Abstract
Background:
The optimal treatments for atrial fibrillation in heart failure patients are controversial. The present study compared the efficacy of catheter ablation and medical therapy in patients with atrial fibrillation and heart failure.
Methods:
Pubmed, Embase, Cochrane Library, and Web of Science were searched until January 15, 2022. Randomized controlled trials comparing catheter ablation for atrial fibrillation with medical therapy in patients with atrial fibrillation and heart failure were enrolled. Primary outcome was all-cause mortality. Secondary outcomes included the heart failure hospitalization and the change in left ventricular ejection fraction, 6-minute walk test distance, peak oxygen consumption, and Minnesota Living with Heart Failure questionnaire score.
Results:
Totally 8 randomized controlled trials involving 1693 patients were included. Compared with medical therapy, catheter ablation significantly reduced all-cause mortality (risk ratios = 0.60, 95% Cl: 0.45 to 0.80, P < .001) and hospitalization due to heart failure (risk ratios = 0.58, 95% Cl: 0.46 to 0.73, P < .001), improved left ventricular ejection fraction (mean difference = 5.25%, 95% CI: 2.78% to 7.71%, P < .001), improved the performance of 6-minute walk test (mean difference = 28.83 m, 95% CI: 8.61 to 49.05 m, P = .005), increased peak oxygen consumption (mean difference = 3.11 mL/kg/min, 95% CI: 1.04 to 5.18 mL/kg/min, P = .003), and reduced Minnesota Living with Heart Failure score (mean difference = −8.45, 95% CI: −16.28 to −0.62, P = .03).
Conclusion:
In heart failure patients with atrial fibrillation, catheter ablation provides more benefits over medical therapy in the important clinical outcomes, exercise capacity, and quality of life.
Keywords: Atrial fibrillation, heart failure, catheter ablation
Highlights
In heart failure (HF) patients with atrial fibrillation, catheter ablation reduced all-cause mortality and HF hospitalization and improved 6-minute walk test distance, peak oxygen consumption, and Minnesota Living with Heart Failure questionnaire score.
About reducing all-cause mortality, catheter ablation is effective in HF patients with preserved ejection fraction, as well as reduced ejection fraction.
This meta-analysis included 8 randomized controlled trials involving 1693 patients with coexisting atrial fibrillation and HF.
Introduction
Atrial fibrillation (AF) and heart failure (HF) are becoming a growing cardiovascular diseases worldwide. The prevalence of AF and HF is increasing exponentially and is associated with an increased burden of cardiac risk factors and improved survival in patients with structural heart disease.1 Emerging evidence suggests that AF may be associated with an increased risk of sudden cardiac death.2 Atrial fibrillation and HF share many predisposing risk factors and coincide in many patients.3 More importantly, AF and HF collaborate to aggravate each other. On the one hand, AF increases in prevalence with HF severity. On the other hand, AF is common pathogenesis of HF decompensation and is associated with poor outcomes in patients with HF.4 Currently, optimal treatments for AF and HF comorbidities are still lacking, and it remains a major challenge for clinicians.
In current guidelines, both heart rate and rhythm control strategies are recommended for patients with AF as the primary means of symptom control.3 In the view of physiology, AF can exacerbate HF through several mechanisms independent of rapid ventricular rates such as irregular rate and loss of atrial kick.4 And given the adverse effects of irregular rhythm, rapid ventricular response, and loss of atrial constriction caused by AF on cardiac output, rhythm control seems to be more reasonable for AF patients with HF. There is an observational study suggesting that rhythm control may produce better outcomes than pharmacological rate control and that pure rate control strategies may even independently increase the risk of all-cause mortality.5 However, long-term use of antiarrhythmic drugs can cause a series of problems. Poor patient adherence to medication has long been a problem for healthcare workers. Inevitably, antiarrhythmic drugs also have arrhythmogenic effects. Therefore, most investigators believe that poor efficacy in maintaining sinus rhythm (SR) and adverse effects of antiarrhythmics offset the advantages of maintaining SR.
Compelling evidence suggests that catheter ablation (CA) is more effective in restoring and maintaining SR than antiarrhythmic drugs.3 Ablation vs. Amiodarone for Treatment of Persistent Atrial Fibrillation in Patients With Congestive Heart Failure and an Implanted Device (AATAC) trial reported that compared with amiodarone, CA was not only more effective in maintaining SR but also improved long-term outcomes in AF patients with HF.6 Therefore, it is speculated that CA might be a better strategy for AF patients with HF, compared with medical therapy. Recently, a sub-analysis of the Catheter Ablation Versus Antiarrhythmic Drug Therapy for Atrial Fibrillation (CABANA) trial showed that CA significantly reduced all-cause mortality in patients with coexisting AF and HF compared with medical therapy.7 Therefore, we performed an updated systematic review and meta-analysis of randomized controlled trials (RCTs) to assess the effects of CA versus medical therapy on all-cause mortality, HF hospitalization, left ventricular ejection fraction (LVEF), peak oxygen consumption (peak VO2), and quality of life in patients with coexisting AF and HF.
Methods
Ethical approval and patient consent were not required for this meta-analysis, as it was based on a secondary analysis of previously published studies guided by Preferred Reporting Item for Systematic Reviews and Meta-Analyses.
Search Strategy and Eligibility Criteria
Four databases including Pubmed, Embase, Cochrane Library, and Web of Science (until January 15, 2022) were searched for RCTs that compared CA against medical therapy in patients with AF and HF. The following Mesh or terms were used: (Heart failure OR cardiac failure OR myocardial failure OR heart decompensation OR left ventricular systolic dysfunction OR left ventricular diastolic dysfunction) AND (atrial fibrillation OR atrial fibrillat* OR auricular fibrillat* OR atrium fibrillat* OR AF) AND (CA OR ablat* OR radiofrequency). We also checked the reference lists of all key articles for any additional eligible papers. The major inclusion criteria were as follows: (a) enrolling patients with AF and HF, (b) comparing CA for AF with medical therapy, (c) RCTs, (d) reporting endpoints of interest, and (e) published by English. We excluded duplicate reports, post hoc analyses, and reports without full text published. The selection process of eligible articles was performed by 2 investigators independently. Disparities between investigators were settled by consensus.
Data Extraction and Quality Assessment
The following data were extracted from each study: the year of publication, sample size, characteristics of the participants, ablation procedure, follow-up duration, outcomes measure, and endpoints of interest. The methodological quality was assessed using the Cochrane Collaboration’s tool which includes 7 domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other bias. Definitions and judgment criteria are provided in Cochrane Handbook.8 Risk of bias was evaluated for the primary endpoint. Quality assessment was also performed independently by 2 authors. All disagreements between 2 authors were settled by consensus.
