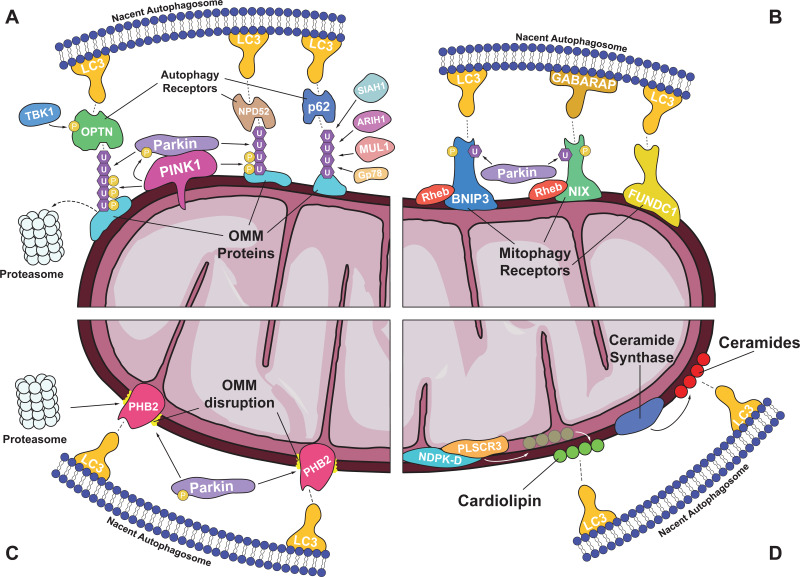
Fig. 1.
Overview of mitophagy mechanisms. (A) Ubiquitin-mediated mitophagy. Dissipation of mitochondrial membrane potential leads to the accumulation of PINK1 kinase on the surface of damaged organelles, where it phosphorylates and activates the E3 ubiquitin ligase Parkin. Ubiquitin moieties attached to OMM proteins, either by Parkin or by other ubiquitin ligases (e.g., MUL1, SIAH1, ARIH1 and Gp78), are stabilised by PINK1-mediated phosphorylation, and are ultimately recognised by autophagy receptors (e.g., OPTN, NPD52 and p62) that drive autophagosome formation through their interaction with LC3 protein. The action of such receptors is enhanced by their TBK1-mediated phosphorylation. Alternatively, ubiquitinated OMM proteins can be degraded by the proteasome, altering mitochondrial dynamics and motility to favor mitophagy. (B) OMM receptor-mediated mitophagy. OMM proteins can act as mitophagy receptors (e.g., BNIP3, NIX and FUNDC1) and induce autophagosome formation through their direct interaction with LC3/GABARAP. The activation of most mitophagy receptors involves their phosphorylation and is facilitated by the translocation of Rheb small GTPase to mitochondria. Further induction of their activity can be achieved by their Parkin-mediated ubiquitination, establishing a cross-talk mechanism between different pathways. (C) IMM receptor-mediated mitophagy. IMM proteins (e.g., PHB2) can act as mitophagy receptors by interacting with LC3 upon the disruption of OMM, due to Parkin-mediated ubiquitination and proteasomal degradation of OMM proteins. (D) Lipid-mediated mitophagy. The presence of certain lipids (e.g., cardiolipin and ceramides) on the OMM can induce mitophagy by driving autophagosome formation through their interaction with LC3. Cardiolipin is externalised from the IMM by the action of PLSCR3 and NDPK-D, while ceramides and their analogues are freshly generated on the OMM by ceramide synthases.