Abstract
Cerebrospinal fluid (CSF) biomarkers, namely total tau, phospho-tau and amyloid beta peptides, have received much attention specifically regarding Alzheimer’s disease (AD), since they can detect the biochemical fingerprint of AD and serve as a diagnostic tool for accurate and early diagnosis during life. In the same way, biomarkers for other neurodegenerative disease pathologies are also needed. We present a case series of six patients with genetic frontotemporal dementia (FTD), with TDP-43 underlying proteinopathy, in an attempt to assess TDP-43 as a novel biomarker alone and in combination with established AD biomarkers for this specific patient group, based on the principles of personalized and precision medicine. Our results indicate that genetic TDP-43-FTD is characterized by increased CSF TPD-43 and increased TDP-43 × τΤ/τP-181 combination. Hence, TDP-43 combined with tau proteins could be a useful tool for the diagnosis of genetic FTD with TDP-43 underling histopathology, supplementing clinical, neuropsychological and imaging data.
Keywords: frontotemporal dementia, genetic FTD, TDP-43 proteinopathy, cerebrospinal fluid, biomarkers, C9orf72, GRN, VCP
1. Introduction
A common neuropathological feature of neurodegenerative diseases is cytoplasmic protein aggregates. Cytoplasmic mislocalization and aggregation of TAR-DNA binding protein 43 (TDP-43) is found in the vast majority of patients with amyotrophic lateral sclerosis (ALS), and in approximately half of patients with frontotemporal dementia (FTD) with ubiquitin inclusions [1].
Recently, great progress in the field of neurogenetics has resulted in the recognition of several gene mutations related to the FTD-ALS spectrum, especially with ubiquitin-TDP-43 predominant histopathology, thus highlighting a new brain proteinopathy. The most recent, but more common discovery, is a genetic mutation in the C9orf72 gene involving a hexanucleotide repeat expansion (GGGGCC) in the first non-coding intron of the gene, responsible for both familial and sporadic FTD and ALS, but more frequently for the combined FTD-ALS phenotype [2,3,4,5].
Heterozygous loss of function mutations in the progranulin gene (GRN) can also lead to FTD with TDP-43 inclusions [6]. GRN mutations have been linked to FTD phenotypes, particularly those of the primary progressive aphasia (PPA) subtype. Mutations in the valosin-containing protein (VCP) gene constitute another rare cause of FTD, which has been recognized to present with the clinical syndrome of “IBMPFD” (inclusion body myopathy, Paget’s disease of bone and FTD), often in combination with ALS in certain families. [7]. Mutations in the gene encoding the TDP-43 protein (TARDBP) are also an extremely rare cause of the FTD phenotype [8].
In recent years, cerebrospinal fluid (CSF) has proven to be an excellent material for biomarker discovery for neurodegenerative diseases [9]. Due to its close proximity to the brain parenchyma, CSF reflects the neurochemical changes taking place in the brain. This approach has been successful in the case of Alzheimer’s disease (AD), where tau proteins, total (τT) and phosphorylated (τP), as well as amyloid-β peptides (Aβ) expressing the pathological features of the disease, have been recognized as biomarkers with high diagnostic accuracy and are now included in research diagnostic criteria for AD [10,11,12,13,14,15]. The combination of increased τT, tau phosphorylated at the threonine 181 site (τP-181) and decreased Aβ with 42 amino acids (Aβ42) in the CSF, has currently been established as the biochemical signature of the disease in vivo [16].
Accordingly, the detection of TDP-43 could be a specific biomarker for TDP-43 proteinopathy. Attempts to quantify TDP-43 in CSF have been successful [17,18,19,20]. However, until now, TDP-43 seemed to lack adequate sensitivity and specificity in order to serve as a meaningful biomarker alone [21]. The combination of TDP-43 with the aforementioned AD biomarkers in the formula TDP-43 × τΤ/τP-181 may increase sensitivity and specificity (>80%) [18], while the τP-181/τT ratio has been proposed as an indirect indicator of a non-tauopathic pathology [22,23].
Since almost all the aforementioned studies lack pathologic confirmation, we isolated and present herein a case series of six patients with genetic FTD and a presumed TDP-43 underlying proteinopathy. We aimed to investigate how the above-mentioned biomarkers perform in a personalized setting, based on the principles of a precision medicine clinical approach. We also include the clinical and imaging data of this group of patients.
2. Materials and Methods
A total of six patients were included in this case series study. All patients were hospitalized at the 1st Department of Neurology of the National and Kapodistrian University of Athens at Eginition Hospital during the years 2011–2018. The study was performed in accordance with the ethical guidelines of the Declaration of Helsinki, and had the approval of the local Ethical and Deontology committee of our hospital (244/5-7-2010). All study subjects and/or their relatives gave informed consent for inclusion in the study.
Patients underwent detailed clinical, neuropsychological, biochemical and neuroimaging examination, to exclude secondary causes of dementia and establish an FTD diagnosis, based on the criteria of either Rascovsky et al. [24] for probable bvFTD or Gorno-Tempini et al. [25] for PPA. The Mini-Mental State examination (MMSE) and the Frontal Assessment Battery (FAB) were used as crude estimates of dementia severity [26,27].
The CSF levels of τT, τP-181 and Aβ42 and Aβ40 were measured in duplicate by commercially available enzyme-linked immunosorbent assay (ELISA) kits (“Innotest hTau antigen”, “phospho-tau181”, “β-amyloid1–42” and “β-amyloid1–40”, respectively, Fujirebio, Gent, Belgium) according to manufacturer’s instructions, and blind to clinical diagnosis. The above biomarkers are measured routinely in the Neurochemistry and Biomarker Unit of our department, as part of an everyday diagnostic approach, as described elsewhere [23]. TDP-43 was measured in triplicate by double sandwich ELISA, using a commercially available kit (Human TAR DNA-binding protein 43 ELISA kit, Cusabio Biotech Co., Ltd., Wuhan, China) according to manufacturer’s instructions, as described elsewhere [18]. The diagnostic performance of TDP-43 was assessed as a biomarker with molecular specificity in patients with an individualized FTD diagnosis, alone and combined with established AD biomarkers (mainly for excluding AD-related dementia). Cut-offs for normal values are shown in Table 1.
Table 1.
| CSF Biomarker | Cut-off Values * |
|---|---|
| total tau protein (τT) | <376 pg/mL |
| tau phosphorylated at threonine-181 (τP-181) | <57 pg/mL |
| amyloid-β peptide with 42 amino acids (Aβ42) | >580 pg/mL |
| Aβ42/Aβ40 | >0.067 |
| τP-181/τT | >0.165 |
| TDP-43 | <3.73 pg/mL |
| TDP-43 × τΤ/τP-181 | <20.5 |
* From Paraskevas et al., 2017; Bourbouli et al., 2017.
