Abstract

Novel derivatives of benzothiazole-2-thiophene S-glycoside were synthesized and tested for their antiviral and anticancer potency and NS3/4A and USP7 enzyme inhibitions. The ring system was formed by first synthesizing new derivatives of 5-mercaptothiophene substituted with the benzothiazole moiety, followed by coupling with various halo sugar derivatives. New compounds were tested in vitro for the cytotoxic effect on five types of normal cell lines and for antiviral activity using a plaque reduction assay against CBV4, HSV-1, HCVcc genotype 4 viruses, HAV HM 175, and HAdV7. Notably, three compounds demonstrated substantial IC50, CC50, and SI values against HSV-1 with a viral reduction of 80% or more. Two substances have demonstrated a reduction of more than 50% in CBV4 and HCVcc viruses. The effectiveness of the compounds against HSV-1 and HCVcc was tested for their capability to inhibit NS3/4A protease and USP7 enzyme. Additionally, a panel of 60 human cancer cells was used to investigate the ability of the newly synthesized compounds to inhibit the in vitro tumor growth. The results revealed that two compounds, 6a and 6c, have an inhibitory effect on most cancer types, whereas 6d and 6f inhibited only three and two cell lines, respectively.
1. Introduction
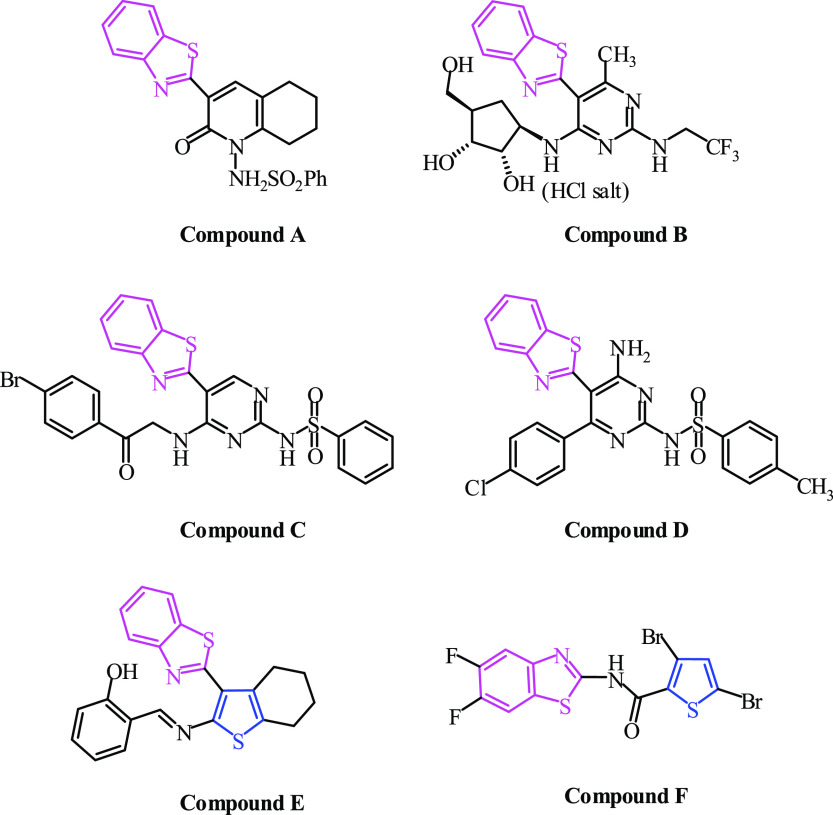
Compounds containing benzothiazole have emerged as a promising pharmacophore with a wide range of biological properties in recent decades including antiinflammatory,1,2 analgesic,3 fungicidal,4 antibactrial,5−7 antidiabetic,8 antitumor,9,10 antileishmanial,11 and anticonvulsant.12 Some drugs such as Riluzole, neuroprotective injury and anticonvulsant drug for the spinal cord, Ethoxzolamine, an antibacterial and diuretic Helicobacter pylori, and Dimazole, antifungal, are containing a benzothiazole ring in their structure. Furthermore, Pittsburgh compound B (PiB) and Flutemetamol are employed for positron emission tomography scans to visualize beta-amyloid plaques in the patient’s brain, which is used as a diagnostic tool for Alzheimer’s disease, as shown in Figure 1.
Figure 1.
Benzothiazole ring as an important moiety as drugs.
Benzothiazole derivatives have remarkable and widespread biological and pharmacological activities against various tumors and cancer cell lines, for example, HepG2 (human liver carcinoma cells), HeLa (human cervical cancer cell line), mammary and ovarian tumor cell lines, colon, SW480 (human colon adenocarcinoma cell line), nonsmall-cell lung, breast subpanels cell lines, and HCC (hepatocellular carcinoma).13 Benzothiazole is also an important component in the creation and producing of antiviral drugs.14 This is clear from the fact that this useful ring presents in a large number of clinically useful agents.15 As an example, 2-pyridone substituted with the benzothiazole moiety, compound A, showed a broad spectrum as an antiviral agent,16Figure 2. Furthermore, pyrimidine substituted with benzothiazole inhibited the growth of various viral cells and enzymes, Figure 2. Compound B showed optimal inhibition against HCV with a low EC50 value and selectivity index greater than 550.17 Additionally, with low IC50 and high CC50 values, compounds C and D demonstrated remarkable viral reduction in the range of 70 to 90% and inhibitory activity against the Hsp90α protein with IC50 comparable to acyclovir, as a standard drug,18Figure 2. Moreover, the thiophene ring and its derivatives are considered to be one of the most significant types of heterocyclic molecules with remarkable applications in the field of medicinal chemistry.19 For example, compounds that contain both benzothiazole and thiophene rings, such as compound E, were found to have highly potent antiviral activities against ADV7 with an EC50 value of 10.8 μg/mL when compared to ribavirin, EC50 value of 27.8 μg/mL,20Figure 2. Furthermore, compound F, 3,5-di-bromothiophene-4,6-difluorobenzothiazole, demonstrated excellent antinorovirus properties with an EC50 value of 0.53 μM.21
Figure 2.
Benzothiazole compounds and hybrid benzothiazole with a thiophene ring as antiviral agents.
On the other hand, S-glycoside derivatives have drawn significant attention in recent years.22−26 Thioethers were discovered to enhance biological activities such as antimicrobial and antitumor activity.27−29 As a result, thioglycosides have a high potential as therapeutics and are becoming more popular as pharmaceutical targets.30 In light of the previous information and our ongoing research into developing new compounds having a thiophene and/or benzothiazole ring as potent drugs, a series of novel benzothiazol-2-yl-5-mercaptothiophene molecules bearing sugar moieties have been synthesized and examined for their cytotoxicity against five normal cells and their reduction percent by a plague reduction assay against five viruses. Additionally, the most potent synthesized compounds were tested for inhibition of both the NS3/4A protease and USP7 enzyme. The produced compounds were also evaluated for their anticancer activities against 60 different cell lines.
2. Results and Discussion
2.1. Chemistry
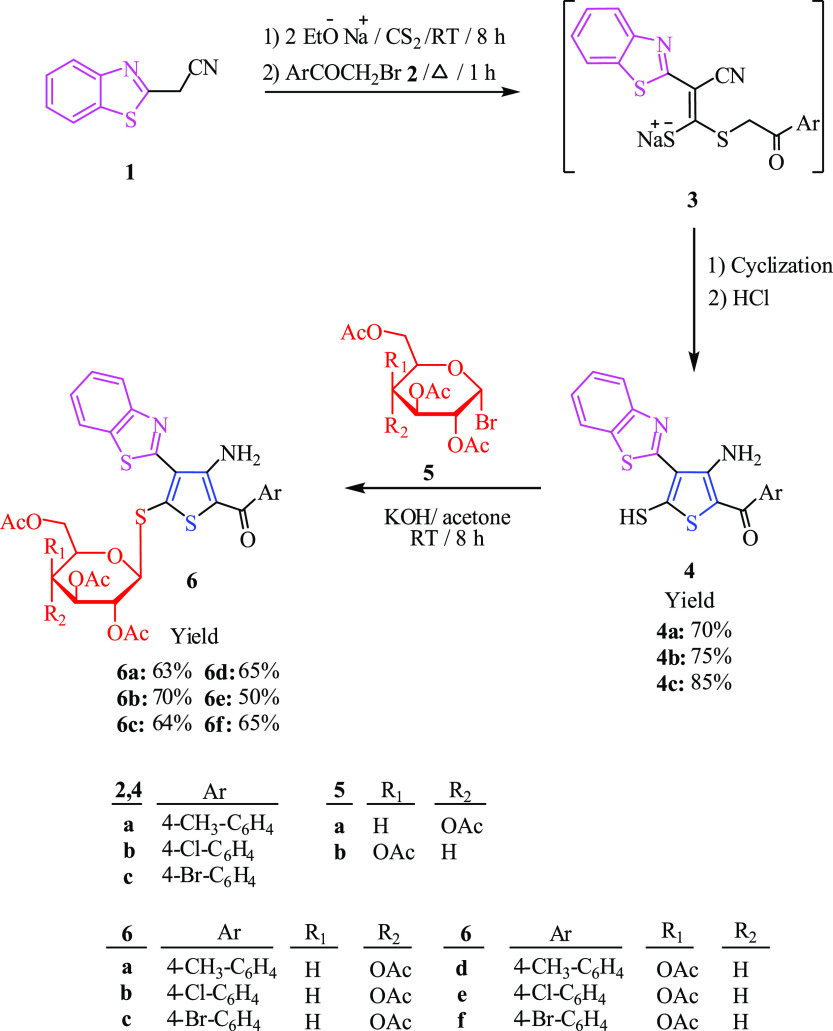
To be able to synthesize our target thiophene derivatives having sugar moieties, the sodium salt of ketene dithioacetal of 2-benzothiazole methyl nitrile was prepared first by the reaction of benzothiazol-2-yl-acetonitrile 1 with carbon disulfide and two equivalents of sodium ethoxide. The obtained salt was then reacted with phenacyl bromide derivatives 2a–2c via nucleophilic substitution reaction forming the intermediate 3a–3c. After cyclization and neutralization of the resulted cyclic compounds with HCl, thiophene derivatives containing benzothiazole ring 4a–4c were isolated in good yield, Scheme 1. Spectroscopic measurement techniques such as 1H NMR, IR, and basic analysis were employed to verify the structure of compounds 4a–4c. The presence of aryl and benzothiazole rings was revealed by the 1H NMR spectra of 4a–4c. For example, the 1H NMR of compound 4b showed four peaks; two doublets of doublets but appear triplets, at 7.32 and 7.44 ppm and two doublets at 7.87 and 8.00 ppm corresponding to the benzothiazole ring as well as two doublet peaks at 7.52 and 7.69 ppm corresponding to the benzene ring substituted with Cl at the p-position.
