Ba[Ba(HF)]6(AuF6)14. For clarity, Ba–F bonds are not shown (yellow octahedra: AuF6; red circles: Ba; green circles: F; small colorless circles: H).
Figure 5.

12-fold coordination of the Ba1 atom in the crystal structure of Ba[Ba(HF)]6(AuF6)14. The thermal ellipsoids are drawn at the 50% probability level. The symmetry operations are (i) 1/3 + x, −1/3 + y, −1/3 + z; (ii) 4/3 – y, 2/3 + x – y, −1/3 + z; (iii) 1/3 – x + y, 2/3 – x, −1/3 + z; (iv) 1 – x, 1 – y, 1 – z; (v) −1/3 + y, 1/3 – x + y, 4/3 – z; (vi) 2/3 – x + y, 4/3 – x, 1/3 + z; (vii) 5/3 – x, 4/3 – y, 4/3 – z.
Figure 6.

12-fold coordination of the Ba2 atom in the crystal structure of Ba[Ba(HF)]6(AuF6)14. The thermal ellipsoids are drawn at the 50% probability level. The symmetry operations are (i) 1 – x, 1 + x – y, z; (ii) −x + y, 1 – x, z; (iii) −1/3 + y, 1/3 – x + y, 4/3 – z; (iv) 2/3 – x, 4/3 – y, 4/3 – z; (v) 2/3 + x – y, 1/3 + x, 4/3 – z; (vi) 1 – y, 1 + x – y, z.
The peculiarity is that one of the crystallographically unique Ba atoms (Ba1) is coordinated by a HF molecule, and the other (Ba2) is not. The Ba1/Ba2 ratio is 1:6, and the chemical formula is therefore Ba[Ba(HF)]6(AuF6)14. Classical chemical elemental analysis of such a salt would give the formula Ba(AuF6)2·0.86HF and based on this formula one would expect some kind of HF-solvate to form. In such cases, only determination of the crystal structure can reveal the true nature of such a compound. Both Ba atoms are coordinated 12-fold by fluorine atoms provided by seven AuF6 units and one HF molecule for Ba1 (Figure 5) and eight AuF6 units for Ba2 (Figure 6). The Ba–F(AuF5) bonds range from 2.748(7) to 3.009(8) Å, while the Ba–F(H) bond is the shortest (2.703(7) Å) and thus the strongest of all Ba–F bonds. The F–H···F–AuF5 hydrogen bond is quite strong with H···F and F···F distances of 1.74(4) and 2.614(10) Å, respectively.
Crystal Structure of [Sr(HF)](H3F4)(AuF6)
The crystal structure of [Sr(HF)](H3F4)(AuF6) crystallizes in the monoclinic space group P21/m. The crystal structure consists of a three-dimensional framework of Sr2+ cations associated with [AuF6]− and [H3F4]− anions and HF molecules (Figure 7).
Figure 7.

Monoclinic unit cell and packing of Sr2+ cations, [AuF6]− and [H3F4]− anions, and HF molecules in the crystal structure of [Sr(HF)](H3F4)(AuF6). For clarity, the Sr–F bonds are not shown (yellow octahedra: AuF6; red circles: Sr; green circles: F; small colorless circles: H).
The coordination of the Sr atom consists of nine fluorine atoms provided by four [AuF6]−, four [H3F4]− anions, and one HF molecule (Figures 8 and S10). The Sr–F(AuF5) bonding contacts are in the range of 2.565(4)–2.612(3) Å. The bond contacts between the Sr atom and the F atoms of the [H3F4]− anions are shorter (2.490(3)–2.505(3) Å), while the Sr–F(H) bond distance (2.522(5) Å) lies between these values. The reported Sr–F(H) bond lengths in the crystal structure of Sr(HF)3(TaF6)2 (200 K) have similar values (2.51(2)–2.55 (2) Å).48
Figure 8.

Ninefold coordination of the Sr atom in the crystal structure of [Sr(HF)](H3F4)(AuF6). The thermal ellipsoids are drawn at the 50% probability level. The symmetry operations are (i) 1 – x, 1 – y, 2 – z; (ii) −x, 1/2 + y, 2 – z; (iii) 1 + x, 1/2 – y, z; (iv) x, 1/2 – y, z; (v) 1 – x, 1/2 + y, 1 – z; (vi) −1 + x, y, z; (vii) 1 – x, −1/2 + y, 1 – z.
In the octahedral AuF6 unit, one Au–F(H) and four Au–Fb(−Sr) (Figure S11) are in the range of 1.880(3)–1.905(4) Å, while the sixth Au–Ft bond is shorter (1.866(4) Å). The HF molecule bonded to the Sr atom is involved in hydrogen bonding with the F atom of the AuF6 unit (Figure S11). The F···F distance between the F atoms involved in the hydrogen bond is 2.596(6) Å.
In the [H3F4]− anion (Figure 9), the H6 atoms are located closer to the two terminal F atoms (F6; 1.02 Å) than to the central ones (F7; 1.45 Å). The H7 atom is located in the middle of the F7 atoms (1.150 Å). Therefore, the appropriate structural formula is [(FH)(FHF)(HF)]−. Previously reported [H3F4]− isomers have branched- or linear-chain geometry.50,52−55 The linear [H3F4]− isomers in KF·2.5HF51 and [Sr(HF)](H3F4)(AuF6) have different conformations. The former has a U-shaped conformation,51 while the latter has a Z-shaped conformation (Figure 9).
Figure 9.
