Abstract
Background:
Chronic dyspepsia's symptoms are frequently seen in primary to tertiary healthcare in Indonesia. This study aimed to describe the potential usability of pepsinogen (PG) values in determining gastric mucosal conditions, including superficial gastritis and atrophic gastritis.
Materials and Methods:
We recruited 646 adult dyspeptic patients and then analyzed PG values (including PGI, PGII, and PGI/II ratio) with endoscopic findings, gastric mucosal damages, and Helicobacter pylori infection. The gastric mucosal damage and H. pylori infection were evaluated using histological examination based on the updated Sydney system.
Results:
Among 646 enrolled patients, 308 (47.2%), 212 (32.8%), 91 (14.1%), 34 (5.2%), and 1 (0.2%) patient were diagnosed with normal mucosa, gastritis, reflux esophagitis, peptic ulcer disease, and gastric cancer, respectively. Significant differences in PGI, PGII, and PGI/II ratio values were observed among ethnic groups (all P < 0.01). The PGI and PGII levels were significantly higher and PGI/II was significantly lower in H. pylori-infected patients than in uninfected ones (all P < 0.001). The optimal cutoff value for PGII and PGI/II was 12.45 ng/mL with an area under the curve (AUC) value of 0.755 (0.702–0.811), sensitivity 59.3%, and specificity 77.1%; and 4.75 with AUC value of 0.821 (0.763–0.855), sensitivity 81.5%, and specificity 78.7%, respectively, to determine moderate–severe atrophy.
Conclusion:
Serum PG levels, a useful biomarker, represent the endoscopic findings, especially for reflux esophagitis. In addition, the benefits of PG values detecting atrophic gastritis were limited to moderate–severe atrophic gastritis. This usefulness requires careful attention for several ethnic groups in Indonesia.
Keywords: Atrophic gastritis, human and disease, Helicobacter pylori, Indonesia, neoplasms, pepsinogens, reflux esophagitis
INTRODUCTION
Chronic dyspepsia's symptoms are frequently seen in primary to tertiary healthcare in Indonesia,[1] with underlying pathologies such as atrophic gastritis, intestinal metaplasia, and peptic ulcers which may act as the cause of the symptoms.[2,3,4] Those pathologies are influenced by Helicobacter pylori infection, which is known as the carcinogenic bacteria and is suffered by approximately half of the world population.[5] Therefore, the diagnosis of H. pylori and gastric mucosa status are still concerning for clinicians. The accuracy of diagnosis and appropriate therapy need to be performed as early as possible to overcome chronic gastritis and prevent more severe clinical manifestations, such as gastric adenocarcinoma, which was reported to have only 25.1% of 5-year survival rate.[6]
The diagnostic methods determining the gastric mucosa condition and H. pylori status include invasive techniques, through endoscopic and biopsy sampling.[7] However, this method is less comfortable, relatively risky for patients, and considered an expensive examination in some places, especially in Indonesia. Serological tests including anti-H. pylori antibodies detection are more convenient and the result can be obtained faster. Pepsinogen (PG) I is mainly produced by the chief cells and mucus neck cells in the gastric fundus, while PGII is produced in the stomach with the addition of the proximal duodenum, and these can be examined from the serum specimen.[8] PGI and PGII are proposed to have good diagnostic values in predicting gastric mucosal status such as atrophic gastritis. Alongside serological tests for H. pylori infection, they may have a benefit as the gastric cancer screening method.[9] The combination of PGs and antibody H. pylori examination was proposed to improve diagnostic accuracy.[10] The ABC method, which was initially introduced by Miki et al., is a classification method to stratify gastric cancer risk based on the serum PG and H. pylori infection status. The ABC method consists of H. pylori-negative/PG-negative (group A), H. pylori-positive/PG-negative (group B), H. pylori-positive/PG-positive (group C), and H. pylori-negative/PG-positive (group D).[11] The use of ABC method is proven useful in countries with a high risk of H. pylori infection and gastric cancer, including China, Japan, Mongolia, and Bhutan.[12,13,14,15] However, the reliability of PGs and ABC method are still questionable when applied to different populations and regions.
Indonesia is the fourth largest populated country worldwide. It occupies a very wide area over numerous islands. In addition, it is inhabited by various ethnic groups with different hygiene and food habits. Although in the national survey, overall Indonesia had a low prevalence of H. pylori (10.4%),[16] there was a difference in ethnic prevalence; a lower prevalence was found in Javanese ethnic who mostly lived in the urban area with endoscopic facilities. However, the prevalence of H. pylori was high in several places, such as Jayapura (Papua Island), Makassar (Sulawesi Island), and Medan (Sumatra Island).[17] These places are considered to have remote areas that have limited access to endoscopic equipment. Therefore, a noninvasive reliable diagnostic method that can detect not only H. pylori infection but also gastric mucosal status, using PGs and/or ABC method, is necessary. In our previous study, the validation of PGs and H. pylori serology has been carried out only in three cities in Indonesia, and we found that PG can be beneficial.[18] However, it may not represent all of the Indonesian population. Therefore, a new survey involving populations in areas with higher H. pylori prevalence should be performed to examine the reliability of PGs use in a nationwide approach. In this study, we aimed to examine the reliability of serum PGs as a biomarker for gastroesophageal disease detection in Indonesia. We also described the diagnostic accuracy of ABC method in Indonesia. In addition, we analyzed the distribution of serum PGs secretion in various determinant factors (H. pylori infection, sex, age, and ethnicity.
MATERIALS AND METHODS
Study population
This cross-sectional study was conducted between October 2014 and March 2017, enrolling adult dyspeptic patients from 13 cities in Indonesia, including Aceh, Bangli, Cimacan, Gunung Sitoli, Kolaka, Kupang Manado, Merauke, Padang, Palembang, Palu, Surabaya, and Ternate. This study population also included 158 samples from our previous study.[18] The inclusion criteria for this study were adult dyspeptic patients. Exclusion factors were as follows: History of H. pylori eradication therapy, partial or total gastrectomy, contraindication to endoscopy, and non-fasting subjects. An experienced endoscopist acquired two gastric biopsy specimens during each endoscopic procedure and made a diagnosis of peptic ulcer, identifying the presence of a mucosal break due to reflux esophagitis. One specimen was obtained from the lesser curvature of the antrum, approximately 3 cm from the pyloric ring, and the other from the greater curvature of the corpus. Both specimens were histologically examined. Furthermore, on the endoscopy's day, fasting serum was collected and stored at −20°C. Furthermore, subjects were interviewed to obtain socio-demographic data: body mass index, smoking and drinking habits, and use of nonsteroidal anti-inflammatory drugs (NSAIDs). All participants signed written informed consent. The study protocol was approved by the ethics committees of Dr. Cipto Mangunkusumo Teaching Hospital (Jakarta, Indonesia), Dr. Soetomo Teaching Hospital (Surabaya, Indonesia), Dr. Wahidin Sudirohusodo Teaching Hospital (Makassar, Indonesia), and Oita University Faculty of Medicine (Yufu, Japan).
