Abstract
Background:
The potential misapplication of current opioid prescribing policies remains understudied and may have substantial adverse implications for patient safety.
Methods:
We used autoregressive integrated moving average models to assess level and trend changes in monthly 1) prescribing rates, 2) days’ supply, and 3) daily morphine milligram equivalents (MME) of incident opioid prescriptions relative to 1) a state medical board initiative to reduce high-dose and -volume opioid prescribing and 2) legislation to limit initial opioid prescriptions for acute and postsurgical pain. We examined outcomes by pain indication overall and by cancer history, using prescribing patterns for benzodiazepines to control for temporal trends. We used large private health insurance claims data to include North Carolina residents, aged 18–64, insured at any point between January 2012 and August 2018.
Results:
After the medical board initiative, prescribing patterns for chronic pain patients did not change; conversely, acute and postsurgical pain patients experienced immediate declines in daily MME. Post-legislation prescription rates did not decline for those with acute, postsurgical, and non-cancer pain, but instead declined among cancer patients with chronic pain. Chronic pain patients experienced the largest days’ supply declines post-legislation, instead of acute and postsurgical pain patients.
Conclusions:
We found mixed evidence on the potential impact of two opioid prescribing policies, with some observed declines in a group not intended to be impacted by the policy. This study provides evidence of the need for clearer opioid prescribing policies to ensure impacts on intended populations and avoid unintended consequences.
Keywords: Opioids, Opioid Prescribing Policies, Pain, Prescriptions, Unintended Consequences
1. INTRODUCTION
Medical prescription opioid use remains common despite increasingly stringent prescribing policies (Lee et al., 2021; National Academies of Sciences, 2017). Current Centers for Disease Control and Prevention (CDC) guidelines (Dowell et al., 2016) suggest prescribing opioids for pain management only if pain and function benefits outweigh risks. Clinical indications for opioid initiation vary widely and commonly include acute, postsurgical, chronic cancer-related, and chronic non-cancer-related pain, with varying risk-benefit relationships across these indications (Liu et al., 2013; Mikosz et al., 2020; Mundkur et al., 2018; Pasricha et al., 2018; Shah et al., 2017). Because prescription opioid use in patients with acute and postsurgical pain has been associated with new-onset long-term opioid use, opioid use disorder (OUD), and overdose (Baker 2017; Hadlandsmyth et al., 2019; Hah et al., 2017), most policy efforts have focused on limiting initial opioid prescription dose and duration for these pain indications.
Although prescribing policies to reduce opioid misuse are necessary, policy efforts may have unintended consequences (Centers for Disease Control and Prevention, 2019; Dowell et al., 2019; Ranapurwala et al., 2021). One unintended consequence is the under-prescribing of opioids in patients with chronic non-cancer and cancer pain, either due to imprecise policy language or an increasingly hostile prescribing environment (Kroenke et al., 2019). Misapplication of guidelines, such as abrupt discontinuation or rapid taper of high-dose opioids (Ranapurwala et al., 2021), may compromise patient safety (Centers for Disease Control and Prevention, 2019; Dowell et al., 2019) by causing patients with sudden uncontrolled pain to seek diverted or illegal opioids, increasing risk of OUD, overdose, and suicide (Binswanger et al., 2020; Bohnert and Ilgen, 2019; Coffin et al., 2020; Dowell et al., 2019; Hartung et al., 2020; Muzzio et al., 2021; Perez et al., 2020). Policies’ unintended prescribing impacts must be understood so that future policies can safely and clearly distinguish between problematic and legitimate prescribing—in particular, addressing appropriate limits for the management of acute and postsurgical pain without negatively impacting clinically indicated management of chronic non-cancer and cancer pain (Kroenke et al., 2019).
In response to the CDC’s 2016 opioid prescribing guidelines for chronic non-cancer pain (Dowell et al., 2016), North Carolina enacted state policies to improve patient care and safety by focusing on prescribers. In May 2016, the state medical board, intending to restrict opioid prescribing at high doses and high volume, launched an investigative initiative to identify clinicians who prescribe large numbers of high-dose opioids or had ≥2 patients die from opioid overdose within a 12-month period (North Carolina Medical Board, 2016; North Carolina Medical Society, 2017). In January 2018, North Carolina enacted legislation intended to, in part, limit the initial prescription of opioids for outpatient acute and postsurgical pain to five and seven days, respectively (General Assembly of North Carolina Session, 2017; North Carolina Medical Board, 2017). We conducted a study to examine the impacts of the two statewide North Carolina policies on opioid prescribing patterns among privately insured postsurgical, acute, and chronic pain patients, overall and by history of cancer.
2. MATERIAL AND METHODS
2.1. Study design, data source, and population
We conducted controlled and single-series interrupted time series analyses to assess the level and trend changes of new opioid prescription rates, daily dose, and daily duration by pain and cancer indication, following a state board medical initiative and a statewide legislative action, from January 2012 through August 2018 in North Carolina.
Deidentified claims data from a large single provider of private health insurance were used in analyses. Eligible persons were North Carolina residents between the ages of 18 and 64 who were insured by the provider for at least six continuous months at any point between July 2011 and August 2018.
Insured persons entered the analysis sample once they had six months of continuous insurance coverage (“washout period”). The earliest entry into the analysis sample after six months of continuous coverage was January 2012, which represented the start of the study period; persons remained in the analysis sample until discontinuation of coverage or the end of the study period (August 2018). Persons who discontinued and then restarted insurance coverage were re-entered in the analysis sample after a new six-month washout period.
2.2. Outcomes
We examined three primary indicators of opioid prescribing practices among persons newly receiving opioid prescriptions: 1) monthly rate of incident opioid prescriptions (opioid prescription fill for someone who had not filled an opioid prescription in the prior six months), out of all persons without an observed opioid prescription in the previous six months; 2) days’ supply of the incident opioid prescription; and 3) daily morphine milligram equivalents (MME) of the incident opioid prescription, based on CDC MME conversion tables (Centers for Disease Control and Prevention, 2021) (additional details in Supplementary Material). We included only insurer-reimbursed outpatient medications, and excluded all opioid co-formulations to treat coughs, colds, or allergies (Supplementary Table 1).
To control for temporal trends (changes in outcomes over time unrelated to the policies of interest) in prescribing of similar medications, we examined rate and days’ supply of incident benzodiazepine prescriptions, a controlled substance that is expected to be unaffected by the opioid policies of interest (Ranapurwala et al., 2021).
2.3. Exposures
We examined two interruption time points for potential impact on opioid prescribing trends: 1) a medical board investigative initiative and 2) state legislation. The first interruption was the Safe Opioids Prescribing Initiative (SOPI) (North Carolina Medical Society, 2017) introduced by the North Carolina Medical Board (NCMB) to identify providers who prescribe high-dose opioids and have had multiple opioid-related patient deaths within a 12-month period. SOPI was implemented on May 1, 2016, and intended to restrict high-dose and high-volume opioid prescribing, with special emphasis placed on persons with chronic pain. The second interruption point was the Strengthen Opioid Misuse Prevention (STOP) Act (North Carolina Medical Board, 2017), state legislation that among other provisions limits the maximum days’ supply of initial prescriptions for acute and postsurgical pain to five and seven days, respectively. STOP Act prescribing limits became effective January 1, 2018, and notably do not apply to chronic or cancer-related pain.