Outcomes
In our study, the primary endpoint was all-cause mortality at the end of the trial. Second endpoints included hospitalization due to HF and the change in LVEF, 6MWT distance, peak VO2, Minnesota Living with Heart Failure (MLWHF) questionnaire score.
Statistical Analysis
We pooled data across trials according to the intention-to-treat principle and calculated weight mean differences with 95% CI to compare the differences for continuous variables. Risk ratios (RR) with 95% CI were used to compare differences in categorical variables. Chi-square based Q test and I2 statistic were used to test heterogeneity among studies. When PQ test (P value from the Q test) <.05 and/or I2 > 50%, significant heterogeneity was presumed. Based on the absence or presence of significant heterogeneity, a fixed-effects or random-effects model was used. We performed sensitivity analyses for each endpoint to further explore the robustness of the results.
Subgroup analyses were conducted to detect the impacts of following clinical heterogeneity on the primary outcome: (1) etiology of HF, (2) sample size, (3) left atrial diameter (LAD), (4) LVEF at baseline, (5) duration of follow-up, and (6) patient’s proportion of NYHA class III/IV. Sensitivity analyses were performed by removing each individual study.
According to Cochrane Handbook, when fewer than 10 studies are analyzed, the power of funnel plots to detect publication bias is weak and is not recommended8 so we did not perform a test for funnel plot asymmetry. The meta-analysis was conducted using Review Manager 5.4 software.
Results
Study Selection
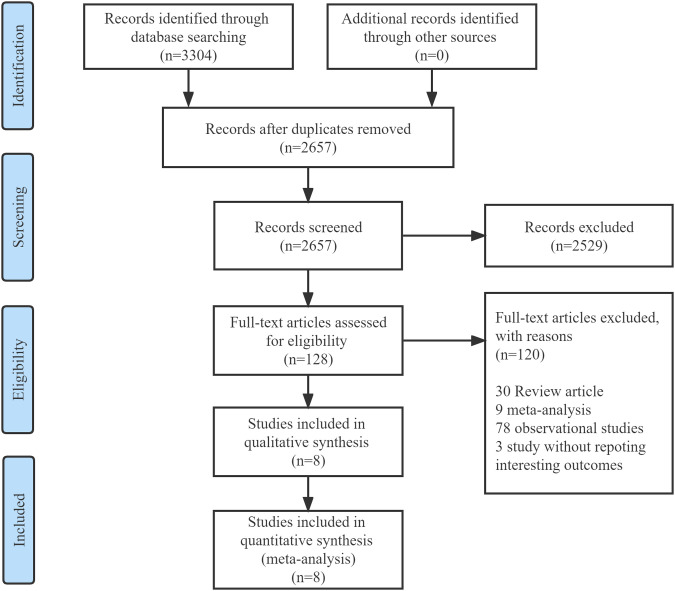
A total of 3304 references were identified through searching databases, of which 128 studies were potentially relevant and were assessed for eligibility. Finally, 8 studies were incorporated in our meta-analysis. The selection process was shown in Figure 1.
Figure 1.
Flow diagram of the selection process of eligible articles.
Characteristics of Studies and Quality Assessment
The enrolled studies were published between 2011 and 2021. A total of 1693 participants were enrolled in the present study (834 in the CA group and 859 in the medical group). The mean age of participants was about 65 years. Most patients were male. Two studies enrolled patients with paroxysmal or persistent AF.7,9 The remaining 6 studies only enrolled patients with persistent AF.6,10-14 In 4 studies, the mean duration of continuous AF was more than 1 year.10,11,13,14 The other 4 studies did not report the duration of continuous AF.6,7,9,12 In most studies, the mean LVEF at baseline was less than 40%, but 1 study enrolled a majority of patients with a mean LVEF greater than 40% at baseline.7 The methods to evaluate LVEF included transthoracic echocardiography (TTE), cardiac magnetic resonance (CMR), and radionuclide ventriculography. In total, 4 studies compared CA with rate control,10,11,13,14 1 study compared CA with amiodarone use,6 and 3 studies compared CA with medical therapy, including rate or rhythm control drugs.7,9,12 Ablation strategies varied widely between studies. All studies included pulmonary vein isolation, and most of the studies reported additional linear ablation and ablation of complex fractionated atrial electrograms. The characteristics of participants are summarized in Table 1 and the characteristics of each study are presented in Table 2. We also summarized the ablation strategies of each study in Table 3.
Table 1.