Histology and immunohistochemistry
Collected biopsy material was stored in 10% buffered formalin and then embedded in paraffin. Hematoxylin–eosin and May–Giemsa staining were performed on serial sections. The degree of inflammation, atrophy, and bacterial density was classified into four grades according to the updated Sydney System: 0, normal; 1, mild; 2, moderate; and 3, marked.[19] Samples with bacterial loads ≥grade 1 were considered positive for H. pylori. In addition, we also performed immunohistochemistry analysis to increase the diagnostic accuracy, as previously described.[20] We incubated the histology specimens using anti-α-H. pylori antibody (DAKO, Glostrup, Denmark) after inactivation of endogenous peroxidase activity. We then incubated the 2nd antibody using biotinylated goat anti-rabbit IgG (Nichirei Co., Tokyo, Japan), followed by avidin-conjugated horseradish peroxidase solution (Vectastain Elite ABC Kit; Vector Laboratories Inc., Burlingame, CA, USA) for attaching the peroxidase. Detection of peroxidase activity on the specimens was performed by H2O2/diaminobenzidine substrate solution. The experienced pathologist was examining our current specimen, who also examines our other works in Myanmar, Vietnam, Bhutan, Dominican Republic, and Indonesia.[21,22,23,24,25,26]
Determination of Helicobacter pylori serology and pepsinogen levels
Using separated sera, we measured both the H. pylori antibody titers with an enzyme linked immunosorbent assay (ELISA) kit (Eiken, Co. Ltd., Tokyo, Japan) and the PGI and PGII levels by using PG ELISA (Eiken), as per the manufacturer's instruction. H. pylori was considered positive if its antibody titers were ≥10U/mL. The PGI and PGI/II ratio were interpreted as PG positive if the PGI levels ≤70 ng/mL and PGI/II ratio ≤3.0, according to the Miki criteria, which is commonly used in Japan.[11] In addition, we performed the ABC method evaluation for gastric cancer screening. Subjects were categorized into four groups: H. pylori-negative/PG-negative (group A), H. pylori-positive/PG-negative (group B), H. pylori-positive/PG-positive (group C), and H. pylori-negative/PG-positive (group D).[11]
Determination of disease
Experienced endoscopists observed the mucosal condition of the upper gastroduodenal tract from esophagus to duodenum. Reflux esophagitis was identified based on the observation of mucosal break on the gastroesophageal junction. The ulcerations in the stomach and duodenum were also identified based on endoscopic examination. Gastric cancer was determined based on endoscopic examination, confirmed by histopathology. The subjects without reflux esophagitis, ulcerations, and gastric cancer, including normal-looking mucosa, were further analyzed based on histological examination. The gastritis individuals were participants with the presence of neutrophil infiltration, monocyte infiltration, atrophy, or intestinal metaplasia. When the subjects did not have any histological gastric mucosal damage, we concluded these as a normal group.
As for atrophic gastritis, we simplified the classification of degree of atrophic gastritis due to a small number of moderate and marked. This classification based on the highest value of atrophic gastritis score in the histological evaluation. Mild atrophic gastritis is defined as score of atrophic gastritis equal to 1 in either antrum or corpus; when we observed score >1 of atrophic gastritis, we defined as moderate–severe. In addition, we also classified atrophic gastritis based on the topographical distribution. An individual was categorized as having antral-predominant gastritis if the atrophic scores in the antrum were greater than those in the corpus, whereas if the atrophic scores in the corpus were greater than those in the antrum, these were categorized as corpus-predominant gastritis. The pan-gastritis was determined if the atrophic gastritis scores both in the antrum and corpus were equal.[27,28,29]
Data analyses
Relationship between discrete variables was tested with a Chi-square test. Association of continuous variables was tested with the Mann–Whitney U- and Kruskal–Wallis tests. Normal distribution test for continuous variables was Shapiro–Wilk test. Discrete variables were presented as n number and percentage, while the continuous variables were presented as mean with standard deviation. The Spearman rank coefficients (r) were determined to evaluate the association between PG levels and gastric-mucosal inflammation and atrophy. A P < 0.05 was considered as statistically significant. Receiver-operating characteristic curves were used to calculate the best cutoff. These included the area under the curve (AUC) and predictive values for discriminating chronic and atrophic gastritis. Utilizing the cut-off value obtained from the ROC analysis, we then calculate the sensitivity, specificity, positive predictive value, negative predictive value, and overall accuracy. The SPSS statistical software package version 18.0 (SPSS Inc., Chicago, IL, USA) was used for all statistical analyses.
RESULTS
Baseline characteristics
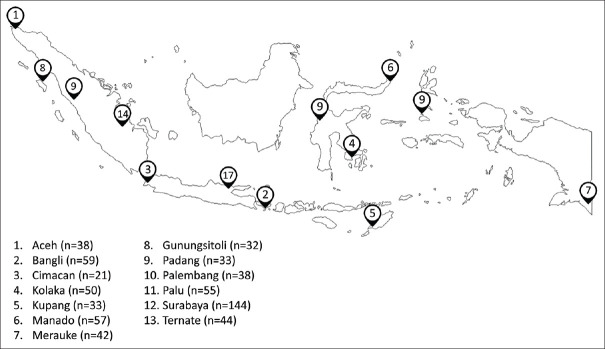
A total of 646 subjects which consisted of 383 males and 263 females were included with an average age of 44.93 ± 12.98 years (range, 14–83 years). Those patients were enrolled from Aceh (n = 38), Padang (n = 33), and Palembang (n = 38) in Sumatra island; Gunungsitoli (n = 32) in Nias island; Cimacan (n = 21) and Surabaya (n = 144) in Java island; Bangli (n = 59) in Bali island; Kolaka (n = 50), Manado (n = 57), and Palu (n = 55) in Sulawesi island; Kupang (n = 33) in Timor island; Merauke (n = 42) in Papua island; and Ternate (n = 44) in Ternate island [Figure 1]. The distribution of ethnicity in the city of endoscopy was described in Supplementary Table 1.
Figure 1.
Map of the enrolled patients in the current study. The map was drawn by the author showing the cities we visited on survey. We performed endoscopy examination to 646 patients from 13 cities in Indonesia
Supplementary Table 1.
The distribution of each ethnic group based on Island and City in our current study
| Ethnic | Island | City |
|---|---|---|
| Aceh | Sumatra | Banda Aceh and Padang |
| Balinese | Bali | Bangli |
| Bataknese | Sumatra | Palembang |
| Bugis | Sulawesi and Java | Kolaka, Palu and Surabaya |
| Chinese | Java and Ternate | Surabaya and Ternate |
| Dayak | Java | Surabaya |
| Javanese | Almost in all largest Island of Indonesia | Cimacan, Kupang, Manado, Palembang, Palu and Surabaya |
| Ternatese | Java and Ternate | Surabaya and Ternate |
| Malay | Sumatra | Palembang |
| Minahasanese | Sulawesi | Manado and Palu |
| Nias | Nias | Gunungsitoli |
| Kaili | Sulawesi | Palu |
| Papuan | Papua | Merauke |
| Timor | Timor | Kupang |
| Tolaki | Sulawesi | Kolaka |
We performed Shapiro–Wilk test and observed that the PGI, PGII, and PGI/II ratio were not normally distributed. Male patients had significantly lower PGI level and PGI/II ratio median values than female patients (42.9 vs. 48.6, P = 0.003 and 5.5 vs. 5.8, P = 0.024, respectively) [Table 1]. In addition, we also observed significant positive correlation between PGI value and PGII value with age (r = 0.377 and r = 0.359, respectively, both P < 0.001). Ethnic group influenced the PGI value and PGI/II ratio (both P < 0.001) with the lowest ethnic group being Tolaki for PGI and Bataknese for PGI/II ratio, whereas the highest was Chinese for both PGI and PGI/II ratio. PGII value was also influenced by the ethnic group (P = 0.015) with the lowest being Tolaki and the highest being Chinese [Table 1].
