Abstract
Objectives
Previous studies ruled out the benefits of azithromycin for treatment of patients with COVID-19 who are hospitalized. However, the effects of azithromycin for treatment of patients with positive SARS-CoV-2 test results in the community remains a matter of debate. This study aimed to assess whether azithromycin, when used in subjects with positive test results for SARS-CoV-2, is associated with a reduced risk of hospitalization, in-hospital COVID-19 outcomes, and death.
Methods
Two study cohorts were selected. Cohort A included subjects with positive test results for SARS-CoV-2 between February 20, 2020 and December 10, 2020; cohort B included subjects infected with SARS-CoV-2 and hospitalized between February 20, 2020 and December 31, 2020. We compared the risk of hospitalization, intensive care unit access, need for mechanical ventilation, and death in azithromycin users versus nonusers. A clustered Fine-Gray analysis was employed to assess the risk of hospitalization; logistic and Cox regressions were performed to assess the risk of intensive care unit access, mechanical ventilation, and death.
Results
In cohort A, among 4861 azithromycin users and 4861 propensity-matched nonusers, azithromycin use was associated with higher risk of hospitalization (hazard ratio [HR] 1.59, 95% confidence interval [CI] 1.45-1.75) compared with nonuse. In cohort B, among 997 subjects selected in both groups, azithromycin use was not significantly associated with intensive care unit access (odds ratio [OR] 1.22, 95% CI 0.93-1.56), mechanical ventilation (OR 1.30, 95% CI 0.99-1.70), 14-day mortality (HR0.88, 95% CI 0.74-1.05), or 30-day mortality (HR 0.89, 95% CI 0.77-1.03).
Conclusion
Our findings confirm the lack of benefits of azithromycin treatment among community patients infected with SARS-CoV-2, raising concern on potential risks associated with its inappropriate use.
Keywords: Azithromycin, COVID-19, Hospitalization, Intensive care unit access, Mechanical ventilation, Mortality, Italy
Abbreviations: ATC, Anatomical Therapeutic Chemical; CI, Confidence Interval; CVD, Cardiovascular Disease; HAD, Healthcare Administrative Database; HIV, Human Immunodeficiency Virus; HPA, Health Protection Agency; HR, Hazard Ratio; ICU, Intensive Care Unit; ID, Index Date; MV, Mechanical Ventilation; NSAIDs, Nonsteroidal Anti-Inflammatory Drugs; OR, Odds Ratio; PSM, Propensity Score Matching; SD, Standard Deviation
Introduction
COVID-19, caused by the new SARS-CoV-2, continues to be widespread, with nearly 600 million cases and >6 million deaths worldwide as of August 29, 2022 (World Health Organization, 2022). Most patients with COVID-19 have flu-like syndrome with a variety of mild symptoms including rhinitis, pharyngitis, cough, and fever. However, some patients experience a more life-threatening disease characterized by respiratory failure, a proinflammatory state, and arterial thromboembolism, which may require hospitalization and intensive care unit (ICU) admission (Bonaventura et al., 2021).
Since the early phase of virus diffusion, several drug classes have been repurposed as potential candidates for treatment of patients with COVID-19, including nonsteroidal anti-inflammatory drugs (NSAIDs), glucocorticoids, and heparins (Perico et al., 2020; Sanders et al., 2020). Among others, azithromycin, a second-generation macrolide, received increased attention because of its antiviral and immunomodulatory activities (Abdelmalek and Mousa, 2022; Langford et al., 2021; Sharma et al., 2021). A body of evidence on the effect of azithromycin on viral infections such as Zika, rhinovirus, and Ebola contributed to raising of the hypothesis of potential efficacy of azithromycin against SARS-CoV-2 infection resulting from multiple possible mechanisms of action (Bosseboeuf et al., 2018; Gielen et al., 2010; Madrid et al., 2015). First, azithromycin may prevent virus entry into human cells by increasing cellular pH and consequently inhibiting endocytotic processes (Echeverría-Esnal et al., 2021; Nitulescu et al., 2020). Another direct effect of azithromycin is driven by its ability to bind and inhibit the spike protein of SARS-CoV-2 (Echeverría-Esnal et al., 2021). Second, the drug may modulate the immune system response by reducing several inflammatory mediators such as inflammatory cytokines, tumor necrosis factor, and interleukins, which have been demonstrated to be major drivers of COVID-19 mortality (Echeverría-Esnal Et al., 2021; Sultana et al., 2020). Finally, some patients with viral infection may develop a secondary bacterial infection or present with a bacterial coinfection for which azithromycin could be an effective treatment.
Previous trials investigated the potential efficacy of azithromycin in patients with COVID-19 who were admitted to the hospital, but no significant results were observed (Furtado et al., 2020; RECOVERY Collaborative Group, 2021). The potential effectiveness of azithromycin was also investigated in several observational studies selectively designed to evaluate azithromycin use in hospitalized patients with severe COVID-19; although results varied, findings from previous trials were confirmed (Arshad et al., 2020; Albani et al., 2020; Kokturk et al., 2021; Ip et al., 2020). However, there is little evidence regarding the effectiveness of azithromycin for treatment of individuals with suspected COVID-19 in the community, in whom earlier treatment may prevent either hospital admission or the occurrence or more severe COVID-19 outcomes. In this regard, one observational study (Szente Fonseca et al., 2020) and two trials (Hinks et al., 2021; Oldenburg et al., 2021) reported no association between treatment with azithromycin in the community and need for hospital admission.
Today, the debate on the potential beneficial effects of this medication is ongoing also because of its use in the general population after positive test results for SARS-CoV-2 infection. Recent studies indicate that azithromycin, along with other medications, is still empirically prescribed by physicians (Jampani and Chandy, 2021) despite several statements issued by regulatory authorities outlining the lack of evidence for its beneficial effects (Bartoletti et al., 2022).
Therefore, this study aimed to assess whether treatment with azithromycin in patients with positive test results for SARS-CoV-2 infection reduces the risk of hospitalization, and whether use of azithromycin before hospitalization is associated with less-severe COVID-19 prognosis (indicated by need for mechanical ventilation (MV), ICU access, and death).
Methods
Study design and data source
The study is part of an Italian regional project that assessed COVID-19 impact on the healthcare system (Valutazione dell'Impatto di COVID-19 ed Elaborazione di Strategie e Strumenti di Mitigazione del Rischio Epidemico [VICES-SMIRE]). The study was funded by the Lombardy Region and is exempt from institutional review board authorization and informed consent (according to the General Authorisation for the Processing of Personal Data for Scientific Research Purposes, issued by the Italian Data Protection Authority).