2.4. Pain indication and cancer history
We analyzed trends in new opioid prescribing for three pain indication groups classified based on Current Procedural Terminology (CPT) (Agency for Healthcare Research and Quality, 2019) and International Classification of Diseases (ICD) codes: 1) postsurgical, 2) acute, and 3) chronic pain (Brummett et al., 2017; Calcaterra et al., 2018; Centers for Disease Control and Prevention, 2013; Mundkur et al., 2018). We assumed that patients with a surgery had a postsurgical indication, patients without surgery who had an acute pain diagnosis had an acute pain indication, and patients without surgery or acute pain who had a chronic pain diagnosis had a chronic pain indication (additional details in Supplementary Material and Supplementary Tables 2 & 3).
We assessed trends for each pain indication group separately for persons with and without cancer history, based on ICD codes and excluding benign neoplasm, non-melanoma skin cancer, neoplasm of uncertain or unspecified behavior, and carcinoma in situ (additional details in Supplementary Material).
2.5. Main analyses
2.5.1. Controlled interrupted time series analyses
We conducted controlled interrupted time series (CITS) analyses using autoregressive integrated moving average (ARIMA) models (Bernal et al., 2017; Bernal et al., 2018) to assess changes in opioid prescribing rates and mean days’ supply relative to the two policies, controlling for temporal trends in benzodiazepines prescribing practices. Since there is little variation in standardized dosing of benzodiazepines, we did not assess mean daily MME with a control series. Additional details can be found in Supplementary Material.
2.5.2. Single-series interrupted time series analyses
To examine the potential underestimation of policy impacts on opioid prescribing rates and days’ supply with the use of benzodiazepines as a control series, we conducted single ITS analyses (without a control series) on all outcomes relative to the policies of interest according to the ARIMA procedures described for the CITS analyses.
All results are reported with 95% CIs to provide a measure of precision, rather than as a test of statistical significance, as recommended the American Statistical Association and major medical journals to de-emphasize drawing conclusions based solely on statistical thresholds (Amrhein, 2019; Wasserstein and Lazar, 2016; International Committee of Medical Journal Editors, 2013).
2.6. Sensitivity analysis
In response to the STOP Act, in April 2018, the private health insurer implemented internal reimbursement policies requiring a limit of seven days’ supply for initial immediate-release opioid prescriptions for acute pain and prior authorization for initial extended-release prescriptions. Since the policy was implemented to comply with the STOP Act, it is viewed as part of the total effect of the STOP Act rather than a separate policy event. To examine the robust findings, we conducted sensitivity analyses for the outcome of days’ supply by changing the exposure date from the STOP Act enactment on January 1, 2018 to the reimbursement policy start date on April 1, 2018.
2.6. Ethical approval
This study was approved by the Institutional Review Board of the University of North Carolina at Chapel Hill.
3. RESULTS
3.1. Medical Board Initiative
3.1.1. Controlled interrupted time series analysis
3.1.1.1. Overall
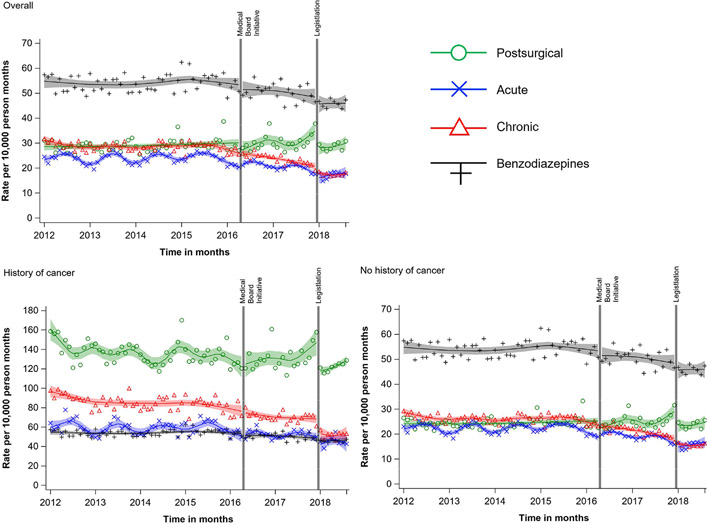
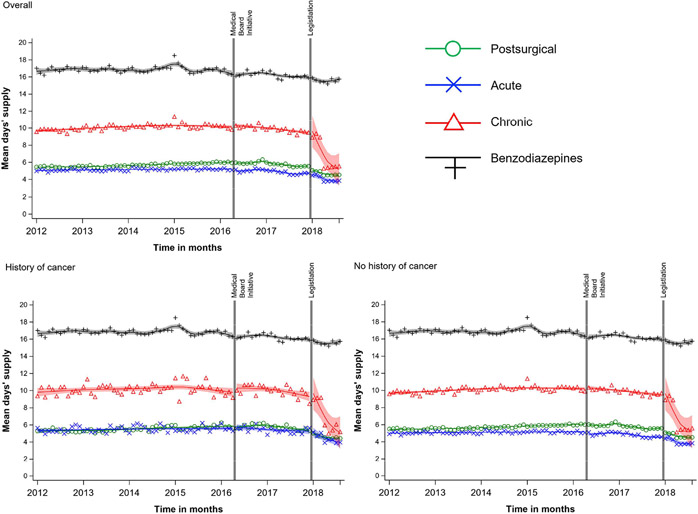
In CITS analyses, following the medical board initiative, there were no notable immediate changes in new opioid prescribing rates for all pain indication groups. However, in the 20 months between the medical board initiative and the legislative action, new opioid prescribing trends in the postsurgical pain group increased at an annualized incidence rate of 3.45 (95% CI: 0.89, 6.01) new prescriptions per 10,000 person months, relative to benzodiazepines. No marked relative trend changes were detected in the other pain indication groups (Table 1; Figure 1). Mean days’ supply of opioids showed no apparent changes before and after the medical board initiative, relative to mean days’ supply of benzodiazepine prescriptions (Table 1; Figure 2).
Table 1.