Characteristics of Patients Included in Our Study
| MacDonald et al 201113 | Jones et al 201311 | Hunter et al 201410 | Di Biase et al 20166 | Prabhu et al 201714 | Marrouche et al 20189 | Kuck et al 201912 | Packer et al 20217 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ablation | Medical | Ablation | Medical | Ablation | Medical | Ablation | Medical | Ablation | Medical | Ablation | Medical | Ablation | Medical | Ablation | Medical | ||
| Sample size | 22 | 19 | 26 | 26 | 26 | 24 | 102 | 101 | 33 | 33 | 179 | 184 | 68 | 72 | 378 | 400 | |
| Age, years | 62.3 ± 6.7 | 64.4 ± 8.3 | 64 ± 10 | 62 ± 9 | 55 ± 12 | 60 ± 10 | 62 ± 10 | 60 ± 11 | 59 ± 11 | 62 ± 9.4 | Median 64 (range, 56-71) |
Median 64 (range, 56-73.5) |
65 ± 8 | 65 ± 8 | Median 68 (range, 62-73) |
Median 67 (range, 62-73) |
|
| Male, n (%) | 17 (77) | 15 (79) | 21 (81) | 24 (92) | 25 (96) | 23 (96) | 77 (75) | 74 (73) | 31 (94) | 29 (88) | 156 (87) | 155 (84) | 60 (80) | 66 (92) | 206 (54.8) | 226 (56.5) | |
| Type of AF | Persistent | Persistent | Persistent | Persistent | Persistent | Paroxysmal (32.5%) or persistent (67.5%) | Persistent | Paroxysmal (31.6%), persistent (55.3%) or Longstanding persistent (13.1%) | |||||||||
| Mean BMI (kg/m2) | 30 ± 5.6 | 30 ± 5.7 | NA | NA | 30 ± 8 | 29 ± 4 | 30 ± 7.5 | 31 ± 4.1 | Median 29 (range, 25.9-32.2) |
Median 29.1 (range, 25.9-32.3) |
29.4 ± 5 | 28.4 ± 4.5 | Median 31 (range, 27-35) |
Median 31 (range, 27-36) |
|||
| Duration of continues AF, months | 44 ± 36.5 | 64 ± 47.6 | 23 ± 22 | 24 ± 29 | 24(17-33) | 24(12-48) | NA | 23 ± 18 | 21 ± 15 | NA | NA | NA | |||||
| NYHA class at baseline, n (%) | I | 0 | 0 | 0 | 0 | 0 | 0 | NA | 2.55 ± 0.62 | 2.45 ± 0.56 | 20 (11) | 19 (11) | 0 | 0 | 0 | 0 | |
| II | 2 (9) | 2 (11) | 14 (54) | 13 (50) | 11 (42) | 12 (50) | 101 (58) | 109 (61) | 28 (41) | 27 (38) | 277 (73.3) | 315 (78.8) | |||||
| III | 20 (91) | 17 (89) | 12 (46) | 13 (50) | 15 (58) | 12 (50) | 50 (29) | 49 (27) | 40 (59) | 45 (62) | 99 (26.2) | 85 (21.3) | |||||
| IV | 0 | 0 | 0 | 0 | 0 | 0 | 3(2) | 2(1) | 0 | 0 | 2(0.5) | 0 | |||||
| Baseline 6MWT (minutes) | 317.5 ± 125.8 | 351.8 ± 117.1 | 416 ± 78 | 411 ± 109 | NA | 348 ± 111 | 350 ± 130 | 491 ± 147 | 489 ± 132 | NA | NA | NA | |||||
| Baseline NT-pro-BNP or BNP (pg/mL) | 2550 ± 2150(NT-pro-BNP) | 1846 ± 1687(NT-pro-BNP) | 412 ± 324(BNP) | 283 ± 285(BNP) | 416 ± 78(BNP) | 411 ± 109(BNP) | NA | 266 ± 210(BNP) | 256 ± 208(BNP) | NA | NA | NA | |||||
| Baseline MLHFQ score | 55.8 ± 19.8 | 59.2 ± 22.4 | 42 ± 23 | 49 ± 21 | 40 ± 10 | 45 ± 10 | 52 ± 24 | 50 ± 27 | NA | NA | NA | NA | |||||
| Baseline peak VO2 (mL/kg/min) | NA | 16.3 ± 5.3 | 18.2 ± 4.8 | 19.2 ± 2.4 | 18.3 ± 3.2 | NA | NA | NA | NA | NA | |||||||
| LAD (mm) | NA | 50 ± 6 | 46 ± 7 | 52 ± 11 | 50 ± 10 | 47 ± 4.2 | 48 ± 4.9 | 48 ± 5.5 | 47 ± 8.2 | NA | 50 ± 6 | 51 ± 5 | NA | ||||
| LVEF (%) | 15.1 ± 6.5 | 19.6 ± 5.9 | 22 ± 8 | 25 ± 7 | 31.8 ± 7.7 | 33.7± 12.1 | 29 ± 5 | 30 ± 8 | 32 ± 9.4 | 34 ± 7.8 | 32 ± 9.4 | 34 ± 7.8 | 27.8 ± 9.5 | 24.8 ± 8.8 | Median 55 (range, 50,60) |
Median 56 (range, 50,62) |
|
| HF etiology (%) | |||||||||||||||||
| Ischemic cardiomyopathy | 49 | 33 | 26 | NA | 0 | 46 | 50 | NA | |||||||||
| past medical history | |||||||||||||||||
| Coronary disease (%) | 50 | 47 | 42 | 50 | 23 | 29 | 62 | 65 | NA | 40 | 52 | NA | 21.2 | 22.5 | |||
| Hypertension (%) | 64 | 58 | NA | 31 | 33 | 45 | 48 | 39 | 36 | 72 | 75 | 82 | 76 | 83.6 | 87.3 | ||
| Diabetes (%) | 32 | 21 | NA | NA | 22 | 24 | 12 | 15 | 28 | 37 | 35 | 31 | 25.7 | 24.5 | |||
| Medical treatment | |||||||||||||||||
| ACEI/ARB (%) | 95 | 95 | 96 | 100 | 100 | 100 | 92 | 88 | 94 | 94 | 94 | 91 | 91 | 94 | NA | ||
| Beta-blocker (%) | 82 | 95 | 92 | 92 | 100 | 100 | 76 | 80 | 88 | 85 | 93 | 95 | 91 | 93 | NA | ||
| Aldosterone antagonist (%) | 45 | 16 | 50 | 23 | NA | 45 | 50 | 33 | 48 | NA | 65 | 67 | NA | ||||
| Digitalis or digoxin (%) | 55 | 47 | 62 | 46 | NA | NA | NA | 18 | 31 | 29 | 29 | NA | |||||
| Amiodarone (%) | NA | 12 | 12 | NA | NA | NA | 31 | 26 | 25 | 34 | NA | ||||||
| Sotalol (%) | NA | NA | NA | NA | NA | 1 | 1 | NA | NA | ||||||||
| Other unspecified antiarrhythmic drugs (%) | NA | NA | NA | NA | 24 | 24 | 32 | 31 | NA | NA | |||||||
AF, atrial fibrillation; BMI, body mass index; 6MWT, 6 minute walk test; BNP, type B natriuretic peptide; NT-proBNP, N-terminal pro-B-type natriuretic peptide; MLWHF, Minnesota Living with Heart Failure Questionnaire; peak VO2, peak oxygen consumption; LAD, left atrial diameter; LVEF, left ventricular ejection fraction; HF, heart failure; ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; NA, no available.
Table 2.