This is a large-scale, retrospective cohort analysis based on the healthcare administrative databases (HADs) of local health protection agencies (HPAs) of Bergamo (HPA-Bergamo) and Brescia (HPA-Brescia) in Lombardy, northern Italy. The two areas covered a population of about 2.3 million inhabitants and were affected by an intense outbreak during the early stage of the pandemic (Conti et al., 2020).
Italy has a universal-coverage healthcare system, and care demand is registered electronically for administrative purposes. In the study, five different administrative healthcare data sources related to the studied areas were linked through an anonymized individual code: (1) the SARS-CoV-2 swab registry, established on February 20, 2020 for monitoring individual data on SARS-CoV-2 infection; (2) the hospital discharge database, which collects information on inpatient care supplied by public or private hospitals; (3) the pharmacy claims database, which includes information on outpatient- dispensed drugs reimbursed by the national healthcare system; (4) the chronic morbidity registry, which includes information on patients’ disease based on pharmacy claims, inpatient records, and disease exemption for copayment records; and (5) the health registry, which reports data on residents with healthcare coverage, including date and reasons for entry (i.e., birth and immigration) and exit (i.e., death and emigration).
Study population
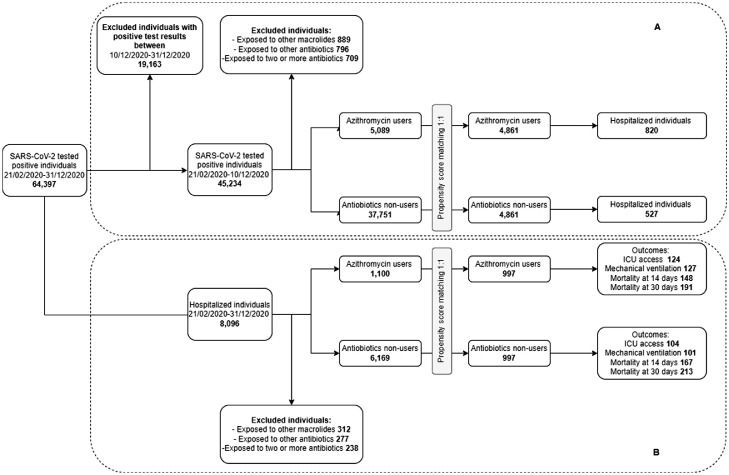
Starting with 64,327 individuals who had positive SARS-CoV-2 test results between February 20, 2020 and December 31, 2020 (Figure 1 ), two cohorts were identified. The first cohort (cohort A), to study the association between azithromycin exposure and risk of hospitalization, included individuals with swabs with positive SARS-CoV-2 test results registered between February 20, 2020 and December 10, 2020. The date of positive test result was considered the index date (ID). Each individual was then followed until the occurrence of the study outcome (i.e., hospitalization), death, or end of follow-up (i.e., 21 days), whichever came first (Figure 1).
Figure 1.
Study cohort selection. (a) Cohort A selection to assess the association between use of azithromycin and hospitalization. (b) cohort B selection to assess the association between previous use of azithromycin and risk of ICU access, need for MV, and death in hospitalized patients infected with SARS-CoV-2.
ICU, intensive care unit; MV, mechanical ventilation.
The second cohort (cohort B), to study disease outcomes (i.e., ICU access, need for MV, and death) in hospitalized patients, included subjects with positive SARS-CoV-2 test results and a hospitalization within 21 days of the test occurring between February 20, 2020 and December 31, 2020 (Figure 1). The date of hospitalization was considered the ID. Individuals were then followed up until the occurrence of each of the study outcomes or the end of the follow-up (i.e., 30 days), whichever came first. Subjects with healthcare coverage beginning after January 1, 2019 were excluded from the study.
Exposure of interest
Exposure to azithromycin (ATC code: J01FA10) alone was assessed in both cohorts by the presence of at least one pharmacy claim from 7 days before to 20 days after the swab with positive test results (exposure period). For hospitalized individuals, the exposure period was truncated to the day before the admission date. Subjects exposed to antibiotics other than azithromycin or to more than one antibiotic were excluded from the study cohorts. Therefore, patients exposed to azithromycin (users) were compared with those not exposed to any antibiotics (nonusers).
Patients’ characteristics
We collected data on sex, age, and comorbidities for each selected individual. Comorbidities were defined using the chronic morbidity registry, updated on January 1, 2020. We included the following main categories: Alzheimer's disease or dementia, respiratory disease, ischemic heart disease, peripheral vascular disease, cerebrovascular disease, hypertension, heart failure, other cardiovascular diseases (CVDs), dyslipidemia, diabetes, chronic liver disease, rheumatic disease, cancer, and infection with Human Immunodeficiency Virus (HIV). We also investigated in both cohorts the exposure to other drugs, such as anticoagulants, NSAIDs, chloroquine or hydroxychloroquine, corticosteroids for systemic use (plain), and immunosuppressants, in the 3 months preceding the ID.
Study outcomes
The outcome of interest for cohort A was hospitalization within 21 days after the ID. The date of hospital admission was retrieved from the hospital discharge database. In cohort B, we investigated inpatient and outpatient 14-day and 30-day mortality from the date of hospitalization (i.e., ID) and the need for MV and ICU access during hospitalization. For the need for MV and ICU access, only the occurrence of these outcomes was available; no information on the date of these events was recorded.
Statistical analysis
Patients’ demographic and clinical characteristics were summarized using frequency and percentage for categorical variables and mean and standard deviation (SD) or median and interquartile range for count variables. Characteristics were compared between the study groups using Pearson chi-square or Fisher's exact test for categorical variables and Student t-test or Wilcoxon test for continuous variables.
Propensity score matching (PSM) was then applied to reduce possible bias due to confounding factors between azithromycin users and nonusers. Propensity scores were computed by age, sex, comorbidities, and concomitant use of one of the drugs listed above. All variables were imputed into the model in a nonparsimonious way. PSM was performed using a 1:1 nearest-neighbor-matching algorithm without replacement and with a caliper width equal to 0.2 of the SD of the propensity scores. PSM balance was tested using standardized differences and variance ratios for all variables included in the propensity score computation.
