Abstract
Noninvasive imaging of the heart plays an important role in the diagnosis and management of pulmonary hypertension (PH), and several well-established techniques are available for assessing performance of the right ventricle, the key determinant of patient survival. While right heart catheterisation is mandatory for establishing a diagnosis of PH, echocardiography is the most important screening tool for early detection of PH. Cardiac magnetic resonance imaging (CMRI) is also a reliable and practical tool that can be used as part of the diagnostic work-up. Echocardiography can measure a range of haemodynamic and anatomical variables (e.g. pericardial effusion and pulmonary artery pressure), whereas CMRI provides complementary information to echocardiography via high-resolution, three-dimensional imaging. Together with echocardiography and CMRI, techniques such as high-resolution computed tomography and positron emission tomography may also be valuable for screening, monitoring and follow-up assessments of patients with PH, but their clinical relevance has yet to be established. Technological advances have produced new variants of echocardiography, CMRI and positron emission tomography, and these permit closer examination of myocardial architecture, motion and deformation. Integrating these new tools into clinical practice in the future may lead to more precise noninvasive determination of diagnosis, risk and prognosis for PH.
Short abstract
Imaging techniques are essential tools for diagnosis and follow-up of patients with pulmonary hypertension http://ow.ly/TI8cQ
Introduction
In patients with pulmonary hypertension (PH), the ability of the right ventricle to adapt to the increased afterload resulting from rising pulmonary vascular resistance is the main determinant of a patient's symptoms, functional class, exercise capacity and survival [1–4]. Although PH can result from a variety of pathological changes to the pulmonary vasculature, it is right ventricular (RV) failure that is often the cause of death [5, 6].
The invasive procedure of right heart catheterisation (RHC) is required for a definitive diagnosis of PH [7, 8]; however, noninvasive imaging can play a role in complementing the findings from RHC. Imaging can be used to screen for possible PH, for quantification and risk stratification, and to monitor treatment response. Common imaging techniques include two-dimensional transthoracic echocardiography (echo) and magnetic resonance imaging (MRI) [9]. Other techniques include computed tomography (CT), ventilation/perfusion scintigraphy and positron emission tomography (PET) [7, 9, 10]. This article will review the imaging techniques that are currently used for the clinical assessment of PH, focusing on echo and cardiac MRI (CMRI), and discuss some newer techniques that are under development that could transform the diagnosis and management of PH in the future.
Imaging techniques
Noninvasive imaging of the right ventricle, especially using echo and CMRI, plays an important role in detecting structural and haemodynamic signs of PH, determining their cause and evaluating the effects of treatment [11]. A comparison of the diagnostic utility of echo and CMRI has found that the indices they measure correlate well with mean pulmonary artery pressure (PAP) and pulmonary vascular resistance at the time of RHC [12]. These two techniques provide complementary information in the assessment of PH [2]; echo provides reliable estimates of PAP [13], whereas CMRI is more suited to functional and morphological assessment of the right ventricle [2]. A comparison of the variables that can be measured by CMRI, echo and RHC are shown in table 1.
TABLE 1.
An overview of measurements achievable by echocardiography (echo), cardiac magnetic resonance imaging (CMRI) and right heart catheterisation (RHC) during the assessment of pulmonary hypertension (PH)
| Variable | Echo | CMRI | RHC |
| Effusion | |||
| Pericardial effusion | Easy to perform, relevant for follow-up [14] | Can be useful as a supplement to echo [15] | NA |
| Pressure | |||
| PAP | Estimation of PAP based on TRV; some controversy versus invasive techniques, and pitfalls have been described, e.g. false interpretation of the spectral wave envelope, sweep velocity set too slow and valve closure artefacts [16] | NA | Fundamental measurement for distinguishing PAH from other forms of PH [7, 17] |
| Anatomy | |||
| Right atrial area | Reference values for right atrial area in healthy adult subjects have been described [18]; easy to perform, accurate | Reference values for right atrial area in healthy adult subjects have been described [19] | NA |
| RV area | Ethnicity, sex and training effects (Asians and females have smaller RV area, sportsmen have higher area [20]); not useful for screening; high variation, even in healthy subjects | Considered the gold standard for measuring ventricular volume, mass and structure [21] | NA |
| RV mass | NA | NA | |
| RV dimensions | RV basal, mid-cavity and longitudinal dimensions on a four-chamber view should be measured in patients with PH [22]; high variation | NA | |
| RV wall thickness | RV thickness can be assessed; thickness >5 mm may suggest RV pressure overload in the absence of other pathologies [22] | Can be measured directly through evaluation of RV mass [23], or indirectly based on the need for increased myocardial perfusion [21] | NA |
| Eccentricity index | Predictive of poor outcomes [24] | NA | NA |
| Central pulmonary artery diameter | Reference values have not been determined | NA | NA |
| Function | |||
| RVEF | Three-dimensional echo is more accurate and reproducible than two-dimensional echo [13] | Can be calculated retrospectively from the stroke volume divided by the end-diastolic volume [25] | NA |
| TAPSE | Directly measures RV function; does not require post hoc analyses or expertise to measure [26] | NA | NA |
| Tei index (RV myocardial performance index) | Calculated as the sum of RV isovolumetric contraction time/ejection time and isovolumetric relaxation time/ejection time [27] | NA | NA |
| RV end-systolic elastance, arterial elastance | NA | CMRI-derived indices have been combined with RHC variables to determine right ventriculo–arterial coupling [28] | NA |
PAP: pulmonary arterial pressure; RV: right ventricular; RVEF: right ventricular ejection fraction; TAPSE: tricuspid annular plane systolic excursion; NA: not applicable; TRV: tricuspid regurgitation velocity; PAH: pulmonary arterial hypertension.
Echocardiography
Echo is the most important screening tool for determining the possibility of PH in patients whose symptoms, signs and medical history are suggestive of this condition [17]. Echo is a reliable, cost-effective and widely available method for noninvasive assessment of the heart [13]. The technique can provide information on several variables that are useful in clinical practice for evaluating RV function, including right atrial area (fig. 1) and RV ejection fraction (RVEF). Echo can also permit determination of RV adaptation to elevated afterload, using the RV myocardial performance index (also known as the Tei index) (table 1) [13]. One of the challenges of using echo for volumetric estimation of the right ventricle and right atrium is that there are still many variables for which the reference value (normal in healthy people) is not known for the right heart, and many need to be standardised [18]. Nevertheless, echo is a key tool at baseline diagnosis, following changes in therapy, during follow-up assessments [18], on clinical worsening of PH [29] and for providing information about prognosis [30]. Furthermore, echo and stress echo can be used to screen for PH in patients who are at high risk of the disease (e.g. those with systemic sclerosis) [31]. Transoesophageal echo can also be used in the diagnostic work-up of patients with complex valvular disease or suspected endocarditis, and in some patients with congenital heart disease [32]. In addition, it can be used to detect left ventricular dysfunction [32] and septal defects [30]. Transthoracic Doppler echo can detect chronic thromboembolic PH (CTEPH) in patients who have had acute pulmonary embolism [33].