Table 1.
Pepsinogen I, pepsinogen II secretion, and the determinant factor
| Factors | n | Pepsinogen levels, mean±SD (median) | ||
|---|---|---|---|---|
|
| ||||
| PG I | PG II | PG I/II | ||
| Helicobacter pylori infection | ||||
| Positive | 59 | 77.5±77.6 (54.4)* | 19.4±16.8 (13.7)* | 4.1±1.2 (4.0)* |
| Negative | 587 | 61.6±50.8 (44.1) | 10.2±7.8 (7.9) | 6.0±1.74 (5.8) |
| P | 0.02 | <0.001 | <0.001 | |
| Sex | ||||
| Male | 383 | 61.5±55.8 (42.9)* | 11.0±10.1 (7.9) | 5.7±1.79 (5.5)* |
| Female | 263 | 65.2±51.0 (48.6) | 11.0±8.28 (8.8) | 6.0±1.75 (5.8) |
| P | 0.003 | 0.074 | 0.023 | |
| Age (years) | ||||
| <18 | 3 | 34.3±8.19 (35.7)‡ | 7.2±3.23 (9.0) | 5.7±2.3 (4.6) |
| 18-29 | 74 | 47.5±44.3 (38.5) | 9.1±7.68 (7.0)‡ | 5.4±1.4 (5.4) |
| 30-39 | 165 | 47.9±42.1 (37.0) | 8.3±6.53 (7.1) | 5.8±1.5 (5.7) |
| 40-49 | 155 | 61.5±48.7 (42.0) | 11.7±11.6 (7.5) | 5.7±1.5 (5.6) |
| 50-59 | 162 | 76.7±67.8 (54.3) | 12.3±9.46 (9.9) | 6.1±2.2 (5.8) |
| ≥60 | 87 | 83.2±49.6 (71.6)¶ | 14.4±9.35 (12.4)¶ | 6.0±1.9 (5.8) |
| P | <0.001 | <0.001 | 0.257 | |
| Ethnic | ||||
| Aceh | 70 | 46.2±28.7 (38.3) | 7.9±3.7 (7.5) | 5.8±1.6 (5.5) |
| Balinese | 61 | 64.8±61.2 (47.7) | 12.1±11.7 (9.0) | 5.6±1.3 (5.5) |
| Bataknese | 2 | 41.4±12.6 (41.4) | 10.1±3.11 (10.1) | 4.1±1.0 (4.1)‡ |
| Bugis | 69 | 64.4±59.5 (39.9) | 10.7±7.3 (7.6) | 5.8±2.1 (5.4) |
| Chinese | 40 | 85.3±55.1 (61.6)¶ | 13.3±8.7 (10.6)¶ | 6.7±2.1 (6.5)¶ |
| Dayak | 6 | 76.5±66.5 (49.6) | 12.1±8.1 (9.2) | 5.8±1.4 (6.1) |
| Javanese | 118 | 77.1±60.2 (62.5) | 12.4±10.9 (9.5) | 6.4±1.7 (6.4) |
| Ternatese | 46 | 59.2±59.2 (44.2) | 9.6±7.3 (7.4) | 6.0±1.4 (5.9) |
| Malay | 36 | 73.0±47.9 (59.1) | 11.8±7.9 (9.7) | 6.4±1.7 (5.8) |
| Minahasanese | 53 | 47.2±23.3 (44.3) | 8.3±3.7 (8.0) | 5.9±1.3 (5.8) |
| Nias | 32 | 61.4±42.8 (44.0) | 10.3±6.6 (8.4) | 6.2±2.1 (5.6) |
| Kaili | 12 | 49.8±31.6 (36.4) | 9.2±4.9 (7.8) | 5.5±1.7 (5.3) |
| Papuan | 43 | 49.6±35.5 (39.0) | 12.5±14.0 (9.1) | 4.7±1.5 (4.4) |
| Timor | 34 | 72.4±90.6 (46.2) | 14.6±14.9 (9.2) | 5.2±1.8 (4.7) |
| Tolaki | 24 | 42.0±30.5 (33.4)‡ | 9.8±9.0 (7.1)‡ | 4.6±0.9 (4.6) |
| P | <0.001 | 0.015 | <0.001 | |
*The calculation showed statistically significant (Wilcoxon rank sum test, all P<0.05), ‡The lowest group among determinant factor, ¶The highest group among determinant factor. PG=Pepsinogen; SD=Standard deviation
According to the H. pylori infection status by histology and immunohistochemistry, PGI level was significantly higher in H. pylori-positive than in H. pylori-negative patients (P = 0.002). In addition, PGII level was significantly higher in H. pylori-positive than in H. pylori-negative patients [P < 0.001, Table 1], whereas the PGI/II ratios were significantly lower in H. pylori-positive than in H. pylori-negative patients (P < 0.001).
PGI of gastritis and reflux esophagitis patients was significantly higher than normal patients (P = 0.010 and P = 0.002, respectively) [Table 2]. Gastritis patients had significantly higher PGII than normal patients (P < 0.001). Reflux esophagitis patients had significantly higher PGI/II than peptic ulcer disease (PUD), normal, and gastritis patients (P = 0.002, P < 0.001, and P < 0.001, respectively) [Table 2].
Table 2.
Pepsinogen level and disease
| PG level | Normal | Gastritis | Reflux esophagitis | PUD | Gastric cancer |
|---|---|---|---|---|---|
| n | 308 | 212 | 91 | 34 | 1 |
| PG I | 55.0±43.0 | 63.4±53.8* | 87.4±75.4* | 65.4±55.6 | 123.0 |
| PG II | 9.4±6.6 | 12.1±10.1 | 12.7±10.6 | 12.6±10.9 | 93.1 |
| PG I/II | 5.9±1.6 | 5.6±1.9 | 6.7±1.8** | 5.4±1.4 | 1.3 |
*PG I level was significantly higher than normal individuals, **PG I/II level was significantly higher compared to normal, gastritis and PUD groups. PUD=Peptic ulcer disease; PG=Pepsinogen
Pepsinogen levels and atrophic gastritis
We observed 122 (20.8%) patients had atrophy either in the antrum or corpus based on histological examination of atrophy score ≥ 1. The patients with atrophic gastritis had significantly higher PGII values and significantly lower PGI/II ratio than nonatrophic gastritis individuals (median = 9.6 vs. 7.9, P = 0.001 and 5.4 vs. 5.7, P = 0.002, respectively), but no significant association was observed on the PGI level [Table 3]. In addition, among those patients with gastritis atrophy, we observed 154 patients (93.0%), 3 patients (1.8%), and 14 patients (8.2%) were classified as antral predominant, corporal predominant, and pan-gastritis, respectively. However, we could not find any significant difference of PG levels among predominant locations.
Table 3.