3. Results
3.1. Patients
3.1.1. Case 1
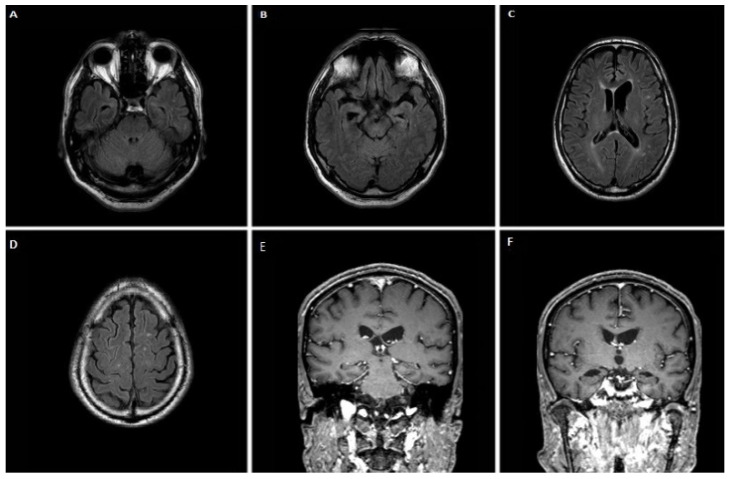
A 61-year-old female patient was admitted to our department for progressive language impairment over the past 1–2 years. She had word-finding difficulty, with intact comprehension, an absence of phonological speech or grammar errors, no other significant cognitive/behavioral disturbances, and preserved activities of daily living. Neuropsychological assessment revealed mild cognitive impairment, with an MMSE score of 25/30 and a FAB score of 12/18. Her writing skills and phrase and sentence repetition were preserved, too. On magnetic resonance imaging (MRI), moderate cortical atrophy in the frontal and temporal lobes, with left predominance and relative preservation of the hippocampus, was apparent (Figure 1). CSF biomarker analysis revealed increased τT = 511.6 pg/mL, normal τP-181 = 28.7 pg/mL, decreased Aβ42 = 504.2 pg/mL, but normal Aβ42/Aβ40 = 0.1 and decreased τP-181/τT = 0.056 ratio. TDP-43 = 4.46 pg/mL and the formula TDP-43 × τΤ/τP-181 = 79.5 were increased. Genetic testing revealed a c.463-2A > G splice site variant—heterozygous mutation in the GRN gene. The patient also had more than a 2-fold decrease in plasma progranulin levels, as described elsewhere [28].
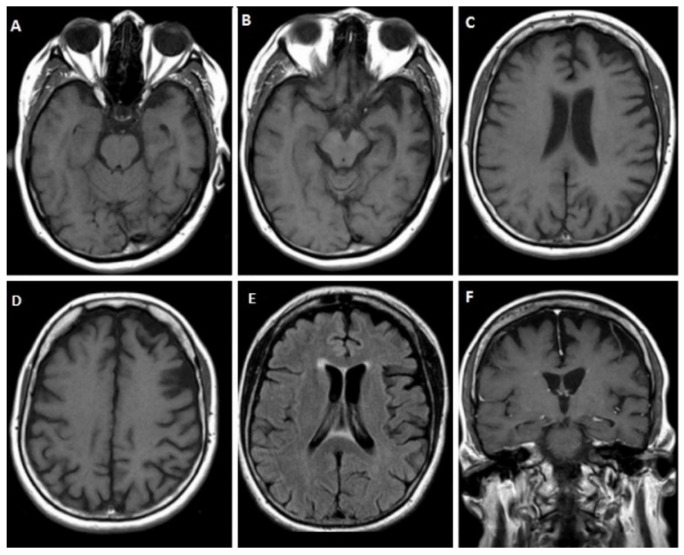
Figure 1.
Brain MRI of case 1. T1 sequence (A–D) and fluid attenuated inversion recovery (FLAIR) (E) showing cortical atrophy in the frontal and temporal lobes, with left hemisphere being more affected, as well as coronal T1 section (F) showing relative preservation of the hippocampus.
3.1.2. Case 2
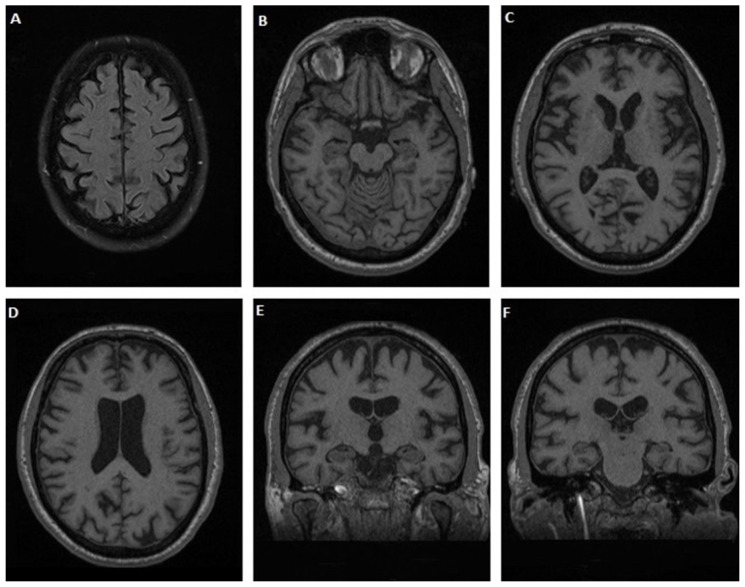
A 60-year-old male patient, with a family history of dementia on his mother and maternal grandmother’s side, presented with language impairment over the last 10 years. Initially, he had difficulty naming objects and writing. Three years later he developed effortful speech with sound errors, decreased fluency, and memory complaints and disorientation. At the age of 55, a dysexecutive syndrome as well as behavioral disturbances (overeating and irritability) appeared. Gradually, he became almost mute, while bradykinesia and tremor on the right hand appeared. At the time of his admission, he was completely mute, and unable to eat. Neurological examination revealed diffuse muscle atrophies without fasciculations, bilateral Parkinsonian syndrome (more severe in the right limbs), pyramidal syndrome with spasticity, hyperreflexia, and Babinski sign bilaterally. Neuropsychological assessment was impossible to perform. MRI performed at the age of 57 revealed significant cortical atrophy in the frontal and temporal lobes with left predominance (Figure 2A–C), with further deterioration three years later, as shown in (Figure 2D–F). CSF biomarker analysis revealed increased τT = 565.12 pg/mL, normal τP-181 = 41.9 pg/mL, Aβ42 = 640 pg/m and Aβ42/Aβ40 = 0.11, while τP-181/τT = 0.074, was decreased. TDP-43 = 5.92 pg/mL and formula TDP-43 × τΤ/τP-181 = 79.9 were increased. Genetic testing revealed a c.934-1G > A splice site variant—heterozygous mutation on the GRN gene, and similarly, a decrease in plasma progranulin levels, as in case 1.