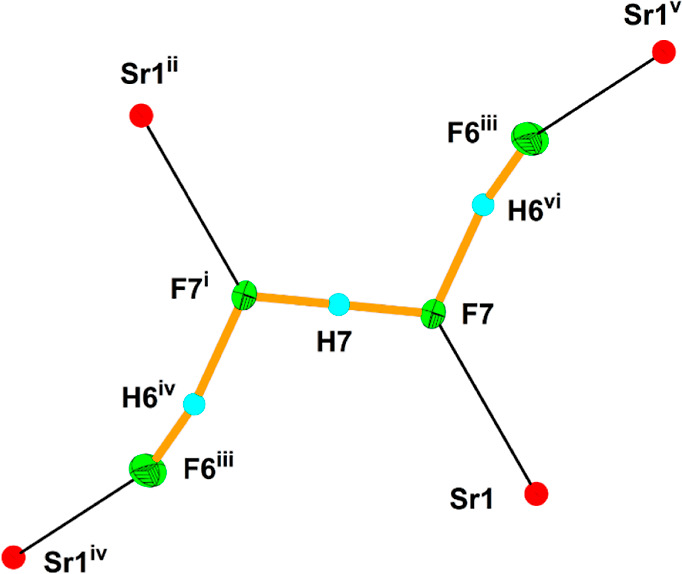
[H3F4]− anion and its interactions with Sr2+ cations in the crystal structure of [Sr(HF)](H3F4)(AuF6). The thermal ellipsoids are drawn at the 50% probability level. The symmetry operations are (i) 1 – x, 1 – y, 2 – z; (ii) 1 – x, 1/2 + y, −z; (iii) 1 + x, 1/2 – y, z; (iv) −x, 1/2 + y, 2 – z; (v) 1 + x, y, z; (vi) 1 + x, 1/2 – y, z.
Crystal Structure of [Ba(HF)]4(AuF4)(AuF6)7
The crystal structure of [Mg(HF)](AuF4)(AuF6)36 was the first example of a [AuF4]−/[AuF6]− salt with mixed anions, while the crystal structure of [Ba(HF)]4(AuF4)(AuF6)7 is the second example. It consists of crystallographically unique Ba2+ cations connected by three crystallographically independent AuF6 octahedra, as well as unique AuF4 plaques and HF molecules (Figures 10 and S12).
Figure 10.

Packing of Ba2+ cations, [AuF6]− and [AuF4]− anions, and HF molecules in the crystal structure of [Ba(HF)]4(AuF4)(AuF6)7. For clarity, the Ba–F bonds are not shown. Only one position is shown for each disordered F atom (yellow octahedra: AuF6; blue plaques: AuF4; red circles: Ba; green circles: F; small blue circles: H).
The fluorine atoms of two AuF6 units (Au3 and Au4) are strongly disordered at several positions. In the case of the Au(3)F6 unit, two cis-located fluorine centers bonded to Ba cations are ordered, while each of the other four F atoms is distributed in three positions (looks like a whole octahedron—with the exception of bridging fluorines—wobble). An even greater disorder was observed for the Au(4)F6 unit: six fluorine atoms are distributed over 24 positions. For this reason, the detailed discussion of some bond distances is not very realistic. Six AuF6 units give eight fluorine ligands bonded to one Ba atom. Two F atoms, one of the AuF4 units, and one of the HF molecules complete the 10-fold coordination of Ba2+ (Figure S13). Each of the fluorine atoms of the [AuF4]− anion serves as a bridging ligand connecting the gold atom (Au1) to the Ba atom (Figure 11). The Au–F bond lengths in the AuF4 unit (4 × 1.900(5) Å) are comparable to those observed in [Mg(HF)](AuF4)(AuF6) (1.898(7)–1.916(7) Å).36 The HF molecule is coordinated to the Ba atom through its fluorine atom. The hydrogen atom is involved in a hydrogen bond with the F atom of the AuF6 group (Au3).
Figure 11.

[AuF4]− anion and its interactions with Ba2+ cations in the crystal structure of [Ba(HF)]4(AuF4)(AuF6)7. The thermal ellipsoids are drawn at the 50% probability level. The symmetry operations are (i) 3/4 + y, 5/4 – x, 5/4 – z; (ii) 2 – x, 1/2 – y, z; (iii) 5/4 – y, −3/4 + x, 5/4 – z. Crystal structures of M(BF4)(AuF6) (M = Sr, Ba).
Crystal Structures of M(BF4)(AuF6) (M = Sr, Ba).
Sr(BF4)(AuF6) crystallizes in two modifications (Figures 12 and S14). A high-temperature β-phase (structure determined at 293 K) crystallized in an orthorhombic unit cell, and a low-temperature α-phase (structure determined at 150 K) crystallized in a monoclinic unit cell (Table 1). No phase transition was observed for Ba(BF4)(AuF6) in the range of 80–230 K. Only one orthorhombic modification was observed, which is isotypic to the high-temperature modification of the Sr salt. This type is isotypic to the already known Ba(BF4)(AsF6) (150 and 200 K).53 Both the monoclinic and orthorhombic modifications have similar features; that is, their crystal structure is a 3-D framework consisting of the M2+ cations (M = Sr, Ba) connected by AuF6 octahedra and BF4 tetrahedra (Figure 12). Since this type of structure has already been well described,53 it will not be discussed in too much detail here.
Figure 12.

Packing of Sr2+ cations and [AuF6]− and [BF4]− anions in the crystal structure of monoclinic α-Sr(BF4)(AuF6) (yellow octahedra: AuF6; blue tetrahedra: BF4; red circles: Sr; green circles: F).
In both modifications, the M2+ cations are ninefold coordinated by fluorine atoms provided by five AuF6 groups and four BF4 groups (Figures S15 and S16). In the β-phase of Sr(BF4)(AuF6), the AuF6– anion exhibits a twofold rotational disorder of the terminal fluorine positions about the F(4)–Sb–F(5) axis with equal occupancy for both orientations. The same type of AuF6– disorder was observed in the crystal structure of the Ba(BF4)(AuF6) salt. In the latter, three atoms of the BF4 unit are disordered over two positions.
Crystal Structure of [Ca(HF)](BF4)2
Single crystals of [Ca(HF)](BF4)2 were obtained in an attempt to grow crystals of Ca(BF4)(AuF6). In [Ca(HF)](BF4)2, the molecule HF is coordinated to the Ca atom through its fluorine atom (Figure 13). An earlier example of this type was [Sr(HF)](BF4)2.48 It is not isotypic to the Ca-salt.
Figure 13.

Packing of Ca2+ cations, [BF4]− anions, and HF molecules in the crystal structure of [Ca(HF)](BF4)2 (blue tetrahedra: BF4; red circles: Sr; green circles: F; small colorless circles: H).
