Significance
Habitat loss has led to widespread range contraction and population declines across taxa. The impact of range contraction on niche occupancy is rarely, if ever, incorporated into conservation assessments. We evaluate range loss in 4,785 terrestrial mammals and changes in niche parameters of range-contracted species. Range contraction results in more homogenous ranges and reduced niche sizes. Some species become restricted to ecologically extreme habitats at the periphery of their historic niche. This ecological marginalization increases extinction risk. Marginalization can result in a “double whammy” where poor performance in marginalized relict populations exacerbates population declines. This phenomenon is an underappreciated global conservation threat that may partially explain the failure of protected areas to buffer species from further decline.
Keywords: marginality, range loss, performance, dynamics, threats
Abstract
Human land-use results in widespread range change across taxa. Anthropogenic pressures can result in species’ realized niches expanding, shifting, or contracting. Marginalization occurs when contraction constrains species to the geographic or ecological extremes of their historic niche. Using 4,785 terrestrial mammal species, we show that range contraction results in niche space and habitat diversity loss. Additionally, ecological marginalization is a common consequence of range contraction caused by human land use change. Remnant populations become located in the climatic and topographic extremes of their historic niche that are more likely to be at the periphery of their historic niche at greater distances from historic niche centroids. This ecological marginalization is associated with poor performance and increased extinction risk independent of geographic range loss. Range loss and marginalization may create a “double whammy” in vulnerable groups, such as large-bodied species and species with small geographical range size. Our results reveal a hitherto unrecognized conservation threat that is vital to incorporate into conservation assessment and management.
Human impacts, such as land-use changes (1), persecution, and community change (2) have globally altered the size, habitat diversity, and connectivity of species’ ranges. Efforts to address pervasive habitat loss and conversion include sustainable harvesting mandates, regulations limiting land clearing and habitat fragmentation, and the creation and expansion of protected areas (PAs) (3). Despite increases in established PAs to ~250,000 terrestrial PAs (22 million km2) and ~20,000 marine PAs (28 million km2) (4), species loss continues. Continuing declines may be the result of extinction debt, or a lag between habitat loss and population extirpation (5). However, the “Protected Area Paradox” (6), whereby declines continue within PAs, may also result from PAs being located in marginal (7), less productive (8) “pristine” lands under little pressure for human conversion (9, 10).
Protected area bias toward marginal habitats results from a global mosaic of human agriculture and settlement that are concentrated in highly productive lands (2, 11) with shallow slopes, fertile soils, and access to surface water (12). Temperate and tropical grasslands and tropical dry, Mediterranean, and temperate broadleaf forests are preferentially converted and fragmented for high-intensity agriculture (13) and SI Appendix, Fig. S1). As most PAs have been created after agricultural intensification, they are more likely to occur in areas that are unattractive for agriculture (8, 9) such as steep slopes or far from water, rather than areas with the most biodiversity. Moreover, biomes such as mountains, deserts, and cold tundra that only account for 10% of global net primary productivity are over-represented in PAs (6, 14).
Species’ ranges encompass abiotic and biotic gradients ranging in quality. In high-quality habitats, individual vital rates (survival and reproduction) and population performance (abundance, density, and growth) are strong, whereas in low-quality, marginal habitats, individual and population metrics are comparatively poor (15). High-quality habitats can support higher population density and produce more emigrants that disperse from “sources” to non-self-sustaining marginal “sinks” (16). Such productive habitats are crucial to maintain meta-population processes and trophic diversity (17). In response to threats, environmental change and persecution, organisms can: 1) retreat into refugia (18), 2) contract into high-quality “core” environments that buffer the threat (19), or 3) move into or co-opt a novel habitat (7).
Range change can lead to ecological niche or geographic range shifts, expansion or collapse (Fig. 1 and SI Appendix, Fig. S2). Random habitat loss will result in a smaller range but will not necessarily impact habitat diversity or the niche center. Habitat loss focused on ecological or geographic margins will lead to niche contraction toward the geographic or ecological “core.” Such contraction to high-quality habitats with high vital rates can lead to increased resilience of the remaining metapopulation (19). In contrast, loss of habitat in the range core can lead to contraction toward ecological extremes at the periphery of the historically occupied niche (Fig. 2). Localized human pressures may extirpate or shift individual populations into marginal conditions (e.g., partial refugee species; ref. (20), whereas marginalization at a macroecological scale can restrict entire species to marginal conditions (7). Given that human activities and land conversion are concentrated in productive accessible lands, range contraction and habitat loss are likely to be associated with shifts toward niche margins. However, the relationship between range contraction and niche shift in mammals has not been evaluated.
Fig. 1.
Dynamics of species response curves and niche spaces following shifts from anthropogenic pressures. (Top) possible outcomes of niche shifts where green to yellow gradients represent high-quality to low-quality conditions, respectively. (Centre) changes in niche space in relation to these shifts where crosses represent niche centroids, blue—contemporary and orange—historic. Ellipses depict niche space, blue—contemporary, orange—historic. (Bottom) predicted changes in average distance to historic niche center with increasing range contraction.
Fig. 2.
Examples of species demonstrating contraction to niche centroid, ecological marginalization and stable niches under range contraction in three-dimensional environmental. (Top) Feathertail glider (Acrobates pygmaeus) as an example of a species with a stable niche under very low levels of range contraction. (Middle) Grevy’s zebra (Equus grevyi) as an example of contraction to niche centroid under very high range contraction (~90%). (Bottom) Red wolf (Canis rufus) as an example of ecological marginalization. All three-dimensional niche spaces are constructed from the first three principal components of contemporary and historic topographic and climatic variables separately. “Historic” niche points (modeled with Mid-Holocene period climate, ~6,000 y ago). Orange dots and ellipse are PCA coordinate points of each species’ contemporary niche and niche space respectively. Blue: historic niche points and niche space.
