Abstract
Introduction
Few formal studies have been performed investigating the frequency of equipment failure during surgery. Surgeons are unable to operate without the plethora of instruments and equipment surrounding them in the operating theatre. As with any mechanical component, instruments and equipment are subject to time- and use-dependent degradation in their performance. Yet no formal requirements exist for the routine inspection or maintenance of instruments. Owing to this lack of information regarding equipment failure we undertook the first investigation of intraoperative equipment malfunction occurring during cardiac surgical procedures.
Methods
Over a 12-month period cardiac surgeons were required to report equipment malfunction during each procedure. Operating theatre equipment was divided into three categories broadly based on equipment portability and function: group 1, theatre infrastructure and components; group 2, large medical equipment; and group 3, surgical instruments.
Results
In a highly significant proportion of operations performed (92%) there was an issue with equipment. The most common issues occurred in group 3 with fine surgical instrument malfunctions; most commonly worn-out needle holders and blunt scissors. Theatre infrastructure and large medical equipment failures (groups 1 and 2) resulted in the cancellation of four cases. Some intraoperative instrument failures were potentially catastrophic.
Conclusions
The incidence of equipment failure during cardiac surgery is unacceptably high. In some instances, cases were cancelled and revenue lost owing to equipment malfunction. A balance between the safety and quality of equipment and cost effectiveness is required. These findings suggest that surgical instruments warrant an annual compulsory inspection.
Keywords: Cardiac equipment malfunction, Risk management, Instrument maintenance, Instrument accountability, Quality control
Introduction
It is well known that the voluntary reporting of medical device failure vastly underestimates the true incidence with which problems occur.1 Instruments represent the interface between the surgeon and the patient. They are subjected to daily wear and tear during surgery and can be exposed to potential for damage caused by dropping, misuse or careless transportation. Furthermore, surgical instruments are required to be sterilised between each case and are therefore vulnerable to heat/cool cycles and erosive cleaning processes that can accelerate degradation.
However, the equipment we use in theatre is not subjected to standardised inspection nor maintenance and in most instances is only repaired or replaced following an intraoperative failure on an ad hoc unplanned basis. Although legislation exists for the regular testing of ubiquitous equipment including electrical appliances, vehicles and home heating, instruments that are used on a daily basis to perform intricate surgery are not similarly assessed. Patient safety, which is of paramount importance, is dependent upon the correct functioning of the equipment that is used in theatre by surgeons.
Among the medical specialties, cardiac surgeons are some of the most avant-garde when it comes to embracing change for the benefit of patient safety.2 Cardiac surgical outcome data have been collected and analysed for decades.3 Surgeon-specific operative results have been published by the press for the public to scrutinise. Cardiac surgeons rapidly adopted the World Health Organization checklist before it was a compulsory requirement. However, despite the safe and transparent nature of international cardiac surgery, some elements of surgical practice are not assessed or audited.
Surgical instruments and equipment used for cardiac operations have escaped scrutiny. These devices can be cycled through operations, passing under the radar of critical examination and inspection. Malperforming instruments are often passed from case to case and can easily be missed owing to increased utilisation or the outsourcing of sterilisation and maintenance services which prevents direct contact with those responsible for maintaining equipment quality. The result is that failure often occurs in situations when instruments are most required, potentially leading to injury to both patient and staff.
The aim of this study is to investigate the frequency with which infrastructural or equipment failure occurs during each cardiac operation and to categorise each failure according to the structure and function of the analysed equipment.
Methods
This study comprised all consecutive first-time elective cardiac surgical cases undertaken by two consultant cardiac surgeons. All cases were classified as open heart surgery and involved the use of cardiopulmonary bypass (eg coronary artery bypass grafting, aortic valve replacement, major aortic surgery, mitral valve surgery) at two UK cardiac centres. Data were collected prospectively for 12 consecutive months.
Equipment failures were recorded in real time during each cardiac operation. A data collection sheet (Figure 1) was devised to enable standardisation of the examination and categorisation of theatre equipment and the types of failures captured. Theatre equipment was categorised into three groups based on size and function.
-
1.