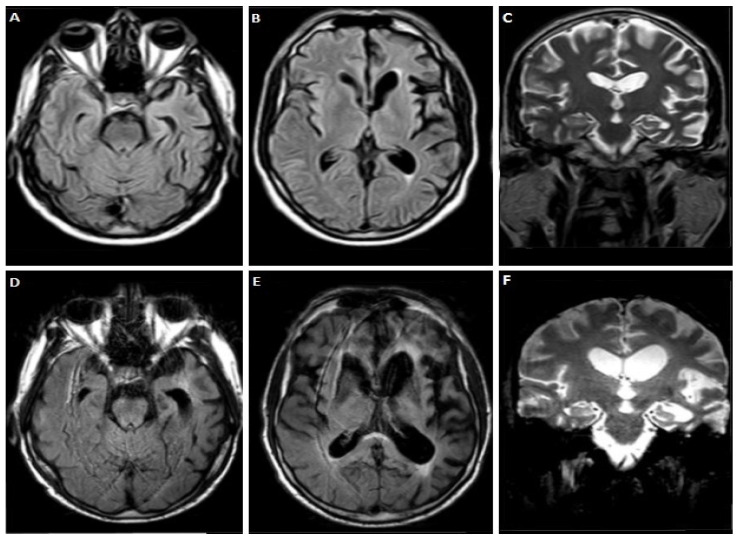
Figure 2.
Brain MRI of case 2 at age 57 (A–C) and 60 (D–F) showing some degree of cortical atrophy in frontal and temporal lobes was observed with left predominance that deteriorated significantly three years later.
3.1.3. Case 3
A 72-year-old male patient, with a family history of motor neuron disease (brother), gradually developed language (phonological speech errors, impaired comprehension, inability to write, severe difficulty in naming objects and reduced phonemic and categorical fluency) and memory impairment, bradykinesia and stereotypical movements of the right hand over the last two years. Three months before admission, his symptoms aggravated. Neurological examination revealed bilateral akinetic-rigid Parkinsonian syndrome, mild pyramidal signs (mainly on the right lower limb with hyperreflexia and extensor plantar response) and apraxia. Neuropsychological assessment revealed severe diffuse cognitive deficits (MMSE score 5/30, FAB score 1/18). On MRI, severe cortical atrophy in frontal and temporal lobes was observed (Figure 3). CSF biomarker analysis revealed normal τT = 316.16 pg/mL and τP-181 = 27.2 pg/mL, decreased Aβ42 = 501.51 pg/mL but normal Aβ42/Aβ40 = 0.14 and low τP-181/τT = 0.086 ratio. TDP-43 levels were 5.74 pg/mL and TDP-43 × τΤ/τP-181 = 66.7, both increased. The genetic testing revealed a pathologic hexanucleotide repeat expansion in the C9orf72 gene.
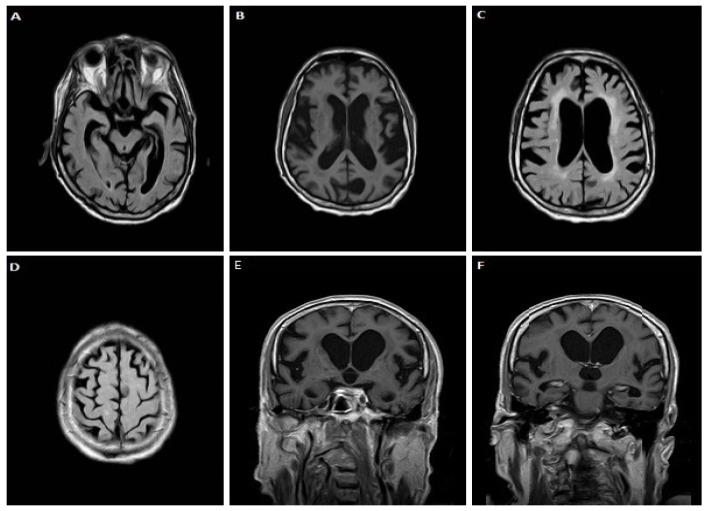
Figure 3.
Brain MRI of case 3. FLAIR images (A,C,D) show severe cortical atrophy in frontal and temporal lobes. In axial (B) and coronal T1 section (E,F) atrophy of the perisylvian and parietal areas is also evident.
3.1.4. Case 4
A 54-year-old female patient, with a medical history of depression with psychotic features was referred for evaluation. She had a family history of dementia (mother) and symptoms of motor neuron disease (two maternal uncles). The patient presented with apathy, inertia, disinhibition, loss of empathy and sympathy, unusual stereotyped and perseverative behaviors, and hyperorality with overeating of carbohydrates during the last two and a half years. One and a half year before her admission, she also developed inappropriate crying and a language impairment. Neurological examination revealed severely impaired non-fluent speech with single-word answers, severe disorientation in time and space, and frontal gait disorder. Neuropsychological assessment revealed moderate cognitive deficits with an MMSE score of 15/30 and a FAB score of 6/18. On MRI, some T2 hyperintense lesions attributed to chronic small vessel ischemia (Fazekas II), and mild cortical atrophy of the temporal lobes was observed (Figure 4). CSF biomarker analysis revealed normal τT = 250.3 pg/mL, τP-181 = 35.45 pg/mL and Aβ42 = 935.95 pg/mL, while τP-181/τT ratio was 0.14. The levels of TDP-43 = 2.48 pg/mL and TDP-43 × τΤ/τP-181 = 17.5 were also normal. Genetic testing, however, revealed a hexanucleotide repeat expansion in the C9orf72 gene.
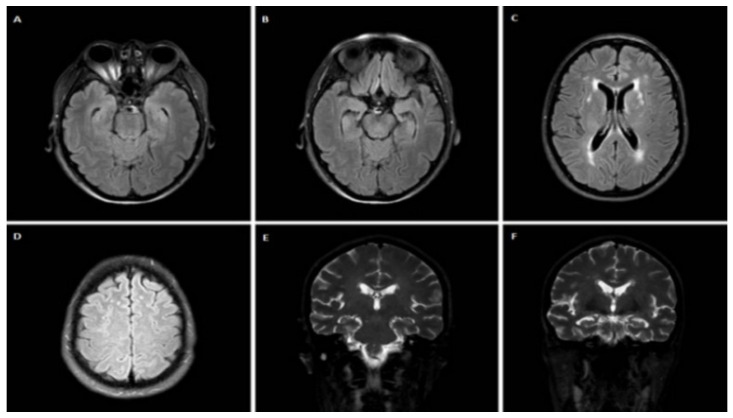
Figure 4.