FIGURE 1.
Measurement of right atrial area by echocardiography. RV: right ventricle; LV: left ventricle; LA: left atrium.
CMRI
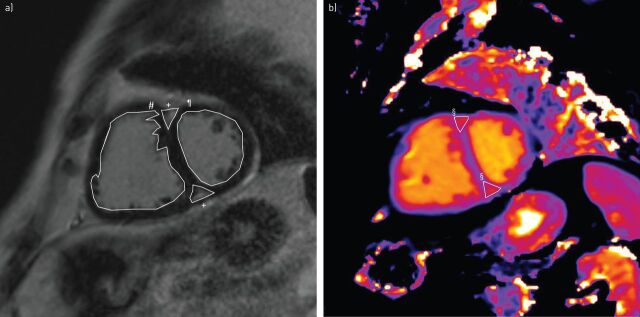
CMRI is a three-dimensional tomographic technique that provides detailed morphology of the cardiac chambers and accurate quantification of chamber volumes, myocardial mass and transvalvular flow (fig. 2) [34]. It is considered to be a particularly useful tool for monitoring the right ventricle, and allowing accurate quantification of ventricular volume, mass, structure and function [21, 35]. Many functional abnormalities of the right ventricle can be detected with CMRI, including the presence of fibrosis, and it may also be employed to evaluate left ventricle/right ventricle interdependence and coupling [36]. CMRI is a valuable technique for identifying inflammatory and infiltrative conditions, and a useful tool in the diagnostic work-up of patients with cardiomyopathy, arrhythmias, cardiac tumours, pericardial diseases or congenital heart disease [32]. Unlike echo, stress tests using CMRI have not yet been established as screening tests for PH.
FIGURE 2.
Cardiac magnetic resonance imaging of the heart in a patient with pulmonary arterial hypertension. a) Short axis image. #: dilated right ventricle; ¶: left ventricle; +: late gadolinium enhancement at right ventricle insertion points. b) Native T1 image of the same patient showing increased native T1 at the right ventricle insertion points (§).
Detection of PH: early diagnosis and identifying the cause
PH is often diagnosed at a late stage of the disease, and is associated with poor survival; early recognition and treatment are important to improve long-term outcomes [37]. PH occurs in multiple clinical conditions, and can be categorised into five distinct groups based on shared characteristics [7]. In pulmonary arterial hypertension (PAH) (defined as group 1 PH [38]), patients in World Health Organization functional class (WHO FC) I or II have been shown to have better long-term survival than patients with more severe functional impairment (WHO FC III or IV), supporting the need for earlier diagnosis and intervention [4, 39].
In order to identify the underlying cause of PH, it is necessary to distinguish between the five categories. Besides PAH, other diagnoses can be PH due to left heart disease (group 2), which includes left ventricular systolic dysfunction, left ventricular diastolic dysfunction (also termed heart failure with preserved ejection fraction) and valvular disease; PH due to chronic lung disease and/or hypoxia (group 3); CTEPH (group 4); and PH due to unclear and/or multifactorial mechanisms (group 5) [38]. Each group has distinct pathological and haemodynamic characteristics. It is therefore important to be able to determine the underlying cause of PH as the prognosis, and management strategies for patients in each of the different groups will differ [38].
The role of echo
Echo has a central role in guidelines for the screening and diagnosis of PH [7], and is the recommended first-line noninvasive tool that should be performed whenever PH is suspected [7, 17, 30, 40]. It can provide an estimate of PAP, evaluate the extent of remodelling and function of the right ventricle, and help to rule out secondary causes of PH [40]. Echo is considered to be the most useful noninvasive tool to determine which patients should undergo RHC [41], and is used as a tool to screen for PH in asymptomatic patients with scleroderma [17, 42]. Regular echo screening can also improve the early detection of PAH amongst those at high risk [40].
Although there is no single echocardiographic measurement that can diagnose PH, estimation of PAP based on tricuspid regurgitation velocity (TRV) can be used if PH is suspected [43]. PH is considered possible when: 1) TRV is ≤2.8 m·s−1, pulmonary arterial systolic pressure is ≤36 mmHg and additional echocardiographic variables suggestive of PH are present; or 2) TRV is 2.9–3.4 m·s−1 and pulmonary arterial systolic pressure is 37–50 mmHg, regardless of whether other signs of PH are present. In their 2015 guidelines for the diagnosis and treatment of PH, the joint task force of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS) recommend grading the probability of PH based on TRV at rest and the presence of additional echo variables [7]. The probability of PH should be considered high if TRV is ≥2.9 m·s−1 and additional echocardiographic variables suggestive of PH are present, or if TRV is >3.4 m·s−1 with no other signs. If the TRV is ≤2.8 m·s−1 (or not measurable) and there are no additional echocardiographic variables suggestive of PH, the probability of PH can be considered low. The probability of PH should be considered intermediate if the TRV is ≤2.8 m·s−1 or immeasurable, accompanied by additional signs of PH, or if the TRV is 2.9–3.4 m·s−1 with no other signs [7].
For measurement of pulmonary arterial systolic pressure, echo shows good correlation with RHC [16]. However, echo cannot be relied on for quantification and assessment of RV systolic function [25], and data suggest patients could be missed when this method is used in isolation from other techniques during screening [44]. When the accuracy of echo was compared with that of RHC for measuring PAP, ∼50% of estimates using echo were deemed accurate (i.e. within 10 mmHg of the value derived from RHC) [44]. For the remaining 50% of estimates that were inaccurate, 38% of pressure overestimates were >20 mmHg and 80% of pressure underestimates were >20 mmHg [44]. Detection rates for PH can be vastly improved when a diagnostic algorithm is used that combines noninvasive assessments, including echo, with RHC, as demonstrated in a recent study of patients with systemic sclerosis [42].
Echo can also help to detect the cause of PH. For example, echo techniques can be used to identify or prompt further investigation for congenital heart disease, sinus venosus-type atrial septal defect, anomalous pulmonary venous return or left heart disease [7].
The role of CMRI
CMRI is an informative tool that can help to identify the cause of PH when used alongside other techniques. In patients with PH, if the echo findings are inconclusive, CMRI may be used to determine the presence of congenital heart disease [7]. The presence or absence of specific CMRI findings might be suggestive of certain underlying diseases [45]. For example, the presence of shunts and aberrant pulmonary veins might be suggestive of PAH, whereas left ventricular impairment, left ventricular hypertrophy or wall motion abnormalities, and dilation of the left atrium could indicate PH due to left heart disease [45]. Moreover, filling defects in the pulmonary arteries might suggest the presence of CTEPH [45].
