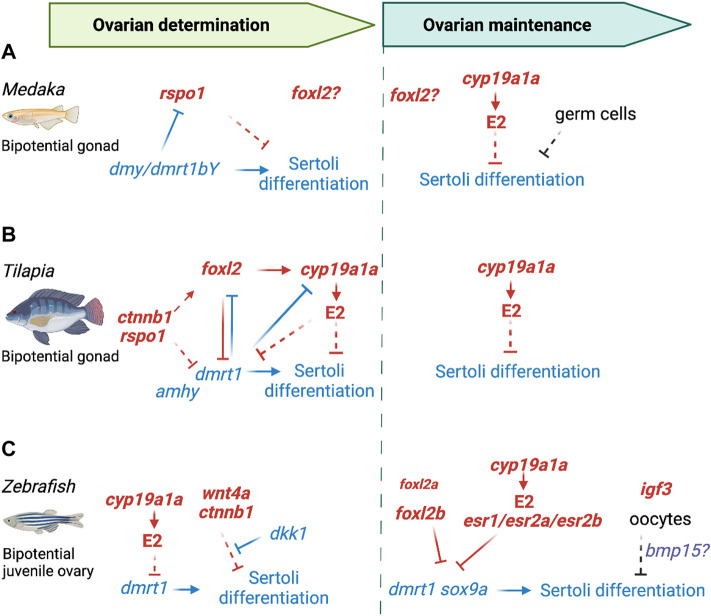
FIGURE 3.
Genetic regulation of pre-granulosa cell differentiation in fish. (A) In medaka, in the absence of the Y-linked sex determining gene dmy, pro-ovarian genes (rspo1 and later foxl2) are upregulated. rspo1, which is directly repressed by Dmy, is sufficient to fully drive ovarian differentiation in XY gonads. The functional role of foxl2 remains to be determined. cyp19a1 is only involved in the maintenance of ovarian differentiation. Loss of germ cells prevents the maintenance of granulosa cell fate. (B) In tilapia, in the absence of the Y-linked sex determining gene amhy, pro-ovarian genes are quickly upregulated. Repression of Wnt/β-Catenin pathway through either rspo1 KO or exposure to inhibitors leads to repression of foxl2 and upregulation of dmrt1, causing Sertoli cell differentiation. Loss of foxl2 results in absence of cyp19a1 expression and upregulation of dmrt1 at time of sex determination. There is mutual antagonism between pro-testis gene dmrt1 and pro-ovary genes foxl2/cyp19a1. cyp19a1 is also involved in the maintenance of ovarian identity. (C) In zebrafish, all larvae first develop a bipotential juvenile ovary. Loss of cyp19a1a leads to upregulation of dmrt1 and Sertoli cell differentiation. Repression of the Wnt/β-Catenin pathway, through either wnt4a KO or overexpression of inhibitor Dkk1 causes Sertoli cell differentiation. Combined loss of either foxl2a/foxl2b, or the three estrogen receptors impairs the maintenance of granulosa cell identity, resulting in complete ovary-to-testis sex reversal. Oocytes are required for the maintenance of granulosa cell fate. Loss of genes involved in PGC/oocyte development, such as igf3, results in ovary-to-testis sex-reversal. Bmp15 is suspected to be the oocyte secreted factor that directly acts on supporting cells.