Brain MRI of case 4. Axial FLAIR images (A–D) and coronal T2 images (E,F) reveal hyperintense lesions attributed to chronic small vessel ischemia (Fazekas II) and some degree of cortical atrophy in temporal lobes.
3.1.5. Case 5
A 62-year-old male patient, with a medical history of arterial hypertension and an operation for a herniated lumbar disc at the L4-L5 level, was referred for evaluation of neuropsychiatric symptoms. He had a family history of “presenile dementia” in his mother, ALS of his older brother, who died at the age of 53, and behavioral symptoms of another brother. The patient presented with progressively deteriorating steppage gait 20 years ago. A year before admission, the patient was unable to walk independently. Additionally, he developed obsessive behavior, aggression, palilalia and apathy. His neuropsychological evaluation revealed severe cognitive impairment, suggestive of predominant frontal lobe dysfunction, MMSE: 2 and FAB: 0 (not applicable). Brain MRI showed mild frontal lobe atrophy (Figure 5). EMG showed diffuse myopathic findings and mild spontaneous activity. Due to the non-AD biomarker profile, the family history of dementia and ALS in members of consecutive generations, which is indicative of a possible autosomal dominant inheritance pattern and the clinical and laboratory findings suggesting myopathy, a high suspicion of a possible VCP-associated syndrome was raised. Genetic testing (DNA isolated from skeletal muscle) revealed a pathogenic heterozygous missense mutation p.R159H (c.476G > A) in the VCP gene, confirming the diagnosis of IBMPFD [29].
Figure 5.
Brain MRI of case 5. Axial FLAIR images (A–D) and coronal T1 images (E,F) reveal mild frontal lobe atrophy.
Lumbar puncture and CSF biomarker analysie were performed one year later and revealed normal τT = 356.75 pg/mL, τP-181 = 32.77 pg/mL, low Aβ42 = 431 pg/mL, but normal Aβ42/Aβ40 = 0.1 and reduced ratio τP-181/τT = 0.091, while TDP-43 levels = 5.27 pg/mL, and TDP-43 × τΤ/τP-181 = 57.3 were increased.
3.1.6. Case 6
A 41-year-old male patient, with a medical history of dyslipidemia was admitted to the neurology department for delusions of religious content over the past 12 years. Moreover, apathy, a change in eating habits with carbohydrate craving and weight gain gradually developed, while dysarthria, executive deficits, stereotypical behaviors and inappropriate laughter appeared over the last two years. Finally, difficulty in swallowing liquids was noticed six months before admission. Neurological examination revealed pseudobulbar dysarthria, an absence of pharyngeal reflex, bilateral pyramidal syndrome and fasciculations in tongue and back muscles. Neuropsychological assessment revealed mild to moderate frontal cognitive deficits with an MMSE score 28/30, and a FAB score 15/18. Neurophysiological examination could not confirm lower motor neuron involvement. On MRI, significant cortical atrophy in frontal, parietal and temporal lobes was observed (Figure 6). CSF biomarker analysis revealed normal τT = 328.05 pg/mL, τP-181 = 32.07 pg/mL, decreased Aβ42 = 397.1 pg/mL, with normal ratio Aβ42/Aβ40 = 0.17 and decreased τP-181/τT = 0.098, while TDP-43 levels 5.74 pg/mL and the formula TDP-43 × τΤ/τP-181 = 57.66 were increased. Genetic testing finally revealed a hexanucleotide repeat expansion in the C9orf72 gene.
Figure 6.
Brain MRI of case 6. Axial (A–D) and coronal (E,F) T1 images showing significant cortical atrophy in frontal, parietal and temporal lobes.
Table 2 summarizes the CSF biomarker profile for all cases. All cases had normal τP-181 and only two out of six cases had increased τT. Four out of six cases had decreased Aβ42; however, all had a normal Aβ42/Aβ40 ratio. Five out of six patients had increased TDP-43 and formula (TDP-43 × τΤ/τP-181) values, while τP-181/τT was abnormal in all.
Table 2.
CSF biomarker levels in the presented cases.
| Case | τT (pg/mL) | τP-181 (pg/mL) | Aβ42 (pg/mL) | Aβ42/Aβ40 | τP-181/τT | TDP-43 (pg/mL) | Formula (f) |
|---|---|---|---|---|---|---|---|
| 1 | 511.6 | 28.7 | 504.2 | 0.10 | 0.056 | 4.46 | 79.5 |
| 2 | 565.1 | 41.9 | 640 | 0.11 | 0.074 | 5.92 | 79.9 |
| 3 | 316.2 | 27.2 | 501.5 | 0.14 | 0.086 | 5.74 | 66.7 |
| 4 | 250.3 | 35.5 | 935.9 | - | 0.140 | 2.48 | 17.5 |
| 5 | 356.8 | 32.8 | 431 | 0.10 | 0.091 | 5.27 | 57.3 |
| 6 | 328.1 | 32.1 | 397.1 | 0.17 | 0.098 | 5.74 | 57.7 |
τT: total tau protein; τP-181: tau phosphorylated at threonine-181; Aβ42: amyloid-β peptide with 42 amino acids; Aβ40: amyloid-β peptide with 40 amino acids; f: TDP-43 × τΤ/τP-181; pathological values are highlighted in bold.
4. Discussion
Herein, we present six patients with clinical, neuropsychological and imaging data fulfilling the criteria of Rascovsky et al. [24] and Gorno-Tempini et al. [25] for probable FTD, in which, due to the genetic findings, patients were classified as definite FTLD with TDP-43 histopathology. Three of our cases had a pathogenic hexanucleotide repeat expansion in the C9orf72 gene; two had a novel mutation in the GRN gene which led to more than a 2-fold decrease of plasma progranulin levels, as expected [28,30]; meanwhile, one patient harbored a pathogenic mutation in the VCP gene. Although small in number, this group of patients is indicative of the most common mutations found in the Greek population [28,31].
In this group of well-characterized genetic FTLD patients, established CSF biomarkers for AD diagnosis (τT, τP-181, Aβ42 and Aβ40) and a novel emerging (TDP-43) biomarker were analyzed. Combinations of these biomarkers as the formula TDP-43 × τΤ/τP-181 and the ratio τP-181/τΤ were also calculated, as previously suggested [18,22].