Scheme 1. Synthesis of Benzothiazole-2-Thiophene S-Glycoside Derivatives 6a−6f.
After confirming the chemical structure of thiophene derivatives 4a–4c, they then reacted with both 2,3,4,6-tetra-O-acetyl-α-D-gluctopyranosyl bromide 5a and 2,3,4,6-tetra-O-acetyl-α-D-galactopyranosyl bromide 5b in the presence of potassium hydroxide to form the corresponding benzothiazole-2-thiophene S-glycoside derivatives 6a–6c and 6d–6f, respectively, in good yield, Scheme 1. The idea is that the corresponding β-products are formed when α-sugars bond through the SN2 reaction by inversion of configuration. The structure of compounds 6a–6f was confirmed based on the spectral measurement techniques such as 13C NMR, 1H NMR, and IR. The 1H NMR spectrum of compound 6f presented the anomeric proton as a doublet at δ = 5.39 ppm with coupling constant J = 8.8 Hz confirming the β-configuration. The remaining six protons of galactose resonated at δ 4.00–5.30 ppm. In order to clearly establish the structure of the new compounds, the crystal structure of 6f was determined,31Figure 3. To the best of our knowledge, this is the first X-ray structure of the new compound type benzothiazole-2-thiophene S-thioglycoside that has been reported.
Figure 3.

X-ray single-crystal structure of compound 6f.
″Reproduced with permission from the International Union of Crystallography under the open-access license.31
Furthermore, compounds 4a–4c were allowed to react with 2,3,4-tri-O-acetyl-α-d-xylosopyranosyl bromide 7a and 2,3,4-tri-O-acetyl-α-l-arabinopyranosyl bromide 7b to furnish the corresponding benzothiazole-2-thiophene S-glycoside derivatives 8a–8c and 8d, respectively, Scheme 2. The structure of products 8a–8d was confirmed through basic analysis and spectroscopic measurement techniques such as 13C NMR, 1H NMR, and IR. The 1H NMR spectrum showed the anomeric proton of 8d as a doublet at δ 5.35 ppm, and the other four protons resonated at 3.97–5.24 ppm. It also showed four acetyl groups as four singlet peaks in the range of 1.95–1.99 ppm. The coupling constant (J = 8.4 Hz) indicated that H-1′is trans-diaxial to H-2’.
Scheme 2. Synthesis of Benzothiazole-2-Thiophene S-Glycoside Derivatives 8a–8d.
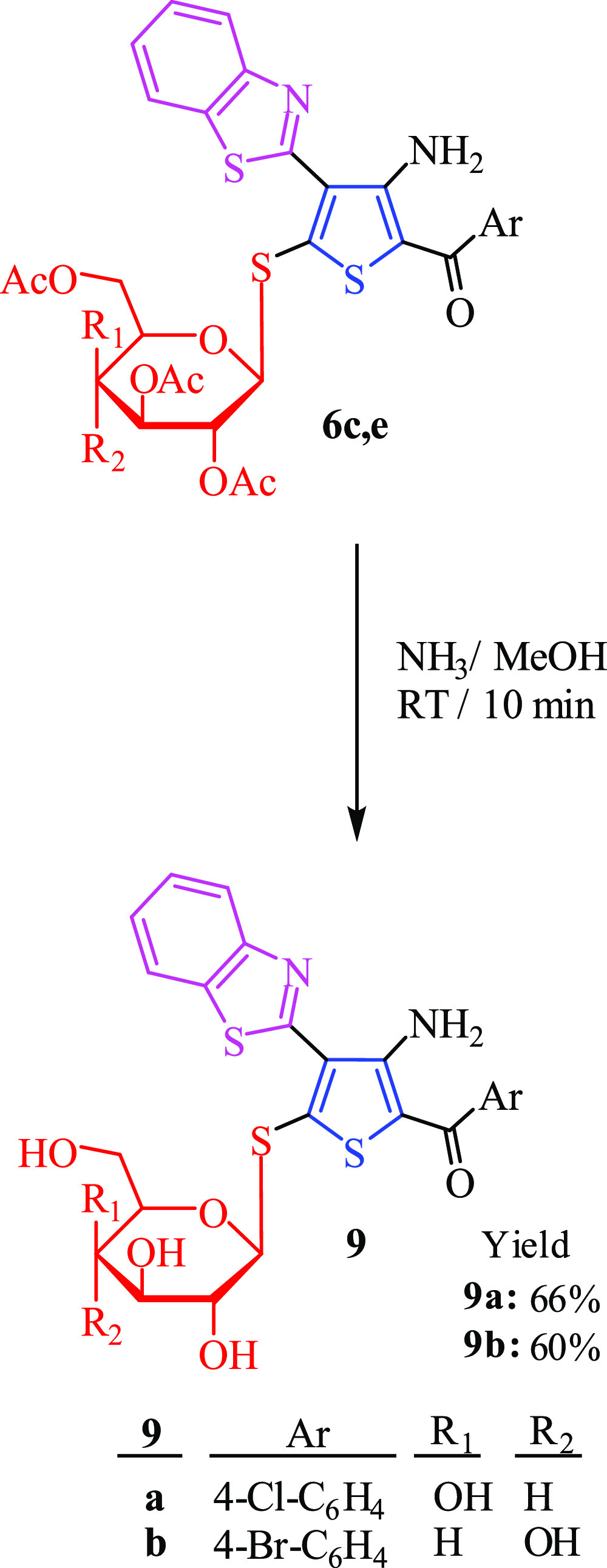
Last but not the least, at room temperature, compounds 6c and 6e were reacted with an NH3–MeOH solution to afford the desired free thioglycosides 9a and 9b. The free hydroxyl derivatives were obtained in good yield, Scheme 3. The structure of de-acetylated compounds was established based on their spectroscopic data. 1H NMR spectra of 9b showed the anomeric proton as a doublet at 4.12 ppm with the J coupling constant equal to 14.4 Hz, which indicated the presence of only β-D-configuration. The remaining six protons of glucose appear as a multiplet signal at 3.38–4.09 ppm, while the four hydroxyl groups of glucose resonated at 3.39–4.16 ppm.
Scheme 3. Synthesis of Free Benzothiazole-2-Thiophene S-Glycoside Derivatives 9a and 9b.
2.2. Biological Evaluation
2.2.1. Antiviral Evaluation
The synthesized compounds were tested in vitro for their antiviral activities against a variety of viruses such as coxsackievirus B4 (CBV4), hepatitis A virus HM 175 (HAV), hepatitis C genotype 4 (HCVcc), adenovirus type 7 (HAdV7), and herpes simplex virus 1 (HSV-1). In order to determine the antiviral activity of the synthesized compounds, both cytotoxicity and inhibition percentage values were determined using plaque reduction assay, and the resulting data are summarized in Tables 1 and 2 and Figure 4.
Table 1. Nontoxic Doses of 4a–4c, 6a–6f, and 8a–8d Compounds on FRHK-4, Hep2, BGM, Vero, and Huh 7.5 Cell Lines.
| nontoxic dose (μg/mL) | |||||
|---|---|---|---|---|---|
| compd. no. | FRHK4 | Hep2 | BGM | Vero | Huh 7.5 |
| 4a | 90 | 90 | 90 | 90 | 90 |
| 4b | 80 | 80 | 70 | 80 | 70 |
| 4c | 100 | 100 | 100 | 100 | 100 |
| 6a | 90 | 90 | 90 | 90 | 90 |
| 6b | 80 | 80 | 80 | 80 | 90 |
| 6c | 100 | 100 | 100 | 100 | 100 |
| 6d | 100 | 100 | 100 | 100 | 100 |
| 6e | 90 | 90 | 80 | 90 | 90 |
| 6f | 90 | 90 | 90 | 100 | 90 |
| 8a | 90 | 90 | 80 | 90 | 90 |
| 8b | 80 | 80 | 80 | 80 | 80 |
| 8c | 100 | 100 | 100 | 100 | 100 |
| 8d | 90 | 90 | 90 | 100 | 90 |
Table 2. CC50, IC50 (μg/mL), and IS of 6c, 6d, and 8c Compounds with Viral Reduction 50% or More.
| herpes simplex virus | |||||
|---|---|---|---|---|---|
| compd no. | mean % of reduction | nontoxic dose (μg/mL) | CC50 (μg/mL) | IC50 (μg/mL) | IS |
| 6c | 83% | 100 | 1.8 | 0.55 | 3.4 |
| 6d | 80% | 100 | 1.6 | 0.57 | 2.8 |
| 8c | 86% | 100 | 2.0 | 0.57 | 3.5 |
| acyclovir | 99% | - | 2.7 | 0.07 | 4.00 |
| HCVcc genotype virus | |||||
| 6c | 63.3% | 100 | 1.9 | 0.76 | 2.5 |
| 8c | 66.7% | 100 | 1.7 | 0.71 | 2.4 |
| coxsackievirus B4 | |||||
| 6c | 63.3% | 100 | 1.9 | 0.76 | 2.5 |
| 6d | 50.0% | 100 | 1.7 | 1.0 | 1.7 |
| 8c | 66.7% | 100 | 1.7 | 0.69 | 2.4 |
Figure 4.
Viral reduction % of 4a–4c, 6a–6f, and 8a–8d compounds against HSV-1, CBV4, HAV, HCVcc, and HAdV7.
2.2.1.1. Cytotoxicity Evaluation
To investigate the antiviral activities of the newly synthesized compounds, the nontoxic dose was first tested against five normal cell lines, including FRHK-4, Hep2, BGM, Vero, and Huh 7.5 cell lines, which serve as specific hosts for the various viruses studied, as shown in Table 1. Based on the data, the nontoxic doses of the tested compounds range between 70 and 100 g/mL, indicating that there are no significant differences between them. In comparison to the other tested compounds, the formed compounds 4c, 6c, 6d, and 8c had the highest safety profile. Compound 4b, on the other hand, had the lowest level of safety in its dosing profile with a nontoxic dose value of 70 μg/mL against the Vero cell line and 80 μg/mL nontoxic dose value against the other examined cell lines.