Here, we evaluate the intrinsic and extrinsic factors associated with range collapse using a combination of phylogenetic and niche modeling (polyhedral convex hull and n-dimensional hypervolumes). We define the niche as a multidimensional volume (hyperspace) where axes correspond to environmental conditions (21). Species historic and contemporary geographic ranges are physical spaces of available environmental conditions with each point (or cell) representing a set of topographic and climatic factors, similar to Hutchinson’s definition of biotope (21). For each species’ niche, we subset range maps to include only cells within habitat types and elevation ranges reported in IUCN assessments. The niche space for each species is described by the first three principal components. We then evaluate factors associated with range loss in 4,785 mammal species and how niche shift impacts extinction risk of range-contracted mammals. We predict that human pressures in ecologically productive landscapes will exclude species from historic niche centers and lead to ecological marginalization. We also predict that ecological marginalization will increase extinction risk where niche centroids shift from core, high-quality habitats to more marginal areas (17, 22).
Results
Thirteen percent, or 627, of 4,785 mammal species have undergone geographic range contraction, but the intensity of range loss varied (SI Appendix, Table S1). Across all species, whether geographic range contraction occurred was explained by the following factors: small geographic range size, large body mass, and the proportion of the historic range transformed for human land-use (Table 1). The extent of range contraction in species which have experienced range contraction was a function of human transformation of the historic range and small geographic range sizes (SI Appendix, Table S2).
Table 1.
Species with more human transformation in their historic range and smaller geographic range sizes are most vulnerable to geographic range loss

|
As species’ geographic ranges contract, their niches shrink and become more homogeneous (higher proportion of habitat diversity loss) (Fig. 3 and Table 2). Large body mass, large geographic ranges, and anthropogenic transformation all predict the extent of habitat diversity loss (SI Appendix, Table S1 and Table 2). Body mass and range loss interact such that larger-bodied species with extensive range loss have disproportionately lost habitat diversity (SI Appendix, Fig. S5).
Fig. 3.
Geographic range loss has severe consequences for species niches—restricting them to smaller, less diverse, ecologically extreme niche spaces. (A) Geographic range loss results in niche shrinkage, (B) Small ranged species are more vulnerable to displacement to less diverse ranges- Range size is in km2, Habitat diversity loss is estimated as proportion of ecoregions lost, Middle): Geographic range contraction restricts species to climatic and topographic extremes, (C) precipitation, (D) elevation, Bottom row) Geographic range loss leads to ecological marginalization. (E) Effect size of Mahalanobis distance to historic niche center, (F) Small ranged species are most vulnerable to ecological marginalization. R and P values presented were calculated from Pearson’s correlation coefficient. Yellow outlines represent 95% confidence interval of Pearson’s CC. Species correspond to range contracted species only. Ecological marginalization against geographic range loss with species names is available SI Appendix, Fig. S4.
Table 2.
For species that have undergone range loss, greater geographic range contraction and human transformation of historic range is associated with more loss of habitat diversity and niche space

|
Most range-contracted species were displaced toward climate or topography extremes (SI Appendix, Table S4). Range collapse and human transformation shift species to more extreme temperature (SI Appendix, Table S8), precipitation (SI Appendix, Table S10), elevation (SI Appendix, Table S12), and slope ranges (SI Appendix, Table S14 and Fig. 3). Species with smaller geographic range are more vulnerable to shifts in temperature and precipitation (Fig. 3 and SI Appendix, Tables S8 and S10). Large-bodied species were also more vulnerable to shifts in temperature (SI Appendix, Table S8), whereas small-bodied species were more vulnerable to shifts in elevation (SI Appendix, Table S12)
For range-contracted species, niche parameters at the range center were different from those at the niche center in the primary principal component for historic (Z = −2.80, df = 524, P = 0.005) and contemporary niches (Z = −1.91, df = 524, P = 0.06, SI Appendix, Table S30). Environmental conditions at the historic geographic center also differ from the historic range average (temperature; Z = −2.70, df = 545, P = 0.007; precipitation Z = −2.02, df = 545, P = 0.04; and elevation Z = 6.44, df = 545, P < 0.001). Environmental conditions at the contemporary geographic center also differ from the contemporary range average (temperature Z = −4.47, df = 545, P < 0.001; elevation Z = 5.28, df = 545, P < 0.001) (SI Appendix, Table S29). Geographic range loss displaces species to the ecological periphery of their historic niche (Fig. 3 and SI Appendix, Table S25) but does not consistently displace species to their geographic range edge (SI Appendix, Table S17 and Fig. S3). Larger range loss results in more ecological marginalization, quantified by a greater Mahalanobis distance between the contemporary niche points and the historic niche centroid (Fig. 3 and SI Appendix, Table S19) and greater distances between historic and contemporary niche centroids (SI Appendix, Table S20). Species with small geographic ranges were most vulnerable to ecological marginalization using both Mahalanobis distance and centroid shifts (Fig. 3). As such, ecological, but not geographic, marginalization is a common consequence of geographic range loss in range-restricted mammals (SI Appendix, Table S28).