In [Ca(HF)](BF4)2, the Ca atom is coordinated by eight fluorine atoms of seven BF4 units and one HF ligand (Figures 14 and S17). The coordination number of the Ca metal in pure Ca(BF4)2 is the same (Figure S17), but, of course, the fluorine atoms are provided only by BF4 units.56 The Ca–F(H) bond distance is longer (2.428(2) Å) than the Ca–F(−BF3) bond lengths (2.322(2)–2.365(2) Å). The B–Fb bond lengths between boron and the bridging fluorine atoms are longer (1.384(4)–1.398(4) Å) compared to the B–Ft bonds between boron and the terminal fluorine atom (1.379(4) Å) involved in hydrogen bonding with the HF molecule (Figure S18). This hydrogen bonding appears to be quite strong, with F···H and F···F distances of 1.90(8) and 2.507(3) Å, respectively.
Figure 14.

Eightfold coordination of the Ca atom in the crystal structure of [Ca(HF)](BF4)2. The thermal ellipsoids are drawn at the 50% probability level. The symmetry operations are (i) 1 + x, y, z; (ii) 1 – x, 1 – y, 1 – z; (iii) 2 – x, 1 – y, −z; (iv) 1 – x, 1 – y, −z; (v) x, −1 + y, z. Crystal structure of [O2]2[Sr(HF)]5[AuF6]12·HF.
Crystal Structure of [O2]2[Sr(HF)]5[AuF6]12·HF
Single crystals of [O2]2[Sr(HF)]5[AuF6]12·HF were accidentally grown in an attempt to grow single crystals of Sr2+/[AuF6]− salts (Table S1). Their crystal structure can be described as a 3-D framework consisting of [Sr(HF)]2+ cations linked by [AuF6]− anions. The O2+ cations and the non-coordinated HF molecules are located inside the cavities (Figure 15). The packing of Sr2+ cations and [AuF6]− anions in [O2]2[Sr(HF)]5[AuF6]12·HF is very similar to the packing of Sr2+ cations and [AsF6]− anions in [Co(HF)2]Sr[Sr(HF)]2[Sr(HF)2]2[AsF6]12 (abbreviated as CoSr5(AsF6)12·8HF; Figure S19). The unit-cell parameters of the latter are also very close to those of [O2]2[Sr(HF)]5[AuF6]12·HF.57
Figure 15.

Packing of [Sr(HF)]2+ and O2+ cations, [AuF6]− anions, and HF molecules in the crystal structure of [O2]2[Sr(HF)]5[AuF6]12·HF (yellow octahedra: AuF6; red circles: Sr; green circles: F; blue circles: O; small colorless circles: H).
There are five crystallographically independent Sr2+ cations (Figure S20) and 12 crystallographically unique AuF6 units. Each of the Sr atoms is ninefold coordinated by fluorine atoms of one HF molecule and six or eight AuF6 units. The Sr–F bond lengths (2.428(7)–2.751(8) Å) are in the same range as in [Sr(HF)](H3F4)(AuF6) and the Au–F bond distances (1.853(8)–1.914(7) Å) are comparable to those in other [AuF6]− salts. The O–O bond lengths (1.03(1) and 1.01(1) Å) are close to those determined in other O2+-salts.58
Conclusions
Attempts to grow solvent-free single crystals of M(AuF6)2 salts (M = Ca, Sr, Ba)38,39 from anhydrous hydrogen fluoride (aHF) solutions failed. Instead, crystals of M2+ salts with coordinated HF (via the F atom) were obtained. From the comparison of the previously reported Raman spectra of M(AuF6)238,39 with the spectra recorded on single crystals (Figures S1 and S2), it can be concluded that the former (which were also prepared in aHF), do not correspond to this chemical formula. The report on the M(AuF6)2 salts appeared in 1990.38 At that time, it was already known that binary fluorides, when dissolved in superacids (i.e., aHF acidified with AsF5, SbF5, etc.), gave solutions of solvated [M(HF)n]x+ cations and [XF6]− anions (X = As, Sb, etc.),59,60 but examples of [M(HF)n]x+([XF6]−)x salts in the condensed state were not known. It was assumed that HF is too weak a coordinating ligand for such salts to be stabilized in the solid state. The crystal structure of La(HF)2(AsF6)3 (published in 1998) was the first example in which HF was coordinated directly to a metal center via its F atom.61 Later, many more examples followed, and today, it is a well-known fact that crystallizations from aHF solutions can give [M(HF)n]x+ salts.48,56 In 2000, two different types of Raman spectra (types A and B) were reported for Ba(AuF6)2, depending on whether KrF2 or UV-irradiated F2 was used to fluorinate a BaF2/2AuF3 mixture in aHF.39 It was suggested that one of the phases might contain [Ba(HF)n]2+ or [Ba(KrF2)n]2+. Since the recent Raman spectra recorded on single crystals of Ba[Ba(HF)]6(AuF6)14 (in the preparation of which KrF2 was not used) are in agreement with the reported spectra,39 the latter possibility is now ruled out. Here, we must mention that the existence of MII(AuVF6)2·nKrF2 (MII = Ca, Sr, Ba; n = 0–4) compounds has been reported in the past.41,62−65 They are reported to be stable at room temperature and have been partially characterized. They were described as clathrates, but the true nature of these compounds was never established. The first real evidence for the existence of compounds with a KrF2 ligand coordinated to a metal center (e.g., [Mg(KrF2)4(AsF6)2] and [Mg(KrF2)4(AsF6)2]·2BrF5) was reported in 2017.66
Another problem in the synthesis of [AuF6]− salts is the possible formation of byproducts, such as [O2]+ salts (i.e., O2AuF6). Their presence can lead to more complex compounds, for example, [O2]2[Sr(HF)]5[AuF6]12·HF. The second problem is the extreme sensitivity of Au(V) compounds to moisture or other impurities, which can lead to partial reduction of Au(V) to Au(III) and the formation of mixed-anion salts [AuF4]−/[AuF6]−.
Attempts to prepare MFAuF6 salts (M = Ca, Sr, Ba) also failed. Instead, [M(HF)n]x+ salts with [AuF6]− and poly(hydrogen fluoride) anions were obtained [e.g., [Sr(HF)](H3F4)(AuF6)]. When a small amount of BF3 is added, [BF4]−/[AuF6]− salts with mixed anions are obtained (M(BF4)(AuF6), M = Sr, Ba). A similar attempt to synthesize the corresponding Ca-salt gave [Ca(HF)](BF4)2.