2.2.1.2. Antiviral Evaluation
The compounds were then tested against five different virus strains with different types of genomes, CBV4, HAV, and HCVcc as RNA viruses and HAdV7 and HSV-1 as DNA viruses. The plaque assay method was used to determine the mean percentage reduction of viral titers (1 × 105, 1 × 106, and 1 × 107) by combining the nontoxic dilutions of tested compounds with 0.1 mL of tested viruses (Tables S1–5, Supporting Information). It was observed that attachment of sugar moieties to the substituted thiophene system enhanced the antiviral activity against the mentioned viruses. Three compounds, in particular, 6c, 6d, and 8c, exhibited interesting antiviral effects that exceed 50% reduction against CBV4, HSV-1, and HCVcc, Figure 5. Compound 6c showed 83.3% reduction against HSV-1 and 63.3% for both CBV4 and HCVcc, while 6d revealed reduction of 80% for HSV-1 and 50% for CBV4. Compound 8c was the most potent one which showed reduction of 86.7% for HSV-1 and 66.7% for both CBV4 and HCVcc.
Figure 5.
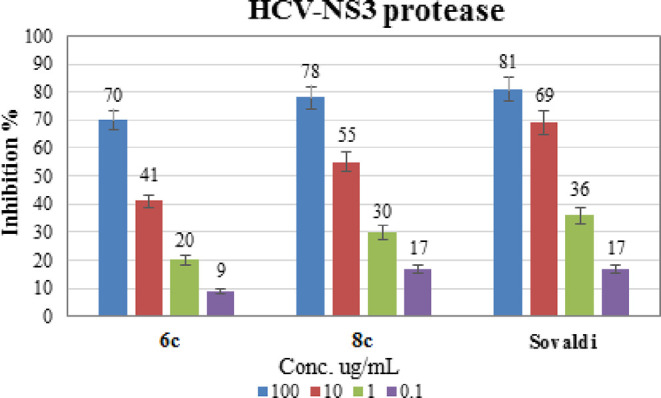
Inhibition percent of compounds 6c, 8c, and Sovaldi against the HCV- NS3 protease enzyme.
CC50, IC50, and SI (the concentration in –μg/mL required to reach 50% cytotoxicity of the uninfected cell, the concentration in μg/mL required to inhibit 50% of the tested viruses, and the selectivity index, CC50/IC50 ratio, respectively) were evaluated for the most potent compounds, Table 2. Based to the data listed in Table 2, the three most potent compounds, 6c, 6d, and 8c, against HSV-1 had IC50 values ranging from 0.55 to 0.57 μg/mL and CC50 values ranging from 1.6 to 2.0 μg/mL. Despite the fact that these three compounds showed a close performance to the standard drug (acyclovir) against HSV-1, two of them, compounds 6c and 8c, had SI values of 3.4 and 3.5, respectively, which are comparable to the standard drug, SI = 4. In the case of the activities against HCVcc, the two most potent compounds, 6c and 8c, had IC50 values of 0.76 to 0.71 μg/mL and CC50 values of 1.9 to 1.7 μg/mL. Both compounds have a comparable SI value. Additionally, the results showed that the three most potent compounds, 6c, 6d, and 8c, against CBV4 had IC50 values ranging from 0.69 to 1.0 μg/mL and CC50 values ranging from 1.7 to 1.9 μg/mL. Moreover, these three compounds displayed SI values ranging from 1.7 to 2.5.
According to the abovementioned data, the structure–activity relationships (SAR) have been showed that the presence of a bromide or methyl group in the para position of the benzoyl ring bonded to the thiophene ring showed higher activity against most of the studied viruses than the corresponding compounds with a nonsubstituted benzoyl ring and with chlorine in the para position of the benzoyl ring.
2.2.2. Enzymatic Assay
2.2.2.1. Hepatitis C Virus NS3/4A Protease
It is known that infection with the hepatitis C virus is a major cause of acute liver disease and hepatocellular carcinoma. The possibility that HCV_NS3 inhibitors would suppress HCV infection directed us to test our newly manufactured compounds as HCV_NS3 inhibitors. The two most potent compounds against HCVcc, compounds 6c and 8c, were evaluated for their efficiency for inhibition of hepatitis C virus NS3/4A protease using EnzoLyte 520 HCV protease assay kit *Fluorimetric*. The kit is designed to detect hepatitis C virus NS3/4A protease activity using a 5-FAM/QXL 520 FRET peptide substrate that can then be observed at Ex/Em = 490 nm/520 nm upon proteolytic cleavage. This cleavage is essential for viral protein maturation. As a result, this protease has emerged as a key target for anti-HCV drug development. The IC50 values of compounds 6c and 8c against HCV NS3 protease in comparison to the standard drug (Sovaldi) is shown in Table 3, and the inhibition percentages obtained from this study were plotted in graph with error bars representing the standard deviation of the obtained data, Figure 5. According to the data, compound 8c has an IC50 value of 5.16 ± 0.26 μg/mL with a xylose moiety and bromine at the para position of the benzoyl group, while compound 6c has an IC50 value of 16.01 ± 0.57 μg/mL with a glucose moiety and bromine at the para position of the benzoyl group. This indicated that compound 8c is more potent than Sovaldi (IC50 = 2.70 ± 0.14 μg/mL).
Table 3. IC50 ug/mL Values for the Synthesized Compounds 6c and 8c Against HCV-NS3 Protease.
| compd no. | IC50 (μg/mL) |
|---|---|
| 6c | 16.01 ± 0.57 |
| 8c | 5.16 ± 0.26 |
| Sovaldi | 2.70 ± 0.14 |
2.2.2.2. Ubiquitin-Specific Protease 7 (USP7)
HSV is a virus that infects the mouth, lips, genitals, and nose mucous membranes. HSV-1 and HSV-2 are the two types of herpes simplex virus. Cold sores and fever blisters on the lips and face are caused by HSV-1, whereas genital herpes is caused by HSV-2. Among the ubiquitin-specific-processing protease (USP) family members, ubiquitin specific protease 7 (USP7) is known as herpes-associated ubiquitin-specific protease (HAUSP) and functions as a gene expression regulator in HSV. The degradation of ICP0, an HSV-associated protein, has been found to be regulated by USP7. ICP0 is an E3-ubiquitin ligase that ubiquitinates itself and certain cellular proteins before degrading them. The three most effective compounds against HSV-1, compounds 6c, 6d, and 8c, were assessed for their efficiency for inhibition of USP7 using the USP7 screening kit which is intended to assess USP7 activity for use in screening and profiling applications. Table 4 presents the IC50 values of compounds 6c, 6d, and 8c and the standard drug (acyclovir) against USP7 protease, and the inhibition percentages obtained from this study were plotted in graph with error bars representing the standard deviation of the evaluated data, Figure 6. The data revealed that compound 8c, containing a xylose moiety and bromine at the para position of the benzoyl group, has the lowest IC50 value of 5.16 ± 0.26 μg/mL. On the other hand, compound 6d, containing a galactose moiety and methyl group at the para position of the benzoyl group, has the highest IC50 value of 30.33 ± 2.03 μg/mL. As a result, when compared to acyclovir (IC50 = 2.480.09 g/mL), compound 8c was found to be the most potent.
Table 4. IC50 ug/mL Values for the Synthesized Compounds 6c, 6d, and 8c Against the HSV-USP7 Enzyme.
| compd no. | IC50 (μg/mL) |
|---|---|
| 6c | 7.68 ± 0.51 |
| 6d | 30.33 ± 2.03 |
| 8c | 7.23 ± 0.26 |
| Acyclovir | 2.48 ± 0.09 |
Figure 6.
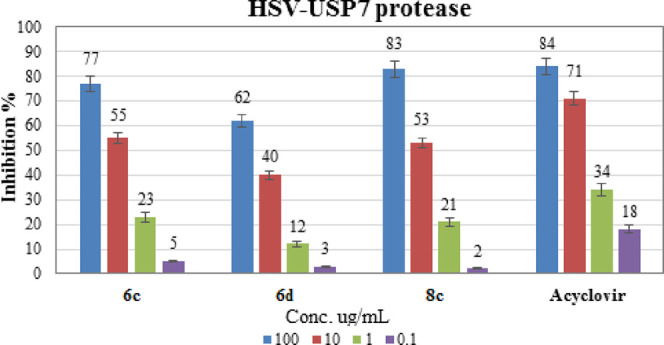
Inhibition percent of compounds 6c, 6d, and 8c and acyclovir against the HSV- USP7 protease enzyme.
2.2.3. Anticancer Evaluation
The in vitro tumor growth inhibitory activities of the newly synthesized compounds were tested at the National Cancer Institute (NCI) against a panel of 60 cancer cell lines of human cancer cells using the standard procedure which involved nine tumor subpanels (leukemia, melanoma, lung, colon, CNS, ovarian, renal, prostate, and breast cancers) at a single concentration of 10–5 M. The potential anticancer effect has been represented as the growth percent (G %) of all the treated 60 cell lines, as shown in Table 5 (Figures S30–42, Supporting Information).