Association of two statewide policies on the prescribing rate and mean days’ supply of initial opioid prescriptions, by pain indication overall and within cancer status – controlled ITS
| pre-Medical Board Initiative |
post-Medical Board Initiative |
post-Legislation | |||
|---|---|---|---|---|---|
| Outcomesa | Trendb (95% CI) |
Absolute differencec (95% CI) |
Change in trendb,d (95% CI) |
Absolute differencec (95% CI) |
Change in trendb,d (95% CI) |
| β7 | β8 | β9 | β10 | β11 | |
| Prescribing rate e | |||||
| Overall | |||||
| Postsurgical pain | 0.08 (−0.44, 0.59) | 1.66 (−1.25, 4.57) | 3.45 (0.89, 6.01) | −0.73 (−6.75, 5.29) | −0.44 (−13.22, 12.35) |
| Acute pain | −0.39 (−0.82, 0.04) | 1.45 (−1.09, 3.99) | 0.20 (−2.04, 2.45) | 0.67 (−4.84, 6.18) | 2.01 (−9.69, 13.71) |
| Chronic pain | −0.59 (−0.85, −0.33) | 1.14 (−0.45, 2.73) | −0.94 (−2.35, 0.47) | 0.01 (−3.71, 3.74) | 1.12 (−6.83, 9.07) |
| History of cancer | |||||
| Postsurgical pain | −3.20 (−4.91, −1.49) | −3.41 (−12.78, 5.97) | 13.65 (5.41, 21.89) | −20.47 (−39.00, −1.93) | 10.99 (−28.49, 50.47) |
| Acute pain | −0.93 (−1.88, 0.02) | −2.74 (−8.30, 2.81) | 2.31 (−2.58, 7.21) | −3.31 (−14.99, 8.37) | −1.84 (−26.71, 23.02) |
| Chronic pain | −2.74 (−3.65, −1.83) | −4.28 (−9.22, 0.66) | 1.28 (−3.07, 5.62) | −12.42 (−22.25, −2.58) | 1.73 (−19.01, 22.46) |
| No history of cancer | |||||
| Postsurgical pain | −0.04 (−0.53, 0.45) | 1.69 (−1.08, 4.46) | 3.23 (0.80, 5.66) | 0.20 (−5.52, 5.92) | −1.31 (−13.45, 10.84) |
| Acute pain | −0.45 (−0.88, −0.03) | 1.58 (−0.93, 4.09) | 0.19 (−2.03, 2.41) | 0.82 (−4.59, 6.24) | 2.19 (−9.30, 13.68) |
| Chronic pain | −0.63 (−0.90, −0.37) | 1.34 (−0.27, 2.95) | −0.92 (−2.35, 0.51) | 0.66 (−3.07, 4.40) | 0.96 (−7.01, 8.92) |
| Mean days’ supply | |||||
| Overall | |||||
| Postsurgical pain | 0.16 (0.05, 0.27) | 0.40 (−0.09, 0.89) | −0.14 (−0.59, 0.31) | −0.23 (−0.93, 0.46) | −0.74 (−2.36, 0.88) |
| Acute pain | 0.05 (−0.05, 0.15) | 0.29 (−0.17, 0.75) | −0.03 (−0.44, 0.39) | 0.15 (−0.53, 0.83) | −1.07 (−2.61, 0.46) |
| Chronic pain | 0.13 (0.03, 0.24) | 0.38 (−0.14, 0.90) | −0.37 (−0.83, 0.09) | 0.40 (−0.44, 1.25) | −6.92 (−8.75, −5.09) |
| History of cancer | |||||
| Postsurgical pain | 0.16 (0.05, 0.27) | 0.48 (−0.03, 0.99) | −0.13 (−0.59, 0.33) | −0.23 (−0.98, 0.53) | −0.76 (−2.47, 0.96) |
| Acute pain | 0.06 (−0.07, 0.19) | 0.39 (−0.22, 0.99) | 0.01 (−0.54, 0.56) | 0.16 (−0.77, 1.10) | −1.74 (−3.80, 0.33) |
| Chronic pain | 0.06 (−0.09, 0.21) | 0.67 (−0.07, 1.40) | −0.41 (−1.06, 0.25) | 0.65 (−0.57, 1.86) | −7.22 (−9.82, −4.61) |
| No history of cancer | |||||
| Postsurgical pain | 0.16 (0.05, 0.27) | 0.39 (−0.10, 0.88) | −0.14 (−0.59, 0.30) | −0.23 (−0.93, 0.47) | −0.72 (−2.35, 0.91) |
| Acute pain | 0.04 (−0.06, 0.14) | 0.29 (−0.17, 0.74) | −0.05 (−0.46, 0.36) | 0.17 (−0.51, 0.85) | −0.99 (−2.51, 0.54) |
| Chronic pain | 0.14 (0.03, 0.25) | 0.33 (−0.20, 0.86) | −0.36 (−0.83, 0.12) | 0.35 (−0.51, 1.21) | −6.87 (−8.74, −5.01) |
Abbreviations: Medical Board Initiative = Safe Opioid Prescribing Initiative; Legislation = Strengthen Opioid Misuse Prevention Act; CI = confidence interval.
New prescription opioid patient population includes person-months where the individual has been insured continuously for >=6 months and has no opioid prescription in the prior 6 months.
Trends calculated per year.
Absolute difference in opioid prescribing relative to benzodiazepine prescribing.
Change in trend in opioid prescribing relative to benzodiazepine prescribing.
Prescribing rates per 10,000 insured person-months.
Figure 1. New prescription opioid rates, per 10,000 person-months, by pain indication overall and within cancer history status.
Medical Board Initiative = Safe Opioid Prescribing Initiative; Legislation = Strengthen Opioid Misuse Prevention Act. New prescription opioid patient population includes person-months where the individual has been insured continuously for ≥6 months and has no opioid prescription in the prior 6 months. Note the change in y-axis for persons with a history of cancer.
Figure 2. Mean days’ supply of new opioid prescriptions, by pain indication overall and within cancer history status.
Medical Board Initiative = Safe Opioid Prescribing Initiative; Legislation = Strengthen Opioid Misuse Prevention Act. New prescription opioid patient population includes person-months where the individual has been insured continuously for ≥6 months and has no opioid prescription in the prior 6 months.
3.1.1.2. By cancer history
Similar to overall trends, postsurgical prescribing rates among persons with and without a cancer history largely increased after the initiative (cancer: 13.65 [95% CI: 5.41, 21.89]; no cancer: 3.23 [95% CI: 0.80, 5.66] new prescriptions per 10,000 per year); we observed no pronounced increases in the other pain groups by cancer history (Table 1; Figure 1). Level and trend changes in mean days’ supply among persons with and without a cancer history were similar to overall trends (Table 1; Figure 2).
3.1.2. Single-series interrupted time series analysis
3.1.2.1. Overall
Post-medical board initiative, prescribing rates in persons with chronic pain decreased (level: −1.99 [95% CI: −3.39, −0.58] new prescriptions per 10,000 person months; trend: −2.57 [95% CI: −3.87, −1.27] new prescriptions per 10,000 per year); we did not observe evidence of immediate or trend declines among postsurgical or acute pain groups within the single-series models (Table 2; Figure 1). Although no immediate changes were observed for mean days’ supply, prescribing trends decreased in all three pain groups (postsurgical: −0.49 [95% CI: −0.65, −0.33; acute: −0.37 [95% CI: −0.54, −0.19]; chronic: −0.68 [95% CI: −1.14, −0.23] days’ supply per year] (Table 2; Figure 2). For mean daily MME in persons with postsurgical pain, the medical board initiative coincided with an immediate drop (−2.39 [95% CI: −3.43, −1.34] daily MME) and a further decline in trend (−1.34 [95% CI: −2.26, −0.41] daily MME per year). In those with acute pain, the initiative coincided with an immediate drop (−0.70 [95% CI: −1.33, −0.08]) in daily MME, but no apparent change in trend. In those with chronic pain, no changes in level or trend were evident at the time of the medical board initiative (Table 2; Figure 3).
Table 2.