The pepsinogen levels between atrophic status, predominant location and severity of atrophic gastritis
| n | PG I | PG II | PG I/PG II | |
|---|---|---|---|---|
| Atrophy status | ||||
| Nonatrophy | 475 | 62.0±60.9 | 10.6±9.4 | 6.0±1.7 |
| Atrophy | 171 | 65.9±59.8 | 12.3±9.2* | 5.6±2.0* |
| P | 0.058 | <0.001 | 0.001 | |
| Predominant location | ||||
| Antral | 154 | 65.9±60.9 | 12.0±8.9 | 5.7±2.0 |
| Corporal | 3 | 36.5±9.4 | 7.4±1.1 | 5.0±1.5 |
| Pan-gastritis | 14 | 72.6±53.4 | 17.2±12.0 | 4.6±1.7 |
| P | 0.135 | 0.098 | 0.587 | |
| Degree of atrophy | ||||
| Mild | 144 | 67.7±64.3 | 11.9±9.6 | 5.8±1.9 |
| Moderate-severe | 27 | 56.3±22.5 | 14.4±6.3** | 4.1±1.2** |
| P | 0.543 | 0.001 | <0.001 |
*The calculation showed statistically significant compared to nonatrophic gastritis patients (Wilcoxon rank-sum test, all P<0.05), **The calculation showed statistically significant compared to mild degree of atrophic gastritis patients (Wilcoxon rank-sum test, all P<0.05). PG=Pepsinogen
We also classified atrophic gastritis based on the severity observed by histological examination. Due to the low number of moderate and severe atrophic gastritis, we combined them into one group. Among all atrophic gastritis patients, we found 144 (84.2%) patients had mild atrophic lesion, while 27 (15.8%) patients had moderate–severe atrophic lesion. The PGII value of moderate–severe atrophy patients was significantly higher than mild atrophy patients (median = 13.5 vs. 9.2, P = 0.001), whereas the PGI/II ratio of moderate–severe atrophy patients was significantly lower than mild atrophy patients (4.1 vs. 5.6, P < 0.001). In addition, we observed a significant positive correlation between PGII value and antral atrophic score based on Sydney System (r = 0.263, P < 0.001), but not in the corpus, and a significant negative correlation between PGI/II and antral atrophic score (r = −0.316, P < 0.001). When we analyzed the presence of inflammation (score of monocyte or neutrophil infiltration ≥1) in the antrum and corpus, the PGI and PGII values were significantly higher in the inflammation group than noninflammation one (both P < 0.001). The PGI/II ratio was significantly lower in inflammation group than noninflammation one (P < 0.001).
Value of pepsinogen for atrophic gastritis
Based on the criteria by Miki et al.,[11] values ≤70 ng/mL for PG I level and ≤3.0 for PG I/II ratio were considered the PG-positive to detect gastric atrophy. By using the cutoff, we observed only 17 patients (2.6%) were considered as PG-positive group. By using histological examination of atrophic either in the antrum or corpus ≥1 as the positive group, we found that sensitivity and specificity were 7.6% (4.5–9.2) and 99.2% (98.2–99.8), respectively [Supplementary Table 2].
Supplementary Table 2.
The classification based on ABC method based on ethnic group
| Ethnic | n | Group A (%) | Group B (%) | Group C (%) | Group D (%) |
|---|---|---|---|---|---|
| Aceh | 70 | 49 (70) | 1 (1.4) | 0 | 20 (28.6) |
| Balinese | 61 | 44 (72.1) | 3 (4.9) | 6 (9.8) | 8 (13.1) |
| Batak | 2 | 0 | 0 | 0 | 2 (100) |
| Bugis | 69 | 42 (60.9) | 4 (5.8) | 8 (11.6) | 15 (21.7) |
| Chinese | 40 | 35 (87.5) | 0 | 2 (5) | 3 (7.5) |
| Dayak | 6 | 5 (83.3) | 0 | 0 | 1 (16.7) |
| Javanese | 118 | 103 (87.3) | 0 | 1 (0.8) | 14 (11.9) |
| Ternatese | 46 | 39 (84.8) | 1 (2.2) | 2 (4.3) | 4 (8.7) |
| Melayu | 36 | 31 (86.1) | 0 | 1 (2.8) | 4 (11.1) |
| Minahasa | 53 | 38 (71.7) | 4 (7.5) | 2 (3.8) | 9 (16.9) |
| Nias | 32 | 24 (75) | 1 (3.1) | 1 (3.1) | 6 (18.8) |
| Kaili | 12 | 8 (66.7) | 1 (8.3) | 1 (8.3) | 2 (16.7) |
| Papuan | 43 | 20 (46.5) | 0 | 9 (20.9) | 14 (32.6) |
| Timor | 34 | 14 (41.2) | 2 (5.9) | 9 (26.5) | 9 (26.5) |
| Tolaki | 24 | 10 (41.7) | 0 | 1 (4.2) | 13 (54.2) |
Considering a low value of sensitivity for the criteria, we recalculated the cutoff value of those measurements. By using atrophy score ≥ 1 as the standard determining positive group, we observed very low AUC value for PGI, PGII, and PGI/II ratio (0.549, 0.589, and 0.581, respectively); thus, we may not consider atrophy score ≥1 as a good standard. When we considered atrophic score ≥2 as the standard determining positive group, we observed that AUC for PGI, PGII, and PGI/II ratio was 0.587, 0.755, and 0.821, respectively [Table 4]. As PGI had a considerably very low AUC value, we only determined the cutoff value for PGII and PGI/II ratio. The optimal cutoff value for PGII was 12.45 ng/mL and PGI/II ratio was 4.75 respectively [Table 4]. With the PGII cutoff of ≥12.45 ng/mL, the sensitivity and specificity were 59.3% and 77.1%, respectively. When utilizing the PGI/II of ≤4.75 as the cutoff, we observed that the sensitivity and specificity were 81.5% and 78.7%, respectively [Table 4]. When we considered using either PGII or PGI/II ratio to determine moderate–severe atrophic data, we found that the sensitivity and specificity were 85.2% and 60.8%, respectively. On the other hand, when using both PGII and PGI/II ratio, we found that the sensitivity and specificity were 55.6% and 94.9%, respectively.
Table 4.
The validation parameters for pepsinogen levels determining moderate-severe atrophy in Indonesia
| Disease group | Parameters | Serum PG (95% CI) | ||
|---|---|---|---|---|
|
| ||||
| PG I | PG II | PG I/II | ||
| Moderate-severe atrophy | Cutoff value | - | ≥12.45 ng/mL | ≤4.75 |
| AUC | 0.587 (0.512-0.622) | 0.755 (0.702-0.811) | 0.821 (0.763-0.855) | |
| Sensitivity (%) | - | 59.3 (38.8-77.6) | 81.5 (61.9-93.7) | |
| Specificity (%) | - | 77.1 (73.0-80.8) | 78.7 (74.3-82.3) | |
| PPV (%) | - | 12.8 (9.3-17.3) | 17.9 (14.5-28.9) | |
| NPV (%) | - | 97.1 (95.5-98.3) | 98.7 (97.1-99.4) | |
| Overall accuracy (%) | - | 76.1 (72.1-79.8) | 78.9 (75.0-82.3) | |
AUC=Area under curve; PG=Pepsinogen; NPV=Negative prediction value; PPV=Positive prediction value; CI=Confidence interval
Validation for Helicobacter pylori IgG and ABC method of Indonesian patients
Following manufacturer's standard for detecting H. pylori infection, we observed that 46 patients (7.1%) were infected by H. pylori. Utilizing the manufacturer's standard, the sensitivity, specificity, and overall accuracy were 69.5%, 99.2%, and 96.4%, respectively when histology results were used as gold standard. Owning considerably low sensitivity value, we determined the new cutoff value. With the AUC 0.934 (95% confidence interval [CI] = 0.890–0.976), the optimal cutoff value for serology test in Indonesia was ≥6.7 U/mL. By using new cutoff, the sensitivity and specificity were 83.5% (95% CI = 71.2–92.3) and 98.7% (97.3–99.4), respectively [Supplementary Table 2].
