Abstract
We investigated the effect of preoperative therapy for non-small cell lung cancer on programmed death-ligand 1 (PD-L1), programmed death-1 (PD-1), poliovirus receptor (CD155), and T cell immunoglobulin and immunoreceptor tyrosine-based inhibitory motif (ITIM) domain (TIGIT) expression and prognosis with the cases of 28 patients received preoperative concurrent chemo-radiotherapy (cCRT) and 27 received preoperative drug therapy. The post-treatment PD-L1 expression was higher in cCRT group than in the drug therapy (50.0% vs 5.0%, p = 0.000), whereas that of CD155 did not significantly differ (40.0% vs 60.0%, p = 0.131). The PD-1 expression was not significantly different between the cCRT and drug therapy groups (51.1% vs 42.9%, p = 0.076), while the TIGIT was significantly higher in the cCRT group (41.5% vs 34.0%, p = 0.008). The patients who received cCRT resulted in elevated PD-L1and TIGIT values had a worse prognosis (p = 0.008). The PD-L1 and TIGIT expression after cCRT was significantly higher than after drug treatment. The cCRT population with high expression of both had a significantly poorer prognosis, indicating elevation of PD-L1 and TIGIT after cCRT as a negative prognostic factor. Combination therapy with anti-PD-L1 and anti-TIGIT antibodies after cCRT may contribute to an improved prognosis.
Subject terms: Cancer microenvironment, Lung cancer
Introduction
Non-small cell lung cancer (NSCLC) accounts for the majority of lung cancer, which is globally the foremost cause of cancer death1. Many newly diagnosed NSCLC patients are with metastatic lesions and traditionally have been treated first with a platinum doublet chemotherapy with limited efficacy. However, the improved prognosis noted with immunotherapies such as programmed death-1/programmed death-ligand 1 (PD-1/PD-L1) axis immune checkpoint inhibitors (ICIs) has changed the treatment strategy2. Despite their durable response, ICI monotherapy has a response rate of at most 20%, leading to the development of combinations of ICIs. Basic research reported good prospects for combination therapy with multiple ICIs3,4, which has also been proven in clinical studies5–8. In the phase II CITYSCAPE trial, anti-T cell immunoglobulin and immunoreceptor tyrosine-based inhibitory motif (ITIM) domain (TIGIT) antibody tiragolumab combined with the PD-L1 inhibitor atezolizumab significantly improved the overall response rate compared to anti-PD-L1 monotherapy (37% vs. 21%) (37% vs. 21%)9. In patients with tumors with high PD-L1 expression (n = 58), the objective response rate was 69.0% (95% CI 50.4–87.5) verus 24.1% (6.8–41.4) and median progression-free survival was 16.6 (95% CI 5.5–22.3) vs 4.1 months [2.1–6.8; HR 0.29 (95% CI 0.15–0.053)]10,11. However, the phase 3 SKYSCRAPER-01 study did not meet its co-primary endpoint of progression-free survival12.
Approximately one-third of newly diagnosed NSCLC patients is locally advanced disease irrelevant to surgical resection13. Concurrent chemo-radiotherapy (cCRT) has been a typical treatment for these patients despite 5-years overall survival (OS) rates of as low as 15%14. Recently, the PACIFIC, a phase III randomized, double-blind trial of durvalumab (an antibody against programmed death ligand-1: PD-L1) vs. placebo, demonstrated that the consolidation treatment with durvalumab for patients without disease progression after cCRT significantly improved their prognosis (4-years OS; 49.6% vs. 36.3%)15–18. As regards an association between the promising combination, anti-TIGIT and anti-PD-L1 therapy, and radiotherapy (RT), RT-induced TIGIT and PD-L1 upregulation and RT in combination with anti-TIGIT and PD-L1 inhibitor showed a complete response (CR) rate of 90% in a mouse model19. RT combined with anti-TIGIT and anti-PD-L1 antibodies is considered to be a hopeful approach. To date, however, no clinical evidence has been reported as concerns the association between RT and alteration in the immune-marker state, TIGIT and PD-L1 expression, and the rationale of RT with anti-TIGIT and anti-PD-L1 antibody drugs.