Association of two statewide policies on the prescribing rate, mean days’ supply, and mean daily MME of initial opioid prescriptions, by pain indication overall and within cancer status – single-series ITS
| pre-Medical Board Initiative |
post-Medical Board Initiative |
post-Legislation | |||
|---|---|---|---|---|---|
| Outcomesa | Trendb (95% CI) |
Absolute difference (95% CI) |
Change in trendb (95% CI) |
Absolute difference (95% CI) |
Change in trendb (95% CI) |
| β1 | β2 | β3 | β4 | β5 | |
| Prescribing rate c | |||||
| Overall | |||||
| Postsurgical pain | 0.19 (−0.31, 0.69) | −0.48 (−1.90, 0.95) | 0.88 (−0.58, 2.33) | −2.49 (−5.58, 0.60) | 1.90 (−4.76, 8.55) |
| Acute pain | −0.23 (−0.87, 0.41) | −1.16 (−3.22, 0.89) | −1.65 (−3.85, 0.56) | −0.67 (−3.57, 2.23) | −0.36 (−7.51, 6.78) |
| Chronic pain | −0.34 (−0.70, 0.02) | −1.99 (−3.39, −0.58) | −2.57 (−3.87, −1.27) | −3.37 (−5.95, −0.78) | 2.05 (−3.46, 7.55) |
| History of cancer | |||||
| Postsurgical pain | −3.79 (−6.70, −0.89) | −3.89 (−14.26, 6.48) | 10.49 (0.41, 20.56) | −16.71 (−35.79, 2.38) | 6.43 (−34.12, 46.98) |
| Acute pain | −0.89 (−2.70, 0.92) | −6.11 (−13.58, 1.35) | 1.35 (−5.43, 8.12) | −6.07 (−17.55, 5.40) | −3.77 (−29.56, 22.02) |
| Chronic pain | −2.07 (−3.96, −1.45) | −6.44 (−12.31, −0.58) | −0.45 (−5.67, 4.77) | −16.58 (−26.59, −6.57) | 4.73 (−16.45, 25.91) |
| No history of cancer | |||||
| Postsurgical pain | 0.09 (−0.35, 0.53) | −0.59 (−1.87, 0.68) | 0.79 (−0.51, 2.09) | −2.01 (−4.77, 0.74) | 1.34 (−4.61, 7.29) |
| Acute pain | −0.29 (−0.89, 0.32) | −0.84 (−2.83, 1.15) | −1.83 (−3.95, 0.30) | −0.42 (−3.19, 2.36) | 0.10 (−6.77, 6.97) |
| Chronic pain | −0.37 (−0.71, −0.03) | −1.85 (−3.20, −0.50) | −2.56 (−3.80, −1.31) | −2.69 (−5.16, −0.23) | 1.81 (−3.44, 7.06) |
| Mean days’ supply | |||||
| Overall | |||||
| Postsurgical pain | 0.15 (0.11, 0.19) | 0.10 (−0.07, 0.27) | −0.49 (−0.65, −0.33) | −0.44 (−0.68, −0.20) | −0.65 (−1.23, −0.08) |
| Acute pain | 0.04 (0.00, 0.08) | −0.03 (−0.23, 0.16) | −0.37 (−0.54, −0.19) | −0.09 (−0.39, 0.21) | −1.01 (−1.69, −0.33) |
| Chronic pain | 0.13 (0.02, 0.24) | 0.02 (−0.49, 0.54) | −0.68 (−1.14, −0.23) | 0.08 (−0.73, 0.89) | −6.80 (−8.61, −4.99) |
| History of cancer | |||||
| Postsurgical pain | 0.15 (0.10, 0.20) | 0.16 (−0.08, 0.40) | −0.47 (−0.68, −0.25) | −0.51 (−0.89, −0.13) | −0.62 (−1.44, 0.21) |
| Acute pain | 0.06 (−0.02, 0.14) | 0.05 (−0.33, 0.43) | −0.33 (−0.67, 0.01) | −0.07 (−0.68, 0.55) | −1.68 (−3.02, −0.35) |
| Chronic pain | 0.06 (−0.09, 0.21) | 0.27 (−0.45, 0.99) | −0.69 (−1.33, −0.04) | 0.26 (−0.93, 1.45) | −7.08 (−9.62, −4.55) |
| No history of cancer | |||||
| Postsurgical pain | 0.15 (0.11, 0.19) | 0.09 (−0.09, 0.26) | −0.50 (−0.66, −0.34) | −0.44 (−0.70, −0.19) | −0.64 (−1.22, −0.05) |
| Acute pain | 0.04 (−0.01, 0.08) | −0.04 (−0.25, 0.17) | −0.38 (−0.57, −0.20) | −0.08 (−0.39, 0.24) | −0.92 (−1.63, −0.21) |
| Chronic pain | 0.14 (0.03, 0.25) | −0.02 (−0.55, 0.50) | −0.67 (−1.14, −0.20) | 0.03 (−0.80, 0.86) | −6.75 (−8.61, −4.89) |
| Mean Daily MME | |||||
| Overall | |||||
| Postsurgical pain | −0.72 (−0.93, −0.50) | −2.39 (−3.43, −1.34) | −1.34 (−2.26, −0.41) | −1.18 (−2.90, 0.54) | −2.80 (−6.50, 0.89) |
| Acute pain | −0.89 (−1.02, −0.76) | −0.70 (−1.33, −0.08) | −0.39 (−0.94, 0.17) | 0.28 (−0.75, 1.31) | −1.11 (−3.32, 1.11) |
| Chronic pain | −0.70 (−0.80, −0.59) | −0.42 (−0.93, 0.10) | −0.16 (−0.62, 0.30) | −0.50 (−1.36, 0.35) | 3.82 (1.99, 5.64) |
| History of cancer | |||||
| Postsurgical pain | −0.84 (−1.13, −0.55) | −1.72 (−3.10, −0.33) | −1.56 (−2.79, −0.32) | −1.68 (−3.97, 0.62) | −1.26 (−6.18, 3.65) |
| Acute pain | −0.74 (−1.10, −0.38) | 0.14 (−1.61, 1.89) | −1.10 (−2.66, 0.45) | 0.75 (−2.14, 3.64) | −0.02 (−6.22, 6.18) |
| Chronic pain | −0.47 (−0.72, −0.23) | −1.22 (−2.41, −0.03) | −0.44 (−1.49, 0.62) | 0.31 (−1.65, 2.28) | 1.22 (−2.98, 5.43) |
| No history of cancer | |||||
| Postsurgical pain | −0.68 (−0.92, −0.44) | −2.53 (−3.71, −1.35) | −1.28 (−2.33, −0.24) | −1.06 (−3.00, 0.88) | −3.15 (−7.32, 1.02) |
| Acute pain | −0.91 (−1.05, −0.78) | −0.82 (−1.48, −0.15) | −0.30 (−0.89, 0.29) | 0.22 (−0.88, 1.31) | −1.24 (−3.60, 1.11) |
| Chronic pain | −0.73 (−0.85, −0.62) | −0.31 (−0.86, 0.24) | −0.13 (−0.62, 0.36) | −0.64 (−1.55, 0.28) | 4.30 (2.33, 6.26) |
Abbreviations: Medical Board Initiative = Safe Opioid Prescribing Initiative; Legislation = Strengthen Opioid Misuse Prevention Act; CI = confidence interval.