Theatre infrastructure and components. This category comprised permanent fixtures and fittings that cannot be removed from the operating theatre but are essential for its functioning. Such items include operating lights, ventilation, computers/imaging screens, temperature regulation, and clocks and timers.
-
2.
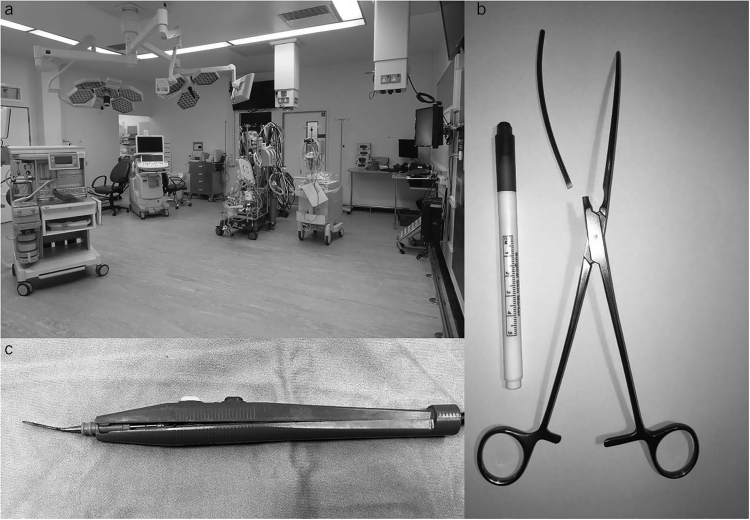
Large medical equipment. This subgroup comprised large but portable medical equipment. Essential machinery that can be wheeled in and out of the operating theatre. Such devices include transoesophageal echocardiography (TOE) machine, diathermy control unit, defibrillator, operating table and suction–irrigation systems (Figure 2a).
-
3.
Surgical instruments. This category of hand-held surgical instruments is routinely under the jurisdiction of the scrub nurse. The instruments are typically light weight, intricate and delicate, are used by the surgeon for direct tissue contact and require sterilisation following every case.
Figure 1 .
Infrastructure and equipment performance recording sheet. Equipment performance from all three categories was recorded and a description of the issue noted.
Figure 2 .
(a) A cardiac operating theatre demonstrating the variety in size and complexity of equipment required for a surgical procedure to be undertaken. (b) Fractured aortic cross-clamp. This instrument fractured mid procedure. This could potentially have had catastrophic consequences. (c) Broken diathermy wand. This device failed during surgery. Plastic fragments were recovered. There was the potential for electrical injury or loss of fragments in the operative field.
At the beginning of the day, a team briefing and the surgical checklist were undertaken by the principal investigators. Any concerns raised by the staff regarding equipment were noted. The investigators then performed an inspection of all equipment in subgroups 1 and 2. The theatre was first inspected to check the functioning of ambient and operating lights, heating and ventilation. All electrical devices were examined including imaging stations and computer terminals used to check patient results.
Major failures such as non-functioning ventilation or operating lights led to an escalation of the problem to the hospital maintenance team and an immediate halt to surgery. Failures such as broken operating tables, diathermy and echocardiography machines were dealt with where possible by swapping for functioning units, reporting the problem and continuing with the surgery.
Equipment in subgroup 3 was initially checked by the principal investigators while the scrub team prepared for the operation. Obvious problems such as lack of correct instrument sets, breaches in the sterility packaging or gross deformities in instrument form or function were observed and recorded. During surgery a predetermined non-scrubbed member of the theatre team was given responsibility for recording prospective instrument failures in subgroup 3 occurring during the operation, as well as additional problems in subgroups 1 and 2. At the end of each case, a debrief was performed and any equipment failures were cross-checked with all disciplines of the theatre team. The completed checklist was the transferred to a database.
Results
Data from 139 patients over a 12-month period revealed that equipment failure occurred in 92% of cardiac cases and resulted in the cancellation of four operations. Of all the recorded malfunctions, failures in group 1 (theatre and infrastructure) accounted for 12% of issues; group 2 (large medical equipment) accounted for 15% of problems; and an 80% failure rate was recorded in group 3 (surgical instruments).