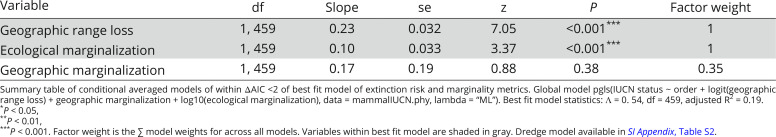
Across all species, increased extinction risk was associated with large body mass and small geographical range (SI Appendix, Table S22). For species that have experienced range contraction, large body mass, and small geographical range (SI Appendix, Table S24) as well as greater geographic range loss and ecological marginalization also increased extinction risk (Table 3). Geographic marginalization did not influence extinction risk (Table 3).
Table 3.
Geographic range loss and both metrics of ecological marginalization independently increase extinction risk
|
Discussion and Conclusion
Human land-use leads to non-random range and habitat diversity losses in terrestrial mammals. As species’ ranges contract, they become increasingly restricted to ecologically marginal habitats and extremes of their historical niche. Ecological, but not geographic, marginalization increases extinction risk independent of geographic range loss. In addition to large body mass and small geographic range predicting extinction risk (23), these same traits render species more vulnerable to habitat diversity loss, niche loss, and ecological marginalization. Our analysis shows that ecological retreat (24) and marginalization is a global phenomenon in terrestrial mammals undergoing range contraction.
The core–periphery hypothesis argues that organism abundance, density, and performance decrease from the geographic or ecological (niche centroid) core to the periphery of a species range/niche (25, 26). Historic niche and range limits form where death rates and emigration exceed birth rates and immigration (27), and further range expansion is unsustainable (28). Niche peripheries can reflect species’ physiological tolerance limits (29) to climatic extremes, resource limitation, disease, predation, or competition (30). Geographic edges are not always associated with reduced population performance (31–33); however, studies have linked peripheral populations to reduced patch occupancy, lower abundance, and higher rates of genetic differentiation and inbreeding (17). Ecologically marginal populations likewise have been associated with patchier occurrence (34), lower abundance and population density (17, 22), lower genetic diversity (35), and lower growth rates (36, 37).
There are instances where demographic (38) trends may not follow the core-periphery hypothesis. As patch quality changes, populations may take time to track more optimal habitats through dispersal, or take time to recover after a marginal period, resulting in a lag between observed demographic metrics and proximity to niche centroid (32, 39).
Ecological marginalization occurs when range contraction restricts populations to suboptimal habitats more often than high-quality ones. Marginalization worsens as niche contraction continues and it is vital to address habitat suitability issues in declining populations before pressures accumulate (40). Partial refugee species have some key populations confined to suboptimal habitat with poor performance (6, 20), whereas refugee species are confined to, and protected in, solely marginal conditions (7). Species are likely to experience increased extinction risk through progressive declines in fitness and resilience as ranges contract toward niche and ecological boundaries (28).
These results highlight that geographic and ecological marginalization are different processes. Historically, the core-periphery model of habitat suitability assumed that range centers represent optimal habitats with suitability declining with distance from the center (25). If true, the environmental conditions at the center of range should be the same as the center of the realized niche. Ecological stresses are distributed throughout a range, such that marginal habitat can occur at either geographic centers or edges (41). The conservation potential of a population can be determined by how the environmental conditions compare to a species ecological niche rather than whether it is in the geographic center or periphery of a historic range. Here, we show that in range-contracted mammals, environmental conditions at range centers differ from range means and niche centroids. Thus, in species contemporary distribution, range centers are not ecologically equivalent to niche centers. A population or species pushed to a range edge may not have been pushed to a niche periphery. Critically, this means that the impact of range loss on long-term resilience is better assessed using niche models than spatial models alone.
Large-bodied animals are vulnerable to extinction or extirpation due to their high energetic requirements, slow reproductive rates, and low population density (23, 42). Large-bodied species are also more likely to be in conflict with humans over areas suitable for rangelands or agricultural purposes and are often selectively harvested by humans (40), pushing them into poor productivity or barren lands (11). Small range size is associated with ecological marginalization as land-use change within their core “primary” habitat can leave low density, capped populations in the remaining marginal habitats (43, 44). We show intrinsic (geographic range size and body mass) and extrinsic factors (geographic range loss) increase extinction risk. We also demonstrate that ecological marginalization increases extinction risk independent of geographic range loss. Large-bodied and small-ranged species are particularly vulnerable to ecological marginalization and geographic range loss. Large-bodied and small-ranged species can experience a form of “double jeopardy” (45) or a “double whammy” where extinction risk is a result of both intrinsic vulnerability and confinement to marginal habitats (40).
Our findings have major implications for evaluating the effectiveness of PAs and identifying conservation priorities for species. PAss are a final stronghold for many species (46). Many PAs are established in areas with little pressure for human land-use, because of low economic value and productivity, rather than for biodiversity value (9, 10). Steep, bare, and sparsely vegetated habitats (e.g., cold mountains, deserts) are overrepresented in PAs (8). Hence, the full spectrum of historical niche space is unprotected for the vast majority (93.1% of amphibians, 89.5% of birds, and 90.9% of terrestrial mammal species) of species (47). Current estimates suggest PAs fail to adequately protect half of the terrestrial mammals globally with hundreds of species having no viable protected area (48). PAs may be inefficient and potentially detrimental when they protect species in marginal habitat (6, 7, 20) and may effectively enforce ecological marginalization (7). This may explain why populations of vulnerable species continue to decline even within PAs (e.g., refs. 49 and 50) and why increasing protected area coverage has not reversed the growing number of threatened species (6). Ecological marginalization, however, is not widely incorporated in conservation management or planning (51). To our knowledge, the only species conservation assessment to discuss ecological marginalization as a conservation threat is that for the European bison (Bison bonasus) (52). At a landscape level, Kerley and Colleagues outline a PA network within the Cape Floristic region, South Africa which prioritizes optimal habitat, in terms of large- and medium-sized mammal density, by estimating species potential distributions and abundances across the landscape (53). Ultimately, incorporating marginality into conservation planning could identify species which are actively protected in marginal areas and prioritize the protection of high-quality habitats which will bolster population size and growth.