New prescription opioid patient population includes person-months where the individual has been insured continuously for >=6 months and has no opioid prescription in the prior 6 months.
Trends calculated per year.
Prescribing rates per 10,000 insured person-months.
Figure 3. Mean daily MME of new opioid prescriptions, by pain indication overall and within cancer history status.
Medical Board Initiative = Safe Opioid Prescribing Initiative; Legislation = Strengthen Opioid Misuse Prevention Act. New prescription opioid patient population includes person-months where the individual has been insured continuously for ≥6 months and has no opioid prescription in the prior 6 months.
3.1.2.2. By cancer history
Persons with chronic pain both with and without cancer history experienced an absolute change in post-initiative prescribing rates, with the largest drop among those with cancer history (cancer: −6.44 [95% CI: −12.31, −0.58]; no cancer: −1.85 (−3.20, −0.50] new prescriptions per 10,000 person months) (Table 2; Figure 1). Trend changes in prescribing rates among those without cancer history were similar to overall trends. Mean days’ supply level and trend changes did not differ by cancer history and were similar to overall (non-stratified) estimates (Table 2; Figure 2). Mean daily MME among those with and without cancer history closely mirrored overall changes in each pain group, with the exception of an immediate decrease in persons with chronic pain (−1.22 [95% CI: −2.42, −0.03] daily MME) (Table 2; Figure 3).
3.2. Legislative action
3.2.1. Controlled interrupted time series analysis
3.2.1.1. Overall
After implementation of the state legislation, relative to benzodiazepines, there were no apparent changes in level and trend of new opioid prescription rates in all pain indication groups overall (Table 1; Figure 1). Although days’ supply changes did not immediately occur after legislative action in the opioid prescription groups, the trend in mean days’ supply for chronic pain fell over the remainder of the study (−6.92 [95% CI: −8.75, −5.09] days’ supply per year relative to benzodiazepines), reflecting the sharp decline three months after the legislation. No marked declines in prescribing trends were observed in the remaining opioid groups (postsurgical: −0.74 [95% CI: −2.36, 0.88]; acute: −1.07 [95% CI: −2.61, 0.46]) days’ supply per year relative to benzodiazepine prescriptions) (Table 1; Figure 2).
3.2.1.2. By cancer history
Following the legislative action, new prescription rates in postsurgical and chronic pain groups with cancer history fell sharply relative to new benzodiazepine prescriptions (postsurgical cRD: −20.47 [95% CI: −39.00, −1.93]; chronic cRD: −12.42 [95% CI: −22.25, −2.58] per 10,000 person months); however, post-legislative trends did not substantially change in these groups (Table 1; Figure 1). Level and trend changes in prescribing rates among persons without a cancer history were similar to overall (non-stratified) trends. When stratifying mean days’ supply by persons with and without a cancer history, similar model estimates were identified for all pain groups for changes in levels and trends (Table 1; Figure 2).
3.2.2. Single-series interrupted time series analysis
3.2.2.1. Overall
After the state legislation, prescribing rates among those with chronic pain immediately decreased (−3.37 [95% CI: −5.95, −0.78] per 10,000 person months) without a notable corresponding trend change (2.05 [95% CI: −3.46, 7.55 per 10,00 person months per year) (Table 2; Figure 1). Mean days’ supply slightly dropped only in the postsurgical pain group (−0.44 [95% CI: −0.68, −0.20]); trends decreased in all three pain groups following the legislation, with the largest change in those with chronic pain (postsurgical: −0.65 [95% CI: −1.23, −0.08]; acute: −1.01 [95% CI: −1.69, −0.33]; chronic: −6.80 [95% CI: −8.61, −4.99] mean days’ supply per year) (Table 2; Figure 2). Postsurgical and acute pain groups evidenced no changes in level and trend of daily MME upon implementation of the legislative action. In the chronic pain group, no immediate differences in daily MME occurred after the state legislation (−0.50 [95% CI: −1.36, 0.35]); however, the daily MME trend increased relative to a previously decreasing change in trend (3.82 [95% CI: 1.99, 5.64]) (Table 2; Figure 3).
3.2.2.2. By cancer history
In stratified analyses, a sharp post-legislative decline was observed in prescribing rates among person with chronic pain and cancer history (−16.58 [95% CI: −26.59, −6.57] per 10,000 person months), with no observed trend changes, similar to the overall analyses (Table 2; Figure 1). For mean days’ supply and daily MME, persons with and without cancer history experienced prescribing trends similar to unstratified analyses (Table 2; Figures 2 & 3).
Results of the sensitivity analyses can be found in Supplementary Material.
4. DISCUSSION
The importance of implementing evidence-based, safe and appropriate opioid prescribing policies is undisputed (Dowell et al., 2019; Lowenstein et al., 2018), however, the potential misapplication of current policies remains understudied and may have substantial adverse implications for patient welfare. To understand the consequences of two opioid prescribing policies, we examined changes in initial opioid prescription rates, days’ supply, and daily MME for privately insured persons with acute, postsurgical, and chronic pain, overall and by cancer history, between 2012 and 2018.
In a prior study examining the impact of North Carolina policies on opioid prescribing practices in the same population (Maierhofer et al., 2021), we demonstrated that prescribing rates and days’ supply declined overall after both the statewide medical board initiative and legislative action. The present analysis presents a more nuanced picture considering pain indication and cancer history: some of the previously reported overall declines may have been driven by declines in unintended or unexpected subpopulations.
The first policy, the NCMB’s investigative initiative or SOPI (North Carolina Medical Society, 2017), intended to curtail opioid prescribing at high doses and high volume, especially in persons with chronic pain. However, we observed no overall changes in daily MME among patients with chronic pain, but immediate and sustained declines among patients with acute and postsurgical pain following the initiative. Post-initiative prescribing rates in single-series models were in accordance with the medical board’s intended consequences with immediate and sustained declines observed among persons with chronic pain overall and by cancer history; postsurgical and acute pain prescribing rates did not appear to be impacted by the initiative. Conversely, in models with a benzodiazepine control series, no apparent changes in prescribing rates occurred for those with chronic pain. For days’ supply, we observed trend declines overall and within most subgroups in single-series models, but there were no notable prescribing changes among those with chronic pain in controlled analyses. These differences between models demonstrate that at the time of the medical board initiative implementation, there were likely other co-interventions that impacted the prescribing rates and days’ supply of both benzodiazepines and opioids. Using another controlled substance as a control series allowed for the control of potential confounding from co-interventions, achieving more unbiased results than those from the single-series models.
The second policy, the STOP Act (North Carolina Medical Board, 2017), is a state legislative action created to address “overprescribing” of opioids, most directly through limiting initial outpatient prescriptions’ days’ supply for acute and postsurgical pain. Notably in CITS analyses, there was no evidence of overall prescribing declines in days’ supply for acute and postsurgical pain groups, but strong evidence of unintended trend declines in all chronic pain groups relative to benzodiazepines. In the single-series models, mean days’ supply trends modestly decreased in postsurgical and acute pain groups as intended by the initiative; however, comparatively, chronic pain prescriptions were more strongly impacted following the policy. Similar to the medical board initiative, the results from controlled analyses were likely more unbiased estimates of the impact of STOP Act in North Carolina.