It appears that it is not possible to prepare M(XF6)2 or MFXF6 (M = Ca, Sr, Ba; X = As, Sb, Au, etc.) from aHF solutions. Ca, Sr, and Ba prefer higher coordination numbers. Achieving such a high coordination number with only [XF6]− ligands would lead to a very crowded environment, which is energetically less favorable than having some HF in the coordination sphere.
Together with the reported crystal structure, the preliminary results of the crystal structures of Sr(HF)(AuF6)2, Sr(H2F3)(AuF6), Ca(AuF4)(AuF6), and [Sr(HF)]2(AuF6)3(AuF4) (Figures S21–S24) suggest that more phases can be expected in the M2+ (M = Ca, Sr, Ba)/[AuF6]− system.
Experimental Section
CAUTION: aHF and some fluorides are highly toxic and must be handled in a well-ventilated hood. Protective clothing must be worn at all times!
Materials and Methods
Reagents
Commercially available reagents BF3 Union Carbide Austria (GmbH, 99.5%), CaF2 (Merck), SrF2 (Alfa Aesar, 99.99%), and BaF2 (Alfa Aesar, 99.995%) were used as supplied. AuF3 was synthesized by the reaction of AuCl3 (Alfa Aesar, 99.99%) with elemental fluorine F2 (Solvay Fluor and Derivate GmbH, 99.98%) in aHF (Linde AG, Pullach, Germany, 99.995%) at room temperature. The “M(AuF6)2“ salts (M = Ca, Sr, Ba) were prepared as previously described.39
Synthetic Apparatus
All manipulations were performed under anhydrous conditions. Nonvolatile materials were handled in an M. Braun glovebox in an argon atmosphere in which the amount of water did not exceed 0.5 ppm. Gaseous F2 and volatile compounds such as aHF and BF3 were handled on a vacuum line constructed from nickel and polytetrafluoroethylene (PTFE).
The vessels used for single-crystal growth were made of tetrafluoroethylene–hexafluoropropylene block-copolymer (FEP; Polytetra GmbH, Germany) tubes. The crystallization vessel consisted of two FEP tubes: one with an inner diameter of 16 mm and an outer diameter of 19 mm and the other with an inner diameter of 4 mm and an outer diameter of 6 mm. Each tube was heat-sealed at one end and connected via linear PTFE connections to a PTFE T-part at 90°. The PTFE valve was attached to the T-part at a 180° angle to the 19 mm o.d. tube. All PTFE parts of the valve were enclosed in brass with threads that prevented deformation of the PTFE parts of the valve and facilitated connection to the reaction vessels and vacuum system. PTFE-coated magnetic stir bars were placed inside the reaction vessels. The temperature gradient between the two arms of the crystallization vessels was maintained by cooling a wider arm of a vessel in the Huber Ministat 230 (to −33 °C).
Before use, all reaction and crystallization vessels were dried under dynamic vacuum and passivated with elemental fluorine F2 at 1 bar for 2 h. aHF was treated with K2NiF6 (Advance Research Chemicals Inc, 99.9%) for several hours before use and stored in FEP vessels over K2NiF6.
Synthesis and Crystal Growth
Solid starting reagents “M(AuF6)2”39 or MF2/nAuF3 (M = Ca, Sr, Ba) mixtures were loaded into reaction vessels in a dry box (Table S1 in the Supporting Information). The solvent (HF, 2–6 mL) was condensed to “M(AuF6)2” (M = Ca, Sr, Ba) at 77 K and warmed to ambient temperature. Similarly, HF (4–6 mL) was condensed onto the MF2/nAuF3 (M = Ca, Sr, Ba; n = 1, 2) reaction mixtures. Fluorine was slowly added to the reaction vessel at room temperature until a pressure of 4 bar was reached. In some experiments, a small amount of BF3 was added to prepare the mixed-anion salts [BF4]−/[AuF6]−. A medium-pressure mercury lamp (Hg arc lamp, 450 W, Ace Glass, USA) was used as the UV source.39 The reaction mixture was stirred at room temperature for 1–5 days. After clear yellow solutions were obtained, the fluorine was pumped off. For the purpose of crystallization, the clear solutions were decanted into the 6 mm o.d. sidearm in all cases. Evaporation of the solvent from this side arm was achieved by maintaining a temperature gradient of about 10–20 °C between the two tubes for several weeks. Slow distillation of aHF from the 6 mm o.d. tube into the 19 mm o.d. tube resulted in crystal growth within the 6 mm o.d. tube.
The crystals were treated in different ways. Some crystals were immersed in perfluorodecalin (melting point 263 K) in a dry box, selected under a microscope, and mounted on the goniometer head of the diffractometer in a cold nitrogen stream (265–273 K). Others were sealed in quartz capillaries used for structure determination at room temperature and for recording Raman spectra at several random positions. A special method was used to isolate the crystals of some batches (Table S3). After pumping out the volatiles at low temperature, a small amount (∼0.5–1 mL) of cold (278 K) perfluorinated oil (perfluorodecaline C10F18) was injected into the narrower FEP tube to cover the crystals. Then, the crystals covered with the cold oil were selected under the microscope and mounted on the goniometer head of the diffractometer in a cold nitrogen stream (265–273 K).
Characterization Methods
Raman Spectroscopy
Raman spectra with a resolution of 0.5 cm–1 were recorded at room temperature using a Horiba Jobin Yvon LabRam-HR spectrometer equipped with an Olympus BXFM-ILHS microscope. Samples were excited with the 632.8 nm emission line of a He–Ne laser with a regulated power in the range of 20–0.0020 mW, resulting in 17–0.0017 mW focused through a 50× microscope objective on a 1 μm spot onto the top surface of the sample.
Single crystals or the powdered material were mounted in the glovebox in previously vacuum-dried quartz capillaries, which were first sealed with Halocarbon 25-5S grease (Halocarbon Corp.) inside the glovebox and later heat-sealed in an oxygen–hydrogen flame outside the glovebox.