Previously, we reported a significant up-regulation of PD-L1 expression on the tumor cells and an increased stromal density of CD8 + tumor infiltrative lymphocytes (TILs) in paired clinical samples before and after cCRT20. In this study, we analyzed 14 more cases in addition to the 41 cases reported in the previous study. Here, we aimed to assess in the same and subsequent clinical samples the effect of RT on PD-L1 and TIGIT expression together with their major counterparts, PD-1 and poliovirus receptor (CD155), and their prognostic impact. We compared the expression and influence of the markers on prognosis in patients undergoing either cCRT or drug therapy before surgery.
Results
Characteristics of patients
Characteristics of the patients and their treatments are summarized in Table 1. We compared 28 patients who underwent cCRT with 27 patients who underwent drug therapy before surgery. All patients of the cCRT group received platinum doublet chemotherapy and radiotherapy concurrently with a median dose of 60 Gy. In the drug therapy group, there were TKI and bronchial artery infusion with cisplatin, as well as platinum doublets.
Table 1.
Characteristics of patients classified by induction therapies.
| cCRT group | Drug-therapy group | p-value | |
|---|---|---|---|
| (n = 28) | (n = 27) | ||
| Sex | 0.931 | ||
| Male | 20 (71.4%) | 19 (70.4%) | |
| Female | 8 (28.6%) | 8 (29.6%) | |
| Age, median (range) | 66 (40–79) | 69 (46–84) | 0.111 |
| Smoking status | 0.937 | ||
| Never | 7 (25.0%) | 7 (25.9%) | |
| Former or current | 21 (75.0%) | 20 (74.1%) | |
| Brinkman index, median (range) | 765 (0–1760) | 940 (0–1880) | 0.734 |
| Comorbidity | 20 (71.4%) | 24 (88.9%) | 0.106 |
| Histology | 0.993 | ||
| Adenocarcinoma | 16 (57.1%) | 15 (55.6%) | |
| Squamous | 10 (35.7%) | 10 (37.0%) | |
| Others | 2 (7.1%) | 2 (7.4%) | |
| Clinical stage | 0.729 | ||
| IIA or IIB | 6 (21.4%) | 4 (14.8%) | |
| ≥ IIIA | 22 (78.6%) | 23 (85.2%) | |
| Drug therapy | 0.001 | ||
| Platina doublet | 28 (100%) | 15 (55.6%) | |
| Other cytotoxic | 0 (0%) | 1 (3.7%) | |
| Bronchial artery infusion | 0 (0%) | 4 (14.8%) | |
| Tyrosine kinase inhibitor | 0 (0%) | 7 (25.9%) | |
| Radiotherapy dose (Gy), median (range) | 60 (44–70) | – | 0.000 |
| Clinical response | 0.587 | ||
| PD or SD | 10 (35.7%) | 12 (44.4%) | |
| PR | 18 (64.3%) | 15 (55.6%) | |
| Resection | 0.469 | ||
| Lobectomy | 25 (89.3%) | 22 (81.5%) | |
| Pneumonectomy | 3 (10.7%) | 5 (18.5%) | |
| Effect of induction therapy | 0.000 | ||
| Ef1 | 7 (25.0%) | 21 (77.8%) | |
| Ef2 | 21 (75.0%) | 6 (22.2%) |
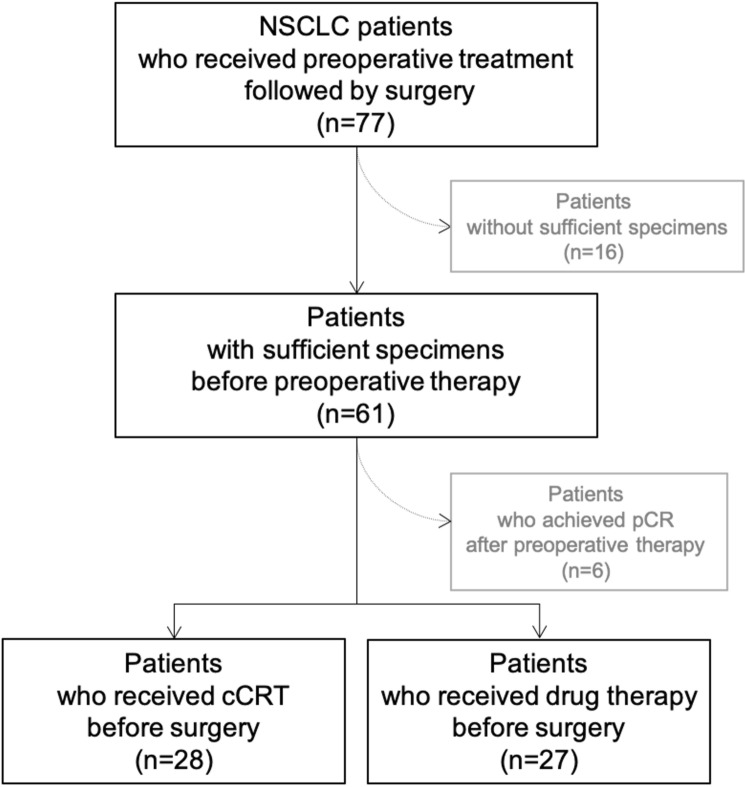
The therapeutic effect of the cCRT was higher than in the drug therapy group. The proportion of Ef2 was 75.0% in the cCRT group vs 22.2% in the drug therapy group (p = 0.000) (Fig. 1).