Pain medicine societies and experts have expressed concerns regarding unintended effects of opioid prescribing guidelines and mandates (Centers for Disease Control and Prevention, 2019; Dowell et al., 2019; Kroenke et al., 2019; Ranapurwala et al., 2021), in particular the rapid tapering of opioid prescriptions in those with chronic pain, which is associated with overdose from illicit opioid use (Binswanger et al., 2020; Coffin et al., 2020; Hartung et al., 2020). Several studies using national databases demonstrate decreasing trends in high-dose opioid prescriptions and prescribing duration following the release of the 2016 CDC Guidelines (Bohnert et al., 2018; Goldstick et al., 2021). For example, a recent study using national claims data (Goldstick et al., 2021) found that opioid-naïve patients were less likely to receive initial opioid prescriptions ≥50 daily MME and received fewer days’ supply after the CDC guidelines. Although important for informing opioid prescribing policies, the bulk of such published studies have focused on the policies’ intended population, and not populations in which unintended prescribing consequences may occur (Agarwal et al., 2019; Hincapie-Castillo et al., 2021; Oueini et al., 2021). SOPI was modeled after the CDC guidelines for chronic pain and was implemented a month after the guidelines’ publication (North Carolina Medical Board, 2016; North Carolina Medical Society 2017), but through a granular examination by pain indication and cancer history, we observed the unintended consequences in prescribing practices described above, as well as those that occurred following the legislative action.
Three months following the STOP Act, in response to that legislation, the private health insurer implemented internal reimbursement policies requiring a limit of seven days’ supply for initial immediate-release opioid prescriptions for acute pain and prior authorization for initial extended-release prescriptions. When shifting the interruption timepoint from the legislation implementation date to the reimbursement policy start date, we observed considerable immediate declines in days’ supply among persons with chronic pain, with no apparent declines in other pain groups. The insurer’s policy changes likely contributed to the post-legislation trend declines observed in persons with chronic pain in the main analyses, and may have unintentionally reduced the days’ supply of chronic pain prescriptions instead of the intended acute and postsurgical pain prescriptions. It may be that chronic pain patients with previous or current opioid prescriptions who change health insurance providers are considered to be opioid-naïve by the insurance provider, thereby limiting prescriptions. While opioid prescribing remains high compared to the pre-2000 years, it is critical to avoid limiting legitimate opioid prescriptions to patients who can safely benefit from pharmacotherapy. Policies and mandates must be well communicated to both prescribers and insurers to avoid potentially harmful unintended effects. Additionally, future studies must examine longer follow-up periods to better understand the impact of the state legislation and reimbursement policy on days’ supply.
The unexpected prescribing reactions to these policies may partly be associated with providers’ perceived risk of regulatory or legal sanction for opioid prescribing (Bolen 2006; Jung and Reidenberg, 2006; Passik and Kirsh, 2006). Given the high frequency of opioid prescribing for acute pain, misapplication of guidelines may occur for this indication if prescribing fear is consistently present in providers’ minds without considering the intended population of a new policy. Prescribing fears may also have contributed to decreased prescribing rates in those with chronic pain after the implementation of the STOP Act, especially since the severity of the opioid epidemic intensified during this period. Additionally, although benzodiazepines were not targeted in these statewide policies, benzodiazepine prescribing may have been unintentionally influenced through provider fear of potential patient overdose with the co-prescribing of opioids and benzodiazepines (U.S. Food and Drug Administration, 2017); however, we assume influence to be minimal without the direct mention of benzodiazepines in SOPI and the STOP Act.
This study has several limitations. We focused analyses on persons for whom we had a reasonable basis of characterizing their pain indication as acute, postsurgical, or chronic using a hierarchical classification based on ICD and CPT codes. A large percentage (45%) of insured persons with a newly prescribed opioid did not have a contemporaneous ICD/CPT code on which to base a pain indication and were therefore excluded from analyses. Because we included only major surgeries in our postsurgical criteria and lacked information on dental procedures (not systematically collected within our data), persons with new opioid prescriptions related to minor surgeries or dental procedures with no other pain indication may account for a proportion of excluded persons. These exclusions may limit generalizability of the results, given this group had a higher rate of new opioid prescriptions than any of the other pain indication groups (data not shown). Results for the overall population, which includes the three pain groups and all other persons without a characterized pain indication, can be found in our prior publication (Maierhofer et al., 2021). Furthermore, the results of this study may not be generalizable to uninsured or publicly insured persons, or to US states with prescription opioid dispensing rates dissimilar to those of North Carolina. During the study period, North Carolina’s overall opioid prescribing trends were higher than the US average (Centers for Disease Control and Prevention, 2020), and consequently these findings are likely more applicable to the privately insured populations in bordering southeastern states with similar opioid prescribing patterns. Because claims data capture information on limited demographics, it was not possible to examine prescribing trends by race and ethnicity. Claims data reflect only prescription claims and not prescription opioid consumption or non-prescription opioid use. Lastly, the cancer group included all persons with a cancer history which may not reflect current cancer diagnosis, and opioid prescriptions in this group may not always be related to cancer pain.
4.1. Conclusions
Between 2016 and 2018, North Carolina introduced two statewide opioid prescribing policies intended to reduce overprescribing by investigating providers who prescribe high volumes of high-dose opioids and limiting the days’ supply of initial outpatient opioid prescriptions for persons with acute and postsurgical pain. We found mixed evidence on the potential impact of these policies. In each case, some prescribing indicators declined after the policy while others did not; where observed, declines were often in a group not intended to be impacted by the policy. The overarching goal of opioid prescribing policies is to improve quality of life through effective pain management while mitigating opioid-related consequences. Because providers face many nuances in both patient care and prescribing guidelines, clear evidence-based policies on volume and dose for the initiation, continuation, and tapered discontinuation of opioid prescriptions, by pain indication and cancer status, are vital to ensure the safe and legitimate prescribing of opioids. Such policies must be elastic in their conception to account for operating psychology of clinical spaces. No matter how well-intentioned and conceived, policies that do not anticipate potential adaptations and responses of insurers and other third-party payers may contribute to outcomes that run counter to the original intentions of the policies.
Supplementary Material
HIGHLIGHTS.
Impact of opioid prescribing policies were examined in privately insured population
Opioid prescribing policies were associated with changes in prescribing behavior
Post-policy prescribing declines were observed in intended and unintended groups
Opioid prescribing policies must consider both intended and unintended consequences
Role of Funding Source.
This work was supported by a grant from the National Institute on Drug Abuse (R21DA046048) and the Centers for Disease Control and Prevention (R01CE003009). Additional support was provided by the National Institute of Allergy and Infectious Diseases (NIAID T32AI007001; T32AI070114). The database infrastructure used for this project was supported by the Cecil G. Sheps Center for Health Services Research and the CER Strategic Initiative of UNC’s Clinical and Translational Science Award (UL1TR001111). The funding sources had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Conflict of Interest.