The filling pressure of the left ventricle is the most critical factor for defining group 2 PH, and this sets it apart from PAH, where there is no concomitant increase in left ventricular filling pressure [46]. The utility of CMRI in distinguishing idiopathic PAH from PH due to heart failure with preserved ejection fraction has been explored. In this study, a single CMRI variable, left atrial volume, was demonstrated as having sufficient discriminatory value, distinguishing idiopathic PAH from PH due to heart failure with preserved ejection fraction with 97% sensitivity and 100% specificity [47].
A further application of CMRI in PAH is to measure blood flow variables. Phase-contrast CMRI can enable the quantification of regurgitant and shunt flow volumes, visualisation of three-dimensional time-resolved flow patterns and assessment of coronary flow reserve, wall shear stress, and turbulence [48]. In PAH, the average blood velocity throughout the cardiac cycle, as measured by phase-contrast CMRI, has been shown to correlate strongly with RHC-derived measurements of pulmonary pressures and resistance [49], thereby making it a useful noninvasive tool for determining the presence of chronic PAH.
Additional techniques to determine the cause of PH
Other imaging techniques that can be used to identify the cause of PH include CT and ventilation/perfusion scintigraphy. Both techniques are helpful for allowing the visualisation of areas with perfusion defects. This feature makes them useful tools during the diagnostic work-up of patients with PH for determining the presence of CTEPH, which should be ruled out in patients with unexplained PH [7]. Different types of CT include high-resolution, contrast-enhanced and multidetector varieties [7, 50]. For diagnosis of CTEPH, ventilation/perfusion scintigraphy has a high sensitivity [50], making it a superior method to CT in ruling out pulmonary embolism as a cause of PH. High-resolution CT scanning can also rule out interstitial lung disease and help to rule out pulmonary veno-occlusive disease [7]. Good agreement between CT and intraoperative findings has been observed for patients with CTEPH [51]. Therefore, a multimodal approach should be used in the diagnostic work-up of patients with CTEPH, involving both CT and ventilation/perfusion scintigraphy.
Quantification of RV function and risk stratification in PH
Once PH has been diagnosed, it is important to establish disease severity. Evaluating the functional and structural limits of the right ventricle is important for determining the severity of disease, prognosis and response to guiding. Imaging can assist the assessment of RV function in the context of PH, and prognostic indicators have been reviewed [52].
Echo
Multiple baseline echo variables, most of them reflecting RV systolic or diastolic function, have been shown to be independently associated with survival [40]. The most useful echo variables that are routinely used to determine the likely prognosis of patients with PH include right atrial area [24, 53], pericardial effusion [24, 52], eccentricity index [24] and tricuspid annular plane systolic excursion (TAPSE) (table 2) [26].
TABLE 2.
Echocardiography and cardiac magnetic resonance imaging (CMRI) variables that have prognostic value in pulmonary hypertension
| Echocardiography [13] | CMRI [54–59] | ||
| Variable | Worse prognosis threshold | Variable | Worse prognosis threshold |
| TAPSE cm | <1.6 | RVEF % | <35 |
| Systolic excursion velocity cm·s−1 | <10 | Stroke volume index mL·m−2 | ≤25 |
| RV fractional area change % | <35 | Ventricular mass index | >0.56 |
| Peak longitudinal RV strain % | ≥19 | LV end-diastolic volume index mL·m−2 | ≤40 |
| Isovolumetric contraction velocity cm·s−1 | <9 | RV end-diastolic volume index mL·m−2 | ≥84 |
| Tei index (pulsed wave) | >0.4 | LV end-systolic volume index mL·m−2 | 15±9 |
| Main pulmonary artery diameter mm | >29 | RV end-systolic volume index mL·m−2 | 47±21 |
| LV eccentricity index | >1.4 | Relative area change % | ≤16 |
| Pericardial effusion | Presence | RV insertion point late gadolinium enhancement | Present |
| PASP during low-dose exercise (<25 W) mmHg | <30 | Full width at half maximum s | >8 |
| Pulmonary transit time s | >6.5 | ||
TAPSE: tricuspid annular plane systolic excursion; RVEF: right ventricular ejection fraction; RV: right ventricular; LV: left ventricular; PASP: pulmonary artery systolic pressure.
The right atrial area is closely related to the degree of pressure in the right atrium, and it is a variable that is relatively easy to determine by echo [53]. Patients with PAH who have a greater right atrial size (RAS) (>27 cm2) have been shown to be at higher risk of mortality or the need for heart transplantation than patients with a lower RAS (<27 cm2, p<0.001), making this a useful prognostic variable [53]. The ESC/ERS guidelines recommend that right atrial area can be used to stratify patients with PAH who are at risk of clinical worsening or death [7]. A right atrial area of <18 cm2 is suggested to confer a low risk (<5%), 18–26 cm2 an intermediate level of risk (5–10%) and a right atrial area of >26 cm2 a high risk [7]. In an outcomes study of patients with severe PH, the prognostic significance of several echocardiographic abnormalities were characterised [24]. Pericardial effusion and indexed right atrial area were shown to be independent predictors of mortality (p=0.003 and p=0.005, respectively) among patients with PAH (WHO FC III–IV) [24]. These two variables, as well as diastolic eccentricity index (degree of septal shift), have also been shown to predict death and/or need for transplantation [24].
TAPSE provides information about the effectiveness of RV systolic function based on the longitudinal function of myocardial RV fibres [40]. In a prospective study, 63 patients with PH underwent RHC followed by transthoracic echo to determine the prognostic significance of TAPSE. A TAPSE of <1.8 cm was identified in patients who had a higher degree of RV systolic dysfunction, right heart remodelling and RV–left ventricular disproportion versus patients with a TAPSE of ≥1.8 cm [26]. Furthermore, patients with a TAPSE of <1.8 cm had much lower rates of survival. Determination of TAPSE has also proved useful for risk stratification in patients with PAH: 1- and 2-year survival estimates were 94% and 88%, respectively, when TAPSE was ≥1.8 cm versus 60% and 50%, respectively, in patients with TAPSE <1.8 cm [26]. This confirms the utility of echocardiographic measurement of TAPSE in predicting risk of severe disease and death in PAH. However, TAPSE can be misleading and often overestimated especially in those patients with very severe impaired right ventricle pump function and a “swinging movement” of the right heart by the moving interventricular septum.
CMRI
CMRI provides an accurate assessment of the right ventricle in PH; it is a reliable tool that can be used to determine disease severity and estimate prognosis. Variables measurable by CMRI that have prognostic value are summarised in table 2 and include RV mass, RV volume and RV function markers. The prognostic indicators for PH have been reviewed previously [21, 60]. CMRI can also be used to supplement echo in the evaluation of pericardial effusion as it provides accurate information on the size and location of the effusion, and on pericardial thickening [15].
The change in RV stroke volume, as measured by CMRI, has been demonstrated to be clinically relevant as it correlates with the change in 6-min walking distance in patients with PH (p<0.0001) [61]. A significant relationship has also been shown between CMRI measures of RV function and the blood biomarker, N-terminal pro-brain natriuretic peptide (NT-proBNP) [62]. NT-proBNP is released from the cardiac ventricle in response to increased pressure or volume overload, and is a highly specific and sensitive measure of RV systolic dysfunction [62]. Quantification of NT-proBNP could be a simple and inexpensive noninvasive alternative to CMRI for evaluating baseline RV systolic dysfunction in PH [62].