Methodological Characteristics of Included Studies
| MacDonald et al 201113 | Jones et al 201311 | Hunter et al 201410 | Di Biase et al 20166 | Prabhu et al 201714 | Marrouche et al 20189 | Kuck et al 201912 | Packer et al 20217 | |
|---|---|---|---|---|---|---|---|---|
| Comparative groups | CA vs. rate control | CA vs. rate control | CA vs. rate control | CA vs. amiodarone | CA vs. rate control | CA vs. rate or rhythm control | CA vs. rate or rhythm control | CA vs. rate or rhythm control |
| Primary endpoint | Change in LVEF measured using CMR from randomization to the last study visit | Change in peak VO2 at 12 months | LVEF between the 2 groups at the 6-month time point on an intention to treat basis | Freedom from AF, atrial flutter or atrial tachycardia of >30 seconds’ duration off AAD at follow-up | Change in LVEF from baseline at 6 months on CMR | Death from any cause or worsening of heart failure that led to an unplanned overnight hospitalization | The absolute increase in LVEF from baseline at 1 year | A composite of death, disabling stroke, serious bleeding, or cardiac arrest |
| Secondary endpoints | Change in LVEF, RVEF, LVESV, LVEDV, LA diameter, BNP, 6-minute walk and quality of life (KCCQ, MLHFQ and SF-36) | Change in MLHFQ score, BNP, and 6-minute walk distance | Percentage reduction in LVESV, change in VO2 max, BNP, NYHA class, MLHFQ and SF-36 scores | Complications, all-cause mortality, AF and HF related unplanned hospitalizations during the post-ablation follow up, change in LVEF, 6-minute walk distance, and MLHFQ score | Improvement in LVEF; change in CMR chamber dimensions, NYHA class, BNP level, 6MWT distance, physical and mental SF-36 scores; AF recurrence; AF burden; procedural complications. | Death from any cause, unplanned hospitalization related to heart failure, death from cardiovascular disease, cerebrovascular accident, unplanned hospitalization for cardiovascular disease, and any hospitalization. In the ablation group, procedure related adverse events and AF-free intervals | 6-minute walk test, quality-of-life, and NT-proBNP | All-cause mortality alone, as well as the composite of all-cause mortality or cardiovascular hospitalization. |
| Method of measuring LVEF | CMR and RNVG | RNVG | TTE | TTE | CMR | TTE | TTE | NA |
| Patients undergoing repeat procedures (%) | 30 | 20 | 65.4 | NA | NA | 24.5 | 15 | NA |
| AF free survival post ablation (%) | 50 | 88 | 73 | 70 | 100 | 63 | 73.5 | 44 |
| Total follow-up time (months) |
6 | 12 | 12 | 24 | 6 | 60 | 12 | 60 |
CA, catheter ablation; LVEF, left ventricular ejection fraction; CMR, cardiac magnetic resonance; peak VO2, peak oxygen consumption; AF, Atrial fibrillation; AAD, antiarrhythmic drugs; RVEF, right ventricular function; LVESV, left ventricular end systolic volume; LVEDV, Left ventricular end diastolic volume; LA, left atrial; BNP, type B natriuretic peptide; KCCQ, The Kansas City Cardiomyopathy Questionnaire; MLHFQ, Minnesota Living with Heart Failure Questionnaire; SF-36, the MOS item short from health survey; 6MWT, 6-minute walk test; NT-proBNP, N-terminal pro-brain natriuretic peptide; RNVG, radionuclide ventriculography; TTE, Transthoracic echocardiography; NA, no available.
Table 3.
Summary of Ablation Strategies for Individual Studies
| Study | PVI | Additional Linear Ablation | Posterior Wall Isolation | SVC Isolation | CFAE Ablation |
|---|---|---|---|---|---|
| MacDonald et al 201113 | √ | √ | √ | ||
| Jones et al 201311 | √ | √ | √ | ||
| Hunter et al 201410 | √ | √ | √ | ||
| Di Biase et al 20166 | √ | √ | √ | √ | √ |
| Prabhu et al 201714 | √ | √ | |||
| Marrouche et al 20189 | √ | √ | √ | √ | √ |
| Kuck et al 201912 | √ | √ | √ | ||
| Packer et al 20217 | √ | √ |
PVI, pulmonary vein isolation; SVC, superior vena cava; CFAE, complex fractionated atrial electrogram.
A risk bias assessment was performed for the primary outcome in each study. All studies reported detailed and reasonable methods to generate the random sequence. Allocation concealment was reported in 4 studies, except 4 open-labeled studies.6,7,11,12 As CA is an invasive procedure, blinding of patients and investigators performing ablation was not permitted. The main endpoint of our study was all-cause mortality. The patient's life or death was absolute so there was no detection bias. One study lost significantly more follow-up in the CA group than the medical group, leading to a high risk of attrition bias.9 No other incomplete outcome data and selective reporting were observed in the remaining studies. Selective reporting was not observed in all studies. In 2 studies, patients in 2 groups were restructured,9,14 9% of patients were moved from the medical group to the CA group in the study by Prabhu et al14 2017. Besides, 10% of patients were moved from the medical group to the CA group, and 16% of patients were transferred from the CA group to the medical group in the study by Marrouche et al9 2018. So both studies were judged as having a high risk of other bias. This condition was not found in the remaining studies, and the remaining studies were judged as having no risk of other bias. The results of the quality assessment are summarized in Figures 2 and 3.
Figure 2.

Risk of bias summary: review authors’ judgments about each risk of bias item for each included study.
Figure 3.
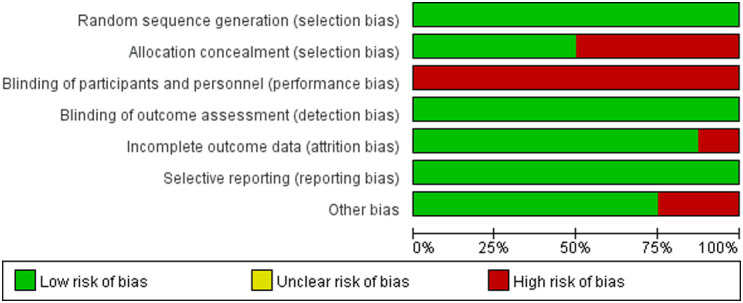
Risk of bias graph: review authors' judgments about each risk of bias item presented as percentages across all included studies.
Outcomes
Primary Outcome
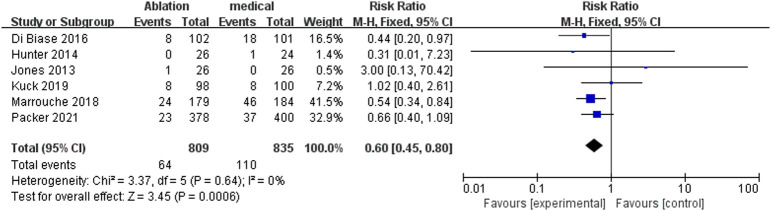
Data for all-cause mortality from baseline were obtained from 6 studies.6,7,9-12 Due to the homogeneity of the included studies, we used a fixed-effects model for data analysis of all-cause mortality (P = .64, I2 = 0%). The pooled results showed that compared with medical therapy, CA significantly reduced all-cause mortality (RR = 0.60, 95% Cl: 0.45-0.80, P < .001) (Figure 4), and sensitivity analysis suggests that the results are consistent.