According to the AT(N) system proposed by the National Institute of Aging and Alzheimer’s Association (NIA–AA) Research Framework group, AD biomarkers are divided into those with molecular specificity for AD, namely τP-181, Aβ42 and Aβ42/Aβ40 ratio, and those with neurodegeneration, τT in the present case [15]. All presented cases had a biomarker profile of either A-T-(N+) or A-T-(N−) according to CSF data, compatible with a “non-Alzheimer’s pathological change” profile. Only one patient (case 4) had a “normal” biomarker profile.
In four out of the six presented cases, Aβ42 was reduced, but the use of Aβ42/Aβ40 ratio restored normality, ruling out amyloidopathy (A). The Aβ42/Aβ40 ratio has been suggested to better reflect the presence or absence of amyloid pathology than Aβ42 alone. Furthermore, the Aβ42/Aβ40 ratio correlates better with amyloid load in PET (positron emission tomography) [32], as it has been found to be reduced in some cases of FTD and vascular dementia (VD), but most commonly in dementia with Lewy bodies (DLB) [33,34,35,36].
The main findings of our study, however, were the increase of CSF TDP-43 levels (higher than the above cut-off value) in five out of six cases, and the increased values of the TDP-43 × τΤ/τP-181 formula. TDP-43 is a protein, which may be suggestive and specific of TDP-43 proteinopathy. Additionally, the τP-181/τT ratio was decreased, which in non-AD (FTD) patients could be considered as an indirect indication for a non-tauopathic pathology [22,23].
TDP-43 aggregation appears in approximately 50% of all FTLD cases (familial and sporadic) and in 97% of all ALS cases [37]. Mutations associated with TDP-43 histopathology have been documented for C9orf72, GRN, VCP, and TARDBP genes for FTLD and the FTD-ALS spectrum [38]. TDP-43 was initially considered as an intracellular/intranuclear protein; however, it is now recognized as an important protein for the existence and wellbeing of cells, through its various functions in RNA metabolism and homeostasis. Apart from FTLD, abnormalities in this protein’s homeostasis are associated with other severe neurodegenerative disorders [39]. Abnormal TDP-43 seems to cause neurotoxicity by various pathogenic processes, although the exact pathophysiological mechanisms that eventually lead to neurodegeneration remain unclear [1].
Although thought to exist as an intracellular/intranuclear protein, recent studies using ELISA and Western blot techniques suggest that TDP-43 can also be detected in extracellular fluids such as plasma and CSF [40].
The results of the present study indicate that CSF TDP-43 levels were increased in five out of six patients with TDP-43 histopathology-associated mutations. Case 4, with a C9orf72 pathological repeat expansion, represents an exception. Nonetheless, the proposed mechanisms through which C9orf72 mutations cause neurodegenerative changes are three: C9orf72 loss-of-function through haploinsufficiency, RNA toxic gain of function, and gain of function through the aggregation of toxic dipeptide repeat proteins [41]. The first two mechanisms cause neurodegeneration related to TDP-43 aggregation. The “gain of function” hypothesis due to the formation and accumulation of dipeptide repeats (DPRs) causes neurodegeneration via repeat-associated non-AUG (RAN) translation of the hexanucleotide GGGGCC from both sense and antisense strands [42]. As a result, there is a production of five distinct DPRs from sense {(poly-GA (glycine–alanine), poly-GP (glycine–proline), poly-GR (glycine–arginine) and antisense poly-GP (glycine–proline), as well as from poly-PR (proline–arginine) and poly-PA (proline–alanine)} strands [43,44]. These DPRs are amyloidogenic, and accumulate in different parts of the central nervous system without TDP-43 aggregation [43,44,45]. The pathogenic contribution of DPR-associated toxicity to disease progression remains unknown. As there is no aggregated TDP-43 in neuronal cytoplasmic inclusions in such patients, we can assume that the TDP-43 concentration in their CSF would be normal. This reasoning provides an explanation for the normal values of TDP-43 and formula in case 4.
TDP-43 abnormalities have also been observed in other neurodegenerative diseases as well, mainly in ALS, the other end of the ALS-FTD spectrum, but also in AD, limbic-predominant age-related TDP-43 encephalopathy (LATE), cerebral age-related TDP-43 with sclerosis (CARTS), and some other even rarer conditions [27,46,47]. Reports on CSF TDP-43 levels are mostly derived from patient cohorts of sporadic FTD cases, with inadequate (74%) clinicopathological concordance rates [48], resulting in relatively low sensitivities and specificities vs. controls. Thus, we enrolled genetic cases in order to reach safer conclusions. Sensitivities and specificities compared with other dementing disorders would have to be investigated as well.
There are also other biomarkers that can be used to differentiate FTD patients from patients with other neurodegenerative diseases, such as NfL [49]. However, NfL, total tau protein and the specific patterns of atrophy, are considered biomarkers of neurodegeneration in general [15]. The aim of the current study was to assess the diagnostic value of CSF TDP-43 protein as a biomarker with molecular specificity for FTD. Therefore, we combined TDP-43 with established AD biomarkers only, mainly to exclude AD, which often enters into a FTD differential diagnosis.
The current study has certain limitations. TDP-43 pathology can only be presumed from genetic mutations, since it is an ante-mortem study. Another disadvantage is the small number of patients, coming from a single-center study that included only patients of Greek origin; however, the mutations that the patients harbored are indicative of the most commonly observed in TDP-43-related FTLD found in the Greek population [28,31].
Our study also has some important advantages. We describe patients that have undergone extended clinical, neuropsychological and imaging examinations, fulfilling the criteria of Rascovsky et al. [24] and Gorno-Tempini et al. [25] for probable FTD, while they are classified as definite FTLD with TDP-43 underlying pathology, due to the genetic findings. This homogeneous genetic FTD cohort may serve as a very good example of how relatively safe conclusions may be reached through personalized and precision medicine. Further studies with larger patient cohorts are nevertheless required to corroborate our results.
5. Conclusions
Based on the results of the present personalized case series study, which is nonetheless indicative of the mutational profile of the population of Greece, the following conclusions can be drawn:
Established AD biomarkers are reliable indicators for the exclusion of AD pathology in genetic FTLD.
A decreased τP-181/τΤ ratio may be useful as an indirect marker for non-tauopathic pathology.
TDP-43 alone and combined with tau proteins, could be a useful tool for the diagnosis of genetic FTD patients with TDP-43 underlying pathology.
Future studies with larger and well characterized patient cohorts are needed to further explore the diagnostic potential of these biomarkers in differentiating TDP-43 proteinopathy from non-TDP-43-related pathologies.
Acknowledgments
We would like to thank the Neurogenetics Unit of the 1st Department of Neurology of NKUA and especially G. Koutsis, G. Karadima and C. Kartanou for performing the genetic analysis regarding the three cases with C9orf72 mutation. We would also like to thank J. Zaganas and the patients and their caregivers for their participation.