The prognostic relevance of planar equilibrium radionuclide angiography (ERNA) of the right ventricle has also been demonstrated in PAH [63]. Patients with a baseline RVEF >25%, as measured by planar ERNA, had better survival than those with an RVEF <25%. Moreover, patients with a stable or increased RVEF at 3–6 months had a trend towards better overall survival and a significantly lower cardiovascular mortality. These results support the recently demonstrated prognostic relevance of baseline and follow-up RVEF measured by MRI in PAH patients [54]. ERNA is affordable and widely available, and therefore may be an alternative to echo or CMRI for measuring RV function in patients with PAH. The limitations of ERNA include the potential for variability in measurements due to the planar measure of a three-dimensional change and the health risks associated with repeated intravenous administration of radioactive tracers [64].
Follow-up assessments
Noninvasive imaging can be used during follow-up to determine clinical deterioration and measure treatment effects, and can also be used to guide treatment decisions. Determining the response to therapy is important so that treatments can be modified as necessary. An approach that is recommended by the ESC and ERS guidelines is “goal-oriented therapy”, a treatment strategy that uses known prognostic indicators as treatment targets [7]. This strategy can delay time to clinical worsening, and extend survival by providing early intervention and therapeutic escalation before the clinical status of the patient deteriorates [52].
Echo can measure right ventricle size and function as a means of evaluating the response to therapy in patients with PAH [52]. Peak tricuspid regurgitation velocity [65], TAPSE, right atrial area [66] and pericardial effusion [14] are further clinically relevant variables that can be measured using echo. Serial transthoracic echocardiograms can measure pericardial effusion during follow-up which, when persistent, can be predictive of poorer outcomes [14]. There are some limitations to using echo as a follow-up assessment to guide therapy, including a lack of consistent methods for reporting RV size and function, and difficulties with obtaining reproducible measurements [67].
A large, prospective, pan-European study (EURO-MR) has demonstrated that CMRI measurements taken at baseline and during follow-up can provide valuable information about patients' responses to PAH-specific therapy [68]. CMRI can measure the effects of treatment on RV volumes [21], RVEF and left ventricular ejection fraction, RV stroke volume index and left ventricular end-diastolic volume index [68]. Contrast-enhanced CMRI, using the extracellular contrasting agent gadopentetate dimeglumine, can be used at follow-up to detect fibrosis at the right ventricle insertion points of the interventricular septum of patients with severe PAH [69]. Contrast-enhanced CMRI can also reveal interventricular septum bowing (i.e. displacement of the intraventricular septum towards the left ventricle) and delayed contrast enhancement (a marker of myocardial abnormalities) [70]. The extent of right ventriculo–arterial coupling can also be measured using both CMRI (for volume assessment) and RHC (for pressure assessment) as part of the follow-up assessment [28, 71]. Severe uncoupling occurs in more severe disease due to a mismatch between increasing arterial load and a failing right ventricle [28].
The future of imaging
New echo techniques
More advanced tools have been developed for noninvasive assessment of PH, some of which are summarised in table 3. New software packages have been developed that can provide further two-dimensional analysis by speckle tracking [9]. Speckle tracking can measure strain (myocardial deformation) and strain rate (the speed at which myocardial deformation occurs) as variables of RV myocardial damage in PH [40]. The extent of myocardial damage in PH, when determined by assessment of strain and strain rate, can be characterised by: 1) normal deformation due to increased afterload and increased contractility; 2) reduced deformation due to increased afterload and preserved contractility; and 3) reduced deformation due to increased afterload and reduced contractility [40].
TABLE 3.
New noninvasive imaging techniques for pulmonary hypertension (PH)
| Variable | Comments |
| Echo | |
| Speckle tracking-derived RV strain and strain rate | RV longitudinal peak systolic strain of ≥ −19% is significantly associated with all-cause mortality [72] |
| RV automated systolic index | Semi-automated and reproducible [73] |
| Three-dimensional echo | Excellent correlation with CMRI, but can underestimate LV volumes [74]; real-time three-dimensional echo could be a time-saving and low-cost alternative to MRI [75] |
| CMRI | |
| RV strain | Systolic strain and strain rate are predictors of right-sided heart failure, clinical deterioration and mortality in patients with PAH [76] |
| Contrast-enhanced CMRI | LGE at the right ventricle insertion point is a marker for advanced disease in PH and is related to clinical worsening [55] |
| Wall sheer stress | Being investigated [77] |
| Exercise CMRI | RV stroke volume does not increase on exercise in patients with PAH (unlike healthy individuals) [78], indicating that these patients need to raise their heart rate to cope with exercise |
| Fast SENC imaging | A through-plane CMRI tagging technique that allows direct measurement of regional function by using a free-breathing single-heartbeat real-time image acquisition [79] |
| Myocardial T1 mapping | Enables quantification of myocardial extracellular volume; may be useful for detecting the early stages of chronic PH, prior to the onset of macroscopic fibrosis [80] |
| PET | |
| Right ventricle 18F-FDG | Higher uptake is associated with greater severity or clinical worsening of PH [81–83] |
| Same-day scanning with 13N-NH3 and 18F-FDG | Feasible for quantifying RV blood flow and metabolism in patients with idiopathic PAH [84] |
Echo: echocardiography; RV: right ventricular; CMRI: cardiac magnetic resonance imaging; SENC: strain-encoded; PET: positron emission tomography; FDG: fluorodeoxyglucose; LV: left ventricular; MRI: magnetic resonance imaging; PAH: pulmonary arterial hypertension; LGE: late gadolinium enhancement.
The advantages of speckle tracking-derived determination of strain and strain rate are that it is dependent on angle, load and geometry. It can accurately measure regional myocardial deformation and there is good correlation between the severity of PH and the degree of RV strain reduction [40]. In comparison to tissue Doppler-based strain imaging, two-dimensional strain imaging by speckle tracking has certain technical advantages; however, the method is volume and load dependent, and not yet standardised. Furthermore, assessments of the right ventricle that are derived from two-dimensional echo alone can show poor consistency [73].
Real-time three-dimensional echo is a superior method to two-dimensional echo that can more accurately analyse RV end-diastolic volume and ejection fraction [30, 85]. The main advantage of three-dimensional echo is that it can provide an evaluation of the right ventricle architecture independent of the geometric assumptions that are inherent to the two-dimensional modality [40]. A new semi-automatic measurement that can be obtained with two-dimensional echo, the RV automated systolic index, generates an apical four-chamber view with automatic endocardial tracking [73]. This method can reduce examiner-dependent variability, but needs to be further evaluated [73]. In patients with chronic disease, three-dimensional echo and two-dimensional speckle-tracking echo are better predictors of RV failure and mortality than existing techniques that evaluate RV variables, such as echo and CMRI [85]. However, in many patients three-dimensional echo cannot be performed since the enlarged right ventricle cannot be seen in its entirety within one view [75].