Table 5. Growth Percent (G %) of 4a–4c, 6a–6f, and 8a–8d Compounds at 10–5 M Concentration.
| growth % |
||||
|---|---|---|---|---|
| tumor cell lines | 6a | 6c | 6d | 6f |
| leukemia | ||||
| CCRFCEM | 60.51 | 44.08 | 79.51 | 85.51 |
| HL-60(TB) | 79.97 | 113.21 | 82.66 | 105.30 |
| K-562 | 68.45 | 66.41 | 90.58 | 92.18 |
| MOLT-4 | 79.96 | – | 69.98 | 79.34 |
| RPMI-8226 | 46.53 | 43.00 | 61.25 | 65.50 |
| nonsmall-cell lung cancer | ||||
| A549/ATCC | 81.90 | 71.51 | 89.15 | 92.17 |
| EKVX | 75.03 | 72.66 | 68.31 | 80.45 |
| HOP-62 | 61.52 | 63.81 | 86.08 | 91.13 |
| HOP-92 | 58.78 | 81.67 | 83.73 | 98.29 |
| NCI–H226 | 30.28 | 28.11 | 49.81 | 65.59 |
| NCI–H23 | 56.06 | 23.82 | 83.72 | 88.36 |
| NCI–H322M | 81.34 | 83.53 | 92.00 | 91.82 |
| NCI–H460 | 43.70 | 44.27 | 108.20 | 107.34 |
| NCI–H522 | 82.14 | 63.78 | 82.60 | 94.07 |
| colon cancer | ||||
| COLO 205 | 82.14 | 63.78 | 82.60 | 94.07 |
| HCC-2998 | 92.18 | 84.98 | 106.44 | 107.74 |
| HCT-116 | 94.15 | 75.89 | 105.80 | 98.76 |
| HCT-15 | 39.31 | 18.65 | 92.50 | 91.26 |
| HT29 | 59.50 | 59.83 | 94.25 | 99.74 |
| KM 12 | 69.02 | 63.11 | 105.46 | 105.73 |
| SW-620 | ||||
| CNS cancer | 80.60 | 78.35 | 86.28 | 95.66 |
| SF-268 | 70.97 | 60.91 | 85.97 | 92.97 |
| SF-295 | 73.57 | 69.60 | 96.89 | 97.06 |
| SF539 | 28.19 | 15.70 | 91.32 | 95.18 |
| SNB-19 | 74.43 | 75.19 | 88.22 | 87.35 |
| SNB-75 | 25.40 | 16.66 | 55.71 | 74.45 |
| U251 | 59.53 | 57.62 | 93.67 | 96.21 |
| melanoma | ||||
| LOX IMVI | 54.51 | 37.10 | 92.00 | 96.76 |
| MALME-3M | 43.88 | 20.04 | 97.98 | 98.05 |
| M14 | 65.68 | 61.62 | 92.69 | 96.10 |
| MDA-MB-435 | 77.42 | 80.65 | 86.32 | 91.67 |
| SK-MEL-2 | 93.61 | 106.50 | 98.57 | 111.89 |
| SK-MEL-28 | 76.43 | 70.66 | 103.42 | 103.85 |
| SK-MEL-5 | 45.50 | 22.05 | 30.64 | 36.29 |
| UACC-257 | 79.26 | 80.15 | 98.45 | 103.96 |
| UACC-62 | 55.85 | 89.28 | 57.28 | 60.39 |
| ovarian cancer | ||||
| IGROV1 | 48.23 | 45.62 | 58.43 | 68.85 |
| OVCAR-3 | 59.41 | 76.24 | 65.91 | 67.13 |
| OVCAR-4 | –13.92 | 12.92. | 30.71 | 46.89 |
| OVCAR-5 | 58.63 | 65.70 | 99.7 | 104.58 |
| OVCAR-8 | 58.68 | 46.10 | 73.63 | 80.31 |
| NCI/ADR-RES | 62.87 | 57.90 | 82.05 | 88.35 |
| SK-OV-3 | 89.92 | 96.59 | 52.61 | 57.30 |
| renal cancer | ||||
| 786-0 | 47.40 | 105.00 | 89.07 | 94.42 |
| A498 | 98.86 | 58.50 | 53.43 | 36.08 |
| ACHN | 23.12 | 97.84 | 86.99 | 90.51 |
| CAKI-1 | 53.68 | 85.49 | 58.55 | 67.61 |
| RXF 393 | 53.54 | 116.32 | 96.29 | 114.72 |
| SN12C | 41.92 | 97.97 | 91.44 | 95.12 |
| TK-10 | 99.32 | 146.81 | 124.58 | 127.82 |
| UO-31 | 25.70 | 73.56 | 57.59 | 61.39 |
| prostate cancer | ||||
| PC-3 | 63.22 | 90.78 | 60.63 | 70.84 |
| DU-145 | 42.11 | 105.71 | 106.81 | 109.77 |
| breast cancer | ||||
| MCF7 | 73.63 | 73.74 | 100.13 | 94.60 |
| MDA-MB-231/ATCC | 30.00 | 84.08 | 72.99 | 80.58 |
| HS 578T | 52.53 83.43 | 93.77 | 82.92 | 95.38 |
| BT-549 | 69.69 | 103.45 | 82.70 | 90.16 |
| T-47D | 75.39 | 71.46 | 56.23 | 67.38 |
| MDA-MB-468 | 73.63 | 112.08 | 78.11 | 91.91 |
Most of the compounds having a thiophene ring paired with a sugar moiety under present investigation showed a range of activities against tested cell lines. The resulted data showed that compounds 6a and 6c revealed an inhibitory effect on most cancer types. Compound 6a was the only compound that exhibited a high inhibition effect on four renal cancer cell lines, 786-0, ACHN, SN12C, and UO-3. It was also the only compound which showed high inhibition on one type of breast cancer, MDA-MB-231/ATCC. Both compounds 6a and 6c showed high inhibition on two of the CNS cancer cell lines, SF-539 and SNB-75, and one of the colon cancer cell lines, HCT-116. However, the inhibition of compound 6c was observed to be higher than that of compound 6a against the previous mentioned cell lines. It was also noticed that compound 6c showed high inhibition against three of lung cancer, melanoma, and ovarian cancer cell lines, while compound 6a showed high inhibition against only two cell lines of the same type of cancer. Moreover, compound 6f revealed high inhibition against three cell lines, SK-MEL-5, OVCAR-4, and A498, while compound 6d showed high inhibition against only two cell lines, SK-MEL-5 and OVCAR-4. Both compounds 6c and 6f were the only ones which showed high inhibition against the renal cancer cell line of type A498.
3. Conclusions
In conclusion, new benzothiazole-2-thiophene S-glycoside derivatives were synthesized by reacting new derivatives of 5-mercaptothiophen substituted with the benzothiazole moiety, followed by coupling with various halo sugar derivatives. All of the compounds’ structures were confirmed using elemental analysis and spectroscopy. The new compounds were tested for their antiviral activity against HSV-1, ED-43/SG-Feo (VYG) replicon of HCV genotype 4a, HAV HM 175, COB4, and HAdV7. Two compounds demonstrated notable antiviral activity against CBV4, HSV-1, and HCVcc viruses, with more than 50% viral reduction. Compounds 6c and 8c, the most potent against HCVcc viruses, were tested against NS3/4A protease, and their activities were compared to those of the reference drug Sovaldi. The three most potent compounds against HSV-1 were also tested against USP7, and their activities were compared to those of the reference drug acyclovir. Additional anticancer evaluation of the synthesized compounds showed that two compounds, 6a and 6c, have an inhibitory effect on most types of cancers, while 6d and 6d only inhibited three and two cell lines, respectively.
4. Experimental Section
4.1. Chemistry
An SMP3 melting point apparatus was used to measure all melting points. At Ain Shams University in Cairo, Egypt, the 1H NMR and 13C NMR spectra were obtained using a Bruker Avance (111)-400 MHz spectrometer in DMSO-d6 using Si(CH3)4, TMS, as an internal criterion. The infrared spectra were captured using a Pye Unicam Spectra-1000 (KBr disk). Elemental analyses were measured in Cairo University’s Micro analytical Data Center. TLC was used to monitor the progress of the reactions using aluminum sheets coated with silica gel F254 (Merck) and inspecting under a short-wavelength UV lamp.
General Procedures for Synthesizing 4a–4c
To a solution of sodium ethoxide (0.001 mol) in 15 mL of ethanol, 2-cyanomethyl benzothiazol 1 (0.001 mol) was added and boiled for 20 min. By subsequently cooling in an ice bath, carbon disulfide (0.0025 mol) was added. The mixture was stirred for 20 min at room temperature and then refluxed for additional 20 min. Subsequently, phenacyl bromide derivatives (2a–2c) (0.001 mol) were added, and the reaction mixture was boiled until the completion of the reaction (TLC, 1-3 h). The reaction mixture was poured into water-ice and neutralized by hydrochloric acid. The resulting solid product was collected by filtration and recrystallized from the proper solvent to provide compounds 4a–4c.
3-Amino-4-(Benzothiazol-2-yl)-5-Mercaptothiophen-2-yl) (-4-Methyphenyl)methanon (4a)
Reddish brown solid; yield (70%); mp 295-297 °C; IR (KBr, cm–1): υ 3394 (NH2), 2924 (ArCH), 1741 (C=O); 1H NMR (400 MHz, DMSO-d6): δ 2.41 (s, 3H, CH3), 7.37–7.43 (m, 3H, 2Ar-H & 1benzothiazole-H), 7.55 (app. t, dd, J = 7.6 Hz, 1H, benzothiazole-H), 7.78 (d, J = 8.4 Hz, 1H, benzothiazole-H), 7.96 (d, J = 7.6 Hz, 2H, Ar-H), 8.01 (d, J = 8.4 Hz, 1H, benzothiazole-H); Anal calcd for chemical formula C19H14N2OS3 (382.51): C %, 59.66; H %, 3.69; N %, 7.32; found, C %, 59.64; H %, 3.67; N %, 7.31.
3-Amino-4-(Benzothiazol-2-yl)-5-Mercaptothiophen-2-yl) (-4-Chlorophenyl) Methanone (4b)
Reddish brown solid; yield (75%); mp 273-274 °C; IR (KBr, cm–1): υ 3409 (NH2), 2926 (ArCH), 1741 (C=O); 1H NMR (400 MHz, DMSO-d6): δ 7.32 (app. t, dd, J = 8.4 Hz, 1H, benzothiazole-H), 7.44 (app. t, dd, J = 8.0 Hz, 1H, benzothiazole-H), 7.52 (d, J = 8.4 Hz, 2H, Ar-H), 7.69 (d, J = 8.4 Hz, 2H, Ar-H), 7.87 (d, J = 7.2 Hz, 1H, benzothiazole-H), 8.00 (d, J = 8.0 Hz, 1H, benzothiazole-H); Anal calcd for chemical formula C18H11ClN2OS3 (402.94): C %, 53.65 H %, 2.75; N %, 6.95; found, C %, 53.63; H %, 2.74; N %, 6.93.
3-Amino-4-(Benzothiazol-2-yl)-5-Mercaptothiophen-2-yl) (-4-Bromophenyl) Methanone (6c)
Brown Solid; Yield (85%); mp 198-200 °C; IR (KBr, cm–1): υ 3396 (NH2), 2975 (ArCH), 1744 (C=O); 1H NMR (400 MHz, DMSO-d6): δ 7.45 (app. t, dd, J = 8.4 Hz, 1H, Benzothiazole-H), 7.53–7.57 (m, 3H, 2Ar-H & 1benzothiazole-H), 7.65 (d, J = 8.4 Hz, 2H, Ar-H), 8.03–8.08 (m, 2H, Benzothiazole-H); Anal calcd for chemical Formula C18H11BrN2OS3 (447.38): C %, 48.33, H %, 2.48; N %, 6.26; found, C %, 48.31; H %, 2.46; N %, 6.23
General Procedures for Synthesizing 6a–6c
To a solution of 4a–4c (0.001 mol) in pulverized KOH (0.01 mol), a solution of 2,3,4,6-tetra-O-acetyl-α-D-gluco- or galactopyranosyl bromide 5a and 5b (0.001 mol) in acetone (20 mL) was dropped within 30 min. The reaction mixture was stirred at room temperature and monitored by TLC (3–8 h) until the reaction completed. The residue was washed with distilled water to remove KBr. The product was dried prior to crystallization from methanol to give compounds 6a–f.