In cohort A, the association between use of azithromycin and risk of hospitalization was analyzed using a clustered Fine-Gray regression model, with death as competing risk. Results were expressed as hazard ratio (HR) with 95% confidence interval (CI). In cohort B, the association between previous azithromycin exposure and 14-day and 30-day mortality was assessed using Cox proportional hazards regression with matched pairs. Patients experiencing ICU access and need for MV were not censored, and stratified analyses were carried out to evaluate their effects on mortality. Results were expressed as HR with 95%CI. The association between use of azithromycin and need for MV or ICU access was assessed using a logistic regression model with matched pairs. Results were expressed as odds ratio (OR) with 95% CI.
Subgroup analyses were built by running multiple PSMs according to baseline patients’ characteristics, such as age (<65 or ≥65 years), sex, and presence of CVD, diabetes, and cancer, which are proven to be associated with poorer COVID-19 prognosis (Bae et al., 2021). Furthermore, we explored the risk of each study outcome by calendar date to account for potential changes in COVID-19 management with time. All statistical analyses were performed using R 4.0.5 (R Foundation for Statistical Computing, Vienna, Austria) and SAS 9.4 (SAS Institute, Cary, North Carolina, USA).
Results
Descriptive statistics
Cohort A included 5089 azithromycin users and 37,751 nonusers (Figure 1). Azithromycin users were more likely to be male (51% vs 46%) and older (mean age 54.5 vs 48.8 years) compared with nonusers (Table 1 ). Azithromycin users also reported significantly higher prevalence of comorbidities and were more likely to use anticoagulants, NSAIDs, chloroquine/hydroxychloroquine, and corticosteroids for systemic use. The PSM led to the selection of 4861 azithromycin users and 4861 nonusers. The study groups showed similar distributions of the main demographic and clinical characteristics; no statistically significant differences were observed (Table 1; Figure 1; Supplementary Table 1; Supplementary Figure 1). Cohort B included 1100 azithromycin users and 6169 nonusers (Figure 1). Nonusers showed a higher prevalence of comorbidities, whereas users were more likely to be treated with anticoagulants (23% vs 15%), chloroquine/hydroxychloroquine (7% vs 2%), and corticosteroids for systemic use (20% vs 9%) (Table 1). The PSM led to the selection of 987 azithromycin users and 987 nonusers; study groups showed no statistically significant differences in main demographic and clinical characteristics (Table 1; Figure 1; Supplementary Table 1; Supplementary Figure 1).
Table 1.
Demographic and clinical characteristics of patients with positive SARS-CoV-2 test results (cohort A) and of individuals hospitalized with COVID-19 (cohort B), before and after PSM
| Cohort A |
Cohort B |
|||||||
|---|---|---|---|---|---|---|---|---|
| Characteristics | Before PSM |
After PSM |
Before PSM |
After PSM |
||||
| Azithromycinusers | Nonusers | Azithromycinusers | Nonusers | Azithromycinusers | Nonusers | Azithromycinusers | Nonusers | |
| Total | 5,089 | 37,751 | 4,861 | 4,861 | 1,100 | 6,169 | 997 | 997 |
| Sex, N (%) | ||||||||
| Female | 2,500 (49.1%) | 20,446 (54.2%)a | 2,409 (49.6%) | 2,447 (50.3%) | 352 (32%) | 2,543 (41.2%) a | 323 (32.4%) | 322 (32.3%) |
| Age | ||||||||
| Age mean ±SD | 54.5 (17.3) | 48.8 (22.7) a | 54.6 (17.2) | 54.6 (17.2) | 66.9 (13.2) | 69.2 (15.6) a | 67.9 (12.6) | 67.9 (12.6) |