We classified each patient based on our modified ABC method, which we only changed the H. pylori infection cutoff point (≥6.7 U/mL) and the PGs value was the same (PG1 <70 ng/mL and PGI/II ratio ≤3). We observed that 585 patients (90.6%) were classified as group A followed by group B (44/646, 6.8%), group C (10/646, 1.5%), and group D (7/646, 1.1%) [Table 5]. When we evaluated ethnicity, we observed a considerably high group C proportion in Papuan and Timor ethnic group (11.6% and 5.8%, respectively) [Supplementary Table 2].
Table 5.
Distribution of modified ABC method classification among Indonesian patients
| n | Group A (%) | Group B (%) | Group C (%) | Group D (%) | P | |
|---|---|---|---|---|---|---|
| Overall | 646 | 585 (90.6) | 44 (6.8) | 10 (1.5) | 7 (1.1) | |
| Sex | ||||||
| Male | 383 | 339 (88.5) | 32 (8.4) | 8 (2.1) | 4 (1.1) | 0.004 |
| Female | 263 | 246 (93.5) | 12 (4.6) | 2 (0.8) | 3 (1.1) | |
| Age group | ||||||
| <18 | 3 | 3 (100) | 0 | 0 | 0 | 0.010 |
| 18-29 | 74 | 69 (93.2) | 3 (4.0) | 1 (1.4) | 1 (1.4) | |
| 30-39 | 165 | 155 (93.9) | 5 (3.0) | 1 (0.6) | 4 (2.4) | |
| 40-49 | 155 | 138 (89.0) | 13 (8.4) | 3 (1.9) | 1 (0.6) | |
| 50-59 | 162 | 145 (89.5) | 13 (8.0) | 4 (2.5) | 0 | |
| ≥60 | 87 | 75 (86.3) | 10 (11.5) | 1 (1.1) | 1 (1.1) | |
| Disease | ||||||
| Gastritis | 212 | 160 (75.5) | 36 (16.9) | 10 (4.7) | 6 (2.8) | 0.001 |
| Reflux esophagitis | 91 | 88 (96.7) | 3 (3.3) | 0 | 0 | |
| PUD | 34 | 29 (85.3) | 4 (11.8) | 0 | 1 (2.9) | |
| Cancer | 1 | 0 | 1 (100) | 0 | 0 |
All carried out statistical analysis were Fischer’s exact test. H. pylori-negative/PG negative (group A); H. pylori positive/PG negative (group B); H. pylori positive/PG positive (group C); and H. pylori-negative/PG positive (group D). H. pylori=Helicobacter pylori; PG=Pepsinogen; PUD=Peptic ulcer disease
DISCUSSION
This study is the nationwide approach study to validate the potential benefit of PG levels to determine if patients require further endoscopic examination. We observed the atrophy group has a significantly lower PGI/II ratio in both H. pylori-positive and -negative in concordance with previous studies.[18,30] The level of PGI in the atrophy group in this study was higher than in the nonatrophy group, which also reported in the previous study,[31] but not in other studies in Korea and Japan.[32,33,34] This was probably due to the higher proportion of mild atrophy. Severe atrophy was related to the loss of glands which caused significant decrease in PG production.[35] This result was supported by remarkably lower PGI in severe atrophy compared to mild atrophy. Furthermore, PGII value has a greater rise as the result of chronic inflammation.[33,35] The presence of inflammation may also give an increase in PGI and PGII[32] as shown by a significant difference between inflammation and no inflammation either in the antrum or corpus, and by the presence of inflammation, it might respond to the high standard deviation values. However, we could not observe any significant difference of PG between the predominant locations of the atrophy. This phenomenon was also reported in several studies in Korea and Europe.[35,36]
PG's ability to distinguish between atrophy diverge according to the country which related to each population's risk of cancer and H. pylori infection rate.[35] Different cutoff value was implemented to determine atrophy.[37,38,39] In this study, first, we applied the cutoff based on the criteria by Miki et al. However, the determined cutoff value from the original study had low sensitivity and specificity as reported in several countries;[40,41] thus, validation is necessary. We calculated the new cutoff adjusted to the Indonesian population. Our newly determined standard cutoff increased the ability to distinguish normal group and moderate–severe atrophy group with improved sensitivity and specificity. This finding suggests that the PG ability to distinguish atrophy group was limited to moderate–severe atrophy only in Indonesia. In addition, the diagnostic benefit of PG to diagnose moderate–severe atrophy in Indonesia was more likely only using PGI/II ratio value alone rather than combination with other values. Indeed, it has lower sensitivity value than the combination of PGII or PGI/II. However, the latter had considerably lower specificity than PGI/II alone. Therefore, PGI/II ratio had better balance between sensitivity and specificity.
We observed that PGI of reflux esophagitis and gastritis patients was significantly higher than patients with normal mucosa. PGI is mainly secreted by the fundus, which might responsible to the development of the esophagitis.[42] Previous study reported the link between PG and endoscopic finding which showed higher PG production in the peptic ulcer and nodular gastritis and also higher PGI/II ratio in erosive esophagitis.[43,44] Other studies in Korean and Japanese population also showed an increasing PGI/II ratio in reflux esophagitis.[44,45] Reflux esophagitis is proposed as the causal pathology of dyspepsia in the area with low prevalence of H. pylori infection as its possible protective mechanism for progression of H. pylori-related disease.[46] This finding may promote the utility of PG for gastroesophageal reflux screening as suggested by prior study.[47] Meanwhile, inflammation occurred in PUD might affect PGII production in the antrum and duodenum resulting a decrease in PGI/II ratio value as reported before.[48] However, our current study found that there was no significant difference in PGI, PGII, or PGI/II ratio between PUD and gastritis. The increase in PGII which lowering the PGI/II ratio was closely associated with H. pylori infection,[44] thus inducing more severe inflammation. In our current study, the rate of H. pylori infection in PUD patients was quite low. Therefore, it may explain the insignificant differences in either PGI, PGII, or PGI/PGII ratio between peptic ulcer and gastritis patients.
However, the measurement of elderly patients should be cautious. We found that PG level was affected by age, it is in concordance with several studies.[49,50] The level of PGI and PGII was increased as the inclining age may be due to the increasing prevalence of superficial gastritis and H. pylori infection in older age individuals.[30] In addition, the decreasing glomerular filtration rate in the elderly reduces PG excretion.[49] In this study, we included 15 ethnics in Indonesia and found a significant association between PGI, PGII, and PGI/PGII ratio with the ethnic. This factor was also mentioned in the previous studies.[51,52] Genetic factors plays role in determining the PG density.[53] We observed that Bataknese had the lowest PGI/II ratio among all ethnic groups we analyzed, suggesting that particular ethnic group more likely to have atrophic gastritis. In addition, our histologic examination analysis showed the Bataknese was grouped as intermediate risk group for gastric cancer.[54] However, the sample number on that particular ethnic group was only 2, a caution that is important to be considered. Therefore, it is necessary to conduct further study with bigger sample size for each ethnic group to see bigger picture of gastric cancer in Indonesia.