Figure 4.
Comparison of the all-cause mortality between catheter ablation group and medical therapy group.
In subgroup analyses, when enrolled studies were stratified according to the etiology of HF, sample size, LAD, LVEF at baseline, duration of follow-up, and the patient’s proportion of NYHA class III/IV, we found no differences across subgroups. The results of subgroup analyses are shown in Table 4.
Table 4.
Subgroup Analyses Comparing Difference of All-Cause Mortality Between Catheter Ablation and Medical Therapy
| Subgroup | Risk Ratios in the All-Cause Mortality | P Value for Subgroup Difference | |
|---|---|---|---|
| Proportion of ischemic cardiomyopathy (%)a | <40 | 0.96, 95% CI: 0.14-6.52, I2 = 0% | .452 |
| ≥40 | 0.61, 95% CI: 0.41-0.91, I2 = 31.8% | ||
| Sample size | <100 | 1.01, 95% CI: 0.43-2.34, I2 = 0% | .644 |
| ≥100 | 0.56, 95% CI: 0.41-0.77, I2 = 0% | ||
| Proportion of left atrium diameter (mm) | <50 | 0.56, 95% CI: 0.36-0.87, I2 = 11.1% | .452 |
| ≥50 | 0.90, 95% CI: 0.37-2.19, I2 = 0% | ||
| Proportion of NYHA class III/IV (%) | <40 | 0.96, 95% CI: 0.14-6.52, I2 = 0% | .452 |
| ≥40 | 0.61, 95% CI: 0.41-0.91, I2 = 31.8% | ||
| Follow-up (month) | ≤24 | 0.64, 95% CI: 0.37-1.13, I2 = 0% | .644 |
| >24 | 0.59, 95% CI: 0.42-0.82, I2 = 0% | ||
aWe stratified the etiology of HF into ischemic and non-ischemic cardiomyopathy.
Secondary Outcomes
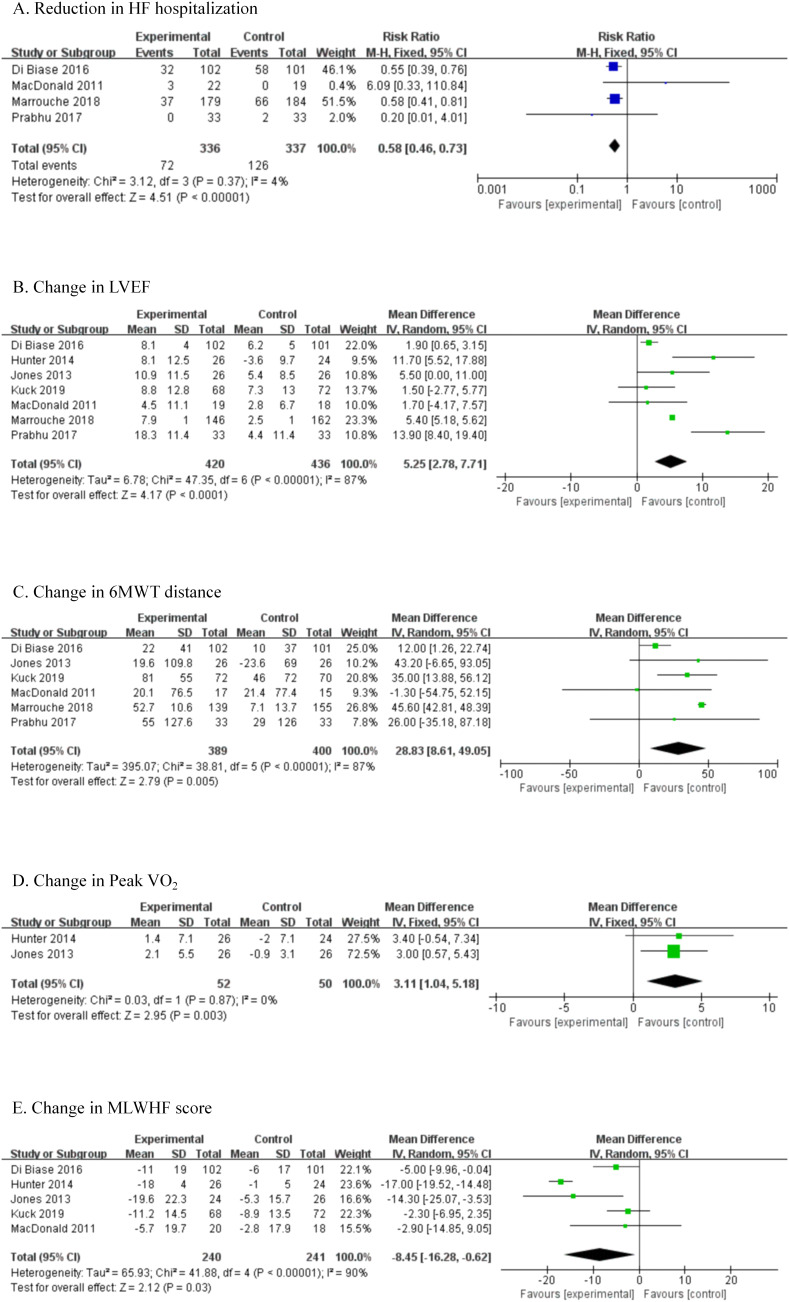
We can extract data for HF hospitalization in 4 studies. We found low heterogeneity among included studies (P = .37, I2=4%). The pooled result from the fixed-effects model demonstrated that compared with medical therapy, CA significantly reduced hospitalization for HF (RR = 0.58, 95% Cl: 0.46-0.73, P < .001).
In terms of LVEF, 7 studies were provided for analysis. There was high heterogeneity among the included studies (P < .001, I2 = 87%). The pooled analysis from the random-effects model demonstrated that the CA had a better improvement than the medical therapy (mean difference [MD] = 5.25%, 95% CI: 2.78%, 7.71%, P < .001).
With regard to the distance of 6MWT, 6 studies provided data on the change in the performance of 6MWT. There was high heterogeneity among the included studies (P < .001, I2 = 87%). The pooled result from the random-effects model indicated that compared with medical therapy, CA significantly improved the performance of 6MWT (MD = 28.83 m, 95% CI: 8.61-49.05 m, P = .005).
Regarding peak VO2, only 2 studies reported the alterations relative to baseline. A fixed-effects model was used to evaluate the difference in peak VO2 due to no heterogeneity (P = .87, I2 = 0%). The pooled result suggested that compared with medical therapy, CA significantly increased peak VO2 (MD = 3.11 mL/kg/min, 5% CI: 1.04-5.18 mL/kg/min, P = .003).