| Age groups | ||||||||
| 0-44 | 1,308 (25.7%) | 15,531 (41.1%) a | 1,227 (25.2%) | 1,227 (25.2%) | 54 (4.9%) | 433 (7%) a | 33 (3.3%) | 33 (3.3%) |
| 45-64 | 2,367 (46.5%) | 13,038 (34.5%) a | 2,292 (47.2%) | 2,292 (47.2%) | 417 (37.9%) | 1,652 (26.8%) a | 367 (36.8%) | 367 (36.8%) |
| ≥65 | 1,414 (27.8%) | 9,182 (24.3%) a | 1,342 (27.6%) | 1,342 (27.6%) | 629 (57.2%) | 4,084 (66.2%) a | 597 (59.9%) | 597 (59.9%) |
| Comorbidity, N (%) | ||||||||
| Alzheimer/dementia | 24 (0.5%) | 510 (1.4%) a | 22 (0.5%) | 17 (0.3%) | 9 (0.8%) | 125 (2%) a | 6 (0.6%) | 4 (0.4%) |
| Respiratory disease | 273 (5.4%) | 1,422 (3.8%) a | 251 (5.2%) | 251 (5.2%) | 63 (5.7%) | 423 (6.9%) | 52 (5.2%) | 61 (6.1%) |
| Ischemic heart disease | 210 (4.1%) | 1,218 (3.2%) a | 201 (4.1%) | 145 (3%) | 84 (7.6%) | 626 (10.1%) a | 79 (7.9%) | 66 (6.6%) |
| Other cardiovascular diseases | 385 (7.6%) | 2,508 (6.6%) a | 365 (7.5%) | 299 (6.2%) | 131 (11.9%) | 1,059 (17.2%) a | 124 (12.4%) | 106 (10.6%) |
| Peripheral vascular disease | 81 (1.6%) | 516 (1.4%) | 73 (1.5%) | 58 (1.2%) | 24 (2.2%) | 234 (3.8%) a | 23 (2.3%) | 20 (2%) |
| Cerebrovascular disease | 88 (1.7%) | 880 (2.3%) a | 81 (1.7%) | 60 (1.2%) | 29 (2.6%) | 341 (5.5%) a | 24 (2.4%) | 23 (2.3%) |
| Hypertension | 1,286 (25.3%) | 7,000 (18.5%) a | 1,212 (24.9%) | 1,169 (24%) | 428 (38.9%) | 2,667 (43.2%) a | 390 (39.1%) | 375 (37.6%) |
| Heart failure | 103 (2%) | 750 (2%) | 97 (2%) | 58 (1.2%) | 45 (4.1%) | 414 (6.7%) a | 43 (4.3%) | 43 (4.3%) |
| Dyslipidemia | 475 (9.3%) | 2,412 (6.4%) a | 447 (9.2%) | 386 (7.9%) | 180 (16.4%) | 1124 (18.2%) | 166 (16.6%) | 146 (14.6%) |
| Diabetes | 430 (8.4%) | 2,402 (6.4%) a | 413 (8.5%) | 360 (7.4%) | 160 (14.5%) | 1063 (17.2%) a | 151 (15.1%) | 123 (12.3%) |
| Chronic liver disease | 141 (2.8%) | 954 (2.5%) | 132 (2.7%) | 114 (2.3%) | 45 (4.1%) | 360 (5.8%) a | 39 (3.9%) | 34 (3.4%) |
| Rheumatic disease | 53 (1%) | 304 (0.8%) | 50 (1%) | 52 (1.1%) | 15 (1.4%) | 96 (1.6%) | 14 (1.4%) | 8 (0.8%) |
| Cancer | 285 (5.6%) | 1,687 (4.5%) a | 273 (5.6%) | 284 (5.8%) | 91 (8.3%) | 568 (9.2%) | 84 (8.4%) | 100 (10%) |
| HIV | 6 (0.1%) | 68 (0.2%) | 6 (0.1%) | 7 (0.1%) | 2 (0.2%) | 20 (0.3%) | 2 (0.2%) | 2 (0.2%) |
| Concomitant therapies within 3 months, N (%) | ||||||||
| Anticoagulants | 560 (11%) | 1,523 (4%) a | 440 (9.1%) | 389 (8%) | 248 (22.5%) | 900 (14.6%) a | 187 (18.8%) | 178 (17.9%) |
| Nonsteroidal anti-inflammatory drugs | 275 (5.4%) | 1,066 (2.8%) a | 238 (4.9%) | 261 (5.4%) | 81 (7.4%) | 409 (6.6%) | 68 (6.8%) | 61 (6.1%) |
| Chloroquine, hydroxychloroquine | 210 (4.1%) | 337 (0.9%) a | 132 (2.7%) | 119 (2.4%) | 73 (6.6%) | 106 (1.7%) a | 37 (3.7%) | 28 (2.8%) |
| Corticosteroids for systemic use | 579-11.4%) | 1,320 (3.5%) a | 430 (8.8%) | 443 (9.1%) | 220 (20%) | 577 (9.4%) | 156 (15.6%) a | 160 (16%) |
| Immunosuppressants | 45 (0.9%) | 263 (0.7%) | 39 (0.8%) | 41 (0.8%) | 16 (1.5%) | 110 (1.8%) | 14 (1.4%) | 9 (0.9%) |
P-value <0.05
PSM, propensity score matching.
Outcomes
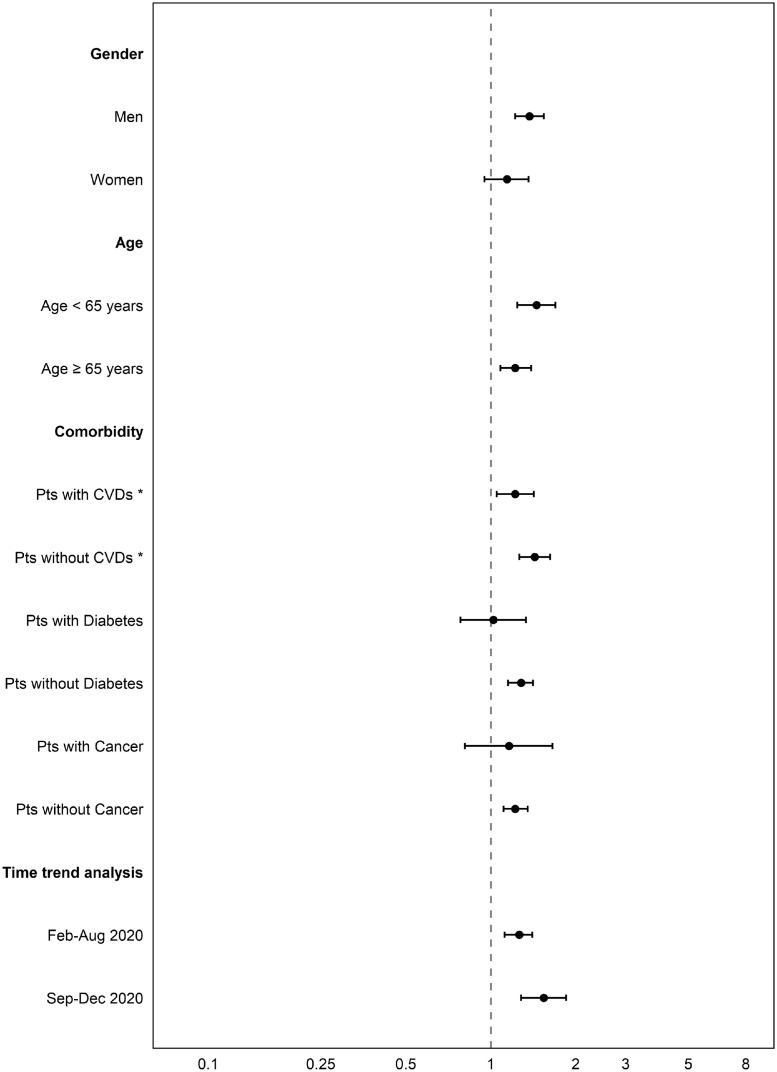
Results reported in Table 2 indicate a significantly higher risk of hospitalization in azithromycin users compared with nonusers (HR 1.59, 95 % CI 1.45-1.75). Results from the subgroup analyses were consistent with the main results, with higher risks observed in males (HR 1.37, 95% CI 1.22-1.54), in those aged <65 years (HR 1.45, 95 %CI 1.24-1.69), and in patients without CVDs (HR 1.43, 95% CI 1.26-1.62), diabetes (HR 1.28, 95% CI 1.15-1.41), and cancer (HR 1.22, 95% CI 1.11-1.35) (Figure 2 ).
Table 2.