Single-Crystal X-ray Diffraction Analysis
Single-crystal X-ray data for [Ca(HF)2](AuF6)2, Ba[Ba(HF)]6(AuF6)14, Sr(H3F4)(AuF6)·HF, [Ba(HF)]4(AuF4)(AuF6)7, M(BF4)(AuF6) (M = Sr, Ba), [Ca(HF)](BF4)2, [O2]2[Sr(HF)]5[AuF6]12·HF, Sr(HF)2(AuF6)2, Sr(H2F3)(AuF6), Ca(AuF4)(AuF6), and [Sr(HF)]2(AuF6)3(AuF4) were collected on a Gemini A diffractometer equipped with an Atlas charge-coupled device (CCD) detector, using graphite monochromated Mo Kα radiation. The data were processed using the CrysAlisPro software suite program package.67 Analytical absorption corrections were applied to all data sets. The structure of Sr(H2F3)(AuF6) was solved using the SHELXS program.68 All other structures were solved using the dual-space algorithm of the SHELXT69 program implemented in the Olex crystallographic software.70 Structure refinement was performed using the SHELXL-2014 software.71 The hydrogen atoms in the structures of Ba[Ba(HF)]6(AuF6)14 and [Ca(HF)](BF4)2 were found on difference Fourier maps, and their positional and thermal parameters were freely refined in [Ca(HF)](BF4)2. In Ba[Ba(HF)]6(AuF6)14, the thermal parameter of the hydrogen atom was constrained to 1.2 U of the connected fluorine atom. The crystals of the salts Sr(HF)(AuF6)2, Sr(H2F3)(AuF6), Ca(AuF4)(AuF6), and [Sr(HF)]2(AuF6)3(AuF4) were of poor quality or multiple twins. Therefore, only the unit-cell parameters are given (Figures S21–S24). Figures were made with Balls & Sticks72 and DIAMOND 4.6 software.73
CCDC 2129976 ([Ba(HF)]4(AuF4)(AuF6)7), 2129977 ([Ca(HF)2](AuF6)2), 2129978 (α-Sr(BF4)(AuF6)), 2129979 (β-Sr(BF4)(AuF6)), 2129980 (Ba[Ba(HF)]6(AuF6)14), 2129981 ([Ca(HF)](BF4)2), 2129982 ([O2]2[Sr(HF)]5[AuF6]12·HF), 2129983 (Ba(BF4)(AuF6)), and 2129984 ([Sr(HF)](H3F4)(AuF6)) contain the supplementary crystallographic data for this work. These data can be obtained free of charge via www.ccdc.cam.ac.uk/data_request/cif, by emailing at data_request@ccdc.cam.ac.uk, or by contacting The Cambridge Crystallographic Data Centre, 12 Union Road, Cambridge CB2 1EZ, UK; fax +44 1223 336033.
Acknowledgments
The authors acknowledge the financial support from the Slovenian Research Agency (research core funding no. P1-0045; Inorganic Chemistry and Technology).
Supporting Information Available
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acs.inorgchem.2c01675.
List of known [AuF6]- salts, additional experimental details, and some results of characterization by Raman spectroscopy and X-ray diffraction analysis (PDF)
Author Contributions
Conceptualization and methodology, validation, formal analysis, synthesis, Z.M.; crystal structure determination, E.G.; writing—original draft preparation, Z.M.; writing—review and editing, Z.M. and E.G.; visualization and supervision, Z.M.; All authors have read and agreed to the published version of the manuscript.
The authors declare no competing financial interest.
Supplementary Material
References
- Cotton S. A.Chemistry of Precious Metals; Blackie Academic & Professional: London, 1997; pp 273–327. [Google Scholar]
- Mohr F. The chemistry of gold-fluoro compounds: a continuing challenge for gold chemists. Gold Bull. 2004, 37, 164–169. 10.1007/bf03215208. [DOI] [Google Scholar]
- Pyykkö P. Theoretical Chemistry of Gold. Angew. Chem., Int. Ed. 2004, 43, 4412–4456. 10.1002/anie.200300624. [DOI] [PubMed] [Google Scholar]
- Bartlett N. Relativistic effects and the chemistry of gold. Gold Bull. 1998, 31, 22–25. 10.1007/bf03215471. [DOI] [Google Scholar]
- Pyykkö P. Relativity, gold, closed-shell interactions, and CsAu•NH3. Angew. Chem., Int. Ed. 2002, 41, 3573–3578. . [DOI] [PubMed] [Google Scholar]
- Zachwieja U. Single-crystal growth and structure refinement of RbAu and CsAu. Z. Anorg. Allg. Chem. 1993, 619, 1095–1097. 10.1002/zaac.19936190621. [DOI] [Google Scholar]
- Müller-Buschbaum H. On the crystal chemistry of oxoaurates. Z. Anorg. Allg. Chem. 2002, 628, 2559–2584. . [DOI] [Google Scholar]
- The Chemistry of Organic Derivatives of Gold and Silver; Patai S., Rapporot Z., Eds.; John Willey & Sons, LTD: Chichester, 1999. [Google Scholar]
- Eklund K.; Kuklin M. S.; Kraus F.; Karttunen A. J. Evolutionary algorithm-based crystal structure prediction for gold(I) fluoride. ChemPhysChem 2020, 21, 802–808. 10.1002/cphc.201901070. [DOI] [PubMed] [Google Scholar]
- Schröder D.; Hrušák J.; Tornieporth-Oetting I. C.; Klapötke T.; Schwarz H. Neutral gold(I) fluoride does indeed exist. Angew. Chem., Int. Ed. 1994, 33, 212–214. 10.1002/anie.199402121. [DOI] [Google Scholar]