Group 1
Two cases were lost secondary to major issues within the basic fabric of the operating theatre complex (Figure 3). Malfunction of the ventilation/laminar flow system led to the cancellation of one operation, and non-functioning theatre lights resulted in the cancellation of another case.
Figure 3 .
Theatre and infrastructure malfunctions. The frequency of reported malfunction in this subgroup was 12%. Issues with lighting, ambient temperature and ventilation were most commonly reported. Two cases (2/139) were cancelled owing to failures of equipment in this group.
Ambient temperature regulation was another common fault in this category, with extremes of temperature being recorded. The theatre temperature was commonly >26°C and considerably higher beneath the surgical lights for gowned surgical staff. Although this did not result in any cancellation of cases, it was dealt with by use of cold water application to the surgeon.
Group 2
Malfunctions in the large medical equipment group affected 15% of cases (Figure 4). One case was cancelled owing to an essential transoesophageal echocardiography (TOE) machine failing to work prior to the commencement of valve repair surgery. Two cases of operating table position and adjustment failure led to patients being transferred to functioning operating tables after they had been intubated and draped for surgery.
Figure 4 .
Large medical equipment malfunctions. The frequency of reported malfunction in this subgroup was 15%. One case was cancelled (1/139) owing to a defective transoesophageal echocardiography (TOE) machine.
Potentially dangerous issues were noted during two separate cases when perished electrical insulation was discovered on internal defibrillation paddles. In both cases, the cardioversion energy was not delivered and new defibrillation paddles were subsequently opened. These issues had the potential to cause harm to both the patient and medical staff.
Group 3
The surgical instrument group (Figure 5) was responsible for the bulk of failures recorded and resulted in one case being cancelled owing to an instrument tray being unavailable prior to the procedure. In 80% of cases a problem with surgical instruments occurred. Needle holders that were unable to grip the needle were the most common issue and accounted for 67% of surgical instrument issues. Slipping tissue forceps and blunt dissecting scissors were the next most frequent issues and occurred in 26% and 20% of cases, respectively. Potentially catastrophic instrument failures included an aortic cross-clamp that fractured mid operation (Figure 2b), a sternal retractor that failed mid procedure while the heart was positioned above the sternum and a diathermy wand that broke, releasing its internal components (Figure 2c).
Figure 5 .
Surgical instrument malfunctions. The recorded frequency of surgical instrument malfunction was 80%. One case was cancelled owing to a lack of the correct instruments (1/139). The graph demonstrates that the main failures were with needle holders, scissors and forceps.
Discussion
Failures in theatre equipment are important to understand and quantify. Operative outcomes following surgery are not only related to the competence of the surgeon performing the case, but may also be correlated to the performance of theatre equipment and surroundings.4,5 During the past two decades increased scrutiny, particularly in cardiac surgery, has been imposed upon hospitals and doctors in an attempt to maintain the quality of care that is provided and improve patient outcomes.3 Conversely, the effect of these inspections and audits that have been imposed may have led to a reduction in the rate of progress of surgical advancement and possibly led to an increase in the practice of risk-averse patient selection.
Mistakes or adverse surgical outcomes are increasingly met with formal complaints, reviews and litigation. Furthermore, several studies have demonstrated that the working environment of a surgeon is also a contributing factor when it comes to operative outcome.6–9 A number of studies in cardiac surgery have investigated a range of ‘system failures’ contributing to adverse surgical outcomes, ranging from communication and coordination issues, staffing, patient-related factors, training issues and other human factors.5,8,10
The aim of our study was to capture specifically the range of equipment and technological factors that can malfunction during cardiac cases. These failures are arguably the most simple to correct, provided we have a greater understanding and adequate investment. Equipment-related malfunctions have been consistently shown to directly contribute to a quarter of adverse events in surgery, and this is an even greater issue in cardiac surgery where more complex technological procedures are associated with greater rates of equipment error.11–13
Of the studies investigating surgical equipment failures, the vast majority focus on specific device errors or specific types of instrument failures.1,4,14,15 These failures form an important component of our study captured within group 3; however, we also sought to encompass a broader and less well-documented range of issues beyond specific equipment failures, including infrastructure within the operating theatre captured in groups 1 and 2.