Five studies provided data on MLWHF and contributed to the analysis. Except for the study by MacDonald et al13 and Kuck et al12, all these studies consistently proven the superiority of CA versus medical therapy in improving the quality of life. Due to the high heterogeneity (P < .001, I2 = 90%), we used a random-effects model. The pooled result demonstrated that compared with medical therapy, CA reduced MLWHF score (MD = −8.45, 95% CI: −16.28 to −0.62, P = .03).
The results regarding secondary outcomes are shown in Figure 5.
Figure 5.
Comparison of secondary outcomes between catheter ablation group and medical therapy group. (A). Reduction in HF hospitalization; (B). Change in LVEF; (C). Change in 6MWT distance; (D). Change in peak VO2; (E). Change in MLWHF score. 6MWT, 6-minute walk test; BNP, brain natriuretic peptide; peak VO2, peak oxygen consumption; MLWHF, Minnesota Living with Heart Failure questionnaire.
Sensitive analyses demonstrated similar results after removing each individual study, except for the results of LVEF.
Discussion
The main findings of this meta-analysis are as follows: (1) compared with medical therapy, CA significantly improved all-cause mortality in HF patients with AF and (2) CA provided additional benefits than medical therapy in improving HF symptoms, exercise capacity, and quality of life.
Comorbidity of AF and HF is a common clinical scenario. Atrial fibrillation and HF collaborate to aggravate each other. On the one hand, optimal HF treatment is necessary. On the other hand, intervention in AF is also a vital approach to break the vicious cycle. According to current guidelines, both rate and rhythm control are recommended for the treatment of AF.3 Several large randomized trials demonstrated that rate and rhythm control had comparable efficacy in all-cause death, stroke, and cardiac mortality in AF patients with or without HF.15 However, in these studies, in the rhythm-control group, the rate of maintaining SR was low and many patients crossed over to the other treatment strategy. Most investigators still believe that AF patients could benefit from SR, especially patients with HF. Some post hoc analyses demonstrated that conversion from AF to SR provided an additional benefit over rate control in both patients with and without HF.16,17 A subgroup analysis of AATAC also demonstrated the importance of SR in patients with reduced ejection fraction.6 When using drugs for heart rhythm control, patients need to take long-term medication, which may cause many side effects, resulting in low medical compliance. Compared with medical therapy, CA is a recognized treatment method for preventing AF. Guidelines recommend CA as a first-line treatment for AF prior to the use of antiarrhythmic drugs, either for paroxysmal AF or persistent AF.3 Therefore, in theory, rhythm control by CA may be reasonable for patients with AF and HF.
Heart failure with preserved ejection fraction (HFpEF) accounts for half of the patient population diagnosed with HF.18 We found that HFpEF may be even more closely related to AF than HF with reduced ejection fraction (HFrEF). The prevalence of AF in HFpEF is approximately 20%-40%, and more than 60% of patients may develop arrhythmia during the development of the disease.19-21 What’s more, AF appears to indicate a worse prognosis in patients with HFpEF than with HFrEF.19 Catheter ablation has turned out to be a safe procedure in patients with HFpEF, with favorable overall outcomes and low complication rates, and the vast majority of patients maintain long-term sinus rhythm.22
In patients with AF and HF, due to the existence of AF, it is not only a high heart rate but also asynchronous myocardial contraction that can cause or aggravate left ventricular dysfunction.23 Therefore, in addition to ischemic cardiomyopathy, tachycardia is one of the causes of HF in such patients. Among the studies we included, 1 study completely excluded patients with ischemic cardiomyopathy and used CMR to observe whether the patients had ventricular fibrosis. In patients with non-ischemic cardiomyopathy and without ventricular fibrosis, the etiology of heart failure is likely to be tachycardiomyopathy (TCMP), and the study noted that restoration of sinus rhythm with CA significantly improved ventricular function, especially in the absence of ventricular fibrosis on CMR.14 No information about TCMP was mentioned in several other included studies.
Evidence suggested that elevated LA diameter may be associated with the recurrence of AF after CA.24 In our study, the average LAD of the patients was about 50 mm, and the sinus rhythm maintenance rate at the end of the study was 44%-100%, which indicates that although the left atrial diameter of some patients is too large, the sinus rhythm maintenance rate after CA is not low. And the patient's endpoints were significantly improved. We speculate that the source of the benefit of CA for patients with AF and HF is not only the restoration of sinus rhythm but also the CA itself. The clinical trial has shown that CA in AF patients can reverse left ventricular structural remodeling and improve left ventricular systolic function, especially in patients with low LVEF and chronic AF.25 We speculate that it may be related to the reduction in the size of the heart chamber by CA.
Efficacy of Catheter Ablation in All-Cause Mortality
Atrial fibrillation and HF are associated with mortality, and there have been 2 previous RCTs with all-cause mortality as the primary endpoint, both of them showed a significant reduction in all-cause mortality with CA compared with medical therapy. There are several RCTs that also report deaths at the end of the trial; however, the sample sizes of these studies were small. A subgroup analysis of a large RCT reported that CA contributed to a lower mortality.7 We pooled it with previous studies and came to similar conclusions.
We have not found any source of heterogeneity in the subgroup analysis. The patients included in the previous study were basically all patients with HFrEF, while the majority of patients included in the new study were with HFpEF.7 This suggested that CA is equally effective in patients with HFpEF and AF.
Our results are important in clinical practice. The coexistence of AF and HF is a challenge for physicians and lacks effective treatments. Our results suggest that CA can reduce all-cause mortality in these patients and reduce the extent to which the disease is detrimental to society. Whether it is HFpEF or HFrEF, CA can significantly reduce all-cause mortality in these patients. Of note, among enrolled trials, drug-refractory symptomatic AF was not in inclusion criteria, which is the main indication for CA according to current guidelines.3 Therefore, our study supports the application of CA in HF patients with AF no matter whether drug-refractory symptomatic is AF or not.