- Laitar D. S.; Müller P.; Gray T. G.; Sadighi J. P. A carbene stabilized gold(I) fluoride: synthesis and theory. Organometallics 2005, 24, 4503–4505. 10.1021/om050619f. [DOI] [Google Scholar]
- Küster R.; Seppelt K. Preparation and structure of F3As–Au+SbF6–, the structures of Au(CO)2+ and Au(PF3)2+. Z. Anorg. Allg. Chem. 2000, 626, 236–240. . [DOI] [Google Scholar]
- Elder S. H.; Lucier G. M.; Hollander F. J.; Bartlett N. Synthesis of Au(II) fluoro complexes and their structural and magnetic properties. J. Am. Chem. Soc. 1997, 119, 1020–1026. 10.1021/ja9630654. [DOI] [Google Scholar]
- Drews T.; Seidel S.; Seppelt K. Gold-xenon complexes. Angew. Chem., Int. Ed. 2002, 41, 454–456. . [DOI] [PubMed] [Google Scholar]
- Wickleder M. S. AuSO4: a true gold(II) sulfate with an Au24+ ion. Z. Anorg. Allg. Chem. 2001, 627, 2112–2114. . [DOI] [Google Scholar]
- Schmidt R.; Müller B. G. Einkristalluntersuchungen an Au[AuF4]2 und CeF4, zwei unerwarteten Nebenprodukten. Z. Anorg. Allg. Chem. 1999, 625, 605–608. . [DOI] [Google Scholar]
- Kurzydłowski D.; Kobyakov S.; Mazej Z.; Pillai S. B.; Chakraborty B.; Jha P. K. Unexpected persistence of cis-bridged chains in compressed AuF3. Chem. Commun. 2020, 56, 4902–4905. 10.1039/d0cc01374a. [DOI] [PubMed] [Google Scholar]
- Bialowons H.; Müller B. G. Synthesis and structure of MII[AuF4]2 with MII = Ni, Pd. Z. Anorg. Allg. Chem. 1997, 623, 434–438. 10.1002/zaac.19976230169. [DOI] [Google Scholar]
- Schmidt R.; Müller B. G. Single crystal investigations on the fluoroaurates Cs[AuF4], Cs[Au2F7] and U2F7[AuF4]. Z. Anorg. Allg. Chem. 2004, 630, 2393–2397. 10.1002/zaac.200400123. [DOI] [Google Scholar]
- Fitz H.; Müller B. G. Synthesis and structure of tetrafluoroaurates(III), TlF2[AuF4], MF2[AuF4]5 (M = Bi, La, Y), Sm[AuF4]3 with an appendix on Sm[AuF4]2. Z. Anorg. Allg. Chem. 2002, 628, 126–132. . [DOI] [Google Scholar]
- Ellwanger M. A.; Steinhauer S.; Golz P.; Beckers H.; Wiesner A.; Braun-Cula B.; Braun T.; Riedel S. Taming the high reactivity of gold(III) fluoride: fluorido gold(III) complexes with N-based ligands. Chem.—Eur. J. 2017, 23, 13501–13509. 10.1002/chem.201702663. [DOI] [PubMed] [Google Scholar]
- Ellwanger M. A.; von Randow C.; Steinhauer S.; Zhou Y.; Wiesner A.; Beckers H.; Braun T.; Riedel S. Tuning the Lewis acidity of difluorido gold(III) complexes: the synthesis of [AuClF2(SIMes)] and [AuF2(OTeF5)(SIMes)]. Chem. Commun. 2018, 54, 9301–9304. 10.1039/c8cc05233f. [DOI] [PubMed] [Google Scholar]
- Pérez-Bitrián A.; Martínez-Salvador S.; Baya M.; Casas J. M.; Martín A.; Menjjn B.; Orduna J. Anionic derivatives of perfluorinated trimethylgold. Chem.—Eur. J. 2017, 23, 6919–6929. 10.1002/chem.201700927. [DOI] [PubMed] [Google Scholar]
- Rindorf G.; Thorup N. Structure of Bis(benzene-l,2-dithiolato)gold(IV). Acta Crystallogr., Sect. C: Cryst. Struct. Commun. 1990, 46, 1437–1439. 10.1107/s0108270189013430. [DOI] [Google Scholar]
- Ray K.; Weyhermüller T.; Goossens A.; Crajé M. W. J.; Wieghardt K.; Do S. S’-coordinated o-dithiobenzosemiquinonate(1-) radicals exist in coordination compounds? The [AuIII(1,2-C6H4S2)2]1-/0 couple. Inorg. Chem. 2003, 42, 4082–4087. 10.1021/ic0207092. [DOI] [PubMed] [Google Scholar]
- Belo D.; Alves H.; Branco Lopes E.; Duarte M. T.; Gama V.; Teives Henriques R.; Almeida M.; Pérez-Benítez A.; Rovira C.; Veciana J. Gold complexes with dithiothiophene ligands: a metal based on a neutral molecule. Chem.—Eur. J. 2001, 7, 511–519. . [DOI] [PubMed] [Google Scholar]
- Lin J.; Zhang S.; Guan W.; Yang G.; Ma Y. Gold with +4 and +6 oxidation states in AuF4 and AuF6. J. Am. Chem. Soc. 2018, 140, 9545–9550. 10.1021/jacs.8b04563. [DOI] [PubMed] [Google Scholar]
- Liu G.; Feng X.; Wang L.; Redfern S. A. T.; Yong X.; Gao G.; Liu H. Theoretical investigation of the valence states in Au via the Au–F compounds under high pressure. Phys. Chem. Chem. Phys. 2019, 21, 17621–17627. 10.1039/c9cp02409c. [DOI] [PubMed] [Google Scholar]
- Riedel S.; Kaupp M. The highest oxidation states of the transition metal elements. Coord. Chem. Rev. 2009, 253, 606–624. 10.1016/j.ccr.2008.07.014. [DOI] [Google Scholar]
- Leary K.; Bartlett N. A new oxidation state of gold: the preparation and some properties of [AuF6]− salts. J. Chem. Soc., Chem. Commun. 1972, 903–904. 10.1039/c39720000903. [DOI] [Google Scholar]
- Holloway J. H.; Schrobilgen G. J. Krypton fluoride chemistry;; a route to AuF5, KrF+AuF6–, Xe2F3+AuF6– , and NO+AuF6–: the KrF2 – XeOF4 system. J. Chem. Soc., Chem. Commun. 1975, 623–624. 10.1039/c39750000623. [DOI] [Google Scholar]
- Graudejus O.; Elder S. H.; Lucier G. M.; Shen C.; Bartlett N. Room temperature syntheses of AuF6– and PtF6– salts, Ag+AuF6–, Ag2+PtF6–, and Ag2+PdF6–, and an estimate for E(MF6–) [M = Pt, Pd]. Inorg. Chem. 1999, 38, 2503–2509. 10.1021/ic981397z. [DOI] [Google Scholar]
