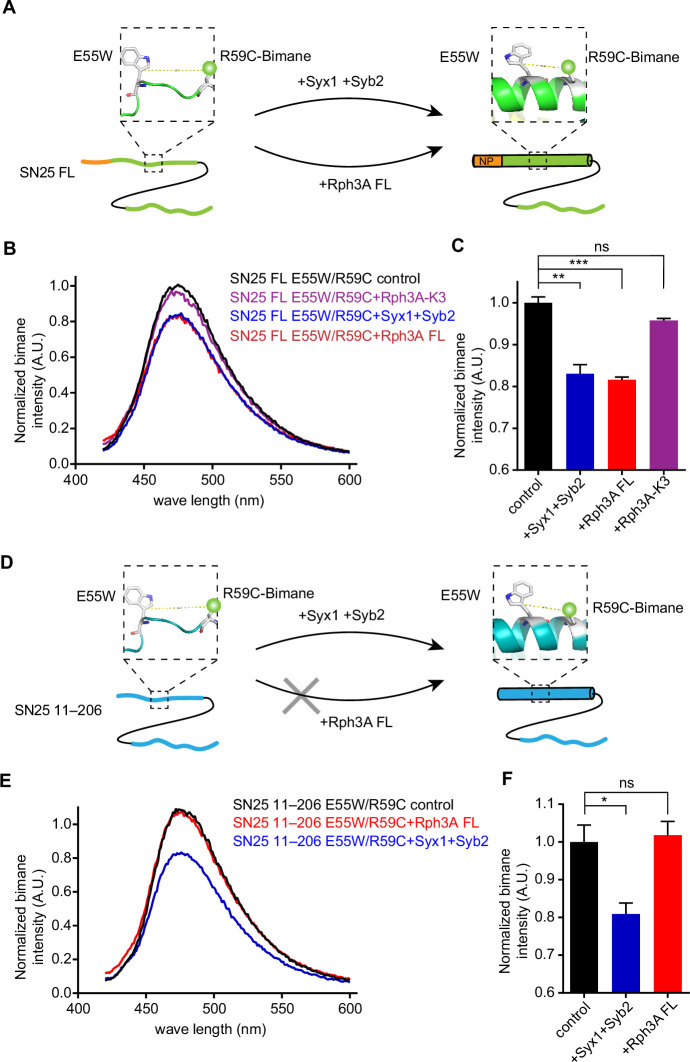
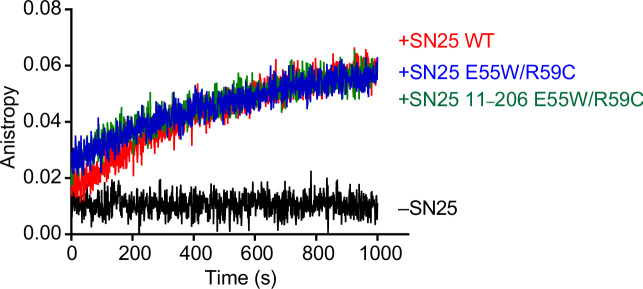
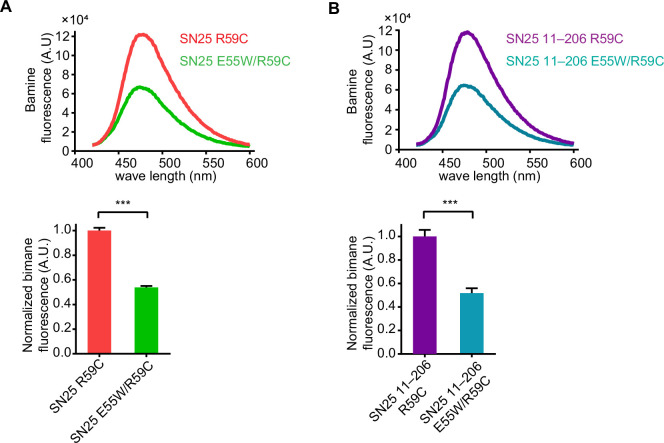
Figure 6. Conformation change of SN25 induced by Rph3A.
(A, D) Illustration of the conformation change from random coils to α-helix in the SN1 of SN25 induced by Syx1 (2–253) and Syb2 (29–93), or by Rph3A, monitored by bimane-tryptophan quenching assay. Tryptophan was introduced at residue E55 and bimane fluorescence was labeled on residue R59C in SN25 FL (A) or SN25 11–206 (D). (B, C) Quenching of bimane fluorescence on the SN25 E55W/R59C with the addition of Syx1 and Syb2, and the addition of Rph3A FL or Rph3A-K3 mutant (B) and quantification of the results observed at 474 nm (C). (E, F) Quenching of bimane fluorescence on the SN25 (11–206) E55W/R59C with the addition of Syx1 and Syb2, or the addition of Rph3A FL (E) and quantification of the results observed at 474 nm (F). Data are presented as mean ± SEM (n=3), technical replicates. The significance were examined by one-way analysis of variance (ANOVA). *p<0.05; **p<0.01; ***p<0.001; ns, not significant.