There have been a range of studies, in different healthcare settings, in which the impact of broader infrastructure issues have been analysed and demonstrate significant detrimental impacts on case outcomes.14,16 Measurement of these types of equipment and infrastructure errors is inherently heterogeneous between different studies, types of surgery and healthcare settings; however, they remain critical components of a successful operation that can potentially be improved.13
Theatre equipment and the instruments used have evaded surveillance and this represents a critical area that needs to be addressed. This deficit has been raised by a number of key opinion holders in the field but is yet to be acted upon despite becoming more important.13,17 In contrast to the aviation industry, which requires that a pre-flight checklist is completed prior to every flight, operations are being performed without the efficiency and function of surgical instruments or theatre infrastructure being checked before a procedure is undertaken.
The results of this study reveal that in over 92% of cases a fault was discovered, 80% of which related to small surgical instruments. The most common culprits were needle holders and forceps, which are the most important interface between the surgeon and the patient. Common issues were blunt scissors, bent forceps and needle holders with worn down tungsten–carbide inserts. Worryingly, some instrument failures were potentially catastrophic, as in the case of an aortic cross-clamp that fractured mid procedure (Figure 2b) and defective electrical insulation (Figure 2c).
In only 9% of cases were there no issues with equipment. In some instances, significant and avoidable clinical risk was introduced, cases were cancelled, and invariably there was an economic impact caused by wasting a theatre slot, the associated manpower and disruption to patient wellbeing.
The impact of defective surgical instruments is serious and has a multitude of adverse effects, ranging from an increase in operating time,1,7 adverse patient outcomes,4 negative impacts on the team and poor surgical morale.5
Our measured incidence of small equipment failure is notably high, substantially so compared with other studies, which have reported specific surgical equipment error rates ranging from 46% for any type of surgical cases in a teaching hospital in The Netherlands18 to 55% of cases in vascular surgery.19 It is well documented that cardiac surgery is more technically challenging and this comes with a greater risk of error rate.13,19 It may be that the cardiac cases in our study were more technically challenging, leading to this higher failure rate. It may also be that wider system issues with suboptimal reporting or maintenance of equipment contributed to the higher prevalence of faults. For example, scrub nurses setting up cases may be unaware of the subtle malfunctions that can be present in surgical instruments before the surgeon is ready to use them. Certain malfunctions are difficult to assess: for example, magnetised needle holders are not obvious before an anastomosis is being performed; Potts scissors may not appear to be blunt or bent to the naked eye; forceps can initially appear to be normal but may not be able to hold the adventitia of an artery without it slipping. Thus damaged instruments may enter an operation insidiously. A surgeon may also ‘make do’ with suboptimal instruments. The investigators often continued the case using, for example, a magnetised needle holder, blunt scissors and slipping forceps, because it was not appropriate to ask for replacement instruments during the middle of a micro-anastomosis. However, such compromise can come at the cost of reduced flow of the operation.
The instrument failure may then go unrecorded because equipment issues are not the first priority of theatre staff during a procedure. The lack of any framework to systematically report instrument malfunctions thus leads to the absence of quantification for this hidden problem. Without formal data for instrument failure the problem may then be undetected.
Sterilisation facilities are also frequently off-site, and direct contact with servicing engineers is not possible. Reporting instrument failure can also be laborious and often requires complex on-line processes. Instruments sent for repair must also be taken out of circulation, further impacting instrument availability for the cases that follow. All these issues represent significant barriers to improving the quality of instruments, which may account for the high fault rate captured in our study and also, importantly, represent key areas for action.
This study was novel in that in addition to small equipment failures, we incorporated the wider infrastructure of the operating theatre based upon size and mobility. Doing so enabled less well-described patterns of equipment failure (in the UK setting) to be discovered and addressed.
Investigating theatre infrastructure uncovered a variety of commonly recurring issues. Defective theatre lights, ventilation and thermostat problems, and non-functioning computers were all observed over the period of the study and resulted in the cancellation of cases.