(3-Amino-4-(benzothiazol-2-yl)-5-(2’,3′,4′,6-tetra-O-acetyl-β-D-glucopyranosylthio)thiophen-2-yl) (-4-methy-phenyl)methanone (6a)
Brown solid; yield (63%); mp 221-223 °C; IR (KBr, cm–1): υ 3388, 3270 (NH2), 2941 (ArCH), 1755 (4CO), 1720 (CO); 1H NMR (400 MHz, DMSO-d6): δ 1.93–2.01 (4 s, 12H, 4xOAc), 2.41 (s, 3H, CH3), 3.95–4.11 (m, 3H, H-5’, 2H-6′), 4.92–5.04 (m, 2H, H-3’& H-4′), 5.37 (t, J = 12.0 Hz, 1H, H-2′), 5.02 (d, J = 8.0 Hz, 1H, H-1′), 7.37 (d, J = 8.0 Hz, 2H, Ar-H), 7.53 (app. t, dd, J = 8.4 Hz, 1H, benzothiazole-H), 7.61 (app. t, dd, J = 8.0 Hz, 1H, benzothiazole-H), 7.73 (d, J = 8.8 Hz, 2H, Ar-H), 8.14 (d, J = 8.8 Hz, 1H, benzothiazole-H), 8.21 (d, J = 9.2 Hz, 1H, benzothiazole-H), 8.86 (s, D2O exch., 2H, NH2); Anal calcd. For chemical formula: C33H32N2O10S3 (712.81): C %, 55.60; H %, 4.52; N %, 3.93; found: C %, 55.62; H %, 4.50; N %, 3.92.
(3-Amino-4-(benzothiazol-2-yl)-5-(2’,3′,4′,6-tetra-O-acetyl--β-D-glucopyranosylthio)thiophen-2-yl) (-4-chloro-phenyl)methanone (6b)
Brown solid; yield (70%); mp 216-218 °C; IR (KBr, cm–1): υ 3421 (NH2), 2987 (ArCH), 1758 (4CO), 1696 (CO); 1H NMR (400 MHz, DMSO-d6): δ 1.64–1.98 (4 s, 12H, 4xOAc), 3.97–4.12 (m, 3H, H-5′, 2H-6′), 4.94 (t, J = 8.0 Hz, 1H, H-4′), 5.00 (t, J = 10.0 Hz, 1H, H-3′), 5.36 (t, J = 8.8 Hz, 1H, H-2′), 5.45 (d, J = 9.2 Hz, 1H, H-1′), 7.53 (app. t, dd, J = 7.2 Hz, 1H, benzothiazole-H), 7.59–7.64 (m, 3H, 2Ar-H & 1benzothiazole-H), 7.84 (d, J = 8.8 Hz, 2H, Ar-H), 8.15 (d, J = 8.4 Hz, 1H benzothiazole-H), 8.90 (s, D2O exch., 2H, NH2); Anal. calcd. For chemical formula: C32H29ClN2O10S3 (733.22): C %, 52.42; H %, 3.99; N %, 3.82; found, C %, 52.40; H %, 3.98; N %, 3.80.
(3-Amino-4-(benzothiazol-2-yl)-5-(2’,3′,4′,6-tetra-O-acetyl--β-D-glucopyranosylthio)thiophen-2-yl) (-4-bromo-phenyl)methanone. (6c)
Yellow solid; yield (64%); mp 222-223 °C; IR (KBr, cm–1): υ 3405,3273 (NH2), 2930 (ArCH), 1745 (4CO), 1685 (CO); 1H NMR (400 MHz, DMSO-d6): δ 1.63–1.98 (4 s, 12H, 4xOAc), 3.96–4.13 (m, 3H, H-5’, 2H-6′), 4.91–5.03 (m, 2H, H-3’ & H4′), 5.36 (t, J = 9.6 Hz, 1H, H-2′), 4.45 (d, J = 9.2 Hz, 1H, H-1′), 7.52 (app. t, dd, J = 8.8 Hz, 1H, benzothiazole-H), 7.61 (app. t, dd, J = 9.6 Hz, 1H, benzothiazole-H), 7.75–7.77 (m, 4H, Ar-H), 8.13 (d, J = 8.8 Hz, 1H benzothiazole-H), 8.20 (d, J = 6.4 Hz, 1H, benzothiazole-H), 8.90 (s, D2O exch., 2H, NH2); 13C NMR (100 MHz, DMSO-d6): δ 20.8, 20.7, 20.8 (4CH3), 62.2, 68.1, 69.7, 73.1, 75.0, 85.0, 109.6, 122.3, 123.8, 125.1, 125.7, 126.5, 127.3, 130.1, 132.1, 133.59, 139.7, 150.9, 155.4, 160.6 (Ar-C), 169.5, 169.7, 169.9, 170.1 (4C=O), 185.7 (C=O); Anal. calcd. For chemical formula: C32H29 BrN2O10S3 (777.68): C %, 49.42; H %, 3.76; N %, 3.60; found, C %, 49.40; H %, 3.74; N %, 3.61.
(3-Amino-4-(benzothiazol-2-yl)-5-(2’,3′,4′,6-tetra-O-acetyl--β-D-galactopyranosylthio)thiophen-2-yl) (-4-methyphenyl)methanone (6d)
Yellow solid; yield (65%); mp 205-207 °C; IR (KBr, cm–1): υ 3405 (NH2), 2941 (ArCH), 1755 (4CO), 1701 (CO); 1H NMR (400 MHz, DMSO-d6): δ 1.70–2.00 (m, 12H, 4xOAc), 2.41 (s, 3H, CH3), 4.00–4.01 (m, 2H, H-6’), 4.32 (t, J = 8.0 Hz, 1H, H-5′), 5.14 (t, J = 8.2 Hz, 1H, H-4’), 5.25–5.29 (m, 2H, H-3′ δ H-2′), 5.37 (d, J = 8.6 Hz, 1H, H-1′), 7.36 (d, J = 8.0 Hz, 2H, Ar-H), 7.52 (app. t, dd, J = 8.0 Hz, 1H, benzothiazole-H), 7.61 (app. t, dd, J = 8.0 Hz, 1H, benzothiazole-H), 7.72 (d, J = 8.7 Hz, 2H, Ar-H), 8.15 (d, J = 9.0 Hz, 1H, benzothiazole-H), 8.20 (d, J = 7.0 Hz, 1H, benzothiazole-H), 8.91 (s, D2O exch., 2H, NH2); Anal. calcd. For chemical formula: C33H32N2O10S3 (712.81): C %, 55.60; H %, 4.52; N %, 3.93; found, C %, 55.62; H %, 4.51; N %, 3.92.
(3-Amino-4-(benzothiazol-2-yl)-5-(2’,3′,4′,6-tetra-O-acetyl--β-D-galactopyranosylthio)thiophen-2-yl) (-4-chlorophenyl)methanone (6e)
Yellow solid; yield (50%); mp 272-274 °C; IR (KBr, cm–1): υ 3404, 3278 (NH2), 2928 (ArCH), 1744 (4CO), 1687 (CO); 1H NMR (400 MHz, DMSO-d6): δ 1.83–2.11 (4 s, 12H, 4xOAc), 3.39–4.15 (m, 3H, H-5’, 2H-6′), 4.85 (d, J = 8.8 Hz, 1H, H-4’), 5.06–5.10 (m, 1H, H-3′), 5.36–5.44 (m, 2H, H-2’, H-1′), 7.44–7.51 (m, 3H, 2Ar-H & 1benzothiazole-H), 7.56 (app. t, dd, J = 8.0 Hz, 1H, benzothiazole-H), 7.80 (d, J = 8.0 Hz, 2H, Ar-H), 7.93 (d, J = 7.2 Hz, 1H, benzothiazole-H), 8.10 (d, J = 8.0 Hz, 1H, benzothiazole-H); Anal. calcd. For chemical formula: C32H29ClN2O10S3 (733.23): C %, 52.42; H %, 3.99; N %, 3.82; found, C %, 52.40; H %, 3.98; N %, 3.80.
(3-Amino-4-(benzothiazol-2-yl)-5-(2’,3′,4′,6′-tetra-O-acetyl-β-D-galactopyranosylthio)thiophen-2-yl) (-4-bromophenyl)methanone (6f)
Yellow solid, yield (65%), mp 130-132 °C; IR (KBr, cm–1): υ 3406, 3281 (NH2), 2923 (ArCH), 1748 (4Ac-CO), 16520 (CO); 1H NMR (400 MHz, DMSO-d6): δ 1.74–1.94 (4 s, 12H, 4xOAc), 4.00–4.02 (m, 2H, H-6′), 4.32 (t, J = 6.0 Hz, 1H, H-5′), 5.15 (t, J = 8.0 Hz, 1H, H-4’), 5.25–5.30 (m, 2H, H-3′, H-2′), 5.39 (d, J = 8.8 Hz, 1H, H-1′), 7.52 (app. t, dd, J = 7.4 Hz, 1H, benzothiazole-H), 7.61 (app. t, dd, J = 7.4 Hz, 1H, benzothiazole-H), 7.77–7.79 (m, 4H, Ar-H), 8.14 (d, J = 7.6 Hz, 1H, benzothiazole-H), 8.21 (d, J = 8.0 Hz, 1H, benzothiazole-H), 8.93 (s, br, D2O exch., 2H, NH2); Anal. calcd. For chemical formula: C32H29 BrN2O10S3 (777.68): C %, 49.42; H %, 3.76; N %, 3.60; found, C %, 49.41; H %, 3.75; N %, 3.61.