Dr. Dasgupta is supported by the US Food and Drug Administration (HHSF223201810183C). Dasgupta is a part-time methods consultant to the RADARS System which had no involvement in or knowledge of this manuscript. The RADARS System is supported by subscriptions from pharmaceutical manufacturers, and governmental and non-governmental agencies for data, research and reporting services. RADARS System is the property of Denver Health and Hospital Authority, a political subdivision of the State of Colorado (United States of America). Dasgupta does not accept compensation from pharmaceutical companies, distributors, or litigants. Courtney N. Maierhofer, Shabbar I. Ranapurwala, Bethany L. DiPrete, Naoko Fulcher, Christopher L. Ringwalt, Paul R. Chelminski, Timothy J. Ives, Vivian F. Go, and Brian W. Pence declare that they have no conflict of interest.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- Agarwal S, Bryan JD, Hu MM, Lee JS, Chua K-P, Haffajee RL, Brummett CM, Englesbe MJ, Waljee JF, 2019. Association of State Opioid Duration Limits With Postoperative Opioid Prescribing. JAMA Netw. Open 2(12), e1918361. 10.1001/jamanetworkopen.2019.18361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Agency for Healthcare Research and Quality, 2019. Healthcare Cost and Utilization Project (HCUP). HCUP Tools and Software. Accessed July 2021. Retrieved from https://www.hcup-us.ahrq.gov/tools_software.jsp. [Google Scholar]
- Amrhein V, Greenland S, McShane B, 2019. Scientists rise up against statistical significance. Nature. 567(7748), 305–307. 10.1038/d41586-019-00857-9. [DOI] [PubMed] [Google Scholar]
- Baker DW, 2017. History of The Joint Commission’s Pain Standards: Lessons for Today’s Prescription Opioid Epidemic. JAMA. 317(11), 1117–1118. 10.1001/JAMA.2017.0935. [DOI] [PubMed] [Google Scholar]
- Bernal JL, Cummins S, Gasparrini A, 2017. Interrupted time series regression for the evaluation of public health interventions: A tutorial. Int. J. Epidemiol 46(1), 348–355. 10.1093/ije/dyw098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernal JL, Cummins S, Gasparrini A, 2018. The use of controls in interrupted time series studies of public health interventions. Int. J. Epidemiol 47(6), 2082–2093. 10.1093/ije/dyy135. [DOI] [PubMed] [Google Scholar]
- Binswanger IA, Glanz JM, Faul M, Shoup JA, Quintana LM, Lyden J, Xu S, Narwaney KJ, 2020. The Association between Opioid Discontinuation and Heroin Use: A Nested Case-Control Study. Drug Alcohol. Depend 217, 108248. 10.1016/j.drugalcdep.2020.108248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohnert ASB, Guy GP, Losby JL, 2018. Opioid prescribing in the United States before and after the centers for disease control and prevention’s 2016 opioid guideline. Ann. Intern. Med 169(6), 367–375. 10.7326/M18-1243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohnert ASB, Ilgen MA, 2019. Understanding Links among Opioid Use, Overdose, and Suicide. NEJM. 380(1), 71–79. 10.1056/NEJMra1802148. [DOI] [PubMed] [Google Scholar]
- Bolen J, 2006. DEA, Administrative Action Statistics, and Pain Management: It Is Time to Get Real. Pain Med. 7(4), 358–359. 10.1111/j.1526-4637.2006.00187.x. [DOI] [PubMed] [Google Scholar]
- Brummett CM, Waljee JF, Goesling J, Moser S, Lin P, Englesbe MJ, Bohnert AS, Kheterpal S, Nallamothu BK, 2017. New Persistent Opioid Use After Minor and Major Surgical Procedures in US Adults. JAMA Surg. 152(6), e170504. 10.1001/jamasurg.2017.0504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calcaterra SL, Scarbro S, Hull ML, Forber AD, Binswanger IA, Colborn KL, 2018. Prediction of Future Chronic Opioid Use Among Hospitalized Patients. J. Gen. Intern. Med 33(6), 898–905. 10.1007/s11606-018-4335-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Centers for Disease Control and Prevention, 2013. Prescription Drug Overdose Data & Statistics: Guide to ICD-9-CM and ICD-10 Codes Related to Poisoning and Pain. Accessed July 2021. Retrieved from http://www.cdc.gov/nchs/icd.htm.
- Centers for Disease Control and Prevention, 2019. CDC Advises Against Misapplication of the Guideline for Prescribing Opioids for Chronic Pain. Accessed July 2021. Retrieved from https://www.cdc.gov/media/releases/2019/s0424-advises-misapplication-guideline-prescribing-opioids.html.
- Centers for Disease Control and Prevention, 2020. US Opioid Dispensing Rate Maps. Accessed July 2021. Retrieved from https://www.cdc.gov/drugoverdose/rxrate-maps/index.html.
- Centers for Disease Control and Prevention, 2021. National Center for Injury Prevention and Control. Opioid Overdose: Data Resources. Analyzing prescription data and morphine milligram equivalents (MME). Accessed July 2021. Retrieved from https://www.cdc.gov/opioids/data-resources/index.html. [Google Scholar]
- Coffin PO, Rowe C, Oman N, Sinchek K, Santos G-M, Faul M, Bagnulo R, Mohamed D, Vittinghoff E, 2020. Illicit opioid use following changes in opioids prescribed for chronic non-cancer pain. PLoS One. 15(5), e0232538. 10.1371/journal.pone.0232538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dowell D, Haegerich T, Chou R, 2016. CDC Guideline for Prescribing Opioids for Chronic Pain - United States, 2016. MMWR Recomm. Rep 65(1), 1–49. 10.15585/MMWR.RR6501E1. [DOI] [PubMed] [Google Scholar]
- Dowell D, Haegerich T, Chou R, 2019. No Shortcuts to Safer Opioid Prescribing. NEJM. 380(24), 2285–2287. 10.1056/NEJMP1904190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- General Assembly of North Carolina Session, 2017. Session Law 2017-74: House Bill 243. Accessed July 2021. Retrieved from https://www.ncmedboard.org/images/uploads/article_images/H243v7.pdf.
- Goldstick JE, Guy GP, Losby JL, Baldwin G, Myers M, Bohnert ASB, 2021. Changes in Initial Opioid Prescribing Practices After the 2016 Release of the CDC Guideline for Prescribing Opioids for Chronic Pain. JAMA Netw. Open 4(7), e2116860. 10.1001/jamanetworkopen.2021.16860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadlandsmyth K, Lund BC, Mosher HJ, 2019. Associations between initial opioid exposure and the likelihood for long-term use. J. Am. Pharm. Assoc 59(1), 17–22. 10.1016/j.japh.2018.09.005. [DOI] [PubMed] [Google Scholar]
- Hah JM, Bateman BT, Ratliff J, Curtin C, Sun E, 2017. Chronic Opioid Use after Surgery: Implications for Perioperative Management in the Face of the Opioid Epidemic. Anesth. Analg 125(5), 1733–1740. 10.1213/ANE.0000000000002458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hartung DM, Johnston KA, Hallvik S, Leichtling G, Geddes J, Hildebran C, Keast S, Chan B, Korthuis PT, 2020. Prescription Opioid Dispensing Patterns Prior to Heroin Overdose in a State Medicaid Program: a Case-Control Study. J. Gen. Intern. Med 35(11), 3188–3196. 10.1007/s11606-020-06192-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hincapie-Castillo JM, Easey T, Hernandez C, Maguire M, Usmani SA, Vouri SM, Goodin A, 2021. Changes in Guantity of Opioids Dispensed following Florida’s Restriction Law for Acute Pain Prescriptions. Pain Med. 22(8), 1870–1876. 10.1093/pm/pnab017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- International Committee of Medical Journal Editors (ICMJE), 2013. The New ICMJE Recommendations (August 2013). Accessed July 2022. Retrieved from http://www.icmje.org/news-and-editorials/new_rec_aug2013.html.