Figure 1.
Flow diagram of the present study participants.
PD-L1 and CD155 expression
To assess the effect of induction therapy on PD-L1 and CD155 expression of the tumor, we evaluated their expression before and after induction therapy. Whereas cCRT group demonstrated a statistically significant upregulation in PD-L1 (p = 0.000) and a downregulated trend in CD155 expression (p = 0.082, Fig. 2A), the drug therapy group showed no significant difference in PD-L1 (p = 0.242) and CD155 expression (p = 0.362, Fig. 2B). The post-treatment median TPS of PD-L1 was tenfold higher in the cCRT group than in the drug therapy (50.0% vs 5.0%, p = 0.000). The post-treatment median TPS of CD155 was not significantly different between the two groups (40.0% vs 60.0%, p = 0.131).
Figure 2.
Alteration in PD-L1 and CD155 expression on tumor cells. The results are shown according to (A) the patients treated with concurrent chemo-radiotherapy and (B) those treated with drug therapy.
PD-1 and TIGIT expression of CD8 + lymphocytes
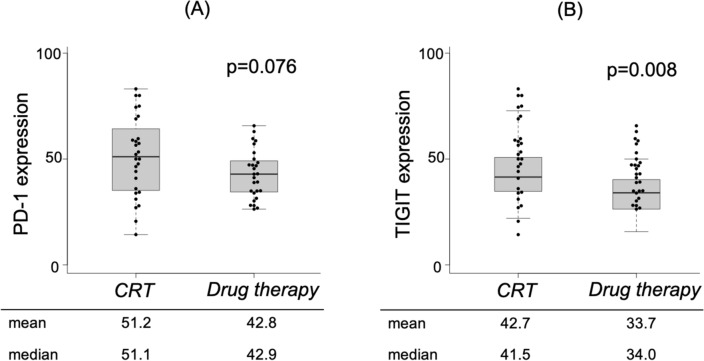
To evaluate the influence of induction therapy on PD-1 and TIGIT expression of CD8 + lymphocytes, we assessed their expression rate in the post-treatment specimens. The PD-1 expression was not statistically different between the cCRT and drug therapy group (51.1% vs 42.9%, p = 0.076, Fig. 3A). The TIGIT expression was statistically higher in the cCRT group compared to the drug therapy group (41.5% vs 34.0%, p = 0.008, Fig. 3B).
Figure 3.
Expression rate of the TIL markers on CD8 + cells after induction therapy. The results are shown according to (A) PD-1 and (B) TIGIT expression.
Cut-off value for prognostic analyses
A receiver operating characteristic curve analysis of PD-L1, CD155, PD-1, and TIGIT for prediction of death was performed, indicating an optimal cut-off value of 25% (AUC = 0.640), 45% (AUC = 0.425), 40% (AUC = 0.483), and 40% (AUC = 0.477), respectively. Based on these results, each case was classified as marker-high (cut-off value or higher) or marker-low (less than the cut-off value).