Efficacy of Catheter Ablation in Second Outcomes
Our study indicated that compared with medical therapy, CA significantly reduced the hospitalization for HF and improved the performance of 6MWT, peak VO2 , and quality of life (reduction of MLWHF score). These clinical parameters reflect subjective HF symptoms and objective exercise capacity and are strongly related to prognosis in HF patients.26-28 It is worth noting that for LVEF, we included data from 7 studies for analysis and 3 of them showed no statistically significant improvement in LVEF from baseline in the CA group compared with the medical group at the end of follow-up,11-13 and the results were highly heterogeneous. Therefore, the interpretation of the difference in LVEF between the 2 groups should be cautious, and the effect of ablation on LVEF is controversial. For 6MWT, we found that the study by Di Biase et al6 was the source of heterogeneity, which may be because more patients with a history of coronary heart disease were included in this study. For MLWHF, we found the study by Hunter et al10 to be a source of heterogeneity, perhaps due to the younger age of the patients included in the study. Therefore, we found that age and past history of coronary heart disease may be factors that can affect patient outcomes. In addition, the study by Prabhu et al14 included patients with suspected TCMP, and the 3-second outcomes including HF hospitalization rate, 6MWT, and LVEF were included in this study. We performed a sensitivity analysis by removing this study, and the result did not change.
Complications
Major complications of CA are rare, mainly including stroke, cardiac tamponade, pulmonary vein stenosis, phrenic nerve palsy, oesophageal injury, and vascular complications. A meta-analysis reported that the major complication rate was 2.9% in the general population.29 In population with HF, complication rate was reported as about 4.3%.30 Among included studies, complication rates ranged from 2.5% to 6.9% after multiple procedures, and most complications could be treated conservatively. In a large clinical study, CA strategies did not increase the incidence of adverse events such as death, disabling stroke, major bleeding, or cardiac arrest in patients with AF compared with medical therapy.31 Overall, CA is a safe method to restore and maintain SR in patients with or without HF.
Limitation
The current metaanalysis presents several limitations: (1) half of the included studies had a follow-up time of less than 24 months, so many studies reported a few deaths, which may have had some impact on the results. (2) There was no standard ablation strategy in the included studies. Different ablation strategies may lead to different outcomes, and today, the optimal ablation strategy for patients with persistent AF remains unclear. (3) In the present study, subgroup analyses were performed based on study-level data. So the results of subgroup analyses should be explained with caution at the individual level. Moreover, we only stratified the etiology of HF into ischemic and non-ischemic cardiomyopathy. Actually, non-ischemic cardiomyopathy is a heterogeneous group of diseases which could be further stratified.
Conclusion
In HF patients with AF, compared with medical therapy, CA reduces all-cause mortality and HF hospitalization and improves HF symptoms, exercise capacity, and quality of life. Therefore, CA should be considered when we choose treatment options for patients with AF and HF, even prior to the conventional rate control. Further larger RCTs are expected to support this treatment strategy.
Footnotes
Peer-review: Externally peer-reviewed.
Author Contributions: Concept – Z.Y., B.X., F.Q.; Design – Z.Y., Z.Z., Y.Qiu; Supervision – Z.Y., Y.X., B.X., F.P.; Funding – Y.X., J.P.; Materials – Z.Y., B.X., Z.Z., F.Q.; Data collection and/or processing – Z.Y., Y.Qi, Z.Z., Y.Qiu; Analysis and/or interpretation – Z.Y., Y.Qi, Y.Qiu, F.P., J.P.; Literature review – Z.Y., Y.Qi, F.P.; Writer – Z.Y., F.Q.; Critical review – Y.X., F.P., J.P.
Acknowledgments: None.
Declaration of Interests: The authors declare that they have no competing interest.
Funding: This work was funded by Science and Technology Department of Zhejiang Province, China, under Grant Agreement No. LGF20H020003.
References
- 1. Batul SA, Gopinathannair R. Atrial fibrillation in heart failure: a therapeutic challenge of our times. Korean Circ J. 2017;47(5):644 662. 10.4070/kcj.2017.0040) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Waldmann V, Jouven X, Narayanan K.et al. Association between atrial fibrillation and sudden cardiac death: pathophysiological and epidemiological insights. Circ Res. 2020;127(2):301 309. 10.1161/CIRCRESAHA.120.316756) [DOI] [PubMed] [Google Scholar]
- 3. Hindricks G, Potpara T, Dagres N.et al. ESC Guidelines for the diagnosis and management of atrial fibrillation developed in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS): the Task Force for the diagnosis and management of atrial fibrillation of the European Society of Cardiology (ESC) Developed with the special contribution of the European Heart Rhythm Association (EHRA) of the ESC. Eur Heart J. 2020;425:373 498. [DOI] [PubMed] [Google Scholar]
- 4. Verma A, Kalman JM, Callans DJ. Treatment of patients with atrial fibrillation and heart failure with reduced ejection fraction. Circulation. 2017;135(16):1547 1563. 10.1161/CIRCULATIONAHA.116.026054) [DOI] [PubMed] [Google Scholar]
- 5. Purmah Y, Proietti M, Laroche C.et al. Rate vs. rhythm control and adverse outcomes among European patients with atrial fibrillation. Europace. 2018;20(2):243 252. 10.1093/europace/euw421) [DOI] [PubMed] [Google Scholar]
- 6. Di Biase L, Mohanty P, Mohanty S.et al. Ablation Versus amiodarone for treatment of persistent atrial fibrillation in patients with congestive heart failure and an implanted device: results from the AATAC multicenter randomized trial. Circulation. 2016;133(17):1637 1644. 10.1161/CIRCULATIONAHA.115.019406) [DOI] [PubMed] [Google Scholar]
- 7. Packer DL, Piccini JP, Monahan KH.et al. Ablation Versus drug therapy for atrial fibrillation in heart failure: results from the CABANA trial. Circulation. 2021;143(14):1377 1390. 10.1161/CIRCULATIONAHA.120.050991) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Cumpston M, Li T, Page MJ.et al. Updated guidance for trusted systematic reviews: a new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst Rev. 2019;10:ED000142. 10.1002/14651858.ED000142) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Marrouche NF, Brachmann J, Andresen D.et al. Catheter ablation for atrial fibrillation with heart failure. N Engl J Med. 2018;378(5):417 427. 10.1056/NEJMoa1707855) [DOI] [PubMed] [Google Scholar]