Echo is commonly carried out while the patient is at rest. Techniques for analysing PAP during exercise, whether by invasive or noninvasive means, have yet to be standardised [86, 87]. Stress (also known as exercise) Doppler echo, which is carried out during dynamic exercise, is a safe and viable tool for screening patients for a pulmonary hypertensive response to exercise; however, it is technically demanding and requires considerable experience [30]. Nevertheless, stress Doppler echo has been used to assess exercise and hypoxia-induced PH, and could be a useful tool in the future for early detection of PH [86]. In summary, unconventional echo methods promise to improve how PH is detected and monitored. Tools such as three-dimensional echo, two-dimensional speckle-tracking and stress Doppler echo can provide an even clearer understanding of right-heart structure and function. In some cases, they can be less observer-dependent; however, they need to be standardised.
New CMRI techniques
New CMRI techniques continue to be developed (table 3). Global measures of ventricular function, as recorded by conventional CMRI, are strong predictors of future heart failure and prognosis. However, they are insensitive to reductions in regional performance (e.g. myocardial strain and torsion) [88]. CMRI myocardial tagging is an established method that can enable measurement of regional left ventricular deformation [79]. This technique tracks differences in CMRI signal intensity between tagged and undisturbed regions of tissue, following on from which mathematical techniques are applied to reconstruct a three-dimensional image of the heart [89]. The alternative tagging technique of fast strain-encoded imaging can analyse regional biventricular strain to identify RV dysfunction in patients with normal global RV function [79]. For measuring extracellular volume fraction, T1 mapping, which measures the longitudinal relaxation time of a tissue, by CMRI has been shown experimentally to correlate with pulmonary haemodynamics, RV arterial coupling and RV performance [80]. Measurement of myocardial T1 before and after the infusion of a contrasting agent allows quantification of the equilibrium contrast extracellular volume. This technique may be used in the future for evaluating myocardial damage and function in patients with PH [80]. The hybrid technique of MRI-guided catheterisation enables simultaneous structural, functional and haemodynamic assessment, and the calculation of variables including RV afterload, contractility and right ventricle–pulmonary artery coupling. This will have benefit for diagnosis and prognosis, as well as for interventional MRI procedures [90].
Positron emission tomography
Evidence suggests that during PH there is a metabolic shift from mitochondrial oxidative phosphorylation to glycolysis, resulting in an increase in glucose uptake [91]. This reliance on glycolysis leads to less efficient production of ATP, and subsequent impairment of RV contractility and reduction in cardiac output [92]. Glucose metabolism in the right heart and pulmonary vasculature can be measured using 2-deoxy-2-18F-fluorodeoxyglucose PET. In PH, 18F-fluorodeoxyglucose accumulation (a measure of glycolytic rate) has been shown to be increased in the right ventricle free wall [81, 82] (table 3). PET has been advocated for assessing PH severity and clinical outcome [10], and could provide a sensitive measure of response to treatment [82]. However, despite studies that show the feasibility of PET in patients with PH, there have been conflicting results, and conclusive evidence of the usefulness of measuring metabolic shift in RV dysfunction has yet to be provided [2]. The functional imaging of PET can be combined with the anatomical imaging of MRI. This hybrid imaging technique provides functional imaging, perfusion, tissue characterisation and flow imaging, giving an improved understanding of the right ventricle compared with PET and MRI used in isolation [2, 93].
Conclusion
Imaging techniques provide anatomical and functional insight into the structure and function of the heart in PH. Echo is an established, reliable and cost-effective tool for enabling early assessment of patients with suspected PH, yet techniques that offer complementary information, such as CMRI, are becoming increasingly important. The development of more advanced imaging tools will increase our understanding of the pathophysiological factors that lead to RV failure in PH, and integrating these tools into diagnostic algorithms will further optimise the detection and management of PH in the future.
Supplementary Material
Acknowledgements
The authors would like to thank Kate Bradford from PAREXEL (Uxbridge, UK) for medical writing assistance, funded by Actelion Pharmaceuticals Ltd (Allschwil, Switzerland).
Footnotes
Conflict of interest: Disclosures can be found alongside the online version of this article at err.ersjournals.com
Provenance: Publication of this peer-reviewed article was sponsored by Actelion Pharmaceuticals Ltd, Allschwil, Switzerland (principal sponsor, European Respiratory Review issue 138).
References
- 1.Chin KM, Kim NH, Rubin LJ. The right ventricle in pulmonary hypertension. Coron Artery Dis 2005; 16: 13–18. [DOI] [PubMed] [Google Scholar]
- 2.van de Veerdonk MC, Marcus JT, Bogaard HJ, et al. State of the art: advanced imaging of the right ventricle and pulmonary circulation in humans (2013 Grover Conference series). Pulm Circ 2014; 4: 158–168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.D'Alonzo GE, Barst RJ, Ayres SM, et al. Survival in patients with primary pulmonary hypertension. Results from a national prospective registry. Ann Intern Med 1991; 115: 343–349. [DOI] [PubMed] [Google Scholar]
- 4.Benza RL, Miller DP, Gomberg-Maitland M, et al. Predicting survival in pulmonary arterial hypertension: insights from the Registry to Evaluate Early and Long-Term Pulmonary Arterial Hypertension Disease Management (REVEAL). Circulation 2010; 122: 164–172. [DOI] [PubMed] [Google Scholar]
- 5.Matthews JC, McLaughlin V. Acute right ventricular failure in the setting of acute pulmonary embolism or chronic pulmonary hypertension: a detailed review of the pathophysiology, diagnosis, and management. Curr Cardiol Rev 2008; 4: 49–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Voelkel NF, Quaife RA, Leinwand LA, et al. Right ventricular function and failure: report of a National Heart, Lung, and Blood Institute working group on cellular and molecular mechanisms of right heart failure. Circulation 2006; 114: 1883–1891. [DOI] [PubMed] [Google Scholar]