Overall Survival regarding each biomarker
The OS curves in respect to PD-L1, CD155, PD-1, and TIGIT expression of all patients are shown in Fig. 4A. Except for PD-L1 expression, there was no significant correlation between CD155, PD-1 and TIGIT expression and OS (PD-L1: p = 0.028, CD155: p = 0.312, PD-1: p = 0.800, TIGIT: p = 0.504). The OS curves for each marker of the cCRT group are demonstrated in Fig. 4B. There was no significant relation between any marker and OS (PD-L1: p = 0.052, CD155: p = 0.306, PD-1: p = 0.952, TIGIT: p = 0.178). The OS curves regarding each marker of the drug therapy group are presented in Fig. 4C. There was no significant correlation between each marker and OS (PD-L1: p = 0.183, CD155: p = 0.513, PD-1: p = 0.787, TIGIT: p = 0.546).
Figure 4.
Overall survival curves compared with the expression of immune markers, PD-L1, CD155, PD-1, and TIGIT. The results are shown according to (A) all patients, (B) patients treated with cCRT, and (C) those treated with drug therapy.
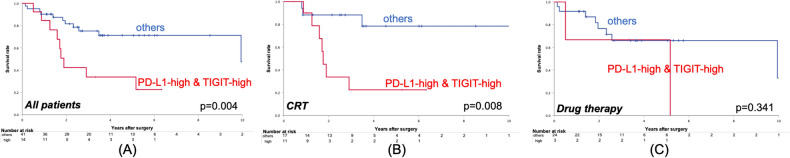
Overall survival regarding PD-L1-high and TIGIT-high
Figure 5 shows the OS in PD-L1-high and TIGIT-high patients. The OS of the patients with elevated PD-L1 and TIGIT was significantly worse (Fig. 5A, p = 0.004). In the CRT group, the survival in cases with elevated PD-L1 and TIGIT was significantly worse (Fig. 5B, p = 0.008), whereas there was no difference in the drug therapy group (Fig. 5C, p = 0.341).
Figure 5.
Overall survival curves regarding the expression of PD-L1 together with TIGIT. The results are shown according to (A) all patients, (B) patients treated with cCRT, and (C) those treated with drug therapy.
Discussion
In this retrospective study, we evaluated the expression of immune markers, PD-L1, CD155, PD-1, and TIGIT after cCRT or drug therapy and their relation to the prognosis in a series of patients with NSCLC. Our results demonstrated that the expression of PD-L1 was upregulated and the expression rate of TIGIT was high after cCRT relative to that after drug therapy, and the patients with NSCLC after cCRT with high expression of both PD-L1 and TIGIT had a significantly poor prognosis. These findings suggest that combination therapy with anti-PD-L1 and anti-TIGIT antibodies after cCRT may contribute to an improved prognosis.
We found that the samples after cCRT demonstrated a significant increase in PD-L1 expression (p = 0.000) in comparison to before the treatment. This result was consistent with our previous study20 and is supported by several preclinical and clinical studies21–23. Regarding CD155, our result showed that the samples after cCRT tend to be a lower expression (p = 0.082) compared to before the therapy. The impact of RT on CD155 expression has not been reported so far. The PD-1 expression rate of CD8 + cells was prone to be high in the cCRT group relative to in the drug therapy group, though the difference was not statistically significant (p = 0.076). While some experiments with mouse models suggested that RT had an effect of increasing PD-1 expression23–26, another study showed no significant difference in PD-121. In our study, the TIGIT expression rate was higher in the cCRT group than in the drug therapy group (p = 0.008). A preclinical study revealed that RT leads to increased TIGIT expression19. Our study may indicate that RT for NSCLC is associated with upregulation of PD-L1, downregulation of CD155, and high expression of PD-1 and TIGIT.
Next, we focused on the prognostic impact of the immune markers. Of the four markers, PD-L1 was the only statistically significant prognostic factor for OS, with a poor prognosis for high PD-L1 expression (Fig. 4A). This trend was prominent in the CRT group and was not observed in the drug therapy group.