Author Contributions
Conceptualization, E.K., E.-S.P., F.B., M.B. and G.P.P.; methodology, F.B., E.-S.P., E.K., V.C.C. and G.P.P.; literature review, E.-S.P., E.K., V.C.C., C.A., F.B. and G.P.P.; data curation, M.B., V.C.C., E.-S.P., G.P.P. and E.K.; writing—original draft preparation, E.-S.P., G.P.P. and E.K.; writing—review and editing, E.K., E.-S.P., M.B., F.B., C.A., G.P.P. and V.C.C.; visualization, F.B., E.-S.P., M.B., V.C.C., C.A. and E.K. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Ethics Committee and the Scientific Board of “Eginition” Hospital (protocol code: 244, date of approval: 5-7-2010).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available upon reasonable request from the corresponding author. The data are not publicly available due to privacy restrictions.
Conflicts of Interest
G.P.P. receives fees from Biogen International as a consultant of the advisory board. The other authors declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding Statement
This research was partially funded by the “Lilian Voudouri Foundation” (grant number: 304/24-04-2017).
Footnotes
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Jo M., Lee S., Jeon Y.M., Kim S., Kwon Y., Kim H.J. The role of TDP-43 propagation in neurodegenerative diseases: Integrating insights from clinical and experimental studies. Exp. Mol. Med. 2020;52:1652–1662. doi: 10.1038/s12276-020-00513-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bennion Callister J., Pickering-Brown S.M. Pathogenesis/genetics of frontotemporal dementia and how it relates to ALS. (Pt B)Exp. Neurol. 2014;262:84–90. doi: 10.1016/j.expneurol.2014.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.DeJesus-Hernandez M., Mackenzie I.R., Boeve B.F., Boxer A.L., Baker M., Rutherford N.J., Nicholson A.M., Finch N.A., Flynn H., Adamson J., et al. Expanded GGGGCC hexanucleotide repeat in noncoding region of C9ORF72 causes chromosome 9p-linked FTD and ALS. Neuron. 2011;72:245–256. doi: 10.1016/j.neuron.2011.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Renton A.E., Majounie E., Waite A., Simon-Sanchez J., Rollinson S., Gibbs J.R., Schymick J.C., Laaksovirta H., van Swieten J.C., Myllykangas L., et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron. 2011;72:257–268. doi: 10.1016/j.neuron.2011.09.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Klotz S., Konig T., Erdler M., Ulram A., Nguyen A., Strobel T., Zimprich A., Stogmann E., Regelsberger G., Hoftberger R., et al. Co-incidental C9orf72 expansion mutation-related frontotemporal lobar degeneration pathology and sporadic Creutzfeldt-Jakob disease. Eur. J. Neurol. 2021;28:1009–1015. doi: 10.1111/ene.14621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gijselinck I., Van Broeckhoven C., Cruts M. Granulin mutations associated with frontotemporal lobar degeneration and related disorders: An update. Hum. Mutat. 2008;29:1373–1386. doi: 10.1002/humu.20785. [DOI] [PubMed] [Google Scholar]
- 7.Wong T.H., Pottier C., Hondius D.C., Meeter L.H.H., van Rooij J.G.J., Melhem S., Netherlands Brain B., van Minkelen R., van Duijn C.M., Rozemuller A.J.M., et al. Three VCP Mutations in Patients with Frontotemporal Dementia. J. Alzheimer’s Dis. JAD. 2018;65:1139–1146. doi: 10.3233/JAD-180301. [DOI] [PubMed] [Google Scholar]
- 8.Kacem I., Sghaier I., Ticozzi N., Mrabet S., Paverelli S., Nasri A., Ratti A., Ben Djebara M., Gargouri-Berrachid A., Silani V., et al. Expanding the phenotype of TARDBP mutation in a Tunisian family with clinical phenotype heterogeneity. Amyotroph. Lateral Scler. Front. Degener. 2022:1–4. doi: 10.1080/21678421.2022.2089856. [DOI] [PubMed] [Google Scholar]
- 9.Mattsson N. CSF biomarkers in neurodegenerative diseases. Clin. Chem. Lab. Med. 2011;49:345–352. doi: 10.1515/CCLM.2011.082. [DOI] [PubMed] [Google Scholar]
- 10.Dubois B., Feldman H.H., Jacova C., Dekosky S.T., Barberger-Gateau P., Cummings J., Delacourte A., Galasko D., Gauthier S., Jicha G., et al. Research criteria for the diagnosis of Alzheimer’s disease: Revising the NINCDS-ADRDA criteria. Lancet Neurol. 2007;6:734–746. doi: 10.1016/S1474-4422(07)70178-3. [DOI] [PubMed] [Google Scholar]
- 11.Albert M.S., DeKosky S.T., Dickson D., Dubois B., Feldman H.H., Fox N.C., Gamst A., Holtzman D.M., Jagust W.J., Petersen R.C., et al. The diagnosis of mild cognitive impairment due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2011;7:270–279. doi: 10.1016/j.jalz.2011.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.McKhann G.M., Knopman D.S., Chertkow H., Hyman B.T., Jack C.R., Jr., Kawas C.H., Klunk W.E., Koroshetz W.J., Manly J.J., Mayeux R., et al. The diagnosis of dementia due to Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2011;7:263–269. doi: 10.1016/j.jalz.2011.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sperling R.A., Aisen P.S., Beckett L.A., Bennett D.A., Craft S., Fagan A.M., Iwatsubo T., Jack C.R., Jr., Kaye J., Montine T.J., et al. Toward defining the preclinical stages of Alzheimer’s disease: Recommendations from the National Institute on Aging-Alzheimer’s Association workgroups on diagnostic guidelines for Alzheimer’s disease. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2011;7:280–292. doi: 10.1016/j.jalz.2011.03.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Dubois B., Feldman H.H., Jacova C., Hampel H., Molinuevo J.L., Blennow K., DeKosky S.T., Gauthier S., Selkoe D., Bateman R., et al. Advancing research diagnostic criteria for Alzheimer’s disease: The IWG-2 criteria. Lancet Neurol. 2014;13:614–629. doi: 10.1016/S1474-4422(14)70090-0. [DOI] [PubMed] [Google Scholar]