Azithromycin use and hospitalization, ICU access, MV, and mortality in patients with COVID-19
| Outcome | Relative risk before PSM | Relative risk after PSM a | ||
|---|---|---|---|---|
| Cohort A – patients tested positive to SARS-CoV-2 | ||||
| Hospitalization | ||||
| Azithromycin nonuser | 3,711 (9.8%) | Reference | 527 (10.8%) | Reference |
| Azithromycin user | 854 (16.8%) | 1.74 (1.62-1.87) | 820 (16.9%) | 1.59 (1.45-1.75) |
| Cohort B – patients hospitalized and tested positive to SARS-CoV-2 | ||||
|---|---|---|---|---|
| ICU access | ||||
| Azithromycin nonuser | 576 (9.3%) | Reference | 104 (10.4%) | Reference |
| Azithromycin user | 132 (12.0%) | 1.32 (1.08-1.62) | 124 (12.4%) | 1.22 (0.93-1.56) |
| MV | ||||
| Azithromycin nonuser | 549 (8.9%) | Reference | 101 (10.1%) | Reference |
| Azithromycin user | 133 (12.1%) | 1.41 (1.15-1.72) | 127 (12.7%) | 1.30 (0.99-1.70) |
| Mortality at 14 days | ||||
| Azithromycin nonuser | 894 (14.5%) | Reference | 167 (16.8%) | Reference |
| Azithromycin user | 153 (13.9%) | 0.96 (0.81-1.14) | 148 (14.8%) | 0.88 (0.74-1.05) |
| Mortality at 30 days | ||||
| Azithromycin nonuser | 1,136 (18.4%) | Reference | 213 (21.4%) | Reference |
| Azithromycin user | 199 (18.1%) | 0.98 (0.84-1.14) | 191 (19.2%) | 0.89 (0.77-1.03) |
Data presented as HR for hospitalization and mortality and as OR for ICU access and MV.
HR, hazard ratio; ICU, intensive care unit; MV, mechanical ventilation; OR, odds ratio; PSM, propensity score matching.
Figure 2.
Subgroup analyses of hospitalization risk in individuals with positive SARS-CoV-2 test results.
CVDs, cardiovascular diseases; Pts, patients.
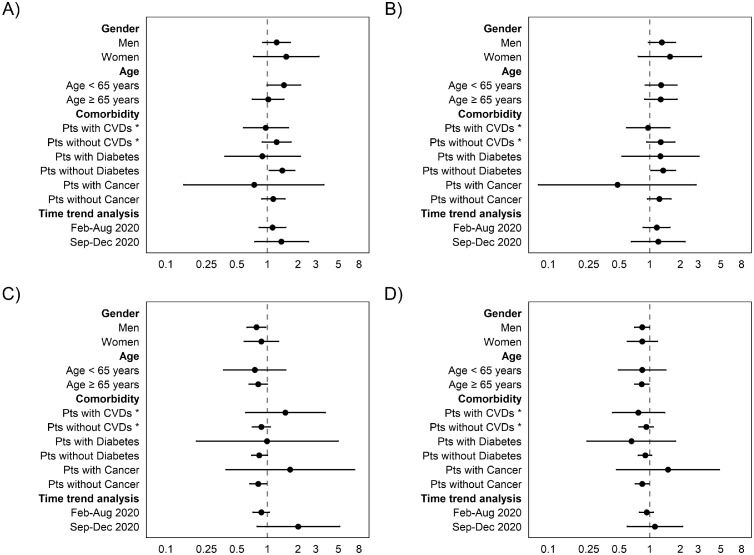
In cohort B, results showed no statistically significant associations between ICU access (OR 1.22, 95% CI 0.93-1.56) or need for MV (OR 1.30, 95% CI 0.99-1.70) and azithromycin use compared with nonuse (Table 2). Similarly, the Cox regression model did not show a statistically significant effect of azithromycin use on 14-day (HR 0.88, 95% CI 0.74-1.05) and 30-day mortality (HR 0.89, 95% CI 0.77-1.03) (Table 2). However, lower 30-day mortality (HR 0.65, 95% CI 0.45-0.92) was observed in azithromycin users with ICU access or subjected to MV. Subgroup analyses showed a significantly increased risk of ICU access (OR 1.40, 95% CI 1.04, 1.86) and MV (OR 1.35, 95% CI 1.02-1.79) in azithromycin users without diabetes (Figure 3 ). Conversely, previous use of azithromycin was associated with decreased risk of 30-day mortality in males (HR 0.78, 95% CI 0.63-0.96), in patients aged ≥65 years (HR 0.83, 95% CI 0.71, 0.98), and in those without cancer (HR 0.84, 95% CI 0.72-0.98) (Figure 3).
Figure 3.
Subgroup analyses of (a) risk of intensive care unit access; (b) risk of need for mechanical ventilation; (c) 14-day all-cause mortality; and (d) 30-day all-cause mortality in individuals hospitalized with COVID-19.
CVDs, cardiovascular diseases; Pts, patients.
Discussion
This population-based study explored the effects of treatment with azithromycin in a community setting on major COVID-19-related outcomes—namely, hospitalization, ICU access, need for MV, and death. Our findings do not support the hypothesis of a protective role of azithromycin in any observed endpoint. Instead, our data outline the concern of potential inappropriate use of this medication, which was empirically prescribed in subjects infected with SARS-CoV-2 during the pandemic period (European Centre for Disease Prevention and Control, 2021; Gonzalez-Zorn, 2021; Huang et al., 2020; Sharma et al., 2021). In fact, despite the massive use of antibiotics observed in subjects who were developing early symptoms of COVID-19 (Khan et al., 2022; Sharma et al., 2021), several studies have shown that coinfection and secondary bacterial infection occurred only in 3.5%-14.3% of patients with COVID-19 (Chedid et al., 2021; European Centre for Disease Prevention and Control, 2021; Hughes et al., 2020; Langford et al., 2020; Lansbury et al., 2020; Sharma et al., 2021).
Among patients with positive test results for SARS-CoV-2, azithromycin was associated with a significantly increased risk of hospitalization during 21 days of follow-up. A possible mechanistic explanation for these results may be related to the effects of this drug on microbiota and on the immune system (Langford et al., 2021; Wypych and Marsland, 2018; Yin et al., 2022). In fact, azithromycin may cause a temporary dysbiosis that can result in the inability of lungs to clear pathogens and can make them more vulnerable to viral infections. The improper use of antibiotics in patients without bacterial infections may also cause a cytokine-mediated overactivation of the immune system and a septic-shock-like phenomenon, thus worsening the hyperinflammation stimulated by COVID-19 (Marsland et al., 2015; McAleer and Kolls, 2018; Wypych and Marsland, 2018). Another possible explanation may be related to patients’ and clinicians’ risk perception and to differences in disease management over time (Arefi et al., 2022; Hayat et al., 2022). In fact, during the early phases of the pandemic, the intensity of care of patients with COVID-19 was based mainly on empirical evidence such as disease severity, clinical symptoms, and risk of disease progression (Jampani and Chandy, 2021). Therefore, clinicians were more likely to use azithromycin in patients with COVID-19 with mild symptoms, with consequent decreased use of hospital care and potential increased risk of disease worsening (Yin et al., 2022). This hypothesis is supported by our findings, which showed higher risk of hospitalization among younger and healthier users compared with older users and those with diabetes, CVDs, and cancer.