Our findings also have significant implications for the establishment of appropriate conservation baselines. We demonstrate that contemporary distributions and niche spaces are shaped through ecological retreat, i.e., withdrawal into marginal refugia to escape human pressures (24). Without reference to historical information, studying species following widespread range and niche loss can lead to stereotyped perceptions of a species and conservation complacency (54). Models that describe the ecology of a species based on these stereotypes, so-called “shifted models,” may result in erroneous conservation recommendations (6, 55) and inaccurate estimates of potential distributions, habitat suitability, and niche breadth (56) or species’ sensitivity to environmental change (57). Ecological marginalization must be considered a hitherto unrecognized threat that compromises conservation efficacy and exacerbates extinction risk.
Methods
Species Traits and Extinction Risk.
Mammal adult body mass (g) and IUCN status were obtained from the PanTHERIA (58) and COMBINE databases (59). “Data Deficient,” extreme outliers, and assumed input errors were excluded. A total of 4785 species’ data were included in the analyses. IUCN status was converted to an ordinal scale from 0 to 5 (least concern = 0, near threatened = 1, vulnerable = 2, endangered = 3, critically endangered = 4, extinct/extinct in wild = 5) as an index of extinction risk.
Species Distributions, Geographic Range Loss, and Habitat Diversity Loss.
For species distributions, we used present natural, i.e., species ranges without human pressures and contemporary ranges from the PHYLACINE dataset (60). The use of the PHYLACINE distributions as estimates of “historic” and contemporary ranges is well established (61). Geographic range loss was assessed from the proportional loss between historic and contemporary ranges. To assess which intrinsic and extrinsic factors were associated with geographic range loss across all 4785 species, geographic range loss was converted to an ordinal scale: zero range loss = 0, 1st quartile of range contraction = 1, 2nd quartile = 2, 3rd quartile = 3, 4th quartile = 4. Range maps were intersected with the Resolve2017 map (62) to assign ecoregions and biomes. Habitat diversity loss was estimated from the proportional loss of ecoregions between historic and contemporary ranges. Freshwater or marine bodies within ranges were excluded.
Anthrome and Land-Use within Ranges.
The Anthropogenic Biomes (anthromes) Version 2 dataset (1,700 to 2,000) (63) was used to assess contemporary land-use within historic species range. Human transformation was calculated as the proportion of a species’ historic range converted to dense settlements (urban, mixed settlements, and village anthromes) and agricultural and rangelands (anthromes of croplands and rangelands with substantial/significant human populations).
Assessing Geographic Marginalization.
For geographic marginalization, 100,000 sampling points were randomly generated throughout both the historic and contemporary ranges of each species. The minimum Euclidean distance from a historic range boundary was then generated for each point. This approach is analogous to the niche margin index (64) but applied to geographic instead of ecological space. A Cohen’s d effect size of the average minimum distance to historic range boundaries for contemporary and historic points was calculated. Positive values depict an increase in distance to the historic range boundary, i.e., contraction to geographic center and negative values depict a decrease in distance, i.e., contraction to edges.
Assessing Ecological Marginalization.
For ecological marginalization, only range-contracted species that lost geographic area between historic and contemporary time points (0 < × < 100%) were assessed. Species’ ranges were trimmed by biome type and elevation limits collated from IUCN assessments. Only biome types and elevation ranges collated from IUCN assessments of each species were used to estimate species’ niches. Conditions within a species’ niche at each time were assessed by sampling all cells for climatic and topographic variables within a species’ trimmed historic and contemporary range.
For present climate conditions, we assessed the period 1970 to 2000 using WorldClim version 2 (65). For past climatic conditions we used the Mid-Holocene period (~6,000 y before present) using WorldClim version 1 (66). We averaged three climatic (general circulation) models (CNRM_CM5, IPSL-CM5A-LR, and the MPI-ESM-P) as estimates of the Holocene climate variability (67). For topographic variables, we evaluated elevation (https://www.worldclim.org/data/2.5m) and calculated slope as the average difference in elevation across eight adjacent cells using the terrain function in the package raster (68). Annual temperature and precipitation, precipitation and temperature seasonality, minimum and maximum temperature (assessed in the coldest and hottest months respectively), minimum and maximum precipitation (assessed in the driest and wettest months, respectively), slope, and elevation were used to represent ecological niches. These factors constitute all major abiotic factors impacting the distribution, diversity, and performance of terrestrial mammals (69). All climatic and topographic conditions were estimated by sampling the centroid of each 2.5 arc-minutes grid cell (approximately 5 × 5 km at the equator) as a compromise between spatial coverage and computational feasibility.
We assessed ecological marginalization by calculating distance between niche centroids and average Mahalanobis distance of contemporary and historic niche points to the historic niche centroid. Firstly, climatic and topographic conditions of historic and contemporary niches were reduced to three principal dimensions using principal component analysis (PCA). PCA coordinates were separated into corresponding historic and contemporary niches. For each, a three-dimensional hypervolume was modeled using a one-class support vector machine (SVM) algorithm (70) (with default γ value of 0.5) using the “hypervolume” package (71). This one-class SVM estimates optimal features that best classify randomly generated points into “inside” or “outside” of the input dataset. Through this procedure, the SVM produces a “tight” fit shape around the input data where boundaries represent niche edges. A one-class SVM is the most suitable algorithm as presence-only data were used and the extremes of the input variables reflect the strict boundaries of species niche space (72). Niche loss was calculated as the proportional loss of hypervolume volume between historic and contemporary hypervolumes. Distance between centroids was calculated as Euclidean distance between contemporary and historic hypervolume centers.