By highlighting these issues and improving awareness to the presence of equipment failure, hospital management can promote more efficient estates maintenance and equipment repair programmes.
Large medical equipment failures were also a significant area of concern. One notable example was the failure of a TOE machine that resulted in the cancellation of mitral valve repair surgery. Although this could not have been foreseen or anticipated, it highlights that departments do not have surplus equipment that can be used in the event of device malfunction.
This study highlights the importance of the system failures surrounding surgeons who operate in a high-complexity and ‘high-consequence’ environment. A substantially high failure rate of surgical instruments across these two hospitals was noted, in addition to a sizeable proportion of infrastructural issues. The consequences of some infrastructure malfunctions were so severe or fundamental to the operation that the procedure had to be cancelled in 2.8% of cases. Other equipment failures were so dangerous that they could potentially have resulted in harm to the both the patient and the surgeon.
Study strengths
To our knowledge, this is the first study to investigate and report the frequency of a range of both infrastructural and equipment malfunctions occurring during cardiac surgery. The study design was prospective data collection, the most robust methodology to capture equipment failure rates, with a transparent and predefined checklist and categorisation to capture equipment failures. We have addressed an important aspect of quality control and accountability that could be significantly improved with certain simple interventions and would have important implications for patient safety, surgical outcomes and efficiency of provision of care.
Limitations of study
The principal limitations of this study relate to standardisation of the definition of equipment failure. To address the issue of equipment grouping an inventory was created of each type of equipment for each group (Figure 1) to allow for reproducibility.
Using the inventory, problems in groups 1 and 2 were quantified in a binary manner and therefore could not be affected by the possibility of subjective analysis. For example, in group 1, the theatre lights either work or do not. In group 2, the operating table either adjusts when required or does not.
Group 3, however, could potentially be subjected to observer bias. A worn-down Castroviejo needle holder may be adequate to pass a needle through a normal aorta in the hands of an adequate surgeon but may be unable to pass a needle through an atheromatous aorta in the hands of a less-experienced surgeon. Likewise, Potts scissors that may easily perform an arteriotomy in one patient may be ineffectual at performing an arteriotomy in another patient owing to tissue-related or ergonomic factors. Regardless of the skill of the surgeon, a bad instrument at the end of its serviceable life must be replaced because we cannot allocate instrument efficacy based upon the skill of the surgeon.
Despite the potential for observer bias in group 3, this was kept to a minimum by photographing broken instruments where possible (Figure 1b,c) and by confirming with the scrub nurse that a defect was present (eg by demonstrating that a needle could rotate while in the grip of a ratcheted needle holder). Although this is not a guaranteed method for standardised reproducibility, photographic evidence of equipment failure provided undisputable proof that a defect is present. Furthermore the investigators both wore 3.5 magnification loupes allowing for accurate inspection of the functioning surfaces of needle holders, forceps and scissors.
The authors were also not blinded to the outcomes collected, potentially leading to selection bias. This could potentially be overcome in future work with involvement of an independent team of data collectors, although this would be resource-intensive.
Conclusions
A balance between safety and quality of equipment and cost effectiveness is required. The introduction of surgical checklists and specific task-orientated de-briefing has been consistently demonstrated as resulting in improved surgical safety outcomes.10 The findings of our study suggest that surgical instruments warrant, as a minimum, an annual inspection for the safety of both the patient and the surgeon. Pilots would not fly an aircraft that had not been maintained correctly, yet surgeons are expected to operate with dangerous equipment and this issue must be addressed. We would also suggest more innovative approaches in the age of rapidly advancing technology; the adoption of a QR labelling system or barcode etched on each instrument would allow a maintenance log book to be compiled, problems to be reported and repairs to be tracked, thus reducing a barrier to measuring and acting on equipment failures.