General Procedures for Synthesizing (8a–8d)
The thiophene derivative 4a–4c (5 mmol) was dissolved in acetone (10 mL) containing 0.5 mL of aq. KOH (5 mmol). The mixture was warmed at 50 °C for 15 min. After cooling, a solution of 2,3,4-tri-O-acetyl-β-L-xylo or arabinopyranosyl bromides 7a and 7b (5 mmol) in acetone (10 mL) was dropped within 30 min. The reaction mixture stirred at room temperature and monitored by TLC until the reaction completed (8 h). The residue was washed with distilled water to remove KBr. The resulting precipitate was dried prior to crystallization from methanol to produce compounds 8a–8d.
(3-Amino-4-(benzothiazol-2-yl)-5-(2,3,5-tri-O-acetyl-β-D-xylopyranosylthio) Thiophen-2-yl) (-4-Methyl-Phenyl) Methanone (8a)
Greenish yellow solid; yield (75%); mp 252-254 °C; IR (KBr, cm–1): υ 3393, 3273 (NH2), 2924 (ArCH), 1741 (4CO), 1701 (CO); 1H NMR (400 MHz, DMSO-d6): δ 1.99–2.02 (3 s, 9H, 3xOAc), 2.42 (s, 3H, CH3), 3.61–3.67 (m, 1H, H-5″), 4.04–4.13 (m, 1H, H-5’), 4.87–4.92 (m, 1H, H-4′), 5.02 (t, J = 8.0 Hz, 1H, H-3′), 5.24 (t, J = 7.2 Hz, 1H, H-2′), 5.43 (d, J = 8.0 Hz, 1H, H-1′), 7.38 (d, J = 7.6 Hz, 2H, Ar-H), 7.52 (app. t, dd, J = 7.6 Hz, 1H, benzothiazole-H), 7.60 (app. t, dd, J = 7.2 Hz, 1H, benzothiazole-H), 7.70 (d, J = 8.4 Hz, 2H, Ar-H), 8.14 (d, J = 10.0 Hz, 1H, benzothiazole-H), 8.20 (d, J = 10.0 Hz, 1H, benzothiazole-H), 8.90 (s, D2O exch., 2H, NH2); Anal. calcd. For chemical formula: C30H28N2O8S3 (640.75): C %, 56.23; H %, 4.40; N %, 4.37; found, C %, 56.25; H %, 4.42; N %, 4.35.
(3-Amino-4-(benzothiazol-2-yl)-5-(2,3,5-tri-O-acetyl-β-D-xylopyranosylthio) Thiophen-2-yl) (-4-Chlorophenyl) Methanone (8b)
Greenish yellow solid; yield (70%); mp 154-156 °C; IR (KBr, cm–1): υ 3402, 3272(NH2), 3024 (ArCH), 1740 (4CO), 1690 (CO); 1H NMR (400 MHz, DMSO-d6): δ 1.99–2.02 (3 s, 9H, 3xOAc), 3.34–3.67 (m, 1H, H-5″), 4.09–4.14 (m, 1H, H-5’), 4.87–4.88 (m, 1H, H-4′), 4.90 (t, J = 8.4 Hz, 1H, H-3′), 5.25 (t, J = 8.4 Hz, 1H, H-2′), 5.47 (d, J = 10.4 Hz, 1H, H-1′), 7.53 (app. t, dd, J = 6.8 Hz, 1H, benzothiazole-H), 7.59–7.66 (m, 3H, 2Ar-H & 1benzothiazole-H), 7.81 (d, J = 9.2 Hz, 2H, Ar-H), 8.15 (d, J = 8.0 Hz, 1H, benzothiazole-H), 8.22 (d, J = 8.4 Hz, 1H, benzothiazole-H), 8.89 (s, D2O exch., 2H, NH2); Anal. calcd. For chemical formula: C29H25ClN2O8S3 (661.17): C %, 52.68; H %, 3.81; N %, 4.24; found, C %, 52.67; H %, 3.80; N %, 4.23.
(3-Amino-4-(benzothiazol-2-yl)-5-(2,3,5-tri-O-acetyl-β-D-xylopyranosylthio) Thiophen-2-yl) (-4-Bromo-Phenyl) Methanone (8c)
Dark yellow solid; yield (75%); mp 244–245.5 °C; IR (KBr, cm–1): υ 3404, 3268 (NH2), 2922 (ArCH), 1750 (4CO), 1735 (CO); 1H NMR (400 MHz, DMSO-d6): δ 1.99–2.02 (3 s, 9H, 3x OAC), 3.62–3.69 (m, 1H, H-5″), 4.11–4.14 (m, 1H, H-5’), 4.86–4.91 (m, 1H, H-4′), 5.03 (t, J = 8.4 Hz, 1H, H-3′), 5.25 (t, J = 8.4 Hz, 1H, H-2′), 5.46 (d, J = 8.4 Hz, 1H, H-1′), 7.53 (app. t, dd, J = 9.2 Hz, 1H, benzothiazole-H), 7.61 (app. t, dd, J = 7.2 Hz, 1H, benzothiazole-H), 7.74 (d, J = 8.8 Hz, 2H, Ar-H), 7.78 (d, J = 8.0 Hz, 2H, Ar-H), 8.14 (d, J = 8.4 Hz, 1H benzothiazole-H), 8.21 (d, J = 8.4 Hz, 1H, benzothiazole-H), 8.90 (s, D2O exch., 2H, NH2); 13C NMR (100 MHz, DMSO-d6): δ 20.8, 20.9, (3CH3), 68.1, 69.5, 71.3, 86.3, 109.2, 122.3, 123.1, 124.6, 125.5, 126.5, 127.3, 129.9, 132.1, 133.5, 139.8, 143.0, 155.4, 160.5 (Ar-C), 169.5, 169.7, 169.9 (3C=O), 185.6 (C=O); Anal. calcd. For chemical formula: C29H25 BrN2O8S3 (705.62): C %, 49.36; H %, 3.57; N %, 3.97; found, C %, 49.35; H %, 3.56; N %, 3.94.
(3-Amino-4-(benzothiazol-2-yl)-5-(2,3,5-tri-O-acetyl-β–L-arabinopyranosylthio)thiophen-2-yl) (-4-bromo-phenyl)methanone (8d)
Yellow solid, yield (65%) mp 206-207 °C; IR (KBr, cm–1): υ 3374, 3266 (NH2), 2989 (ArCH), 1746 (4CO); 1H NMR (400 MHz, DMSO-d6): 1.95–1.99 (3 s, 9H, 3x0Ac), δ 3.90–3.97 (m, 2H, H-5’), 4.00–4.10 (m, 1H, H-4’), 5.13–5.24 (m, 2H, H-3’, H-2′), 5.35 (d, J = 8.4 Hz, 1H, H-1′), 7.53 (app. t, dd, J = 9.2 Hz, 1H, benzothiazole-H), 7.61 (app. t, dd, J = 8.0 Hz, 1H, benzothiazole-H), 7.73 (d, J = 9.2 Hz, 2H, Ar-H), 7.78 (d, J = 8.0 Hz, 2H, Ar-H), 8.14 (d, J = 8.4 Hz, 1H, benzothiazole-H), 8.20 (d, J = 8.4 Hz, 1H, benzothiazole-H), 8.94 (s, D2O exch., 2H, NH2). 13C NMR (100 MHz, DMSO-d6): δ 20.8, 20.8, 20.9 (3CH3), 67.6, 68.1, 70.5, 86.0, 109.4, 122.3, 123.1, 124.6, 125.5, 126.5, 127.3, 129.9, 132.2, 133.5, 139.9, 143.0, 150.9, 155.5, 160.6 (Ar-C), 169.6, 169.8, 170.1 (4C=O), 185.7 (C=O); Anal calcd. For Chemical Formula: C29H25BrN2O8S3 (705.62): C %, 49.36; H %, 3.57; N %, 3.97; found, C %, 49.34; H %, 3.56; N %, 3.98.
General Procedure for the Synthesis of (9a, 9b)
On a solution of protected nucleosides 6c and 6e (0.001 mol) in dry methanol (20 mL), dry gaseous ammonia was passed for 10 min at 0 °C with stirring. The reaction combination was then moved at room temperature and monitored by TLC (9–10 h) until the reaction completed to afford a solid residue. The resulting mixture was crystallized from the appropriate solvent to give the corresponding products (9a and 9b).
(3-Amino-4-(benzothiazol-2-yl)-5-((2S,3S,4R,5R)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-ylthio)thiophen-2-yl) (4-chlorophenyl)methanone (9a)
Yellow solid; yield (66%); mp 210-212 °C; IR (KBr, cm–1): υ 3374-3266 (NH2), 2989 (ArCH), 1746 (CO); 1H NMR (400 MHz, DMSO-d6): δ 4.85–4.92 (4 s, 4H, 2′-OH, 3′-OH, 4′-OH, 6′-OH), δ 3.38–3.44 (m, 1H, H-5″), 3.54–3.62 (m, 1H, H-5′), 3.98–4.13 (m, 3H, H-4’, H-3′, H-2′), 4.09 (d, J = 14.4 Hz, 1H, H-1′), 7.50–7.55 (m, 3H, 2Ar-H & 1benzothiazole-H), 7.68 (app. t, dd, J = 8.0 Hz, 1H, benzothiazole-H), 7.85 (d, J = 8.8 Hz, 2H, Ar-H), 8.14 (d, J = 11.6 Hz, 1H, benzothiazole-H), 8.22 (d, J = 7.2 Hz, 1H, benzothiazole-H), 8.94 (s, D2O exch., 2H, NH2); Anal. calcd. For chemical formula: C24H21ClN2O6S3 (565.08): C, 51.01; H, 3.75; N, 4.96; found, C, 51.05; H, 3.72; N, 4.98.