For Mahalanobis distances, we used convex hull modeling (73) to model ecological niches at each time point as minimum volume ellipsoids (MVE). A MVE for both historic and contemporary niche spaces was modeled using the first three principal components of environmental variables (as per hypervolumes). We then calculated the centroid of historic MVE using the cov_center function in the ntbox package (74). The Mahalanobis distance of all points to the historic niche centroid was calculated. The difference in average Mahalanobis distance between historic and contemporary MVEs was calculated as a Cohen’s d effect size. Negative effect sizes occur when the species contracts to its historic core, and positive effect sizes occur when species contracts to its historic periphery. Mahalanobis distances are calculated on the observed distribution only and account for the covariance of multiple environmental axes giving a more accurate representation of the distance in environmental space (75).
Ecological Variables Shift.
To investigate variable shifts to ecological extremes, we also investigated the Cohen’s d effect size shift in elevation, slope, annual temperature, and annual precipitation. These variables represent key abiotic characteristics that dictate species’ distributions, niche dynamics, and habitat selection across geographic space and time (76).
Comparing Conditions at Niche and Range Centers in Range-Contracted Species.
We investigate the similarity between individual environmental variables and niche parameters at the niche center and geographic center in both historic and contemporary time periods. We compare the conditions at the geographic center with the mean of the frequency distribution of environmental variables found across the whole of the species range. We also compare the position of the geographic center within the principal component with the niche center. We calculate Z scores as a measure of standardized difference between range center and niche center. For environmental variables, Z scores were calculated as (condition at geographic center minus range mean)/standard deviation (all cells of each condition within range). For niche centers, Z scores were calculated as (Principal component coordinate at geographic center minus mean of principal component coordinates)/standard deviation (all of principal component coordinates in principal component). For individual variables, we extracted annual temperature and precipitation, slope and elevation from climatic maps at the coordinates of the geographic center. We calculate the species range mean for each condition by extracting all cells within occupied biome types and elevation ranges collated from IUCN assessments for each species. Geographic centers for historic and contemporary ranges were calculated separately and the respective environmental variables were extracted from each period to account for the changing range centers and climatic conditions across the time periods. Geographic center was estimated using getSpPolygonsLabbptSlots function in sp package (77). We used the same niche models (identical climatic and topographic factors) as those in the ecological marginalization analysis for niche center analysis (PCA). Species with geographic centers within oceans or freshwater bodies were removed from the analysis. We conducted a meta-analysis of resultant Z scores using the Stouffer’s aggregate z-score for each ecological variable and three principal components (76).
Modeling and Data Analysis.
All data analyses were performed in R (77). We used phylogenetic generalized least squares (PGLS) models to assess the effect of intrinsic variables and human transformation on the metrics of loss, marginalization, shift, and extinction risk. PGLS models use phylogenetic relationships to account for the expected covariance between species from close phylogeny (78). PGLS models were conducted using “caper” package (79). We calculated Mahalanobis distances using the Mahalanobis function in the package stats (77). All GIS operations were conducted using the raster package (80). Random points were generated using sp package (77). All Cohen’s d estimates at 95% confidence intervals were conducted using rstatix (77). Model selection maximized AICc of global models using dredge function in MuMIn package in R (77). The explanatory variables were transformed to Gaussian distributions to conform to model assumptions. Contemporary range size, body mass, effect size of Mahalanobis distance (ecological marginalization) and distance between niche centroids were log-transformed. Geographic range loss, habitat diversity loss and human transformation were logit transformed. Geographic marginalization was asin-transformed. Absolute effect size of shift of annual precipitation, temperature, slope, and elevation were log-transformed. We used conditional model averaging for models within ΔAIC < 2 of the best fit model. We calculated factor weight across all models using MuMIn::importance function from MuMIn package in R (77). Aggregate Z scores at were calculated using stouffer_z function in auctestr package (76).
Supplementary Material
Appendix 01 (PDF)
Acknowledgments
Author contributions
S.S. proposed the manuscript, J.A.B. and S.S. designed research; J.A.B. and Y.Z. performed research; J.A.B. and S.S. analyzed data; and J.A.B., G.I.H.K., and S.S. wrote the paper.
Competing interest
The authors declare no competing interest.
Footnotes
This article is a PNAS Direct Submission.
Contributor Information
Jake Alan Britnell, Email: jake.britnell@manchester.ac.uk.
Susanne Shultz, Email: susanne.shultz@manchester.ac.uk.
Data, Materials, and Software Availability
Code for generating niche hypervolumes, minimum convex hulls and effect size changes, extracting climatic variables range contracted species, CSVs of species niche parameters for data analysis, PGLS data analysis across species, and code for niche and range center analysis data have been deposited in Dryad (81).