- 7.Galiè N, Humbert M, Vachiery JL, et al. 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension. Eur Respir J 2015; 46: 903–975. [DOI] [PubMed] [Google Scholar]
- 8.Rosenkranz S, Preston IR. Right heart catheterisation: best practice and pitfalls in pulmonary hypertension. Eur Respir Rev 2015; 24: 642–652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gerges M, Gerges C, Lang IM. Advanced imaging tools rather than hemodynamics should be the primary approach for diagnosing, following, and managing pulmonary arterial hypertension. Can J Cardiol 2015; 31: 521–528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ahmadi A, Ohira H, Mielniczuk LM. FDG PET imaging for identifying pulmonary hypertension and right heart failure. Curr Cardiol Rep 2015; 17: 555. [DOI] [PubMed] [Google Scholar]
- 11.Bossone E, Nanda NC, Naeije R. Imaging the right heart: a challenging road map. Echocardiography 2015; 32: Suppl. 1, S1–S2. [DOI] [PubMed] [Google Scholar]
- 12.Rajaram S, Swift AJ, Capener D, et al. Comparison of the diagnostic utility of cardiac magnetic resonance imaging, computed tomography, and echocardiography in assessment of suspected pulmonary arterial hypertension in patients with connective tissue disease. J Rheumatol 2012; 39: 1265–1274. [DOI] [PubMed] [Google Scholar]
- 13.Moceri P, Baudouy D, Chiche O, et al. Imaging in pulmonary hypertension: focus on the role of echocardiography. Arch Cardiovasc Dis 2014; 107: 261–271. [DOI] [PubMed] [Google Scholar]
- 14.Batal O, Dardari Z, Costabile C, et al. Prognostic value of pericardial effusion on serial echocardiograms in pulmonary arterial hypertension. Echocardiography 2015; 32: 1471–1476. [DOI] [PubMed] [Google Scholar]
- 15.Klein AL, Abbara S, Agler DA, et al. American Society of Echocardiography clinical recommendations for multimodality cardiovascular imaging of patients with pericardial disease. J Am Soc Echocardiogr 2013; 26: 965–1012. [DOI] [PubMed] [Google Scholar]
- 16.Greiner S, Jud A, Aurich M, et al. Reliability of noninvasive assessment of systolic pulmonary artery pressure by Doppler echocardiography compared to right heart catheterization: analysis in a large patient population. J Am Heart Assoc 2014; 3: e001103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hoeper MM, Bogaard HJ, Condliffe R, et al. Definitions and diagnosis of pulmonary hypertension. J Am Coll Cardiol 2013; 62: Suppl. 25, D42–D50. [DOI] [PubMed] [Google Scholar]
- 18.Grünig E, Henn P, D'Andrea A, et al. Reference values for and determinants of right atrial area in healthy adults by 2-dimensional echocardiography. Circ Cardiovasc Imaging 2013; 6: 117–124. [DOI] [PubMed] [Google Scholar]
- 19.Maceira AM, Cosin-Sales J, Roughton M, et al. Reference right atrial dimensions and volume estimation by steady state free precession cardiovascular magnetic resonance. J Cardiovasc Magn Reson 2013; 15: 29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Grünig E, Biskupek J, D'Andrea A, et al. Reference ranges for and determinants of right ventricular area in healthy adults by two-dimensional echocardiography. Respiration 2015; 89: 284–293. [DOI] [PubMed] [Google Scholar]
- 21.Peacock AJ, Vonk Noordegraaf A. Cardiac magnetic resonance imaging in pulmonary arterial hypertension. Eur Respir Rev 2013; 22: 526–534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Rudski LG, Lai WW, Afilalo J, et al. Guidelines for the echocardiographic assessment of the right heart in adults: a report from the American Society of Echocardiography. J Am Soc Echocardiogr 2010; 23: 685–713. [DOI] [PubMed] [Google Scholar]
- 23.Kawut SM, Lima JA, Barr RG, et al. Sex and race differences in right ventricular structure and function: the multi-ethnic study of atherosclerosis – right ventricle study. Circulation 2011; 123: 2542–2551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Raymond RJ, Hinderliter AL, Willis PW, et al. Echocardiographic predictors of adverse outcomes in primary pulmonary hypertension. J Am Coll Cardiol 2002; 39: 1214–1219. [DOI] [PubMed] [Google Scholar]
- 25.Vonk-Noordegraaf A, Souza R. Cardiac magnetic resonance imaging: what can it add to our knowledge of the right ventricle in pulmonary arterial hypertension? Am J Cardiol 2012; 110: Suppl. 6, 25S–31S. [DOI] [PubMed] [Google Scholar]
- 26.Forfia PR, Fisher MR, Mathai SC, et al. Tricuspid annular displacement predicts survival in pulmonary hypertension. Am J Respir Crit Care Med 2006; 174: 1034–1041. [DOI] [PubMed] [Google Scholar]
- 27.Kim WH, Otsuji Y, Yuasa T, et al. Evaluation of right ventricular dysfunction in patients with cardiac amyloidosis using Tei index. J Am Soc Echocardiogr 2004; 17: 45–49. [DOI] [PubMed] [Google Scholar]
- 28.Sanz J, Garcia-Alvarez A, Fernández-Friera L, et al. Right ventriculo-arterial coupling in pulmonary hypertension: a magnetic resonance study. Heart 2012; 98: 238–243. [DOI] [PubMed] [Google Scholar]
- 29.Grünig E, Barner A, Bell M, et al. Non-invasive diagnosis of pulmonary hypertension: ESC/ERS Guidelines with Updated Commentary of the Cologne Consensus Conference 2011. Int J Cardiol 2011; 154: Suppl. 1, S3–S12. [DOI] [PubMed] [Google Scholar]
- 30.Bossone E, D'Andrea A, D'Alto M, et al. Echocardiography in pulmonary arterial hypertension: from diagnosis to prognosis. J Am Soc Echocardiogr 2013; 26: 1–14. [DOI] [PubMed] [Google Scholar]
- 31.Nagel C, Henn P, Ehlken N, et al. Stress Doppler echocardiography for early detection of systemic sclerosis-associated pulmonary arterial hypertension. Arthritis Res Ther 2015; 17: 165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.McMurray JJ, Adamopoulos S, Anker SD, et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2012. Eur Heart J 2012; 33: 1787–1847. [DOI] [PubMed] [Google Scholar]
- 33.Ribeiro A, Lindmarker P, Johnsson H, et al. Pulmonary embolism: one-year follow-up with echocardiography doppler and five-year survival analysis. Circulation 1999; 99: 1325–1330. [DOI] [PubMed] [Google Scholar]
- 34.Marrone G, Mamone G, Luca A, et al. The role of 1.5T cardiac MRI in the diagnosis, prognosis and management of pulmonary arterial hypertension. Int J Cardiovasc Imaging 2010; 26: 665–681. [DOI] [PubMed] [Google Scholar]
- 35.Barrier M, Meloche J, Jacob MH, et al. Today's and tomorrow's imaging and circulating biomarkers for pulmonary arterial hypertension. Cell Mol Life Sci 2012; 69: 2805–2831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kuehne T, Yilmaz S, Steendijk P, et al. Magnetic resonance imaging analysis of right ventricular pressure-volume loops: in vivo validation and clinical application in patients with pulmonary hypertension. Circulation 2004; 110: 2010–2016. [DOI] [PubMed] [Google Scholar]