- 10. Hunter RJ, Berriman TJ, Diab I.et al. A randomized controlled trial of catheter ablation versus medical treatment of atrial fibrillation in heart failure (the CAMTAF trial). Circ Arrhythm Electrophysiol. 2014;7(1):31 38. 10.1161/CIRCEP.113.000806) [DOI] [PubMed] [Google Scholar]
- 11. Jones DG, Haldar SK, Hussain W.et al. A randomized trial to assess catheter ablation versus rate control in the management of persistent atrial fibrillation in heart failure. J Am Coll Cardiol. 2013;61(18):1894 1903. 10.1016/j.jacc.2013.01.069) [DOI] [PubMed] [Google Scholar]
- 12. Kuck KH, Merkely B, Zahn R.et al. Catheter ablation versus best medical therapy in patients with persistent atrial fibrillation and congestive heart failure: the randomized AMICA trial. Circ Arrhythm Electrophysiol. 2019;12(12):e007731. 10.1161/CIRCEP.119.007731) [DOI] [PubMed] [Google Scholar]
- 13. MacDonald MR, Connelly DT, Hawkins NM.et al. Radiofrequency ablation for persistent atrial fibrillation in patients with advanced heart failure and severe left ventricular systolic dysfunction: a randomised controlled trial. Heart. 2011;97(9):740 747. 10.1136/hrt.2010.207340) [DOI] [PubMed] [Google Scholar]
- 14. Prabhu S, Taylor AJ, Costello BT.et al. Catheter ablation Versus medical rate control in atrial fibrillation and systolic dysfunction: the CAMERA-MRI study. J Am Coll Cardiol. 2017;70(16):1949 1961. 10.1016/j.jacc.2017.08.041) [DOI] [PubMed] [Google Scholar]
- 15. Al-Khatib SM, Allen LaPointe NMA, Chatterjee R.et al. Rate- and rhythm-control therapies in patients with atrial fibrillation: a systematic review. Ann Intern Med. 2014;160(11):760 773. 10.7326/M13-1467) [DOI] [PubMed] [Google Scholar]
- 16. Corley SD, Epstein AE, DiMarco JP.et al. Relationships between sinus rhythm, treatment, and survival in the Atrial Fibrillation Follow-Up Investigation of Rhythm Management (AFFIRM) Study. Circulation. 2004;109(12):1509 1513. 10.1161/01.CIR.0000121736.16643.11) [DOI] [PubMed] [Google Scholar]
- 17. Suman-Horduna I, Roy D, Frasure-Smith N.et al. Quality of life and functional capacity in patients with atrial fibrillation and congestive heart failure. J Am Coll Cardiol. 2013;61(4):455 460. 10.1016/j.jacc.2012.10.031) [DOI] [PubMed] [Google Scholar]
- 18. Dunlay SM, Roger VL, Redfield MM. Epidemiology of heart failure with preserved ejection fraction. Nat Rev Cardiol. 2017;14(10):591 602. 10.1038/nrcardio.2017.65) [DOI] [PubMed] [Google Scholar]
- 19. Lam CS, Rienstra M, Tay WT.et al. Atrial fibrillation in heart failure with preserved ejection fraction: association with exercise capacity, left ventricular filling pressures, natriuretic peptides, and left atrial volume. JACC Heart Fail. 2017;5(2):92 98. 10.1016/j.jchf.2016.10.005) [DOI] [PubMed] [Google Scholar]
- 20. Patel RB, Vaduganathan M, Shah SJ, Butler J. Atrial fibrillation in heart failure with preserved ejection fraction: insights into mechanisms and therapeutics. Pharmacol Ther. 2017;176:32 39. 10.1016/j.pharmthera.2016.10.019) [DOI] [PubMed] [Google Scholar]
- 21. Santhanakrishnan R, Wang N, Larson MG.et al. Atrial fibrillation begets heart failure and vice versa: temporal associations and differences in preserved versus reduced ejection fraction. Circulation. 2016;133(5):484 492. 10.1161/CIRCULATIONAHA.115.018614) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Androulakis E, Sohrabi C, Briasoulis A.et al. Catheter ablation for atrial fibrillation in patients with heart failure with preserved ejection fraction: a systematic review and meta-analysis. J Clin Med. 2022;11(2);112. 10.3390/jcm11020288) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Martin CA, Lambiase PD. Pathophysiology, diagnosis and treatment of tachycardiomyopathy. Heart. 2017;103(19):1543 1552. 10.1136/heartjnl-2016-310391) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Jin X, Pan J, Wu H, Xu D. Are left ventricular ejection fraction and left atrial diameter related to atrial fibrillation recurrence after catheter ablation? A meta-analysis. Med (Baltim). 2018;97(20):e10822. 10.1097/MD.0000000000010822) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Zhu P, Zhang Y, Jiang P.et al. Effects of radiofrequency catheter ablation on left ventricular structure and function in patients with atrial fibrillation: a meta-analysis. J Interv Card Electrophysiol. 2014;40(2):137 145. 10.1007/s10840-014-9903-1) [DOI] [PubMed] [Google Scholar]
- 26. Passantino A, Lagioia R, Mastropasqua F, Scrutinio D. Short-term change in distance walked in 6 min is an indicator of outcome in patients with chronic heart failure in clinical practice. J Am Coll Cardiol. 2006;48(1):99 105. 10.1016/j.jacc.2006.02.061) [DOI] [PubMed] [Google Scholar]
- 27. Swank AM, Horton J, Fleg JL.et al. Modest increase in peak VO2 is related to better clinical outcomes in chronic heart failure patients: results from heart failure and a controlled trial to investigate outcomes of exercise training. Circ Heart Fail. 2012;5(5):579 585. 10.1161/CIRCHEARTFAILURE.111.965186) [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Huang TY, Moser DK, Hwang SL. Identification, associated factors, and prognosis of symptom clusters in Taiwanese patients with heart failure. J Nurs Res. 2018;26(1):60 67. 10.1097/JNR.0000000000000199) [DOI] [PubMed] [Google Scholar]
- 29. Gupta A, Perera T, Ganesan A.et al. Complications of catheter ablation of atrial fibrillation: a systematic review. Circ Arrhythm Electrophysiol. 2013;6(6):1082 1088. 10.1161/CIRCEP.113.000768) [DOI] [PubMed] [Google Scholar]
- 30. Anselmino M, Matta M, Castagno D, Giustetto C, Gaita F. Catheter ablation of atrial fibrillation in chronic heart failure: state-of-the-art and future perspectives. Europace. 2016;18(5):638 647. 10.1093/europace/euv368) [DOI] [PubMed] [Google Scholar]
- 31. Packer DL, Mark DB, Robb RA.et al. Effect of catheter ablation vs antiarrhythmic drug therapy on mortality, stroke, bleeding, and cardiac arrest Among patients with atrial fibrillation: the CABANA randomized clinical trial. JAMA. 2019;321(13):1261 1274. 10.1001/jama.2019.0693) [DOI] [PMC free article] [PubMed] [Google Scholar]



 Content of this journal is licensed under a
Content of this journal is licensed under a