Supporting Information
References
- 1.Ellis E. C., et al. , People have shaped most of terrestrial nature for at least 12,000 years. Proc. Natl. Acad. Sci. U.S.A. 118, e2023483118 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ellis E. C., Anthropogenic transformation of the terrestrial biosphere. Philos. Trans. R. Soc. A Math. Phys. Eng. Sci. 369, 1010–1035 (2011). [DOI] [PubMed] [Google Scholar]
- 3.Tilman D., et al. , Future threats to biodiversity and pathways to their prevention. Nature 546, 73–81 (2017). [DOI] [PubMed] [Google Scholar]
- 4.UNEP-WCMC and IUCN, “UNEP-WCMC and IUCN protected planet report 2020” (UNEP-WCMC and IUCN, Cambridge UK, Switzerland, Gland, 2020). [Google Scholar]
- 5.Kuussaari M., et al. , Extinction debt: A challenge for biodiversity conservation. Trends Ecol. Evol. 24, 564–571 (2009). [DOI] [PubMed] [Google Scholar]
- 6.Kerley G. I. H., Beest M., Cromsigt J. P. G. M., Pauly D., Shultz S., The protected area paradox and refugee species: The giant panda and baselines shifted towards conserving species in marginal habitats. Cons. Sci. Pract. 2, e11 (2020). [Google Scholar]
- 7.Kerley G. I. H., Kowalczyk R., Cromsigt J. P. G. M., Conservation implications of the refugee species concept and the European bison: King of the forest or refugee in a marginal habitat? Ecography 35, 519–529 (2012). [Google Scholar]
- 8.Sayre R., et al. , An assessment of the representation of ecosystems in global protected areas using new maps of World climate regions and World ecosystems. Glob. Ecol. Conserv. 21, e00860 (2020). [Google Scholar]
- 9.Venter O., et al. , Bias in protected-area location and its effects on long-term aspirations of biodiversity conventions. Conserv. Biol. 32, 127–134 (2018). [DOI] [PubMed] [Google Scholar]
- 10.Joppa L. N., Pfaff A., High and far: Biases in the location of protected areas. PLoS One 4, e8273 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Huston M. A., The three phases of land-use change: Implications for biodiversity. Ecol. Appl. 15, 1864–1878 (2005). [Google Scholar]
- 12.Ellis E. C., Ramankutty N., Putting people in the map: Anthropogenic biomes of the world. Front Ecol. Environ. 6, 439–447 (2008). [Google Scholar]
- 13.Jacobson A. P., Riggio J., Tait A. M., Baillie J. E. M., Global areas of low human impact (‘Low Impact Areas’) and fragmentation of the natural world. Sci. Rep. 9, 1–13 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Ellis E. C., Goldstein M. I. , DellaSala D. A., “Anthromes” in Encyclopedia of the World’s Biomes Elsevier; (Michael I. Goldstein & Dominick A. Della Sala Imprint: Elsevier ISBN 978-0-12-816097-8, 2020), vol. 5–5, pp. 5–11. [Google Scholar]
- 15.Kawecki T. J., Adaptation to marginal habitats. Annu. Rev. Ecol. Evol. Systemat. 39, 321–342 (2008). [Google Scholar]
- 16.Pulliam H. R., Sources, sinks, and population regulation. Am. Nat. 132, 652–661 (1988). [Google Scholar]
- 17.Pironon S., et al. , Geographic variation in genetic and demographic performance: New insights from an old biogeographical paradigm. Biol. Rev. 92, 1877–1909 (2017). [DOI] [PubMed] [Google Scholar]
- 18.Scheele B. C., Foster C. N., Banks S. C., Lindenmayer D. B., Niche contractions in declining species: Mechanisms and consequences. Trends Ecol. Evol. 32, 346–355 (2017). [DOI] [PubMed] [Google Scholar]
- 19.Channell R., Lomolino M. V., Trajectories to extinction: Spatial dynamics of the contraction of geographic ranges of endangered species. J. Biogeogr. 27, 169–180 (2000). [Google Scholar]
- 20.Lea J. M., Kerley G. I., Hrabar H., Barry T. J., Shultz S., Recognition and management of ecological refugees: A case study of the Cape mountain zebra. Biol. Conserv. 203, 207–215 (2016). [Google Scholar]
- 21.Cowell R., Rangel T., Hutchinson’s duality: The once and future niche. Proc. Natl. Acad. Sci. 106, 19651–19658 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Osorio-Olvera L., Yañez-Arenas C., Martínez-Meyer E., Peterson A. T., Relationships between population densities and niche-centroid distances in North American birds. Ecol. Lett. 23, 555–564 (2020). [DOI] [PubMed] [Google Scholar]
- 23.Ripple W. J., et al. , Extinction risk is most acute for the world’s largest and smallest vertebrates. Proc. Natl Acad. Sci. U.S.A. 114, 10678–10683 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Crowley B. E., et al. , Extinction and ecological retreat in a community of primates. Proc. R. Soc. B 279, 3597–3605 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Brown J. H., On the relationship between abundance and distribution of species. Am. Nat. 124, 255–279 (1984). [Google Scholar]
- 26.Martínez-Meyer E., Díaz-Porras D., Peterson A. T., Yáñez-Arenas C., Ecological niche structure and rangewide abundance patterns of species. Biol. Lett. 9, 20120637 (2013). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Holt R. D., et al. , Theoretical models of species’ borders: Single species approaches. Oikos 108, 18–27 (2005). [Google Scholar]
- 28.Sagarin R. D., Gaines S. D., Gaylord B., Moving beyond assumptions to understand abundance distributions across the ranges of species. Trends Ecol. Evol. 21, 524–530 (2006). [DOI] [PubMed] [Google Scholar]