- Lucier G. M.; Chen S.; Elder S. H.; Bartlett N. Facile routes to NiF62–, AgF4–, AuF6–, and PtF6–, salts using O2+ as a source of O2F in anhydrous HF. Inorg. Chem. 1998, 37, 3829–3834. 10.1021/ic971603n. [DOI] [PubMed] [Google Scholar]
- Hwang I.-C.; Seppelt K. Gold pentafluoride: structure and fluoride ion affinity. Angew. Chem., Int. Ed. 2001, 40, 3690–3693. . [DOI] [PubMed] [Google Scholar]
- Lehmann J. F.; Schrobilgen G. J. Structural and vibrational characterization of [KrF][AuF6] and α-[O2][AuF6] using single crystal X-ray diffraction, Raman spectroscopy and electron structure calculations. J. Fluorine Chem. 2003, 119, 109–124. 10.1016/s0022-1139(02)00220-8. [DOI] [Google Scholar]
- Leary K.; Zalkin A.; Bartlett N. Crystal structure of Xe2F11+AuF6– and the Raman spectrum of Xe2F11+. Inorg. Chem. 1974, 13, 775–779. 10.1021/ic50134a003. [DOI] [Google Scholar]
- Mazej Z.; Goreshnik E. X-ray single crystal structures of Cd(AuF6)2, Mg(HF)AuF4AuF6 and KAuF6 and vibrational spectra of Cd(AuF6)2 and KAuF6. Solid State Sci. 2006, 8, 671–677. 10.1016/j.solidstatesciences.2006.02.032. [DOI] [Google Scholar]
- Mazej Z.; Goreshnik E.; Tavčar G. X-ray single crystal structures of Hg(AuF6)2 and AgFAuF6. J. Fluorine Chem. 2011, 132, 686–689. 10.1016/j.jfluchem.2011.03.022. [DOI] [Google Scholar]
- Popov A. I.; Val’kovskii M. D.; Kiselev Yu. M.; Tchumaevsky N. A.; Sokolov V. B.; Spirin S. N. Structures of fluoroaurates(V) of alkaline earth elements. Zh. Neorg. Khim. 1990, 35, 1970–1977. [Google Scholar]
- Mazej Z. Recent achievements in the synthesis and characterization of metal hexafluoroantimonates and hexafluoroaurates. J. Fluorine Chem. 2004, 125, 1723–1733. 10.1016/j.jfluchem.2004.09.008. [DOI] [Google Scholar]
- Mazej Z.Photochemical Syntheses of Fluorides in Liquid Anhydrous Hydrogen Fluoride. In Modern Synthesis Processes and Reactivity of Fluorinated Compounds; Groult H., Leroux F., Tressaud A., Eds.; Elsevier: London, 2017; p 587. [Google Scholar]
- Nabiev S. S.; Sokolov V. B.; Spirin S. N.; Chaivanov B. B. Synthesis and spectral properties of hexafluoroaurates. Russ. J. Phys. Chem. A 2011, 85, 1931–1941. 10.1134/S0036024411110215. [DOI] [Google Scholar]
- Mazej Z.; Goreshnik E. A. Crystal structures of dioxonium lanthanoid(III) pentakis(tetrafluoridoborates) of lanthanum and cerium. J. Fluorine Chem. 2017, 196, 3–6. 10.1016/j.jfluchem.2016.05.014. [DOI] [Google Scholar]
- Baxter A. F.; Schaab J.; Christe K. O.; Haiges R. Perfluoroalcohols: the preparation and crystal structures of heptafluorocyclobutanol and hexafluorocyclobutane-1,1-diol. Angew. Chem., Int. Ed. 2018, 57, 8174–8177. 10.1002/anie.201804101. [DOI] [PubMed] [Google Scholar]
- Kostiuk N.; Goettel J. T.; Gerken M. Synthesis and characterization of SF4 adducts with polycyclic amines. Inorg. Chem. 2020, 59, 8620–8628. 10.1021/acs.inorgchem.0c01105. [DOI] [PubMed] [Google Scholar]
- Bortolus M. R.; Mercier H. P. A.; Schrobilgen G. J. Group 6 oxyfluoro-anion salts of [XeF5]+ and [Xe2F11]+: syntheses and structures of [XeF5][M2O2F9] (M=Mo, W), [Xe2F11][M’OF5] (M’=Cr, Mo, W), [XeF5][HF2]·CrOF4, and [XeF5][WOF5]·XeOF4. Chem.—Eur. J. 2020, 26, 8935–8950. 10.1002/chem.202000826. [DOI] [PubMed] [Google Scholar]
- Tramšek M.; Benkič P. Calcium hexafluoroarsenates with metal coordinated HF molecules. Acta Chim. Slov. 2004, 51, 769–778. [Google Scholar]
- Turičnik A.; Benkič P.; Žemva B. Syntheses and crystal structures of [Mg(HF)2](SbF6)2 and [Ca(HF)2](SbF6)2: new examples of HF acting as a ligand to metal centers. J. Fluorine Chem. 2003, 121, 245–251. 10.1016/s0022-1139(03)00052-6. [DOI] [Google Scholar]
- Tramšek M.; Goreshnik E.; Lozinšek M.; Žemva B. HF molecules and poly(hydrogen fluoride) anions as ligands to metal centers. J. Fluorine Chem. 2009, 130, 1093–1098. 10.1016/j.jfluchem.2009.06.008. [DOI] [Google Scholar]
- Tramšek M.; Mazej Z.; Goreshnik E.; Tavčar G. Crystal structure determination of Pb2F2(HF)(SbF6)2, PbFSbF6 and Ba(HF)(AF6)2: (A = As, Sb). J. Fluorine Chem. 2015, 175, 18–21. 10.1016/j.jfluchem.2015.03.002. [DOI] [Google Scholar]
- Bunič T.; Tramšek M.; Goreshnik E.; Žemva B. Barium hydrogen tetrafluoride of the composition Ba(H3F4)2: The first example of homoleptic HF metal environment. Solid State Sci. 2006, 8, 927–931. 10.1016/j.solidstatesciences.2006.02.045. [DOI] [Google Scholar]
- Mootz D.; Boenigk D. Fluorides and fluoroacids. 12. Complex-anion homology and isomerism in the crystal structures of two potassium poly(hydrogen fluorides), KF•2.5HF and KF•3HF. J. Am. Chem. Soc. 1986, 108, 6634–6636. 10.1021/ja00281a030. [DOI] [Google Scholar]