- 37.Humbert M, Coghlan JG, Khanna D. Early detection and management of pulmonary arterial hypertension. Eur Respir Rev 2012; 21: 306–312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Simonneau G, Gatzoulis MA, Adatia I, et al. Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol 2013; 62: Suppl. 25, D34–D41. [DOI] [PubMed] [Google Scholar]
- 39.Humbert M, Sitbon O, Chaouat A, et al. Survival in patients with idiopathic, familial, and anorexigen-associated pulmonary arterial hypertension in the modern management era. Circulation 2010; 122: 156–163. [DOI] [PubMed] [Google Scholar]
- 40.D'Alto M, Romeo E, Argiento P, et al. Pulmonary arterial hypertension: the key role of echocardiography. Echocardiography 2015; 32: Suppl. 1, S23–S37. [DOI] [PubMed] [Google Scholar]
- 41.Habib G, Torbicki A. The role of echocardiography in the diagnosis and management of patients with pulmonary hypertension. Eur Respir Rev 2010; 19: 288–299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Coghlan JG, Denton CP, Grünig E, et al. Evidence-based detection of pulmonary arterial hypertension in systemic sclerosis: the DETECT study. Ann Rheum Dis 2014; 73: 1340–1349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Howard LS, Grapsa J, Dawson D, et al. Echocardiographic assessment of pulmonary hypertension: standard operating procedure. Eur Respir Rev 2012; 21: 239–248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fisher MR, Forfia PR, Chamera E, et al. Accuracy of Doppler echocardiography in the hemodynamic assessment of pulmonary hypertension. Am J Respir Crit Care Med 2009; 179: 615–621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Bradlow WM, Gibbs JS, Mohiaddin RH. Cardiovascular magnetic resonance in pulmonary hypertension. J Cardiovasc Magn Reson 2012; 14: 6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fang JC, DeMarco T, Givertz MM, et al. World Health Organization Pulmonary Hypertension group 2: Pulmonary hypertension due to left heart disease in the adult – a summary statement from the Pulmonary Hypertension Council of the International Society for Heart and Lung Transplantation. J Heart Lung Transplant 2012; 31: 913–933. [DOI] [PubMed] [Google Scholar]
- 47.Crawley SF, Johnson MK, Dargie HJ, et al. LA volume by CMR distinguishes idiopathic from pulmonary hypertension due to HFpEF. JACC Cardiovasc Imaging 2013; 6: 1120–1121. [DOI] [PubMed] [Google Scholar]
- 48.Nayak KS, Nielsen JF, Bernstein MA, et al. Cardiovascular magnetic resonance phase contrast imaging. J Cardiovasc Magn Reson 2015; 17: 71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Sanz J, Kuschnir P, Rius T, et al. Pulmonary arterial hypertension: noninvasive detection with phase-contrast MR imaging. Radiology 2007; 243: 70–79. [DOI] [PubMed] [Google Scholar]
- 50.Tunariu N, Gibbs SJ, Win Z, et al. Ventilation-perfusion scintigraphy is more sensitive than multidetector CTPA in detecting chronic thromboembolic pulmonary disease as a treatable cause of pulmonary hypertension. J Nucl Med 2007; 48: 680–684. [DOI] [PubMed] [Google Scholar]
- 51.Ley S, Kauczor HU, Heussel CP, et al. Value of contrast-enhanced MR angiography and helical CT angiography in chronic thromboembolic pulmonary hypertension. Eur Radiol 2003; 13: 2365–2371. [DOI] [PubMed] [Google Scholar]
- 52.Baldi F, Fuso L, Arrighi E, et al. Optimal management of pulmonary arterial hypertension: prognostic indicators to determine treatment course. Ther Clin Risk Manag 2014; 10: 825–839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Bustamante-Labarta M, Perrone S, De La Fuente RL, et al. Right atrial size and tricuspid regurgitation severity predict mortality or transplantation in primary pulmonary hypertension. J Am Soc Echocardiogr 2002; 15: 1160–1164. [DOI] [PubMed] [Google Scholar]
- 54.van de Veerdonk MC, Kind T, Marcus JT, et al. Progressive right ventricular dysfunction in patients with pulmonary arterial hypertension responding to therapy. J Am Coll Cardiol 2011; 58: 2511–2519. [DOI] [PubMed] [Google Scholar]
- 55.Freed BH, Gomberg-Maitland M, Chandra S, et al. Late gadolinium enhancement cardiovascular magnetic resonance predicts clinical worsening in patients with pulmonary hypertension. J Cardiovasc Magn Reson 2012; 14: 11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.van Wolferen SA, Marcus JT, Boonstra A, et al. Prognostic value of right ventricular mass, volume, and function in idiopathic pulmonary arterial hypertension. Eur Heart J 2007; 28: 1250–1257. [DOI] [PubMed] [Google Scholar]
- 57.Hagger D, Condliffe R, Woodhouse N, et al. Ventricular mass index correlates with pulmonary artery pressure and predicts survival in suspected systemic sclerosis-associated pulmonary arterial hypertension. Rheumatology 2009; 48: 1137–1142. [DOI] [PubMed] [Google Scholar]
- 58.Gan CT, Lankhaar JW, Westerhof N, et al. Noninvasively assessed pulmonary artery stiffness predicts mortality in pulmonary arterial hypertension. Chest 2007; 132: 1906–1912. [DOI] [PubMed] [Google Scholar]
- 59.Swift AJ, Telfer A, Rajaram S, et al. Dynamic contrast-enhanced magnetic resonance imaging in patients with pulmonary arterial hypertension. Pulm Circ 2014; 4: 61–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Swift AJ, Wild JM, Nagle SK, et al. Quantitative magnetic resonance imaging of pulmonary hypertension: a practical approach to the current state of the art. J Thorac Imaging 2014; 29: 68–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.van Wolferen SA, van de Veerdonk MC, Mauritz GJ, et al. Clinically significant change in stroke volume in pulmonary hypertension. Chest 2011; 139: 1003–1009. [DOI] [PubMed] [Google Scholar]
- 62.Blyth KG, Groenning BA, Mark PB, et al. NT-proBNP can be used to detect right ventricular systolic dysfunction in pulmonary hypertension. Eur Respir J 2007; 29: 737–744. [DOI] [PubMed] [Google Scholar]
- 63.Courand PY, Pina Jomir G, Khouatra C, et al. Prognostic value of right ventricular ejection fraction in pulmonary arterial hypertension. Eur Respir J 2015; 45: 139–149. [DOI] [PubMed] [Google Scholar]
- 64.Naeije R, Ghio S. More on the right ventricle in pulmonary hypertension. Eur Respir J 2015; 45: 33–35. [DOI] [PubMed] [Google Scholar]
- 65.Grünig E, Tiede H, Enyimayew EO, et al. Assessment and prognostic relevance of right ventricular contractile reserve in patients with severe pulmonary hypertension. Circulation 2013; 128: 2005–2015. [DOI] [PubMed] [Google Scholar]
