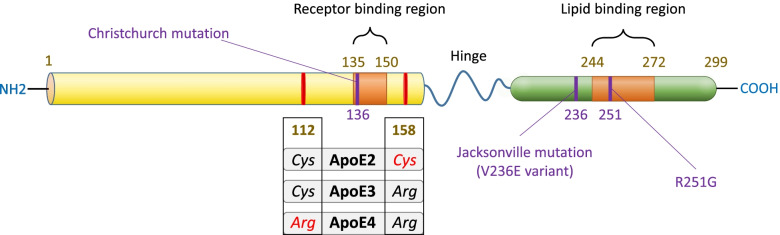
Fig. 1.
ApoE protein structure and mutations. The human apoE protein is characterized by three main domains, an N-terminal containing the receptor-binding region and a C-terminal domain where the lipid-binding region is located. A flexible hinge region joins N-terminal and C-terminal domains. Mutations at positions 112 and 158 will give rise to the most prevalent apoE isoforms, apoE2, apoE3, and apoE4. Other apoE variants characterized are the Christchurch mutation, which presents a Ser-136 variant [R136S]. ApoE Jacksonville mutation, also called V236E, variant is located at position 236 proximal to the lipid-binding region and reduces apoE self-aggregation. Lastly, the apoE R251G variant induces a single amino acid switch at position 251 and has been initially linked with decreased risk of suffering AD