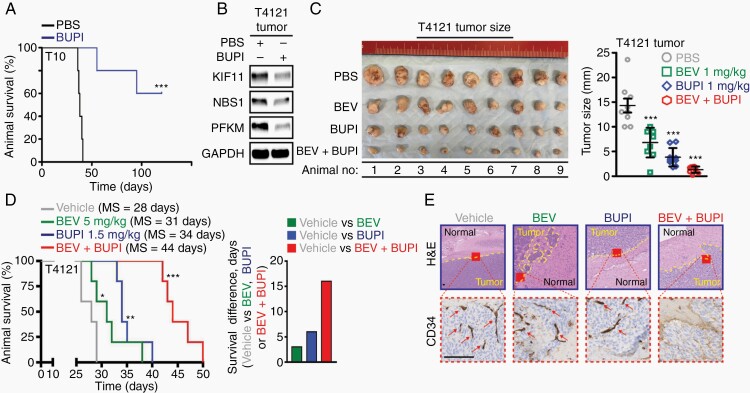
Fig. 6.
Bupivacaine impairs glioblastoma growth and augments bevacizumab efficacy in vivo. (A) Bupivacaine pre-treatment (GI50 for 24 hours) in vitro reduces tumorigenicity of T10 GBM cells intracranially implanted in the frontal lobe of immunocompromised mice (n = 5 per arm). (B) Bupivacaine treatment (intratumoral injection at 1 mg/kg on alternate days for 2 weeks) reduces PFKM, KIF11, and NBS1 expression in GBM tumors grown in immunocompromised mice. Loading control: GAPDH. Combinational bupivacaine and bevacizumab therapy inhibited tumor growth with a greater efficacy than either of the monotherapies in subcutaneous (C) as well as (D) in orthotopic GBM xenograft model. Treatment was given on alternate days for 3 weeks: (i) vehicle PBS intraperitoneally and vehicle artificial cerebral spinal fluid intratumorally via a guide screw; (ii) 5 mg/kg bevacizumab/BEV intraperitoneally; (iii) 1.5 mg/kg bupivacaine/BUPI intratumorally via a guide screw; and (iv) the combination of bevacizumab/BEV and bupivacaine/BUPI. (E) H&E analysis and immunohistochemistry assessment of CD34 biomarker in representative coronal brain sections confirmed reduced tumor burden, parenchymal invasion, and vascularization (arrows) in animals co-treated with bevacizumab and bupivacaine. Scale bar = 100 µm.
Significance is determined by log-rank test (A, D) or t test (C), where data are shown as mean ± SEM (*P < .05, **P < .01, ***P < .001).