Findings from cohort B confirm the evidence from previous studies that found no beneficial effect of in- and out-of-hospital treatment with azithromycin (Arshad et al., 2020; Albani et al., 2020; Kokturk et al., 2021; Ip et al., 2020; RECOVERY Collaborative Group, 2021). Additionally, we were able to provide powerful new evidence on the potential role of this drug in patients with COVID-19 who are admitted to the hospital but were treated before hospitalization. Although not statistically significant, results on ICU access and need for MV mirrored what was observed in cohort A, thus suggesting that perception of lower risk among azithromycin users may have influenced not only the risk of hospitalization but also hospital-based outcomes such as ICU access and need for MV. Conversely, the intensity of care within the hospitals may have influenced the data on mortality, as demonstrated by the statistically significant lower risk observed in azithromycin users who accessed ICU or were subjected to MV.
This study has limitations. First, the Italian healthcare administrative databases does not include data on in-hospital patient management or clinical data such as severity of COVID-19 disease or results from biological samples (i.e., microbiological tests). Second, the use of PSM analysis (Austin, 2011) allowed us to balance measured baseline demographic and clinical characteristics between exposed and unexposed groups, as already proposed in similar studies (Trifirò et al., 2020), and thus to minimize potential confounding by indication. However, we were unable to completely rule out the presence of residual unmeasured confounders because of the lack of some relevant information in the Italian healthcare administrative databases, such as COVID-19 disease severity (National Institutes of Health, 2022) and possible risk factors for disease outcomes and death in patients with COVID-19, such as obesity and smoking. Lastly, we were not able to identify the reasons for antibiotic use, as the Italian pharmacy claims database does not include information on drug indications.
Conclusion
Our findings are in line with available evidence that does not recommend the use of azithromycin as effective treatment for patients with SARS-CoV-2 infection and raise concern on risks associated with inappropriate use of this drug. This highlights the importance of following antibiotic stewardship principles—even during challenging times such as COVID-19 pandemic—because the use of antibiotics without solid microbiological evidence on bacterial coinfection may result in both no beneficial effects for patients with COVID-19 and increased risk of adverse events (Chedid et al., 2021; Sharma et al., 2021). These aspects are crucial for preserving the effectiveness of these medications and preventing the spread of drug-resistant organisms.
Funding
The VICES-SMIRE project was funded by Lombardy Region (DGR n. XI/3017/2020 and D.G. Welfare nr. 7082/2020).
Ethical approval statement
The study is exempt from institutional review board authorization and informed consent (according to General Authorisation for the Processing of Personal Data for Scientific Research Purposes, issued by the Italian Data Protection Authority).
Author contributions
Conceptualization: ICA, CF, LGM, and GM; methodology: ICA, CF, GM; data curation: CF, DR, SC, and SK; writing (original draft preparation): ICA, CF and GM; writing (review and editing): all authors. All authors have read and agreed to the published version of the manuscript.
Data availability statement
Data sharing is not applicable to this article.
Declarations of competing interest
ICA, CF, DR, SC, PF, PC, RDP, SK, AZ, GM, AS, GC and GM have no conflicts of interest to declare. LGM reported receiving grants from Bayer, Daiiki-Sankyo, and Boehringer Ingelheim outside the submitted work and speaker fees from Pfizer and Bayer.
Footnotes
Supplementary material associated with this article can be found, in the online version, at doi:10.1016/j.ijid.2022.09.005.
Appendix. Supplementary materials
References
- Abdelmalek SMA, Mousa A. Azithromycin misuse during the COVID-19 pandemic: a cross-sectional study from Jordan. Infect Drug Resist. 2022;15:747–755. doi: 10.2147/IDR.S351827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Albani F, Fusina F, Giovannini A, Ferretti P, Granato A, Prezioso C, Divizia D, Sabaini A, Marri M, Malpetti E, Natalini G. Impact of azithromycin and/or hydroxychloroquine on hospital mortality in COVID-19. J Clin Med. 2020;9:2800. doi: 10.3390/jcm9092800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arefi MF, Babaei AP, Barzanouni S, Ebrahimi S, Salehi AR, Khajehnasiri F, Poursadeghian M. Risk perception in the COVID-19 pandemic; a health promotion approach. J Educ Health Promot. 2022;11:118. doi: 10.4103/jehp.jehp_1162_21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arshad S, Kilgore P, Chaudhry ZS, Jacobsen G, Wang DD, Huitsing K, Brar I, Alangaden GJ, Ramesh MS, McKinnon JE, O'Neill W, Zervos M. Henry Ford COVID-19 Task Force. Treatment with hydroxychloroquine, azithromycin, and combination in patients hospitalized with COVID-19. Int J Infect Dis. 2020;97:396–403. doi: 10.1016/j.ijid.2020.06.099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Austin PC. An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011;46:399–424. doi: 10.1080/00273171.2011.568786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bae S, Kim SR, Kim MN, Shim WJ, Park SM. Impact of cardiovascular disease and risk factors on fatal outcomes in patients with COVID-19 according to age: a systematic review and meta-analysis. Heart. 2021;107:373–380. doi: 10.1136/heartjnl-2020-317901. [DOI] [PubMed] [Google Scholar]
- Bartoletti M, Azap O, Barac A, Bussini L, Ergonul O, Krause R, Paño-Pardo JR, Power NR, Sibani M, Szabo BG, Tsiodras S, Verweij PE. Zollner-Schwetz I, Rodríguez-Baño J. ESCMID COVID-19 living guidelines: drug treatment and clinical management. Clin Microbiol Infect. 2022;28:222–238. doi: 10.1016/j.cmi.2021.11.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonaventura A, Vecchié A, Dagna L, Martinod K, Dixon DL, Tassell BWV, Dentali F, Montecucco F, Massberg S, Levi M, Abbate A. Endothelial dysfunction and immunothrombosis as key pathogenic mechanisms in COVID-19. Nat Rev Immunol. 2021;21:319–329. doi: 10.1038/s41577-021-00536-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bosseboeuf E, Aubry M, Nhan T, de Pina JJ, Rolain JM, Raoult D, Musso D. Azithromycin inhibits the replication of Zika virus. J Antivir Antiretrovir. 2018;10:6–11. [Google Scholar]
- Chedid M, Waked R, Haddad E, Chetata N, Saliba G, Choucair J. Antibiotics in treatment of COVID-19 complications: a review of frequency, indications, and efficacy. J Infect Public Health. 2021;14:570–576. doi: 10.1016/j.jiph.2021.02.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Conti S, Ferrara P, Mazzaglia G, D'Orso MI, Ciampichini R, Fornari C, Madotto F, Magoni M, Sampietro G, Silenzi A, Sileo CV, Zucchi A, Cesana G, Manzoli L, Mantovani LG. Magnitude and time-course of excess mortality during COVID-19 outbreak: population-based empirical evidence from highly impacted provinces in northern Italy. ERJ Open Res. 2020;6:00458–02020. doi: 10.1183/23120541.00458-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Echeverría-Esnal D, Martin-Ontiyuelo C, Navarrete-Rouco ME, De-Antonio Cuscó M, Ferrández O, Horcajada JP, Grau S. Azithromycin in the treatment of COVID-19: a review. Expert Rev Anti Infect Ther. 2021;19:147–163. doi: 10.1080/14787210.2020.1813024. [DOI] [PubMed] [Google Scholar]
- European Centre for Disease Prevention and Control (ECDC). Treatment and pharmaceutical prophylaxis of COVID-19. https://www.ecdc.europa.eu/en/covid-19/latest-evidence/treatment#:~:text=Use%20of%20antibiotics%20in%20patients,COVID%2D19%20%5B10%5D, 2021 (accessed 25 August 2022).