- 29.Lee J. E., Janion C., Marais E., van Vuuren B. J., Chown S. L., Physiological tolerances account for range limits and abundance structure in an invasive slug. Proc. R. Soc. B 276, 1459 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Caughley G., Grice D., Barker R., Brown B., The edge of the range. J. Anim. Ecol. 57, 771 (1988). [Google Scholar]
- 31.Chevalier M., Broennimann O., Guisan A., Using a robust multi-settings inference framework on published datasets still reveals limited support for the abundant centre hypothesis: More testing needed on other datasets. Glob. Ecol. Biogeogr. 30, 2211–2228 (2021). [Google Scholar]
- 32.Osorio-Olvera L., Soberón J., Falconi M., On population abundance and niche structure. Ecography 42, 1415–1425 (2019). [Google Scholar]
- 33.Santini L., Pironon S., Maiorano L., Thuiller W., Addressing common pitfalls does not provide more support to geographical and ecological abundant-centre hypotheses. Ecography 42, 696–705 (2019). [Google Scholar]
- 34.Burner R. C., Styring A. R., Rahman M. A., Sheldon F. H., Occupancy patterns and upper range limits of lowland Bornean birds along an elevational gradient. J. Biogeogr. 46, 2583–2596 (2019). [Google Scholar]
- 35.Lira-Noriega A., Manthey J. D., Relationship of genetic diversity and niche centrality: A survey and analysis. Evolution 68, 1082–1093 (2014). [DOI] [PubMed] [Google Scholar]
- 36.Manthey J. D., et al. , A test of niche centrality as a determinant of population trends and conservation status in threatened and endangered North American birds. Endangered Species Res. 26, 201–208 (2015). [Google Scholar]
- 37.Martínez-Gutiérrez P. G., Martínez-Meyer E., Palomares F., Fernández N., Niche centrality and human influence predict rangewide variation in population abundance of a widespread mammal: The collared peccary (Pecari tajacu). Divers. Distrib. 24, 103–115 (2018). [Google Scholar]
- 38.Dallas T., Decker R. R., Hastings A., Species are not most abundant in the centre of their geographic range or climatic niche. Ecol. Lett. 20, 1526–1533 (2017). [DOI] [PubMed] [Google Scholar]
- 39.Holt R. D., Reflections on niches and numbers. Ecography 43, 387–390 (2020). [Google Scholar]
- 40.Caughley G., Directions in conservation biology. J. Animal Ecol. 63, 215 (1994). [Google Scholar]
- 41.Parmesan C., Singer M Mosaics of climatic stress across species’ ranges: Tradeoffs cause adaptive evolution to limits of climatic tolerance. Philos. Trans. R. Soc. B Biol. Sci. 377, 20210003 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Purvis A., Gittleman J. L., Cowlishaw G., Mace G. M., Predicting extinction risk in declining species. Proc. Biol. Sci. 267, 1947–1952 (2000). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Benítez-López A., et al. , The impact of hunting on tropical mammal and bird populations. Science 356, 180–183 (2017). [DOI] [PubMed] [Google Scholar]
- 44.Fisher D. O., Trajectories from extinction: Where are missing mammals rediscovered? Glob. Ecol. Biogeogr. 20, 415–425 (2011). [Google Scholar]
- 45.Hughes T. P., Bellwood D. R., Connolly S. R., H. v. Cornell, R. H. Karlson, Double jeopardy and global extinction risk in corals and reef fishes. Curr. Boil. 24, 2946–2951 (2014). [DOI] [PubMed] [Google Scholar]
- 46.Pacifici M., di Marco M., Watson J. E. M., Protected areas are now the last strongholds for many imperiled mammal species. Conserv. Lett. 13, e12748 (2020). [Google Scholar]
- 47.Hanson J. O., et al. , Global conservation of species’ niches. Nature 580, 232–234 (2020). [DOI] [PubMed] [Google Scholar]
- 48.Williams D., Rondinini C., Tilman D., Global protected areas seem insufficient to safeguard half of the world’s mammals from human-induced extinction. Proc. Natl. Acad. Sci. U.S.A. 119, e2200118119 (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Robson A., et al. , Over 80% of Africa’s savannah conservation land is failing or deteriorating according to lions as an indicator species. Conserv. Lett. 15, e12844 (2021). [Google Scholar]
- 50.Rada S., et al. , Protected areas do not mitigate biodiversity declines: A case study on butterflies. Divers. Distrib. 25, 217–224 (2019). [Google Scholar]
- 51.Margules C. R., Pressey R. L., Systematic conservation planning. Nature 405, 243–253 (2000). [DOI] [PubMed] [Google Scholar]
- 52.Plumb G., Kowalczyk R., Hernandez-Blanco J. A., Bison bonasus. The IUCN Red List of Threatened Species 2020, E.T2814A45156279. 10.2305/IUCN.UK.2020-3.RLTS.T2814A45156279.en (2020). [DOI]
- 53.Kerley G., Pressey R., Cowling R., Boshoff A., Sims-Castley R., Options for the conservation of large and medium-sized mammals in the Cape Floristic Region hotspot, South Africa. Biol. Conserv. 112, 169–190 (2003). [Google Scholar]
- 54.Britnell J. A., et al. , Species stereotypes as a result of unconscious research biases compromise conservation efficacy. Biol. Conserv. 261, 109275 (2021). [Google Scholar]
- 55.Kerley G. I. H., Monsarrat S., Shifted models cannot be used for predicting responses of biodiversity to global change: The African elephant as an example. Afr. Zool. 57, 70–73 (2022). [Google Scholar]
