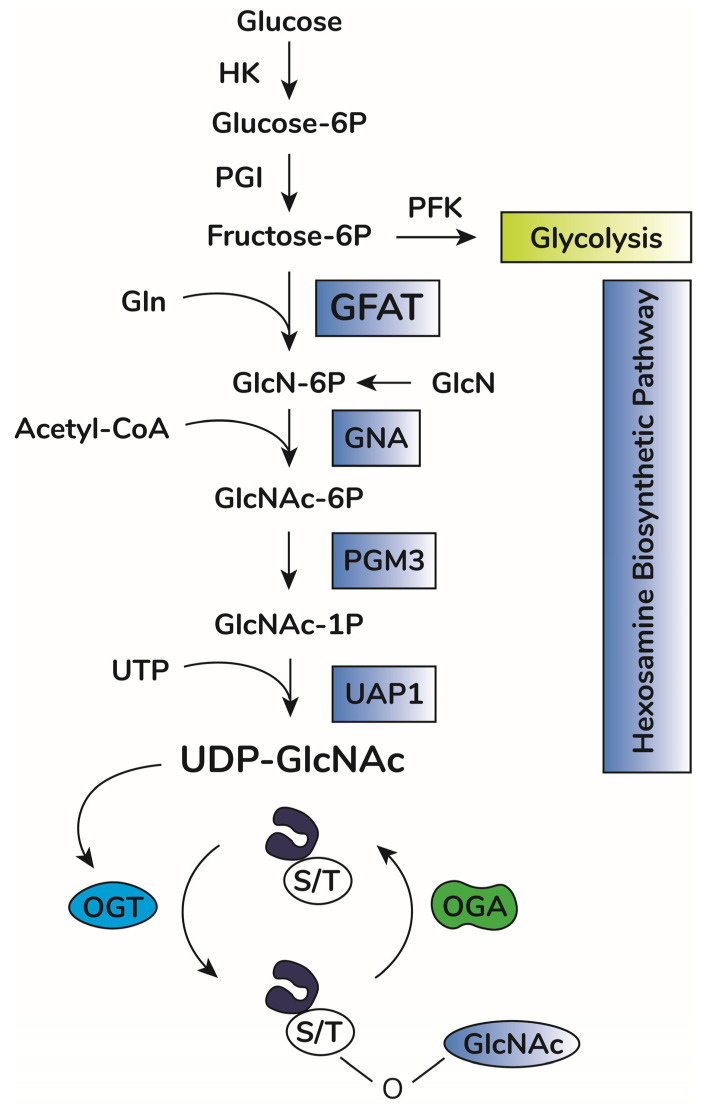
Figure 1.
O-GlcNAc cycling scheme. Upon entry in cells, glucose is rapidly phosphorylated to glucose-6-phosphate (Glucose-6P) by hexokinase (HK). Glucose-6P is isomerized by phosphoglucose isomerase (PGI), producing fructose-6-phosphate (Fructose-6P), a substrate for both phosphofructokinase (PFK) of the glycolytic pathway or glucosamine-fructose amino transferase (GFAT), the rate-limiting reaction of the hexosamine biosynthetic pathway (HBP). GFAT requires glutamine (Gln) as the amine donor for generating glucosamine-6-phosphate (GlcN-6P), which is then N-acetylated by glucosamine-6-phosphate N-acetyltransferase (GNA1), producing N-acetyl-glucosamine-6-phosphate (GlcNAc-6P). This step requires acetyl-CoA as the acetyl donor. GlcNAc-6P is transformed in GlcNAc-1P by phosphoacetylglucosamine mutase (PGM3). Using UTP as the nucleotide donor, UDP-N-acetylglucosamine pyrophosphorylase (UAP1) produces uridine-diphosphate N-acetyl glucosamine (UDP-GlcNAc). This molecule is the substrate of O-GlcNAc transferase (OGT) for protein O-GlcNAcylation by adding O-linked GlcNAc moieties at serine or threonine residues of target proteins. O-GlcNAcase (OGA) removes GlcNAc residues, counteracting OGT activity.