- Jung B, Reidenberg MM, 2006. The Risk of Action by the Drug Enforcement Administration Against Physicians Prescribing Opioids for Pain. Pain Med. 7(4), 353–357. 10.1111/j.1526-4637.2006.00164.x. [DOI] [PubMed] [Google Scholar]
- Kroenke K, Alford D, Argoff C, Canlas B, Covington E, Frank J, Haake KJ, Hanling S, Hooten WM, Kertesz SG, Kravitz RL, Krebs EE, Stanos SP, Sullivan M, 2019. Challenges with Implementing the Centers for Disease Control and Prevention Opioid Guideline: A Consensus Panel Report. Pain Med. 20(4), 724–735. 10.1093/PM/PNY307. [DOI] [PubMed] [Google Scholar]
- Lee B, Zhao W, Yang K-C, Ahn Y-Y, Perry BL, 2021. Systematic Evaluation of State Policy Interventions Targeting the US Opioid Epidemic, 2007-2018. JAMA Netw. Open 4(2), e2036687–e2036687. 10.1001/JAMANETWORKOPEN.2020.36687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y, Logan J, Paulozzi L, Zhang K, Jones C, 2013. Potential Misuse and Inappropriate Prescription Practices Involving Opioid Analgesics. Am. J. Manag. Care 19(8), 648–658. [PubMed] [Google Scholar]
- Lowenstein M, Grande D, Delgado MK, 2018. Opioid Prescribing Limits for Acute Pain — Striking the Right Balance. NEJM. 379(6), 504–506. 10.1056/NEJMp1803661. [DOI] [PubMed] [Google Scholar]
- Maierhofer CN, Ranapurwala SI, DiPrete BL, Fulcher N, Ringwalt CL, Chelminski PR, Ives TJ, Dasgupta N, Go VF, Pence BW, 2021. Association Between Statewide Opioid Prescribing Interventions and Opioid Prescribing Patterns in North Carolina, 2006–2018. Pain Med. 10.1093/pm/pnab181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mikosz CA, Zhang K, Haegerich T, Xu L, Losby JL, Greenspan A, Bladwin G, Dowell D, 2020. Indication-Specific Opioid Prescribing for US Patients with Medicaid or Private Insurance, 2017. JAMA Netw. Open 3(5), e204514. https://doi.org/doi: 10.1001/jamanetworkopen.2020.4514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mundkur ML, Rough K, Huybrechts KF, Levin R, Gagne JJ, Desai RJ, Patorno E, Choudhry NK, Bateman BT, 2018. Patterns of opioid initiation at first visits for pain in United States primary care settings. Pharmacoepidemiol. Drug Saf 27(5), 495–503. 10.1002/pds.4322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muzzio K, Chandler M, Painter JT, Dragic L, 2021. Characterizing Patients after Opioid Taper in a VA Medical Center. J. Pain. Palliat. Care Pharmacother 35(2), 84–90. 10.1080/15360288.2021.1900492. [DOI] [PubMed] [Google Scholar]
- National Academies of Sciences, Engineering, and Medicine, 2017. Pain Management and the Opioid Epidemic: Balancing Societal and Individual Benefits and Risks of Prescription Opioid Use. Washington, DC: The National Academies Press. Accessed July 2021. Retrieved from 10.17226/24781. [DOI] [PubMed] [Google Scholar]
- North Carolina Medical Board, 2016. North Carolina Medical Board Annual Report. Accessed July 2021. Retrieved from https://www.ncmedboard.org/images/uploads/disciplinary_reports/2016_Annual_Report1.pdf.
- North Carolina Medical Board, 2017. Bill Summary: The Strengthen Opioid Misuse Prevention (STOP) Act of 2017. Accessed July 2021. Retrieved from https://www.ncmedboard.org/images/uploads/article_images/The_STOP_Act_summary-OnLetterhead.pdf.
- North Carolina Medical Society, 2017. NC Medical Board Refines Safe Opioid Prescribing Initiative Criteria. Accessed July 2021. Retrieved from https://www.ncmedsoc.org/nc-medical-board-refines-safe-opioid-prescribing-initiative-criteria/.
- Oueini R, Goodin A, Vouri SM, Park H, Lo-Ciganic W-H, Hincapie-Castillo JM, 2021. Changes in Schedule II oral opioid volume dispensed in a private health plan following Florida’s Acute Pain Opioid Restriction Law. J. Manag. Care Spec Pharm 27(6), 779–784. 10.18553/jmcp.2021.27.6.779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pasricha SV, Tadrous M, Khuu W, Juurlink DN, Mamdani MM, Paterson JM, Gomes T, 2018. Clinical indications associated with opioid initiation for pain management in Ontario, Canada: a population-based cohort study. Pain. 159(8), 1562. 10.1097/J.PAIN.0000000000001242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Passik SD, Kirsh KL, 2006. Fear and Loathing in the Pain Clinic. Pain Med. 7(4), 363–364. 10.1111/j.1526-4637.2006.00189.x. [DOI] [PubMed] [Google Scholar]
- Perez HR, Buonora M, Cunningham CO, Heo M, Starrels JL, 2020. Opioid Taper Is Associated with Subsequent Termination of Care: a Retrospective Cohort Study. J. Gen. Intern. Med 35(1), 36–42. 10.1007/s11606-019-05227-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ranapurwala SI, Ringwalt CL, Pence BW, Schiro S, Fulcher N, McCort A, DiPrete BL, Marshall SW, 2020. State Medical Board Policy and Opioid Prescribing: A Controlled Interrupted Time Series. Am. J. Prev. Med 10.1016/j.amepre.2020.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shah A, Hayes C, Martin B, 2017. Factors Influencing Long-Term Opioid Use Among Opioid Naive Patients: An Examination of Initial Prescription Characteristics and Pain Etiologies. J. Pain 18(11), 1374–1383. 10.1016/J.JPAIN.2017.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- U.S. Food and Drug Administration, 2017. FDA Drug Safety Communication: FDA warns about serious risks and death when combining opioid pain or cough medicines with benzodiazepines; requires its strongest warning. FDA. Accessed July 2022. Retrieved from https://www.fda.gov/drugs/drug-safety-and-availability/fda-drug-safety-communication-fda-warns-about-serious-risks-and-death-when-combining-opioid-pain-or. [Google Scholar]
- Wasserstein RL and Lazar NA, 2016. The ASA Statement on p-Values: Context, Process, and Purpose, The American Statistician. 70(2), 129–133. 10.1080/00031305.2016.1154108 [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.