- 56.Monsarrat S., Novellie P. A., Rushworth I., Kerley G. I. H., Shifted distribution baselines: Neglecting long-term biodiversity records risks overlooking potentially suitable habitat for conservation management. Philos. Trans. R. Soc. B 374, 937 (2019). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sales L. P., et al. , The effect of past defaunation on ranges, niches, and future biodiversity forecasts. Glob. Change Biol. 28, 3683–3693 (2022). [DOI] [PubMed] [Google Scholar]
- 58.Jones K. E., et al. , PanTHERIA: A species-level database of life history, ecology, and geography of extant and recently extinct mammals. Ecology 90, 2648–2648 (2009). [Google Scholar]
- 59.Soria C. D., et al. , COMBINE: A coalesced mammal database of intrinsic and extrinsic traits. Ecology 102, e03344 (2021). [DOI] [PubMed] [Google Scholar]
- 60.Faurby S., et al. , Phylacine 1.2: The phylogenetic atlas of mammal macroecology. Ecology 99, 2626 (2018). [DOI] [PubMed] [Google Scholar]
- 61.di Marco M., Pacifici M., Maiorano L., Rondinini C., Drivers of change in the realised climatic niche of terrestrial mammals. Ecography 44, 1180–1190 (2021). [Google Scholar]
- 62.Dinerstein E., et al. , An ecoregion-based approach to protecting half the terrestrial realm. BioScience 67, 534–545 (2017). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ellis E. C., Goldewijk K. K., Siebert S., Lightman D., Ramankutty N., Anthropogenic transformation of the biomes, 1700 to 2000. Glob. Ecol. Biogeogr. 19, 589–606 (2010). [Google Scholar]
- 64.Broennimann O., et al. , Distance to native climatic niche margins explains establishment success of alien mammals. Nat. Commun. 12, 1–8 (2021). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Fick S. E., Hijmans R. J., WorldClim 2: New 1-km spatial resolution climate surfaces for global land areas. Int. J. Climatol. 37, 4302–4315 (2017). [Google Scholar]
- 66.Hijmans R. J., Cameron S. E., Parra J. L., Jones P. G., Jarvis A., Very high resolution interpolated climate surfaces for global land areas. Int. J. Climatol. 25, 1965–1978 (2005). [Google Scholar]
- 67.Knutti R., Masson D., Gettelman A., Climate model genealogy: Generation CMIP5 and how we got there. Geophys. Res. Lett. 40, 1194–1199 (2013). [Google Scholar]
- 68.Hijmans R. J., van Etten J., raster: Geographic Data Analysis and Modeling (R package version 2.2-31, The R Foundation for Statistical Computing, Vienna, 2014). https://cran.r-project.org/. [Google Scholar]
- 69.Visconti P., et al. , Projecting global biodiversity indicators under future development scenarios. Conserv. Lett. 9, 5–13 (2016). [Google Scholar]
- 70.Drake J. M., Randin C., Guisan A., Modelling ecological niches with support vector machines. J. Appl. Ecol. 43, 424–432 (2006). [Google Scholar]
- 71.Blonder B., Lamanna C., Violle C., Enquist B. J., The n-dimensional hypervolume. Glob. Ecol. Biogeogr. 23, 595–609 (2014). [Google Scholar]
- 72.Blonder B., et al. , New approaches for delineating n-dimensional hypervolumes. Methods in Ecol. Evol. 9, 305–319 (2018). [Google Scholar]
- 73.Qiao H., et al. , NicheA: Creating virtual species and ecological niches in multivariate environmental scenarios. Ecography 39, 805–813 (2016). [Google Scholar]
- 74.Osorio-Olvera L., et al. , ntbox: An r package with graphical user interface for modelling and evaluating multidimensional ecological niches. Methods in Ecol. Evol. 11, 1199–1206 (2020). [Google Scholar]
- 75.Soberón J., Peterson A. T., Osorio-Olvera L., A comment on “Species are not most abundant in the centre of their geographic range or climatic niche”. Rethinking Ecol. 3, 13–18 (2018). [Google Scholar]
- 76.Tingley M. W., Monahan W. B., Beissinger S. R., Moritz C., Birds track their Grinnellian niche through a century of climate change. Proc. Natl. Acad. Sci. U.S.A. 106, 19637–19643 (2009). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.R Core Team, R: A Language and Environment for Statistical Computing, Vienna, Austria. (R Foundation for Statistical Computing, Vienna, Austria, 2021), https://www.R-project.org/. [Google Scholar]
- 78.Symonds M. R., Blomberg S. P., “A primer on phylogenetic generalised least squares” in Modern Phylogenetic Comparative Methods and Their Application in Evolutionary Biology (Springer, Berlin, Heidelberg, 2014), pp. 105–130. [Google Scholar]
- 79.Orme D., et al. , caper: Comparative Analyses of Phylogenetics and Evolution in R. R package version 1.0.1. (2013). http://CRAN.R-project.org/package=caper.
- 80.Paradis E., Claude J., Strimmer K., APE: Analyses of phylogenetics and evolution in R language. Bioinformatics 20, 289–29 (2004). [DOI] [PubMed] [Google Scholar]
- 81.Britnell J. A., Yichun Z., G.I.H, Kerley, Shultz S., Ecological marginalization is widespread and increases extinction risk in mammals, Dryad, Dataset. 10.5061/dryad.k3j9kd5bv. Deposited 4 December 2022. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix 01 (PDF)
Data Availability Statement
Code for generating niche hypervolumes, minimum convex hulls and effect size changes, extracting climatic variables range contracted species, CSVs of species niche parameters for data analysis, PGLS data analysis across species, and code for niche and range center analysis data have been deposited in Dryad (81).