The eradication of H. pylori significantly decrease the level of PGI in the peptic ulcer patients.[55] In our current study, it was also significantly increase the PGI but greater rise in PGII, thus decrease PGI/II ratio as stated in many studies.[34,35] Several possible reasons are that H. pylori infection induces somatostatin deficiency, and inflammation by producing cytokines such as leukotriene and TNFα thus increases the gastrin and gastric acid secretion.[50,56] The increased gastrin secretion would later increase PG secretion. Infection process may also damage the mucous and chief cells which resulted in the leakage of zymogen cells and released the PG before converting to pepsin.[55] Therefore, the combination of PG with the H. pylori serological test is also proposed to improve gastric cancer screening, known as the ABC method.[10,11] Group A has the lowest risk so may not require further endoscopy examination and the higher risk was group B, C, and D and require endoscopy examination every 3, 2, and 1 years, respectively.[57] Our overall observation showed an enormous number of group A and very low group C and D, suggesting the atrophic condition in Indonesia, even with the presence of H. pylori in Indonesia is very low. As comparison to countries with higher gastric cancer incidence, including Japan and Bhutan, the group showed those countries had considerably higher number of group C and group D than Indonesia,[13,15] suggesting that Indonesia is far less having gastric cancer risk than those countries. These findings were in concordance with the age-standardized ratio of gastric cancer incidence from GLOBOCAN that showing 1.5/100,000 population (GLOBOCAN, 2018, available from: https://gco.iarc.fr/). In addition, we also observed that Papuan and Timor ethnic group had a considerably proportion of group C, which in concordance with our previous study which reported a high prevalence of H. pylori infection among those ethnicity.[16]
There were several limitations in this study. First, the patients included in this study was only dyspeptic patients without the addition of healthy subjects combined as a whole population. Therefore, it might decrease the predictive value of serum PGs analyzed in this study; therefore, it would be cautions in application for general population including asymptomatic subjects. Second, the low sample number was also a limitation in this current study, which resulted in a low sample number when our analysis deemed us to divide our samples into several ethnic groups. Therefore, our current works might only be regarded as preliminary generating study. Further study with larger sample number and including a healthy individual is necessary. In addition, even though histology and culture result appeared to be normal, it does not exclude possibility of the atrophy and inflammation due to sampling bias during endoscopy.
CONCLUSION
Validation of indirect methods is essential before their application. We showed that serum PG levels are useful biomarkers for atrophic gastritis. However, the beneficial of PG values determining atrophic gastritis only limited to moderate–severe atrophic gastritis in Indonesia. In addition to atrophic gastritis, serum PG levels also have benefits that represent the endoscopy finding, especially for reflux esophagitis. This usefulness needs to carefully take attention for older age.
Financial support and sponsorship
This work was supported in part by grants from the National Institutes of Health (DK62813) and Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) of Japan (16H05191, 16H06279, 18KK0266 and 19H03473) (YY). It was also supported by the Japan Society for the Promotion of Science (JSPS) Institutional Program for Core-to-Core Program; B. Africa-Asia Science Platform (YY). Support was also provided by the JSPS Institutional Program for Young Researcher Overseas Visits (YY), the Strategic Funds for the Promotion of Science and Technology from the Japan Science and Technology Agency (YY). LAW, DD, and KAF are PhD students supported by the Japanese government (MEXT) scholarship program for 2015, 2016, and 2017, respectively. Program Penelitian Kolaborasi Indonesia (PPKI) 2021 (MM, AFS, GAS). The funders had no role in the study design, data collection, and data analysis, interpretation of data and writing of the manuscript.
Conflicts of Interest
There are no conflicts of interest.
Acknowledgments
This report is based on work supported in part by the National Institutes of Health (DK62813) and Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) of Japan (16H05191, 16H06279, 18KK0266, and 19H03473) (YY).
REFERENCES
- 1.Syam AF, Simadibrata M, Makmun D, Abdullah M, Fauzi A, Renaldi K, et al. National consensus on management of dyspepsia and Helicobacter pylori infection. Acta Med Indones. 2017;49:279–87. [PubMed] [Google Scholar]
- 2.Tavakoli T, Hoseini M, Tabatabaee TS, Rostam Z, Mollaei H, Bahrami A, et al. Comparison of dialectical behavior therapy and antianxiety medication on anxiety and digestive symptoms in patients with functional dyspepsia. J Res Med Sci. 2020;25:59. doi: 10.4103/jrms.JRMS_673_19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Addula M, Wilson VE, Reddymasu S, Agrawal DK. Immunopathological and molecular basis of functional dyspepsia and current therapeutic approaches. Expert Rev Clin Immunol. 2018;14:831–40. doi: 10.1080/1744666X.2018.1524756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hooi JK, Lai WY, Ng WK, Suen MM, Underwood FE, Tanyingoh D, et al. Global Prevalence of Helicobacter pylori infection: Systematic review and meta-analysis. Gastroenterology. 2017;153:420–9. doi: 10.1053/j.gastro.2017.04.022. [DOI] [PubMed] [Google Scholar]
- 5.McColl KE. Clinical practice.Helicobacter pylori infection. N Engl J Med. 2010;362:1597–604. doi: 10.1056/NEJMcp1001110. [DOI] [PubMed] [Google Scholar]
- 6.De Angelis R, Sant M, Coleman MP, Francisci S, Baili P, Pierannunzio D, et al. Cancer survival in Europe 1999-2007 by country and age: Results of EUROCARE-5-a population-based study. Lancet Oncol. 2014;15:23–34. doi: 10.1016/S1470-2045(13)70546-1. [DOI] [PubMed] [Google Scholar]
- 7.Yin YF, Li Y, Chen YL, Zhang DK. Application of digestive endoscopy in diagnosis of Helicobacter pylori infection. Zhongguo Yi Xue Ke Xue Yuan Xue Bao. 2018;40:563–7. doi: 10.3881/j.issn.1000-503X.10192. [DOI] [PubMed] [Google Scholar]
- 8.Kono S. International Encyclopedia of Public Health. 2nd ed. Vol. 3. Cambridge, Massachusetts, United States: Elsevier; 2016. [Last accessed on 2022 Sep 22]. Gastric cancer; pp. 215–22. Available from: http://dx.doi.org/10.1016/B978-0-12-803678-5.00167-3 . [Google Scholar]