- 15.Jack C.R., Jr., Bennett D.A., Blennow K., Carrillo M.C., Dunn B., Haeberlein S.B., Holtzman D.M., Jagust W., Jessen F., Karlawish J., et al. NIA-AA Research Framework: Toward a biological definition of Alzheimer’s disease. Alzheimer’s Dement. J. Alzheimer’s Assoc. 2018;14:535–562. doi: 10.1016/j.jalz.2018.02.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Biscetti L., Salvadori N., Farotti L., Cataldi S., Eusebi P., Paciotti S., Parnetti L. The added value of Abeta42/Abeta40 in the CSF signature for routine diagnostics of Alzheimer’s disease. Clin. Chim. Acta Int. J. Clin. Chem. 2019;494:71–73. doi: 10.1016/j.cca.2019.03.001. [DOI] [PubMed] [Google Scholar]
- 17.Kasai T., Tokuda T., Ishigami N., Sasayama H., Foulds P., Mitchell D.J., Mann D.M., Allsop D., Nakagawa M. Increased TDP-43 protein in cerebrospinal fluid of patients with amyotrophic lateral sclerosis. Acta Neuropathol. 2009;117:55–62. doi: 10.1007/s00401-008-0456-1. [DOI] [PubMed] [Google Scholar]
- 18.Bourbouli M., Rentzos M., Bougea A., Zouvelou V., Constantinides V.C., Zaganas I., Evdokimidis I., Kapaki E., Paraskevas G.P. Cerebrospinal Fluid TAR DNA-Binding Protein 43 Combined with Tau Proteins as a Candidate Biomarker for Amyotrophic Lateral Sclerosis and Frontotemporal Dementia Spectrum Disorders. Dement. Geriatr. Cogn. Disord. 2017;44:144–152. doi: 10.1159/000478979. [DOI] [PubMed] [Google Scholar]
- 19.Kojima Y., Kasai T., Noto Y.I., Ohmichi T., Tatebe H., Kitaoji T., Tsuji Y., Kitani-Morii F., Shinomoto M., Allsop D., et al. Amyotrophic lateral sclerosis: Correlations between fluid biomarkers of NfL, TDP-43, and tau, and clinical characteristics. PLoS ONE. 2021;16:e0260323. doi: 10.1371/journal.pone.0260323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Junttila A., Kuvaja M., Hartikainen P., Siloaho M., Helisalmi S., Moilanen V., Kiviharju A., Jansson L., Tienari P.J., Remes A.M., et al. Cerebrospinal Fluid TDP-43 in Frontotemporal Lobar Degeneration and Amyotrophic Lateral Sclerosis Patients with and without the C9ORF72 Hexanucleotide Expansion. Dement. Geriatr. Cogn. Disord. Extra. 2016;6:142–149. doi: 10.1159/000444788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.The Ronald and Nancy Reagan Research Institute of the Alzheimer’s Association and the National Institute on Aging Working Group Consensus report of the Working Group on: “Molecular and Biochemical Markers of Alzheimer’s Disease”. Neurobiol. Aging. 1998;19:109–116. doi: 10.1016/S0197-4580(98)00022-0. [DOI] [PubMed] [Google Scholar]
- 22.Borroni B., Benussi A., Archetti S., Galimberti D., Parnetti L., Nacmias B., Sorbi S., Scarpini E., Padovani A. Csf p-tau181/tau ratio as biomarker for TDP pathology in frontotemporal dementia. Amyotroph. Lateral Scler. Front. Degener. 2015;16:86–91. doi: 10.3109/21678421.2014.971812. [DOI] [PubMed] [Google Scholar]
- 23.Paraskevas G.P., Kasselimis D., Kourtidou E., Constantinides V., Bougea A., Potagas C., Evdokimidis I., Kapaki E. Cerebrospinal Fluid Biomarkers as a Diagnostic Tool of the Underlying Pathology of Primary Progressive Aphasia. J. Alzheimer’s Dis. JAD. 2017;55:1453–1461. doi: 10.3233/JAD-160494. [DOI] [PubMed] [Google Scholar]
- 24.Rascovsky K., Grossman M. Clinical diagnostic criteria and classification controversies in frontotemporal lobar degeneration. Int. Rev. Psychiatry. 2013;25:145–158. doi: 10.3109/09540261.2013.763341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Gorno-Tempini M.L., Miller B.L. Primary progressive aphasia as a model to study the neurobiology of language. Brain Lang. 2013;127:105. doi: 10.1016/j.bandl.2013.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Folstein M.F., Folstein S.E., McHugh P.R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J. Psychiatr. Res. 1975;12:189–198. doi: 10.1016/0022-3956(75)90026-6. [DOI] [PubMed] [Google Scholar]
- 27.Dubois B., Slachevsky A., Litvan I., Pillon B. The FAB: A Frontal Assessment Battery at bedside. Neurology. 2000;55:1621–1626. doi: 10.1212/WNL.55.11.1621. [DOI] [PubMed] [Google Scholar]
- 28.Bourbouli M., Paraskevas G.P., Rentzos M., Mathioudakis L., Zouvelou V., Bougea A., Tychalas A., Kimiskidis V.K., Constantinides V., Zafeiris S., et al. Genotyping and Plasma/Cerebrospinal Fluid Profiling of a Cohort of Frontotemporal Dementia-Amyotrophic Lateral Sclerosis Patients. Brain Sci. 2021;11:1239. doi: 10.3390/brainsci11091239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Papadimas G.K., Paraskevas G.P., Zambelis T., Karagiaouris C., Bourbouli M., Bougea A., Walter M.C., Schumacher N.U., Krause S., Kapaki E. The multifaceted clinical presentation of VCP-proteinopathy in a Greek family. Acta Myol. Myopathies Cardiomyopathies Off. J. Mediterr. Soc. Myol. 2017;36:203–206. [PMC free article] [PubMed] [Google Scholar]
- 30.Finch N., Baker M., Crook R., Swanson K., Kuntz K., Surtees R., Bisceglio G., Rovelet-Lecrux A., Boeve B., Petersen R.C., et al. Plasma progranulin levels predict progranulin mutation status in frontotemporal dementia patients and asymptomatic family members. Brain J. Neurol. 2009;132:583–591. doi: 10.1093/brain/awn352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Ramos E.M., Koros C., Dokuru D.R., Van Berlo V., Kroupis C., Wojta K., Wang Q., Andronas N., Matsi S., Beratis I.N., et al. Frontotemporal dementia spectrum: First genetic screen in a Greek cohort. Neurobiol. Aging. 2019;75:224.e1–224.e8. doi: 10.1016/j.neurobiolaging.2018.10.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Janelidze S., Zetterberg H., Mattsson N., Palmqvist S., Vanderstichele H., Lindberg O., van Westen D., Stomrud E., Minthon L., Blennow K., et al. CSF Abeta42/Abeta40 and Abeta42/Abeta38 ratios: Better diagnostic markers of Alzheimer disease. Ann. Clin. Transl. Neurol. 2016;3:154–165. doi: 10.1002/acn3.274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kapaki E., Paraskevas G.P., Papageorgiou S.G., Bonakis A., Kalfakis N., Zalonis I., Vassilopoulos D. Diagnostic value of CSF biomarker profile in frontotemporal lobar degeneration. Alzheimer Dis. Assoc. Disord. 2008;22:47–53. doi: 10.1097/WAD.0b013e3181610fea. [DOI] [PubMed] [Google Scholar]