(3-Amino-4-(benzothiazol-2-yl)-5-((2S,3S,4R,5S)-3,4,5-trihydroxy-6-(hydroxymethyl)tetrahydro-2H-pyran-2-ylthio)thiophen-2-yl) (4-bromophenyl)methanone (9b)
Yellow solid; yield (60%); mp 210-212 °C; IR (KBr, cm–1): υ 3374, 3266 (NH2), 2989 (ArCH), 1746 (CO); 1H NMR (400 MHz, DMSO-d6): δ 4.87–4.90 (4 s, 4H, 2′-OH, 3′-OH, 4′-OH, 6′-OH), δ 3.39–3.46 (m, 1H, H5″), 3.55–3.65 (m, 1H, H5′), 4.00–4.16 (m, 3H, H-4’, H-3′, H-2′), 4.12 (d, J = 14.4 Hz, 1H, H-1′), 7.51–7.57 (m, 3H, 2Ar-H & 1benzothiazole-H), 7.68 (app. t, dd, J = 8.0 Hz, 1H, Benzothiazole-H), 7.85 (d, J = 8.8 Hz, 2H, Ar-H), 8.14 (d, J = 11.6 Hz, 1H, Benzothiazole-H), 8.22 (d, J = 7.2 Hz, 1H, 1benzothiazole-H), 8.94 (S, D2O Exch., 2H, NH2); Anal. calcd. For chemical Formula: C24H21BrN2O6S3 (609.53): C, 47.29; H, 3.47; N, 4.60; found, C, 47.32; H, 3.44; N, 4.63
4.2. Antiviral Activity
4.2.1. Cytotoxicity Test
The National Research Center in Cairo, Egypt, conducted cytotoxicity and antiviral tests.32,33 In 1 mL of DMSO, 50 mg of each sample was dissolved. One milliliter of each sample was added to 24 μL of an antibiotic-antimycotic solution to decontaminate them. To obtained the nontoxic dose of the tested samples, 100 μL of the original dissolved samples were bifold diluted, and 100 μL of each dilution was added to cell lines of the Hep-2, Vero, BGM, FRHK4, and Huh 7.5 which were obtained from VACSERA. These cell lines had previously been cultivated in 96 multiwell plates from Greiner-Bio one, Germany. A reversed light microscope was used to assess the morphology of the cells, and the trypan blue dye exclusion method was used to gauge their vitality.
4.2.1.1. Cell morphology Evaluation by Inverted Light Microscopy
Separate 96 tissue culture dishes, from Greiner-Bio one, Germany, with Vero, Hep-2, FRHK4, Huh, and BGM 7.5 cell cultures “2*105 cells/mL” were arranged alone. Cell monolayers were confluent after 24 h of incubation at 37 degrees Celsius in a humidified atmosphere of 5 percent (v/v) CO2, at which point the media was uninvolved from each well and replaced with 100 L of bifold dilutions of the various samples examined and prepared in Dulbecco’s modified Eagle’s medium (DMEM, GIBCO BRL). DMEM (100 mL) devoid of samples was added for cell controls. For 72 h, all cultures were incubated in a wetted environment containing 5% (v/v) CO2 at 37 °C. Daily checks of cell morphology for microscopically discernible morphological changes, such as cell turning and shrinkage, loss of union, and vacuolization and cytoplasm granulation, were carried out. Morphological changes were scored.32
4.2.2. Cell Viability Assay
The assay was complete using the pan blue dye exclusion method.33 In 12 tissue culture dishes, Hep-2, BGM, Vero, Huh, and FRHK4 7.5 cell cultures (2*105 cells/mL) were developed. Following a 24 h incubation period, the identical assay defined above for experienced sample cytotoxicity was performed, using 100 μL of experienced sample dilutions (bifold dilutions) per well. After 72 h, the medium was uninvolved, the cells were trypsinzed, and an identical volume of 0.4 percent (w/v) was added. Aqueous solution of pan blue dye was added to the cell suspension. Under the part contrast microscope, viable cells were calculated.
4.2.3. Determination of Coxsackievirus B4, Adenovirus 7, Herpes Simplex Virus Type 1, and HAV HM175 Titers Using Plaque Assay
Nontoxic dilutions were mixed (100 μL) with 100 μL of different doses of herpes simplex virus type 1, HAV HM175, adenovirus 7, and coxsackievirus B4 (1 × 10,5 1 × 10,6 and 1 × 107). The mixture was left for 30 min. In 12 multiwell plates, (100 μL) 10-fold dilutions of treated and untouched adenovirus 7, coxsackievirus B4, HAV HM175, and herpes simplex virus type 1 were inoculated independently into Vero cell lines, Hep-2, BGM, and FRHK4 at 37 °C. Later, 1 h of adsorption at 37 °C in a 5% water–CO2 cloud atmosphere was carried out. The dishes were shaken on a regular basis to retain the cells from dehydrating out. Following adsorption, 1 mL of 2X media of DMEM (Gibco-BRL) was added, 1 mL of 1 percent agars was added to each well, and the dishes were left at 37 °C in a 5% water–CO2 cloud atmosphere. The cells were immersed with 0.4 percent crystal violet after the appropriate incubation period and formalin fixation, and the sum of plaques was calculated. Following that, the viral titers were counted and stated as plaque-forming units per milliliter (u/mL).34 For the hopeful materials having viral drop 50 percent or more, the CC50 and IC50 were calculated. The CC50, or 50% cytotoxic concentration of the test extract, is well-defined as the concentration that reduces the OD492 of the treated uninfected cells to half that of the untreated uninfected cells. The concentration at which the compound plaque reduction rate touches halfway between the reference line and the maximum is referred to as the IC50. All records were calculated as the mean of three measurements (triplicates).
4.2.4. Antiviral Assay of Synthesized Compounds Against the ED43/SGFeo (VYG) Replicon of Hepatitis C Virus Genotype 4a
The nontoxic dosage of the synthesized compounds was given to the ED-43/SG-Feo (VYG) replicon of HCV genotype 4a. According to the literature, HCV was calculated in algal extracts treated with Huh 7.5 infected cells using qRT-PCR and agreeing to the manufacturer’s directions to display a dose-dependent reduction in subgenomic RNA copies.35
4.3. Enzyme Assay
4.3.1. HCV_NS3 Protease Assay
NS3 inhibitor screening kit (Colorimetric) hepatitis C virus nonstructural protein 3 (HCV_ NS3) comprises an RNA helicase 1 and serine protease. Hepatitis C virus (HCV) infection is a chief cause of hepatocellular carcinoma and chronic liver disease. The probability that inhibitors of HCV_ NS3 would be inhibitors of HCV infection had motivated us to test our recently produced compounds as probable new HCV_ NS3 inhibitors. A mixture of each powerful compound with acyclovir (reference drug), in a 1:1 ratio, was also synthesized. Results were counted as IC50.The comparison of the three compounds 8c, 8d, and 10c against the standard drug Sovaldi showed that the three compounds had a potent inhibitory effect toward HCV_ NS3.
4.3.2. Ubiquitin-Specific Protease (USP7) Assay
This assay was performed by means of the USP7 inhibitor run assay kit, BPS Bioscience, Catalog 79256. The USP7 assay kit was used to perform the assay for the inhibitory result of the new compounds on the USP7 enzyme. The 42USP7 supplied with the kit is human USP7. Herpes simplex virus recombinant stated. In the beginning, stabilization of the enzyme was carried out by adding 13 μL of 0.5 M DTT. By diluting 5X USP7 test buffer in purified water, store solutions of the ubiquitin particular protease7 (1X USP7) assay buffer were prepared. Afterward, the Ub-AMC substrate was weakened 400 times in a 1X USP7 assay barrier before adding 20 μL to each well labeled “Check Inhibitor,” “Positive Control,” and “Blank.” A total of 5 μL of inhibitor solution was added to “Check Inhibitor,” “Positive Control,” and ″Blank,″ followed by 5 μL of the same solution without inhibitor (inhibitor buffer). Following that, 25 μL of the 1X USP7 assay barrier was added to the well labeled ″Blank.″ Following that, 25 μL of 1X USP7 assay buffer was added to the well labeled “Blank”. The reaction was started by adding 25 μL of diluted USP7 enzyme, 0.4 ng/μL (10 ng/25 μL) with 1x USP7 assay buffer, to the wells labeled “Check Inhibitor” and “Positive Control”. The fluorescence power was directly delivered in a microtiter plate or luminometer reader, which is skilled of reading at 460 nm emissions and 360 nm excitation, after 30 min of incubation on a rotating platform at room temperature. The value “Blank” was subtracted from all readings.
4.4. Anticancer
Among the prepared compounds, 13 compounds were selected for in vitro anticancer action evaluation. Anticancer assays were made according to the US NCI protocol based on the processes of the assay defined at http://www.dtp.nci.nih.gov. The tested compounds were evaluated counter to a panel of 60 cancer cell appearances resulting from various tumors, such as colon, leukemia, lung, melanoma, ovarian, breast, prostate, CNS, and renal cancer. The compounds were left with the cells for 48 h at a concentration of 10–5 M, and cell density was valued using sulforhodamine B (SRB), a protein binding dye. The outcomes were expressed as the growing % (G %) of the cells tested with each compound in comparison to the cells tested with vehicle and in relation to the amount of cells existing when the compound handling began.
Supporting Information Available
The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsomega.2c03444.
13C NMR, 1H NMR, and IR spectra of prepared compounds; tables of the antiviral activities of new compounds against HAV HM 175, COB4, HSV-1, HCV genotype 4a, and HAdV7; and figures of growth percent (G %) of the synthesized compounds (PDF)
The authors declare no competing financial interest.
