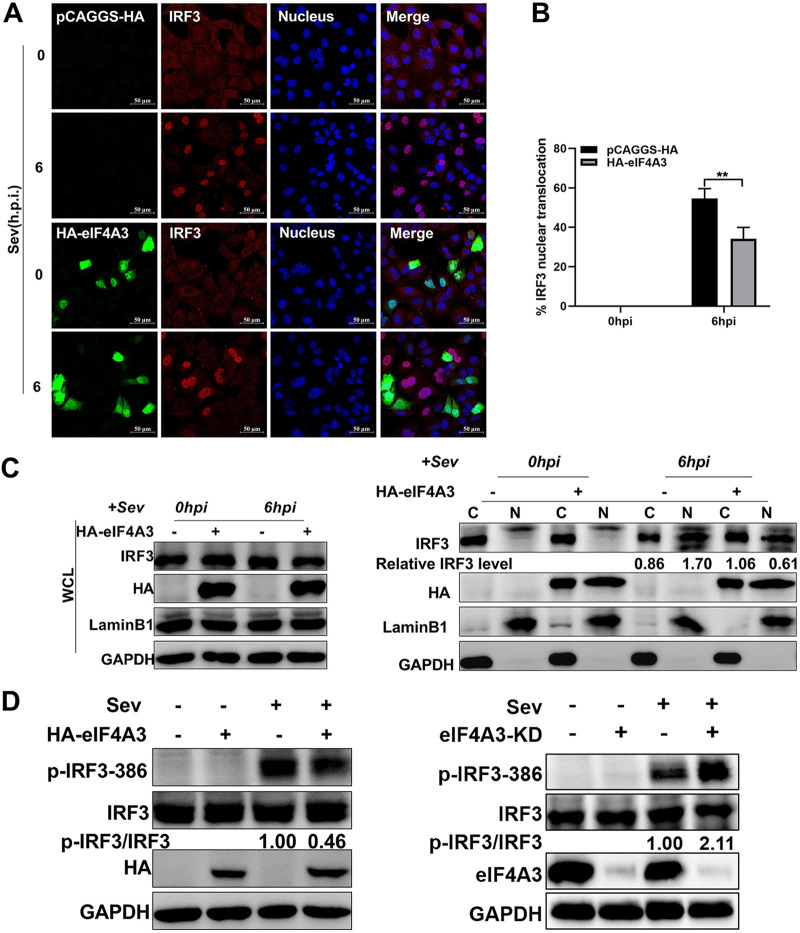
FIG 5.
The inhibition of eIF4A3 on IRF3 activation. (A to C) Effect of eIF4A3 on nuclear translocation of IRF3. A549 cells were transfected with HA-eIF4A3 or vector (HA) and simulated with SeV for 6 h after 24 h of transfection. (A) Confocal microscopy was performed using an anti-IRF3 rabbit antibody (red) and anti-HA mouse antibody (green). DAPI was used to stain for the nucleus (blue). Samples were examined with a confocal microscope (LSM 880; Zeiss). Images are representative of three independent experiments. Scale bar = 50 μM. (B) Quantitative analysis of IRF3 translocation. At least 100 cells in each group were scored (*, P < 0.05; **, P < 0.01; ***, P < 0.001; ****, P < 0.0001; two-tailed Student’s t test). (C) Western blot analysis of the distribution of IRF3 in the cytoplasmic and nuclear factions. The indicated cells were subjected to isolation of cytoplasmic and nuclear fractions. Lamin B1 and GAPDH were used as the loading control for nuclear and cytoplasmic fractions, respectively. The band intensities were quantified, and relative cytoplasmic IRF3 levels (IRF3/GAPDH) and relative nuclear IRF3 levels (IRF3/lamin B1) are shown below. (D) Effect of eIF4A3 on the phosphorylation of IRF3 induced by SeV. A549 cells were transfected with HA-eIF4A3 or vector (HA), and simulated with SeV for 6 h after 24 h of transfection or eIF4A3 knockdown A549 cells and WT A549 cells infected with SeV for 6 h. The cell lysates were harvested for the Western blot analysis using anti-IRF3 antibody and anti-Ser386 phosphorylated IRF3 antibody. The band intensities were quantified, and relative IRF3 levels (p-IRF3-386/IRF3) are shown below.