- 9.Miki K. Gastric cancer screening using the serum pepsinogen test method. Gastric Cancer. 2006;9:245–53. doi: 10.1007/s10120-006-0397-0. [DOI] [PubMed] [Google Scholar]
- 10.Sasazuki S. The ABC method and gastric cancer: Evidence from prospective studies. J Epidemiol. 2016;26:611–2. doi: 10.2188/jea.JE20160140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Miki K. Gastric cancer screening by combined assay for serum anti-Helicobacter pylori IgG antibody and serum pepsinogen levels-“ABC method”. Proc Jpn Acad Ser B Phys Biol Sci. 2011;87:405–14. doi: 10.2183/pjab.87.405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Chen XZ, Huang CZ, Hu WX, Liu Y, Yao XQ. Gastric cancer screening by combined determination of serum Helicobacter pylori antibody and pepsinogen concentrations: ABC method for gastric cancer screening. Chin Med J (Engl) 2018;131:1232–9. doi: 10.4103/0366-6999.231512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yamaguchi Y, Nagata Y, Hiratsuka R, Kawase Y, Tominaga T, Takeuchi S, et al. Gastric cancer screening by combined assay for serum anti-Helicobacter pylori IgG antibody and serum pepsinogen levels - The ABC method. Digestion. 2016;93:13–8. doi: 10.1159/000441742. [DOI] [PubMed] [Google Scholar]
- 14.Gantuya B, Oyuntsetseg K, Bolor D, Erdene-Ochir Y, Sanduijav R, Davaadorj D, et al. Evaluation of serum markers for gastric cancer and its precursor diseases among high incidence and mortality rate of gastric cancer area. Gastric Cancer. 2019;22:104–12. doi: 10.1007/s10120-018-0844-8. [DOI] [PubMed] [Google Scholar]
- 15.Shiota S, Mahachai V, Vilaichone RK, Ratanachu-Ek T, Tshering L, Uchida T, et al. Seroprevalence of Helicobacter pylori infection and gastric mucosal atrophy in Bhutan, a country with a high prevalence of gastric cancer. J Med Microbiol. 2013;62:1571–8. doi: 10.1099/jmm.0.060905-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Miftahussurur M, Syam AF, Nusi IA, Makmun D, Waskito LA, Zein LH, et al. Surveillance of Helicobacter pylori antibiotic susceptibility in Indonesia: Different resistance types among regions and with novel genetic mutations. PLoS One. 2016;11:e0166199. doi: 10.1371/journal.pone.0166199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Syam AF, Miftahussurur M, Makmun D, Nusi IA, Zain LH, Zulkhairi, et al. Risk factors and prevalence of Helicobacter pylori in five largest islands of Indonesia: A preliminary study. PLoS One. 2015;10:e0140186. doi: 10.1371/journal.pone.0140186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Miftahussurur M, Nusi IA, Akil F, Syam AF, Wibawa ID, Rezkitha YA, et al. Gastric mucosal status in populations with a low prevalence of Helicobacter pylori in Indonesia. PLoS One. 2017;12:e0176203. doi: 10.1371/journal.pone.0176203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dixon MF, Genta RM, Yardley JH, Correa P. Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol. 1996;20:1161–81. doi: 10.1097/00000478-199610000-00001. [DOI] [PubMed] [Google Scholar]
- 20.Uchida T, Kanada R, Tsukamoto Y, Hijiya N, Matsuura K, Yano S, et al. Immunohistochemical diagnosis of the cagA-gene genotype of Helicobacter pylori with anti-East Asian CagA-specific antibody. Cancer Sci. 2007;98:521–8. doi: 10.1111/j.1349-7006.2007.00415.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Vilaichone RK, Mahachai V, Shiota S, Uchida T, Ratanachu-ek T, Tshering L, et al. Extremely high prevalence of Helicobacter pylori infection in Bhutan. World J Gastroenterol. 2013;19:2806–10. doi: 10.3748/wjg.v19.i18.2806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Shiota S, Murakami K, Fujioka T, Yamaoka Y. Population-based strategies for Helicobacter pylori-associated disease management: A Japanese perspective. Expert Rev Gastroenterol Hepatol. 2010;4:149–56. doi: 10.1586/egh.10.7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Shiota S, Cruz M, Abreu JA, Mitsui T, Terao H, Disla M, et al. Virulence genes of Helicobacter pylori in the Dominican republic. J Med Microbiol. 2014;63:1189–96. doi: 10.1099/jmm.0.075275-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Nguyen LT, Uchida T, Tsukamoto Y, Trinh TD, Ta L, Mai HB, et al. Clinical relevance of cagPAI intactness in Helicobacter pylori isolates from Vietnam. Eur J Clin Microbiol Infect Dis. 2010;29:651–60. doi: 10.1007/s10096-010-0909-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Nguyen TL, Uchida T, Tsukamoto Y, Trinh DT, Ta L, Mai BH, et al. Helicobacter pylori infection and gastroduodenal diseases in Vietnam: A cross-sectional, hospital-based study. BMC Gastroenterol. 2010;10:114. doi: 10.1186/1471-230X-10-114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Miftahussurur M, Shiota S, Suzuki R, Matsuda M, Uchida T, Kido Y, et al. Identification of Helicobacter pylori infection in symptomatic patients in Surabaya, Indonesia, using five diagnostic tests. Epidemiol Infect. 2015;143:986–96. doi: 10.1017/S095026881400154X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bodger K, Wyatt JI, Heatley RV. Variation in serum pepsinogens with severity and topography of Helicobacter pylori-associated chronic gastritis in dyspeptic patients referred for endoscopy. Helicobacter. 2001;6:216–24. doi: 10.1046/j.1523-5378.2001.00031.x. [DOI] [PubMed] [Google Scholar]
- 28.Capurso G, Carnuccio A, Lahner E, Panzuto F, Baccini F, Delle Fave G, et al. Corpus-predominant gastritis as a risk factor for false-negative 13C-urea breath test results. Aliment Pharmacol Ther. 2006;24:1453–60. doi: 10.1111/j.1365-2036.2006.03143.x. [DOI] [PubMed] [Google Scholar]
- 29.Uchida T, Miftahussurur M, Pittayanon R, Vilaichone RK, Wisedopas N, Ratanachu-Ek T, et al. Helicobacter pylori infection in Thailand: A nationwide study of the CagA phenotype. PLoS One. 2015;10:e0136775. doi: 10.1371/journal.pone.0136775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shan JH, Bai XJ, Han LL, Yuan Y, Sun XF. Changes with aging in gastric biomarkers levels and in biochemical factors associated with Helicobacter pylori infection in asymptomatic Chinese population. World J Gastroenterol. 2017;23:5945–53. doi: 10.3748/wjg.v23.i32.5945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sjomina O, Pavlova J, Daugule I, Janovic P, Kikuste I, Vanags A, et al. Pepsinogen test for the evaluation of precancerous changes in gastric mucosa: A population-based study. J Gastrointestin Liver Dis. 2018;27:11–7. doi: 10.15403/jgld.2014.1121.271.pep. [DOI] [PubMed] [Google Scholar]
- 32.Kang JM, Kim N, Yoo JY, Park YS, Lee DH, Kim HY, et al. The role of serum pepsinogen and gastrin test for the detection of gastric cancer in Korea. Helicobacter. 2008;13:146–56. doi: 10.1111/j.1523-5378.2008.00592.x. [DOI] [PubMed] [Google Scholar]