In NSCLC, reports of PD-L1 as a prognostic factor have been inconclusive27–33. This study differs from other studies in that all the patients had undergone the preoperative treatments. Tumors that can express PD-L1 due to the high density of CD8 + cells after cCRT and their IFN-γsecretion34,35 may be associated with poor prognosis through their ability to escape the immune system. Furthermore, in this study, the prognosis was significantly poor in the subgroup with high expression group of both PD-L1 and TIGIT, and this event was also remarkable in the cCRT group and was not observed in the drug therapy group (Fig. 5). In this subgroup, exhaustion of T cells and NK cells by TIGIT36, in addition to immune escape through PD-L1, may have resulted in tumor progression and a worse prognosis. These results indicate that the combined use of anti-PD-L1 antibody and anti-TIGIT antibody after cCRT for NSCLC may improve their prognosis.
Several limitations of our study warrant mention. First, the study was conducted under a retrospective design at a single center. Although we decided upon an ideal cut-off value for each marker using ROC analysis, this should be confirmed in a larger cohort. Second, the patients in this study had a wide variety of backgrounds. The RT doses ranged widely and the kinds of the drug therapy were diverse and included TKIs. Third, it would be desirable to have a baseline assessment in evaluating the impact of preoperative treatment, which is not being done. Finally, the present study was conducted on a small number of subjects. These might affect the results. Allowing for these issues, however, we believe that our findings are valuable in this age of ICIs combination therapy considering the scarcity of specimens after preoperative therapy.
Conclusion
PD-L1 expression after cCRT was higher than before the therapy and TIGIT expression rate was higher in the cCRT group than in the drug therapy group. The prognosis of patients with elevation of both PD-L1- and TIGIT patients undergoing cCRT was poorer than that of the others in the cCRT group. These findings may indicate a clinical rationale for combining anti-PD-L1 and anti-TIGIT antibodies after cCRT.
Materials and methods
Patients
In this retrospective study, we reviewed cases of NSCLC patients who had received preoperative therapy followed by surgery from April 2006 to October 2020 in our department (Fig. 1). Inclusion criteria were available tumor specimens before and after preoperative therapy satisfactory for pathological evaluation of PD-L1 and CD155, and surgical specimens satisfactory for assessment of PD-1 and TIGIT expression of stromal CD8 + TIL. Of 77 patients who had undergone preoperative therapy. Sixteen cases were excluded because of insufficient formalin-fixed paraffin-embedded samples before therapy, and six were excluded because of a lack of viable tumor cells in surgical specimens due to pathological CR. We reviewed the remaining 55 patients, of whom 28 underwent preoperative cCRT and 27 underwent preoperative drug therapy alone.
The study protocol was approved by the University of Occupational and Environmental Health, Japan (UOEHCRB21-154), and the study was performed in accordance with the Declaration of Helsinki. Informed consent was obtained from all subjects or their legal guardians.
Pathological effect of induction therapy
The effect of induction therapy was assessed based on the criteria in the classification of lung cancer by the Japan lung cancer society. Ef3 means that there are no viable tumor cells. Ef2 means that less than one-third of tumor cells were viable. Ef1 means that more than one-third of tumor cells were viable despite the existence of a therapeutic effect.