- 66.Tonelli AR, Conci D, Tamarappoo BK, et al. Prognostic value of echocardiographic changes in patients with pulmonary arterial hypertension receiving parenteral prostacyclin therapy. J Am Soc Echocardiogr 2014; 27: 733–741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.McLaughlin VV, Gaine SP, Howard LS, et al. Treatment goals of pulmonary hypertension. J Am Coll Cardiol 2013; 62: Suppl. 25, D73–D81. [DOI] [PubMed] [Google Scholar]
- 68.Peacock AJ, Crawley S, McLure L, et al. Changes in right ventricular function measured by cardiac magnetic resonance imaging in patients receiving pulmonary arterial hypertension-targeted therapy: the EURO-MR study. Circ Cardiovasc Imaging 2014; 7: 107–114. [DOI] [PubMed] [Google Scholar]
- 69.McCann GP, Beek AM, Vonk-Noordegraaf A, et al. Delayed contrast-enhanced magnetic resonance imaging in pulmonary arterial hypertension. Circulation 2005; 112: e268. [DOI] [PubMed] [Google Scholar]
- 70.Blyth KG, Groenning BA, Martin TN, et al. Contrast enhanced-cardiovascular magnetic resonance imaging in patients with pulmonary hypertension. Eur Heart J 2005; 26: 1993–1999. [DOI] [PubMed] [Google Scholar]
- 71.Trip P, Kind T, van de Veerdonk MC, et al. Accurate assessment of load-independent right ventricular systolic function in patients with pulmonary hypertension. J Heart Lung Transplant 2013; 32: 50–55. [DOI] [PubMed] [Google Scholar]
- 72.Haeck ML, Scherptong RW, Marsan NA, et al. Prognostic value of right ventricular longitudinal peak systolic strain in patients with pulmonary hypertension. Circ Cardiovasc Imaging 2012; 5: 628–636. [DOI] [PubMed] [Google Scholar]
- 73.Greiner S, André F, Heimisch M, et al. Non-invasive quantification of right ventricular systolic function by echocardiography: a new semi-automated approach. Clin Res Cardiol 2013; 102: 229–235. [DOI] [PubMed] [Google Scholar]
- 74.Kühl HP, Schreckenberg M, Rulands D, et al. High-resolution transthoracic real-time three-dimensional echocardiography: quantitation of cardiac volumes and function using semi-automatic border detection and comparison with cardiac magnetic resonance imaging. J Am Coll Cardiol 2004; 43: 2083–2090. [DOI] [PubMed] [Google Scholar]
- 75.Leibundgut G, Rohner A, Grize L, et al. Dynamic assessment of right ventricular volumes and function by real-time three-dimensional echocardiography: a comparison study with magnetic resonance imaging in 100 adult patients. J Am Soc Echocardiogr 2010; 23: 116–126. [DOI] [PubMed] [Google Scholar]
- 76.Sachdev A, Villarraga HR, Frantz RP, et al. Right ventricular strain for prediction of survival in patients with pulmonary arterial hypertension. Chest 2011; 139: 1299–1309. [DOI] [PubMed] [Google Scholar]
- 77.Truong U, Fonseca B, Dunning J, et al. Wall shear stress measured by phase contrast cardiovascular magnetic resonance in children and adolescents with pulmonary arterial hypertension. J Cardiovasc Magn Reson 2013; 15: 81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Holverda S, Gan CT, Marcus JT, et al. Impaired stroke volume response to exercise in pulmonary arterial hypertension. J Am Coll Cardiol 2006; 47: 1732–1733. [DOI] [PubMed] [Google Scholar]
- 79.Shehata ML, Harouni AA, Skrok J, et al. Regional and global biventricular function in pulmonary arterial hypertension: a cardiac MR imaging study. Radiology 2013; 266: 114–122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Garcia-Alvarez A, Garcia-Lunar I, Pereda D, et al. Association of myocardial T1-mapping CMR with hemodynamics and RV performance in pulmonary hypertension. JACC Cardiovasc Imaging 2015; 8: 76–82. [DOI] [PubMed] [Google Scholar]
- 81.Fang W, Zhao L, Xiong CM, et al. Comparison of 18F-FDG uptake by right ventricular myocardium in idiopathic pulmonary arterial hypertension and pulmonary arterial hypertension associated with congenital heart disease. Pulm Circ 2012; 2: 365–372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Oikawa M, Kagaya Y, Otani H, et al. Increased [18F]fluorodeoxyglucose accumulation in right ventricular free wall in patients with pulmonary hypertension and the effect of epoprostenol. J Am Coll Cardiol 2005; 45: 1849–1855. [DOI] [PubMed] [Google Scholar]
- 83.Tatebe S, Fukumoto Y, Oikawa-Wakayama M, et al. Enhanced [18F]fluorodeoxyglucose accumulation in the right ventricular free wall predicts long-term prognosis of patients with pulmonary hypertension: a preliminary observational study. Eur Heart J Cardiovasc Imaging 2014; 15: 666–672. [DOI] [PubMed] [Google Scholar]
- 84.Bokhari S, Raina A, Rosenweig EB, et al. PET imaging may provide a novel biomarker and understanding of right ventricular dysfunction in patients with idiopathic pulmonary arterial hypertension. Circ Cardiovasc Imaging 2011; 4: 641–647. [DOI] [PubMed] [Google Scholar]
- 85.Vitarelli A, Mangieri E, Terzano C, et al. Three-dimensional echocardiography and 2D-3D speckle-tracking imaging in chronic pulmonary hypertension: diagnostic accuracy in detecting hemodynamic signs of right ventricular (RV) failure. J Am Heart Assoc 2015; 4: e001584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Grünig E, Weissmann S, Ehlken N, et al. Stress Doppler echocardiography in relatives of patients with idiopathic and familial pulmonary arterial hypertension: results of a multicenter European analysis of pulmonary artery pressure response to exercise and hypoxia. Circulation 2009; 119: 1747–1757. [DOI] [PubMed] [Google Scholar]
- 87.Lewis GD, Bossone E, Naeije R, et al. Pulmonary vascular hemodynamic response to exercise in cardiopulmonary diseases. Circulation 2013; 128: 1470–1479. [DOI] [PubMed] [Google Scholar]
- 88.Shehata ML, Cheng S, Osman NF, et al. Myocardial tissue tagging with cardiovascular magnetic resonance. J Cardiovasc Magn Reson 2009; 11: 55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Moore CC, Lugo-Olivieri CH, McVeigh ER, et al. Three-dimensional systolic strain patterns in the normal human left ventricle: characterization with tagged MR imaging. Radiology 2000; 214: 453–466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Rogers T, Ratnayaka K, Lederman RJ. MRI catheterization in cardiopulmonary disease. Chest 2014; 145: 30–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Cottrill KA, Chan SY. Metabolic dysfunction in pulmonary hypertension: the expanding relevance of the Warburg effect. Eur J Clin Invest 2013; 43: 855–865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Archer SL, Fang YH, Ryan JJ, et al. Metabolism and bioenergetics in the right ventricle and pulmonary vasculature in pulmonary hypertension. Pulm Circ 2013; 3: 144–152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Nensa F, Poeppel TD, Beiderwellen K, et al. Hybrid PET/MR imaging of the heart: feasibility and initial results. Radiology 2013; 268: 366–373. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.