- Furtado RHM, Berwanger O, Fonseca HA, Corrêa TD, Ferraz LR, Lapa MG, Zampieri FG, Veiga VC, Azevedo LCP, Rosa RG, Lopes RD, Avezum A, Manoel ALO, Piza FMT, Martins PA, Lisboa TC, Pereira AJ, Olivato GB, Dantas VCS, Milan EP, Gebara OCE, Amazonas RB, Oliveira MB, Soares RVP, Moia DDF, Piano LPA, Castilho K, Momesso RGRAP, Schettino GPP, Rizzo LV, Neto AS, Machado FR, Cavalcanti AB. COALITION COVID-19 Brazil II Investigators. Azithromycin in addition to standard of care versus standard of care alone in the treatment of patients admitted to the hospital with severe COVID-19 in Brazil (COALITION II): a randomised clinical trial. Lancet. 2020;396:959–967. doi: 10.1016/S0140-6736(20)31862-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gielen V, Johnston SL, Edwards MR. Azithromycin induces anti-viral responses in bronchial epithelial cells. Eur Respir J. 2010;36:646–654. doi: 10.1183/09031936.00095809. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Zorn B. Antibiotic use in the COVID-19 crisis in Spain. Clin Microbiol Infect. 2021;27:646–647. doi: 10.1016/j.cmi.2020.09.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayat K, Mustafa ZU, Ikram MN, Ijaz-Ul-Haq M, Noor I, Rasool MF, Ishaq HM, Rehman AU, Hasan SS, Fang Y. Perception, attitude, and confidence of physicians about antimicrobial resistance and antimicrobial prescribing among COVID-19 patients: a cross-sectional study from Punjab, Pakistan. Front Pharmacol. 2022;12 doi: 10.3389/fphar.2021.794453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hinks TSC, Cureton L, Knight R, Wang A, Cane JL, Barber VS, Black J, Dutton SJ, Melhorn J, Jabeen M, Moss P, Garlapati R, Baron T, Johnson G, Cantle F, Clarke D, Elkhodair S, Underwood J, Lasserson D, Pavord ID, Morgan S, Richards D. Azithromycin versus standard care in patients with mild-to-moderate COVID-19 (ATOMIC2): an open-label, randomised trial. Lancet Respir Med. 2021;9:1130–1140. doi: 10.1016/S2213-2600(21)00263-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang JT, Ran RX, Lv ZH, Feng LN, Ran CY, Tong YQ, Li D, Su HW, Zhu CL, Qiu SL, Yang J, Xiao MY, Liu MJ, Yang YT, Liu SM, Li Y. Chronological changes of viral shedding in adult inpatients with COVID-19 in Wuhan, China. Clin Infect Dis. 2020;71:2158–2166. doi: 10.1093/cid/ciaa631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hughes S, Troise O, Donaldson H, Mughal N, Moore LSP. Bacterial and fungal coinfection among hospitalized patients with COVID-19: a retrospective cohort study in a UK secondary-care setting. Clin Microbiol Infect. 2020;26:1395–1399. doi: 10.1016/j.cmi.2020.06.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ip A, Berry DA, Hansen E, Goy AH, Pecora AL, Sinclaire BA, Bednarz U, Marafelias M, Berry SM, Berry NS, Mathura S, Sawczuk IS, Biran N, Go RC, Sperber S, Piwoz JA, Balani B, Cicogna C, Sebti R, Zuckerman J, Rose KM, Tank L, Jacobs LG, Korcak J, Timmapuri SL, Underwood JP, Sugalski G, Barsky C, Varga DW, Asif A, Landolfi JC, Goldberg SL. Hydroxychloroquine and tocilizumab therapy in COVID-19 patients-an observational study. PLoS One. 2020;15 doi: 10.1371/journal.pone.0237693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jampani M, Chandy SJ. Increased antimicrobial use during COVID-19: the risk of advancing the threat of antimicrobial resistance. Health Sci Rep. 2021;4:e459. doi: 10.1002/hsr2.459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Khan S, Hasan SS, Bond SE, Conway BR, Aldeyab MA. Antimicrobial consumption in patients with COVID-19: a systematic review and meta-analysis. Expert Rev Anti Infect Ther. 2022;20:749–772. doi: 10.1080/14787210.2022.2011719. [DOI] [PubMed] [Google Scholar]
- Kokturk N, Babayigit C, Kul S, Cetinkaya PD, Nayci SA, Baris SA, Karcioglu O, Aysert P, Irmak I, Yuksel AA, Sekibag Y, Toprak OB, Azak E, Mulamahmutoglu S, Cuhadaroglu C, Demirel A, Kerget B, Ketencioglu BB, Ozger HS, Ozkan G, Ture Z, Ergan B, Oguz VA, Kilinc O, Ercelik M, Ciftci TU, Alici O, Temel EN, Ataoglu O, Aydin A, Bahcetepe DC, Gullu YT, Fakili F, Deveci F, Kose N, Tor MM, Gunluoglu G, Altin S, Turgut T, Tuna T, Ozturk O, Dikensoy O, Gulhan PY, Basyigit I, Boyaci H, Oguzulgen IK, Borekci S, Gemicioglu B, Bayraktar F, Elbek O, Hanta I, Okur HK, Sagcan G, Uzun O, Akgun M, Altinisik G, Dursun B, Edis EC, Gulhan E, Eyuboglu FO, Gultekin O, Havlucu Y, Ozkan M, Coskun AS, Sayiner A, Kalyoncu AF, Itil O, Bayram H. The predictors of COVID-19 mortality in a nationwide cohort of Turkish patients. Respir Med. 2021;183 doi: 10.1016/j.rmed.2021.106433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langford BJ, So M, Raybardhan S, Leung V, Soucy JPR, Westwood D, Daneman N, MacFadden DR. Antibiotic prescribing in patients with COVID-19: rapid review and meta-analysis. Clin Microbiol Infect. 2021;27:520–531. doi: 10.1016/j.cmi.2020.12.