- 33.Kim HY, Kim N, Kang JM, Park YS, Lee DH, Kim YR, et al. Clinical meaning of pepsinogen test and Helicobacter pylori serology in the health check-up population in Korea. Eur J Gastroenterol Hepatol. 2009;21:606–12. doi: 10.1097/MEG.0b013e3283086757. [DOI] [PubMed] [Google Scholar]
- 34.Asaka M, Kimura T, Kudo M, Takeda H, Mitani S, Miyazaki T, et al. Relationship of Helicobacter pylori to serum pepsinogens in an asymptomatic Japanese population. Gastroenterology. 1992;102:760–6. doi: 10.1016/0016-5085(92)90156-s. [DOI] [PubMed] [Google Scholar]
- 35.Kim N, Jung HC. The role of serum pepsinogen in the detection of gastric cancer. Gut Liver. 2010;4:307–19. doi: 10.5009/gnl.2010.4.3.307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sipponen P, Maaroos HI. Chronic gastritis. Scand J Gastroenterol. 2015;50:657–67. doi: 10.3109/00365521.2015.1019918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Miki K, Morita M, Sasajima M, Hoshina R, Kanda E, Urita Y. Usefulness of gastric cancer screening using the serum pepsinogen test method. Am J Gastroenterol. 2003;98:735–9. doi: 10.1111/j.1572-0241.2003.07410.x. [DOI] [PubMed] [Google Scholar]
- 38.Tong Y, Wu Y, Song Z, Yu Y, Yu X. The potential value of serum pepsinogen for the diagnosis of atrophic gastritis among the health check-up populations in China: A diagnostic clinical research. BMC Gastroenterol. 2017;17:88. doi: 10.1186/s12876-017-0641-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kim EH, Kang H, Park CH, Choi HS, Jung DH, Chung H, et al. The optimal serum pepsinogen cut-off value for predicting histologically confirmed atrophic gastritis. Dig Liver Dis. 2015;47:663–8. doi: 10.1016/j.dld.2015.05.014. [DOI] [PubMed] [Google Scholar]
- 40.Chae H, Lee JH, Lim J, Kim M, Kim Y, Han K, et al. Clinical utility of serum pepsinogen levels as a screening test of atrophic gastritis. Korean J Lab Med. 2008;28:201–6. doi: 10.3343/kjlm.2008.28.3.201. [DOI] [PubMed] [Google Scholar]
- 41.Broutet N, Plebani M, Sakarovitch C, Sipponen P, Mégraud F, Eurohepygast Study Group Pepsinogen A, pepsinogen C, and gastrin as markers of atrophic chronic gastritis in European dyspeptics. Br J Cancer. 2003;88:1239–47. doi: 10.1038/sj.bjc.6600877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gritti I, Banfi G, Roi GS. Pepsinogens: Physiology, pharmacology pathophysiology and exercise. Pharmacol Res. 2000;41:265–81. doi: 10.1006/phrs.1999.0586. [DOI] [PubMed] [Google Scholar]
- 43.Emami MH, Ataie-Khorasgani M, Jafari-Pozve N. Diagnostic value of alarm symptoms for upper GI malignancy in patients referred to GI clinic: A 7 years cross sectional study. J Res Med Sci. 2017;22:76. doi: 10.4103/jrms.JRMS_450_15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lee SP, Lee SY, Kim JH, Sung IK, Park HS, Shim CS. Link between serum pepsinogen concentrations and upper gastrointestinal endoscopic findings. J Korean Med Sci. 2017;32:796–802. doi: 10.3346/jkms.2017.32.5.796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Yamaji Y, Mitsushima T, Ikuma H, Okamoto M, Yoshida H, Kawabe T, et al. Inverse background of Helicobacter pylori antibody and pepsinogen in reflux oesophagitis compared with gastric cancer: Analysis of 5732 Japanese subjects. Gut. 2001;49:335–40. doi: 10.1136/gut.49.3.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Enomoto S, Oka M, Ohata H, Mukoubayashi C, Watanabe M, Moribata K, et al. Assessment of gastroesophageal reflux disease by serodiagnosis of Helicobacter pylori-related chronic gastritis stage. World J Gastrointest Endosc. 2011;3:71–7. doi: 10.4253/wjge.v3.i4.71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Miftahussurur M, Doohan D, Nusi IA, Adi P, Rezkitha YA, Waskito LA, et al. Gastroesophageal reflux disease in an area with low Helicobacter pylori infection prevalence. [Last accessed on 2022 Sep 22];PLoS One. 2018 13:e0205644. doi: 10.1371/journal.pone.0205644. Available from: https://www.scopus.com/inward/record.uri?eid=2-s2.0-85056547032&doi=100.1371%2Fjournal.pone. 0205644&partnerID=40&md5=6ee1e6c545c7b6994011800026a81159 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Samloff IM, Stemmermann GN, Heilbrun LK, Nomura A. Elevated serum pepsinogen I and II levels differ as risk factors for duodenal ulcer and gastric ulcer. Gastroenterology. 1986;90:570–6. doi: 10.1016/0016-5085(86)91110-8. [DOI] [PubMed] [Google Scholar]
- 49.Pilotto A, Vianello F, Di Mario F, Plebani M, Farinati F, Azzini CF. Effect of age on gastric acid, pepsin, pepsinogen group A and gastrin secretion in peptic ulcer patients. Gerontology. 1994;40:253–9. doi: 10.1159/000213593. [DOI] [PubMed] [Google Scholar]
- 50.Huang RG, Xiao HL, Zhou B, Song XH, Zhang J, Wang CM, et al. Serum pepsinogen levels are correlated with age, sex and the level of Helicobacter pylori infection in healthy individuals. Am J Med Sci. 2016;352:481–6. doi: 10.1016/j.amjms.2016.08.005. [DOI] [PubMed] [Google Scholar]
- 51.Fahey MT, Hamada GS, Nishimoto IN, Kowalski LP, Iriya K, Gama-Rodrigues JJ, et al. Ethnic differences in serum pepsinogen levels among Japanese and non-Japanese Brazilian gastric cancer patients and controls. Cancer Detect Prev. 2000;24:564–71. [PubMed] [Google Scholar]
- 52.Ang TL, Fock KM, Dhamodaran S, Teo EK, Tan J. Racial differences in Helicobacter pylori, serum pepsinogen and gastric cancer incidence in an urban Asian population. J Gastroenterol Hepatol. 2005;20:1603–9. doi: 10.1111/j.1440-1746.2005.03898.x. [DOI] [PubMed] [Google Scholar]
- 53.Bebelman JP, Evers MP, Zelle B, Bank R, Pronk JC, Meuwissen SG, et al. Family and population studies on the human pepsinogen A multigene family. Hum Genet. 1989;82:142–6. doi: 10.1007/BF00284047. [DOI] [PubMed] [Google Scholar]
- 54.Miftahussurur M, Waskito LA, Syam AF, Nusi IA, Dewa Nyoman Wibawa I, Rezkitha YA, et al. Analysis of risks of gastric cancer by gastric mucosa among Indonesian ethnic groups. PLoS One. 2019;14:e0216670. doi: 10.1371/journal.pone.0216670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Park SM, Park J, Chang SK, Yoo BC, Kim HJ. Helicobacter pylori infection and serum pepsinogen I concentration in peptic ulcer patients: Effect of bacterial eradication. Korean J Intern Med. 1996;11:1–8. doi: 10.3904/kjim.1996.11.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jiang HX, Pu H, Huh NH, Yokota K, Oguma K, Namba M. Helicobacter pylori induces pepsinogen secretion by rat gastric cells in culture via a cAMP signal pathway. Int J Mol Med. 2001;7:625–9. doi: 10.3892/ijmm.7.6.625. [DOI] [PubMed] [Google Scholar]
- 57.Tatemichi M, Sasazuki S, Inoue M, Tsugane S JPHC Study Group. Clinical significance of IgG antibody titer against Helicobacter pylori. Helicobacter. 2009;14:231–6. doi: 10.1111/j.1523-5378.2009.00681.x. [DOI] [PubMed] [Google Scholar]