- 34.Paraskevas G.P., Kapaki E., Papageorgiou S.G., Kalfakis N., Andreadou E., Zalonis I., Vassilopoulos D. CSF biomarker profile and diagnostic value in vascular dementia. Eur. J. Neurol. 2009;16:205–211. doi: 10.1111/j.1468-1331.2008.02387.x. [DOI] [PubMed] [Google Scholar]
- 35.Lewczuk P., Riederer P., O’Bryant S.E., Verbeek M.M., Dubois B., Visser P.J., Jellinger K.A., Engelborghs S., Ramirez A., Parnetti L., et al. Cerebrospinal fluid and blood biomarkers for neurodegenerative dementias: An update of the Consensus of the Task Force on Biological Markers in Psychiatry of the World Federation of Societies of Biological Psychiatry. World J. Biol. Psychiatry Off. J. World Fed. Soc. Biol. Psychiatry. 2018;19:244–328. doi: 10.1080/15622975.2017.1375556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Paraskevas G.P., Bougea A., Constantinides V.C., Bourbouli M., Petropoulou O., Kapaki E. In vivo Prevalence of Alzheimer Biomarkers in Dementia with Lewy Bodies. Dement. Geriatr. Cogn. Disord. 2019;47:289–296. doi: 10.1159/000500567. [DOI] [PubMed] [Google Scholar]
- 37.Tan R.H., Yang Y., Kim W.S., Dobson-Stone C., Kwok J.B., Kiernan M.C., Halliday G.M. Distinct TDP-43 inclusion morphologies in frontotemporal lobar degeneration with and without amyotrophic lateral sclerosis. Acta Neuropathol. Commun. 2017;5:76. doi: 10.1186/s40478-017-0480-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sellami L., Saracino D., Le Ber I. Genetic forms of frontotemporal lobar degeneration: Current diagnostic approach and new directions in therapeutic strategies. Rev. Neurol. 2020;176:571–581. doi: 10.1016/j.neurol.2020.02.008. [DOI] [PubMed] [Google Scholar]
- 39.de Boer E.M.J., Orie V.K., Williams T., Baker M.R., De Oliveira H.M., Polvikoski T., Silsby M., Menon P., van den Bos M., Halliday G.M., et al. TDP-43 proteinopathies: A new wave of neurodegenerative diseases. J. Neurol. Neurosurg. Psychiatry. 2021;92:86–95. doi: 10.1136/jnnp-2020-322983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Foulds P., McAuley E., Gibbons L., Davidson Y., Pickering-Brown S.M., Neary D., Snowden J.S., Allsop D., Mann D.M. TDP-43 protein in plasma may index TDP-43 brain pathology in Alzheimer’s disease and frontotemporal lobar degeneration. Acta Neuropathol. 2008;116:141–146. doi: 10.1007/s00401-008-0389-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Babic Leko M., Zupunski V., Kirincich J., Smilovic D., Hortobagyi T., Hof P.R., Simic G. Molecular Mechanisms of Neurodegeneration Related to C9orf72 Hexanucleotide Repeat Expansion. Behav. Neurol. 2019;2019:2909168. doi: 10.1155/2019/2909168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Freibaum B.D., Taylor J.P. The Role of Dipeptide Repeats in C9ORF72-Related ALS-FTD. Front. Mol. Neurosci. 2017;10:35. doi: 10.3389/fnmol.2017.00035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Mori K., Arzberger T., Grasser F.A., Gijselinck I., May S., Rentzsch K., Weng S.M., Schludi M.H., van der Zee J., Cruts M., et al. Bidirectional transcripts of the expanded C9orf72 hexanucleotide repeat are translated into aggregating dipeptide repeat proteins. Acta Neuropathol. 2013;126:881–893. doi: 10.1007/s00401-013-1189-3. [DOI] [PubMed] [Google Scholar]
- 44.Zu T., Liu Y., Banez-Coronel M., Reid T., Pletnikova O., Lewis J., Miller T.M., Harms M.B., Falchook A.E., Subramony S.H., et al. RAN proteins and RNA foci from antisense transcripts in C9ORF72 ALS and frontotemporal dementia. Proc. Natl. Acad. Sci. USA. 2013;110:E4968–E4977. doi: 10.1073/pnas.1315438110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Ash P.E., Bieniek K.F., Gendron T.F., Caulfield T., Lin W.L., Dejesus-Hernandez M., van Blitterswijk M.M., Jansen-West K., Paul J.W., 3rd, Rademakers R., et al. Unconventional translation of C9ORF72 GGGGCC expansion generates insoluble polypeptides specific to c9FTD/ALS. Neuron. 2013;77:639–646. doi: 10.1016/j.neuron.2013.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Nelson P.T., Trojanowski J.Q., Abner E.L., Al-Janabi O.M., Jicha G.A., Schmitt F.A., Smith C.D., Fardo D.W., Wang W.X., Kryscio R.J., et al. “New Old Pathologies”: AD, PART, and Cerebral Age-Related TDP-43 with Sclerosis (CARTS) J. Neuropathol. Exp. Neurol. 2016;75:482–498. doi: 10.1093/jnen/nlw033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Nelson P.T., Dickson D.W., Trojanowski J.Q., Jack C.R., Boyle P.A., Arfanakis K., Rademakers R., Alafuzoff I., Attems J., Brayne C., et al. Limbic-predominant age-related TDP-43 encephalopathy (LATE): Consensus working group report. Brain J. Neurol. 2019;142:1503–1527. doi: 10.1093/brain/awz099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Brunnstrom H., Englund E. Clinicopathological concordance in dementia diagnostics. Am. J. Geriatr. Psychiatry Off. J. Am. Assoc. Geriatr. Psychiatry. 2009;17:664–670. doi: 10.1097/JGP.0b013e3181a6516e. [DOI] [PubMed] [Google Scholar]
- 49.Delaby C., Alcolea D., Carmona-Iragui M., Illan-Gala I., Morenas-Rodriguez E., Barroeta I., Altuna M., Estelles T., Santos-Santos M., Turon-Sans J., et al. Differential levels of Neurofilament Light protein in cerebrospinal fluid in patients with a wide range of neurodegenerative disorders. Sci. Rep. 2020;10:9161. doi: 10.1038/s41598-020-66090-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data presented in this study are available upon reasonable request from the corresponding author. The data are not publicly available due to privacy restrictions.