References
- 1.Samore MH, Evans RS, Lassen Aet al. Surveillance of medical device-related hazards and adverse events in hospitalized patients. JAMA 2004; 291: 325–334. [DOI] [PubMed] [Google Scholar]
- 2.Wahr JA, Prager RL, Abernathy JH 3rdet al. Patient safety in the cardiac operating room: human factors and teamwork: a scientific statement from the American Heart Association. Circulation 2013; 128: 1139–1169. [DOI] [PubMed] [Google Scholar]
- 3.Bridgewater B. Society for cardiothoracic surgery in GB, Ireland. cardiac registers: the adult cardiac surgery register. Heart 2010; 96: 1441–1443. [DOI] [PubMed] [Google Scholar]
- 4.Courdier S, Garbin O, Hummel Met al. Equipment failure: causes and consequences in endoscopic gynecologic surgery. J Minim Invasive Gynecol 2009; 16: 28–33. [DOI] [PubMed] [Google Scholar]
- 5.Diaz-Perez A, Vega-Ochoa A, Dominguez-Lozano Bet al. Factors attributable to the cancellation of programmed surgeries. Cir Cir 2020; 88: 489–499. [DOI] [PubMed] [Google Scholar]
- 6.Calland JF, Guerlain S, Adams RBet al. A systems approach to surgical safety. Surg Endosc 2002; 16: 1005–1014; discussion 1015. [DOI] [PubMed] [Google Scholar]
- 7.Healey AN, Sevdalis N, Vincent CA. Measuring intra-operative interference from distraction and interruption observed in the operating theatre. Ergonomics 2006; 49: 589–604. [DOI] [PubMed] [Google Scholar]
- 8.Lingard L, Garwood S, Poenaru D. Tensions influencing operating room team function: does institutional context make a difference? Med Educ 2004; 38: 691–699. [DOI] [PubMed] [Google Scholar]
- 9.Vincent C, Moorthy K, Sarker SKet al. Systems approaches to surgical quality and safety: from concept to measurement. Ann Surg 2004; 239: 475–482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Catchpole KR, Giddings AE, de Leval MRet al. Identification of systems failures in successful paediatric cardiac surgery. Ergonomics 2006; 49: 567–588. [DOI] [PubMed] [Google Scholar]
- 11.Buzink SN, van Lier L, de Hingh IH, Jakimowicz JJ. Risk-sensitive events during laparoscopic cholecystectomy: the influence of the integrated operating room and a preoperative checklist tool. Surg Endosc 2010; 24: 1990–1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Treadwell JR, Lucas S, Tsou AY. Surgical checklists: a systematic review of impacts and implementation. BMJ Qual Saf 2014; 23: 299–318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Weerakkody RA, Cheshire NJ, Riga Cet al. Surgical technology and operating-room safety failures: a systematic review of quantitative studies. BMJ Qual Saf 2013; 22: 710–718. [DOI] [PubMed] [Google Scholar]
- 14.Graafland M, Bemelman WA, Schijven MP. Game-based training improves the surgeon's situational awareness in the operation room: a randomized controlled trial. Surg Endosc 2017; 31: 4093–4101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tapper A, Leale D, Megahan Get al. Robotic instrument failure-A critical analysis of cause and quality improvement strategies. Urology 2019; 131: 125–129. [DOI] [PubMed] [Google Scholar]
- 16.Prin M, Eaton J, Mtalimanja O, Charles A. High elective surgery cancellation rate in Malawi primarily due to infrastructural limitations. World J Surg 2018; 42: 1597–1602. [DOI] [PubMed] [Google Scholar]
- 17.Henrickson SE, Wadhera RK, Elbardissi AWet al. 3rd. Development and pilot evaluation of a preoperative briefing protocol for cardiovascular surgery. J Am Coll Surg 2009; 208: 1115–1123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wubben I, van Manen JG, van den Akker BJet al. Equipment-related incidents in the operating room: an analysis of occurrence, underlying causes and consequences for the clinical process. Qual Saf Health Care 2010; 19: e64. [DOI] [PubMed] [Google Scholar]
- 19.Albayati MA, Gohel MS, Patel SRet al. Identification of patient safety improvement targets in successful vascular and endovascular procedures: analysis of 251 hours of complex arterial surgery. Eur J Vasc Endovasc Surg 2011; 41: 795–802. [DOI] [PubMed] [Google Scholar]