- Mootz D.; Poll W. Fluorides and fluoro acids. V. Crystal structure of the 1:4 phase in the system water-hydrogen fluoride and a new investigation of one of the 1:2 phases. Z. Anorg. Allg. Chem. 1982, 484, 158–164. 10.1002/zaac.19824840113. [DOI] [Google Scholar]
- Lozinšek M.; Bunič T.; Goreshnik E.; Meden A.; Tramšek M.; Tavčar G.; Žemva B. Syntheses, crystal structures and Raman spectra of Ba(BF4)(PF6), Ba(BF4)(AsF6) and Ba2(BF4)2(AsF6)(H3F4); the first examples of metal salts containing simultaneously tetrahedral BF4– and octahedral AF6– anions. J. Solid State Chem. 2009, 182, 2897–2903. 10.1016/j.jssc.2009.08.004. [DOI] [Google Scholar]
- Ivlev S. I.; Soltner T.; Karttunen A. J.; Mühlbauer M. J.; Kornath A. J.; Kraus F. Syntheses and crystal strucutres of sodium hydrogen fluorides NaF•nHF ( n = 3, 4). Z. Anorg. Allg. Chem. 2017, 643, 1436–1442. 10.1002/zaac.201700228. [DOI] [Google Scholar]
- Jordan T. H.; Dickens B.; Schroeder L. W.; Brown E. E. The crystal structure of Ca(BF4)2. Acta Crystallogr., Sect. B: Struct. Crystallogr. Cryst. Chem. 1975, 31, 669–672. 10.1107/s0567740875003597. [DOI] [Google Scholar]
- Mazej Z.; Goreshnik E. Crystal structures of [SbF6]− salts of di- and tetrahydrated Ag +, tetrahydrated Pd2+ and hexahydrated Cd2+ cations. Z. Kristallogr.-Cryst. Mater. 2017, 232, 339–347. 10.1515/zkri-2016-2031. [DOI] [Google Scholar]
- Mazej Z.; Goreshnik E. Syntheses of Dioxygenyl Salts by Photochemical Reactions in Liquid Anhydrous Hydrogen Fluoride: X-ray Crystal Structures of α- and β-O2Sn2F9, O2Sn2F9·0.9HF, O2GeF5·HF, and O2[Hg(HF)]4(SbF6)9. Inorg. Chem. 2020, 59, 2092–2103. 10.1021/acs.inorgchem.9b03518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Donnell T. A. Hydrogen fluoride – a simpler solvent than water?. J. Fluorine Chem. 1978, 11, 467–480. 10.1016/S0022-1139(00)82460-4. [DOI] [Google Scholar]
- O’Donnell T. A. Stabilization of unusual cationic species in protonic superacids and acidic melts. Chem. Soc. Rev. 1987, 16, 1–43. 10.1039/cs9871600001. [DOI] [Google Scholar]
- Mazej Z.; Borrmann H.; Lutar K.; Žemva B. Metal coordinated HF—a unique case. Inorg. Chem. 1998, 37, 5912–5914. 10.1021/ic980716j. [DOI] [Google Scholar]
- Chaivanov B. B.; Nabiev S. S.; Sokolov V. B.; Spirin S. N. “Clathrates” (?) of noble-gas fluorides. J. Fluorine Chem. 1991, 54, 12. 10.1016/s0022-1139(00)83522-8. [DOI] [Google Scholar]
- Nabiev S. S.; Sokolov V. B.; Chaivanov B. B. Structure of simple and complex noble gas fluorides. Crystallogr. Rep. 2011, 56, 774–791. 10.1134/s1063774511050191. [DOI] [Google Scholar]
- Nabiev S. S.; Sokolov V. B.; Chaivanov B. B. Molecular and crystal structures of noble gas compounds. Russ. Chem. Rev. 2014, 83, 1135–1180. 10.1070/rcr4475. [DOI] [Google Scholar]
- Solov’ev S. N.; Korunov A. A.; Zubkov K. G.; Minasyan K. A. Standard Enthalpy of the Formation of Ba[AuF6]2·4KrF2 (cr.). Russ. J. Phys. Chem. A 2012, 86, 320–322. 10.1134/s003602441202029x. [DOI] [Google Scholar]
- Lozinšek M.; Mercier H. P. A.; Brock D. S.; Žemva B.; Schrobilgen G. J. Coordination of KrF2 to a naked metal cation, Mg2+. Angew. Chem., Int. Ed. 2017, 56, 6251–6254. 10.1002/anie.201611534. [DOI] [PubMed] [Google Scholar]
- CrysAlisPro, Version 1.171.37.31; Agilent Technologies; (release Jan 14, 2014. CrysAlis171.NET).
- Sheldrick G. M. A short history of SHELX. Acta Crystallogr., Sect. A: Found. Crystallogr. 2008, 64, 112–122. 10.1107/s0108767307043930. [DOI] [PubMed] [Google Scholar]
- Sheldrick G. M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr., Sect. A: Found. Adv. 2015, 71, 3–8. 10.1107/s2053273314026370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dolomanov O. V.; Bourhis L. J.; Gildea R. J.; Howard J. A. K.; Puschmann H. OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Crystallogr. 2009, 42, 339–341. 10.1107/s0021889808042726. [DOI] [Google Scholar]
- Sheldrick G. M. Crystal structure refinement with SHELXL. Acta Crystallogr., Sect. C: Struct. Chem. 2015, 71, 3–8. 10.1107/s2053229614024218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozawa T. C.; Kang S. J. Balls & Sticks: easy-to-use structure visualization and animation program. J. Appl. Crystallogr. 2004, 37, 679. 10.1107/s0021889804015456. [DOI] [Google Scholar]
- Diamond—Crystal and Molecular Structure Visualization Crystal Impact; Dr. H. Putz & Dr. K. Brandenburg GbR: Kreuzherrenstr. 102, 53227 Bonn, Germany. http://www.crystalimpact.com/diamond.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.