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Langford BJ, So M, Raybardhan S, Leung V, Westwood D, MacFadden DR, Soucy JPR, Daneman N. Bacterial co-infection and secondary infection in patients with COVID-19: a living rapid review and meta-analysis. Clin Microbiol Infect. 2020;26:1622–1629. doi: 10.1016/j.cmi.2020.07.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lansbury L, Lim B, Baskaran V, Lim WS. Co-infections in people with COVID-19: a systematic review and meta-analysis. J Infect. 2020;81:266–275. doi: 10.1016/j.jinf.2020.05.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Madrid PB, Panchal RG, Warren TK, Shurtleff AC, Endsley AN, Green CE, Kolokoltsov A, Davey R, Manger ID, Gilfillan L, Bavari S, Tanga MJ. Evaluation of Ebola virus inhibitors for drug repurposing. ACS Infect Dis. 2015;1:317–326. doi: 10.1021/acsinfecdis.5b00030. [DOI] [PubMed] [Google Scholar]
- Marsland BJ, Trompette A, Gollwitzer ES. The gut-lung axis in respiratory disease. Ann Am Thorac Soc. 2015;12:S150–S156. doi: 10.1513/AnnalsATS.201503-133AW. [DOI] [PubMed] [Google Scholar]
- McAleer JP, Kolls JK. Contributions of the intestinal microbiome in lung immunity. Eur J Immunol. 2018;48:39–49. doi: 10.1002/eji.201646721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- National Institutes of Health (NIH). Clinical spectrum of SARS-CoV-2 infection. https://www.covid19treatmentguidelines.nih.gov/overview/clinical-spectrum/, 2022 (accessed 24 August 2022).
- Nitulescu GM, Paunescu H, Moschos SA, Petrakis D, Nitulescu G, Ion GND, Spandidos DA, Nikolouzakis TK, Drakoulis N, Tsatsakis A. Comprehensive analysis of drugs to treat SARS-CoV-2 infection: mechanistic insights into current COVID-19 therapies (review) Int J Mol Med. 2020;46:467–488. doi: 10.3892/ijmm.2020.4608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oldenburg CE, Pinsky BA, Brogdon J, Chen C, Ruder K, Zhong L, Nyatigo F, Cook CA, Hinterwirth A, Lebas E, Redd T, Porco TC, Lietman TM, Arnold BF, Doan T. Effect of oral azithromycin vs placebo on COVID-19 symptoms in outpatients with SARS-CoV-2 infection: a randomized clinical trial. JAMA. 2021;326:490–498. doi: 10.1001/jama.2021.11517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Perico N, Suter F, Remuzzi G. A recurrent question from a primary care physician: how should I treat my COVID-19 patients at home? Clin. Med Investig. 2020;5:1–8. [Google Scholar]
- RECOVERY Collaborative Group Azithromycin in patients admitted to hospital with COVID-19 (RECOVERY): a randomised, controlled, open-label, platform trial. Lancet. 2021;397:605–612. doi: 10.1016/S0140-6736(21)00149-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanders JM, Monogue ML, Jodlowski TZ, Cutrell JB. Pharmacologic treatments for coronavirus disease 2019 (COVID-19): a review. JAMA. 2020;323:1824–1836. doi: 10.1001/jama.2020.6019. [DOI] [PubMed] [Google Scholar]
- Sharma S, Singh A, Banerjee T. Antibacterial agents used in COVID-19: a systematic review and meta-analysis. Environmental Sustainability. 2021;4:503–513. doi: 10.1007/s42398-021-00194-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sultana J, Cutroneo PM, Crisafulli S, Puglisi G, Caramori G, Trifirò G. Azithromycin in COVID-19 patients: pharmacological mechanism, clinical evidence and prescribing guidelines. Drug Saf. 2020;43:691–698. doi: 10.1007/s40264-020-00976-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szente Fonseca SN, de Queiroz Sousa A, Wolkoff AG, Moreira MS, Pinto BC, Takeda CFV, Rebouças E, Abdon APV, Nascimento ALA, Risch HA. Risk of hospitalization for Covid-19 outpatients treated with various drug regimens in Brazil: comparative analysis. Travel Med Infect Dis. 2020;38 doi: 10.1016/j.tmaid.2020.101906. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trifirò G, Massari M, Da Cas R, Ippolito FM, Sultana J, Crisafulli S, Rossi PG, Marino M, Zorzi M, Bovo E, Leoni O, Ludergnani M, Alegiani SS. ITA-COVID-19: RAAS inhibitor group. Renin-angiotensin-aldosterone system inhibitors and risk of death in patients hospitalised with COVID-19: a retrospective Italian cohort study of 43,000 patients. Drug Saf. 2020;43:1297–1308. doi: 10.1007/s40264-020-00994-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organization. WHO coronavirus (COVID-19) dashboard. https://covid19.who.int/, 2022 (accessed 29 August 2022).
- Wypych TP, Marsland BJ. Antibiotics as instigators of microbial dysbiosis: implications for asthma and allergy. Trends Immunol. 2018;39:697–711. doi: 10.1016/j.it.2018.02.008. [DOI] [PubMed] [Google Scholar]
- Yin X, Xu X, Li H, Jiang N, Wang J, Lu Z, Xiong N, Gong Y. Evaluation of early antibiotic use in patients with non-severe COVID-19 without bacterial infection. Int J Antimicrob Agents. 2022;59 doi: 10.1016/j.ijantimicag.2021.106462. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data sharing is not applicable to this article.