Supplementary Material
References
- Zheng X. J.; Li C. S.; Cui M. Y.; Song Z. W.; Bai X. Q.; Liang C. W.; Wang H. Y.; Zhang T. Y. Synthesis, biological evaluation of benzothiazole derivatives bearing a 1,3,4-oxadiazole moiety as potential anti-oxidant and anti-inflammatory agents. Bioorg. Med. Chem. Lett. 2020, 30, 127237. 10.1016/j.bmcl.2020.127237. [DOI] [PubMed] [Google Scholar]
- Jaiswal S.; Verma N. K. Benzothiazole moiety with sulphonamide as anti-inflammatory and analgesic activity. Rev. Abstr. 2021, 4142, 90–102. [Google Scholar]
- Kumar G.; Singh N. P. Synthesis, anti-inflammatory and analgesic evaluation of thiazole/oxazole substituted benzothiazole derivatives. Bioorg. Chem. 2021, 107, 104608. 10.1016/j.bioorg.2020.104608. [DOI] [PubMed] [Google Scholar]
- Ballari M. S.; Herrera Cano N.; Wunderlin D. A.; Feresin G. E.; Santiago A. N. One-pot sequential synthesis and antifungal activity of 2-(benzylsulfonyl)benzothiazole derivatives. RSC Adv. 2019, 9, 29405–29413. 10.1039/c9ra04488d. [DOI] [Google Scholar]
- Azzam R. A.; Elsayed R. E.; Elgemeie G. H. Design, synthesis, and antimicrobial evaluation of a new series of N-sulfonamide 2-pyridones as dual inhibitors of DHPS and DHFR enzymes. ACS Omega 2020, 5, 10401–10414. 10.1021/acsomega.0c00280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azzam R. A.; Elsayed R. E.; Elgemeie G. H. Design and synthesis of a new class of pyridine-based n-sulfonamides exhibiting antiviral, antimicrobial, and enzyme inhibition characteristics. ACS Omega 2020, 5, 26182–26194. 10.1021/acsomega.0c03773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elsayed R. E.; Madkour T. M.; Azzam R. A. Tailored-design of electrospun nanofiber cellulose acetate/poly(lactic acid) dressing mats loaded with a newly synthesized sulfonamide analog exhibiting superior wound healing. Int. J. Biol. Macromol. 2020, 164, 1984–1999. 10.1016/j.ijbiomac.2020.07.316. [DOI] [PubMed] [Google Scholar]
- Bhagdev K.; Sarkar S. B. As an antidiabetic agent. Ann. Romanian Soc. Cell Biol. 2021, 25, 20269–20285. [Google Scholar]
- Kamal A.; Syed M. A. H.; Mohammed S. M. Therapeutic potential of benzothiazoles: A patent review (2010-2014). Expert Opin. Ther. Pat. 2015, 25, 335–349. 10.1517/13543776.2014.999764. [DOI] [PubMed] [Google Scholar]
- Irfan A.; Batool F.; Zahra Naqvi S. A.; Islam A.; Osman S. M.; Nocentini A.; Alissa S. A.; Supuran C. T. Benzothiazole derivatives as anticancer agents. J. Enzyme Inhib. Med. Chem. 2020, 35, 265–279. 10.1080/14756366.2019.1698036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Er M.; Özer A.; Direkel Ş.; Karakurt T.; Tahtaci H. Novel substituted benzothiazole and imidazo[2,1-b][1,3,4]thiadiazole derivatives: Synthesis, characterization, molecular docking study, and investigation of their in vitro antileishmanial and antibacterial activities. J. Mol. Struct. 2019, 1194, 284–296. 10.1016/j.molstruc.2019.05.104. [DOI] [Google Scholar]
- Kale A.; Kakde R.; Pawar S.; Thombare R. Recent development in substituted benzothiazole as an anticonvulsant agent. Mini-Reviews Med. Chem. 2020, 21, 1017–1024. [DOI] [PubMed] [Google Scholar]
- Tariq S.; Kamboj P.; Amir M. Therapeutic advancement of benzothiazole derivatives in the last decennial period. Arch. Pharm. (Weinheim). 2019, 352, e1800170–17. 10.1002/ardp.201800170. [DOI] [PubMed] [Google Scholar]
- Azzam R. A.; Elgemeie G. H.; Osman R. R. Synthesis of novel pyrido[2,1-b]benzothiazole and N-substituted 2-pyridylbenzothiazole derivatives showing remarkable fluorescence and biological activities. J. Mol. Struct. 2020, 1201, 127194. 10.1016/j.molstruc.2019.127194. [DOI] [Google Scholar]
- Asiri Y. I.; Alsayari A.; Muhsinah A. B.; Mabkhot Y. N.; Hassan M. Z. Benzothiazoles as potential antiviral agents. J. Pharm. Pharmacol. 2020, 72, 1459–1480. 10.1111/jphp.13331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azzam R. A.; Elboshi H. A.; Elgemeie G. H. Novel synthesis and antiviral evaluation of new benzothiazole-bearing n-sulfonamide 2-pyridone derivatives as USP7 enzyme inhibitors. ACS Omega 2020, 5, 30023–30036. 10.1021/acsomega.0c04424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arasappan A.; Bennett F.; Girijavallabhan V.; Huang Y.; Huelgas R.; Alvarez C.; Chen L.; Gavalas S.; Kim S. H.; Kosinski A.; Pinto P.; Rizvi R.; Rossman R.; Shankar B.; Tong L.; Velazquez F.; Venkatraman S.; Verma V. A.; Kozlowski J.; Shih N.-Y.; Piwinski J. J.; MacCoss M.; Kwong C. D.; Clark J. L.; Fowler A. T.; Geng F.; Kezar H. S.; Roychowdhury A.; Reynolds R. C.; Maddry J. A.; Ananthan S.; Secrist J. A.; Li C.; Chase R.; Curry S.; Huang H.-C.; Tong X.; Njoroge F. G. 5-Benzothiazole substituted pyrimidine derivatives as HCV replication (replicase) inhibitors. Bioorg. Med. Chem. Lett. 2012, 22, 3229–3234. 10.1016/j.bmcl.2012.03.036. [DOI] [PubMed] [Google Scholar]
- Azzam R. A.; Osman R. R.; Elgemeie G. H. Efficient synthesis and docking studies of novel benzothiazole-based pyrimidinesulfonamide scaffolds as new antiviral agents and hsp90α inhibitors. ACS Omega 2020, 5, 1640–1655. 10.1021/acsomega.9b03706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jha K. K.; Kumar S.; Tomer I.; Mishra R. Thiophene: The molecule of diverse medicinal importance. J. Pharm. Res. 2012, 5, 560–566. [Google Scholar]
- Ke S.; Wei Y.; Yang Z.; Wang K.; Liang Y.; Shi L. Novel cycloalkylthiophene-imine derivatives bearing benzothiazole scaffold: Synthesis, characterization and antiviral activity evaluation. Bioorg. Med. Chem. Lett. 2013, 23, 5131–5134. 10.1016/j.bmcl.2013.07.023. [DOI] [PubMed] [Google Scholar]
- Ohba M.; Oka T.; Ando T.; Arahata S.; Ikegaya A.; Takagi H.; Ogo N.; Owada K.; Kawamori F.; Wang Q.; Saif L. J.; Asai A. Discovery and synthesis of heterocyclic carboxamide derivatives as potent anti-norovirus agents. Chem. Pharm. Bull. 2016, 64, 465–475. 10.1248/cpb.c16-00001. [DOI] [PubMed] [Google Scholar]
- Abu-Zaied M. A.; Elgemeie G. H.; Mahmoud N. M. Anti-Covid-19 Drug analogues: Synthesis of novel pyrimidine thioglycosides as antiviral agents against SARS-COV-2 and avian influenza H5N1 viruses. ACS Omega 2021, 6, 16890–16904. 10.1021/acsomega.1c01501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abu-Zaied M. A.; Loutfy S. A.; Hassan A. E.; Elgemeie G. H. Novel purine thioglycoside analogs: Synthesis, nanoformulation and biological evaluation in in vitro human liver and breast cancer models. Drug Des. Devel. Ther. 2019, Volume 13, 2437–2457. 10.2147/dddt.s201249. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Elgemeie G.; Abu-Zaied M.; Azzam R. Antimetabolites: A first synthesis of a new class of cytosine thioglycoside analogs. Nucleosides, Nucleotides Nucleic Acids 2016, 35, 211–222. 10.1080/15257770.2015.1127961. [DOI] [PubMed] [Google Scholar]
- Abu-Zaied M. A.; Mahmoud N. M.; Elgemeie G. H. Toward developing therapies against Corona virus: Synthesis and anti-avian influenza virus activity of novel cytosine thioglycoside analogues. ACS Omega 2020, 5, 20042–20050. 10.1021/acsomega.0c01537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Abu-Zaied M. A.; Hammad S. F.; Halaweish F. T.; Elgemeie G. H. Sofosbuvir thio-analogues: Synthesis and antiviral evaluation of the first novel pyridine- and pyrimidine-based thioglycoside phosphoramidates. ACS Omega 2020, 5, 14645–14655. 10.1021/acsomega.0c01364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang G. L.; Gadi M. R.; Cui X.; Liu D.; Zhang J.; Saikam V.; Gibbons C.; Wang P. G.; Li L. Protecting-group-freeS-glycosylation towards thioglycosides and thioglycopeptides in water. Green Chem. 2021, 23, 2907–2912. 10.1039/d1gc00098e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen P.; Větvička V.; Crich D. Synthesis and evaluation of oligomeric thioether-linked carbacyclic β-(1?3)-glucan mimetics. J. Org. Chem. 2019, 84, 5554–5563. 10.1021/acs.joc.9b00504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Compañón I.; Guerreiro A.; Mangini V.; Castro-López J.; Escudero-Casao M.; Avenoza A.; Busto J. H.; Castillón S.; Jiménez-Barbero J.; Asensio J. L.; Jiménez-Osés G.; Boutureira O.; Peregrina JM; Hurtado-Guerrero R.; Fiammengo R.; Bernardes GJL; Corzana F. Structure-based design of potent tumor-associated antigens: Modulation of peptide presentation by single-atom O/S or O/Se substitutions at the glycosidic linkage. J. Am. Chem. Soc. 2019, 141, 4063–4072. 10.1021/jacs.8b13503. [DOI] [PubMed] [Google Scholar]
- Scott K. A.; Njardarson J. T.. Analysis of US FDA-Approved Drugs Containing Sulfur Atoms. Top. Curr. Chem. 2018, 376. 10.1007/s41061-018-0184-5 [DOI] [PubMed] [Google Scholar]
- Azzam R. A.; Elgemeie G. H.; Gad M.; Jones P. G. (4-bromobenzoyl)-3-(benzo[d]thiazol-2-yl)-2-[(20,30,40,60-tetra-O-acetyl-b-D-galactopyranosyl) sulfanyl]thiophene. IUCrData 2022, 7, x220412. 10.1107/s2414314622004126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simões C. M. O.; Amoros M.; Girre L. Mechanism of antiviral activity of triterpenoid saponins. Phyther. Res. 1999, 13, 323–328. [DOI] [PubMed] [Google Scholar]
- Walum E.Understanding cell toxicology; Ellis Horwood, 1990. [Google Scholar]
- Schmidtke M.; Knorre C.; Blei L.; Stelzner A.; Birch-Hirschfeld E. Penetration and antiviral activity of coxsackievirus B3 (CVB3)-specific phosphorothioate oligodeoxynucleotides (PS-ODN). Nucleosides and Nucleotides 1998, 17, 1557–1566. 10.1080/07328319808004686. [DOI] [Google Scholar]
- Miura T.; Fukami T. A.; Hasegawa K.; Ono N.; Suda A.; Shindo H.; Yoon D. O.; Kim S. J.; Na Y. J.; Aoki Y.; Shimma N.; Tsukuda T.; Shiratori Y. Lead generation of heat shock protein 90 inhibitors by a combination of fragment-based approach, virtual screening, and structure-based drug design. Bioorg. Med. Chem. Lett. 2011, 21, 5778–5783. 10.1016/j.bmcl.2011.08.001. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.