Evaluation of tumor immune microenvironment
Consecutive 4-μm tissue sections were used for hematoxylin–eosin staining and immunohistochemistry (IHC) staining. The objects of IHC in this study were PD-L1, CD155, PD-1 of CD8 + lymphocytes, and TIGIT of CD8 + lymphocytes. PD-L1 staining was performed as described previously37. Tissue sections were deparaffinized and incubated in ethylenediaminetetraacetic acid (pH 8.0) at 98 °C for antigen retrieval. Endogenous peroxidase was blocked using 3% H2O2. After nonspecific reaction blocking with Protein Block Serum-Free (Agilent Technologies, Palo Alto CA, USA), we incubated sections with rabbit anti–PD-L1 monoclonal antibody (clone E1L3N, Cell Signaling Technology Japan, Tokyo, Japan) diluted at 1:200 and washed and incubated with a detection reagent (SignalStain Boost IHC Detection Reagent HRP Rabbit, Cell Signaling Technology, Tokyo, Japan). The sections were visualized with DAB + Liquid (Agilent Technologies) and counterstained with Mayer’s hematoxylin. Retrieval of primary antibody and secondary antibody of the remaining IHC were as follows: as described previously5, IHC of CD155 was performed with citrate buffer (pH 6.0) at 98 °C for antigen retrieval and mouse anti-CD155 monoclonal antibody (clone B6, Santa Cruz Biotechnology, Texas, US) and Histofine Simple Stain, MAX-PO (Nichirei Bioscience, Tokyo, Japan). The other than these the same as described above. For CD8 and PD-1 multistaining, we used citrate buffer (pH 6.0) at 98 °C as antigen retrieval buffer and rabbit anti-CD8 antibody (clone 1779R, Novus Biologicals, Centennial CO, US) diluted at 1:300 and mouse anti-PD-1 antibody (clone NAT105, Abcam, Cambridge, UK) diluted at 1:50 as the primary antibody and Alexa Flour 488-conjugated goat anti-rabbit IgG (Thermo Fisher Scientific, Massachusetts, US) diluted at 1:500 and Alexa Fluor 594-conjugated goat anti-mouse IgG (Thermo Fisher Scientific, Waltham MA, US) diluted to 1:500 as the secondary antibodies. For CD8 and TIGIT multistaining, the antigen retrieval buffer was Tris-ethylenediaminetetraacetic acid (pH 9.0) at 98 °C. The primary anti- TIGIT antibody was mouse anti-TIGIT antibody (clone TG1, Dianova, Hamburg, Germany) diluted to 1:50. The secondary antibody was the same as above.
We examined the expression of the tumor markers PD-L1, and CD155 in paired samples obtained before and after preoperative therapy. For the TIL markers, PD-1 and TIGIT, the samples after preoperative therapy alone were evaluated because of the inability to evaluate tiny specimens before preoperative therapy with the above methods. Expression of PD-L1, CD155, PD-1, and TIGIT was independently evaluated by two of the investigators who were blinded to clinical data. PD-L1 and CD155 were recoded as tumor proportion scores (TPS). The expression rate of PD-1 or TIGIT was calculated as the ratio of CD8-positive and PD-1- or TIGIT-positive cells to CD8-positive cells. A discrepant result between two investigators was resolved by consensus on simultaneous examination.
Statistical analysis
Proportions of categorical variables were compared by the chi-squared test or Fisher’s exact test, as appropriate. Continuous variables were compared using a Wilcoxon signed-rank test for paired data or the Mann–Whitney U-test for unpaired data. Receiver operating characteristic (ROC) curve analyses were done to decide the optimal cut-off value of the TPS and expression rate. The Kaplan–Meier method was used to assess the probability of OS, and survival differences were analyzed with the log-rank test. Differences were considered statistically significant when p values were < 0.05. All statistical analyses were performed with commercial software (SPSS version 27, IBM, Armonk, New York, USA).
Ethics declarations
The institutional review board of the University of Occupational and Environmental Health, Japan approved the present study.
Acknowledgements
We are grateful to all the clinical and laboratory staff and administrators in our department. This study was supported in part by the Japan Society for the Promotion of Science (JSPS) [Grants-in-Aid for Scientific Research Grant Number 20K07688].
Author contributions
M.T., K.Y., K.K., and F.T. conceived and planned the experiments. M.M., M.K., T.K., T.M., Y. N., and N.N. carried out the experiments. M.T., K.Y., and K.K. planned and carried out the simulations. Y. N., N.N., R.O., H.M., Y.N., A.T., and M.T. contributed to sample preparation. M.K., T.K., R.O., H.M contributed to the interpretation of the results. M.M. took the lead in writing the manuscript. All authors provided critical feedback and helped shape the research, analysis, and manuscript.
Data availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2021. CA Cancer J. Clin. 2021;71:7–33. doi: 10.3322/caac.21654. [DOI] [PubMed] [Google Scholar]
- 2.Reck M, et al. Five-year outcomes with pembrolizumab versus chemotherapy for metastatic non–small-cell lung cancer with PD-L1 tumor proportion score ≥ 50% J. Clin. Oncol. 2021;39:2339–2349. doi: 10.1200/jco.21.00174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chauvin J-M, et al. TIGIT and PD-1 impair tumor antigen–specific CD8+ T cells in melanoma patients. J. Clin. Invest. 2015;125:2046–2058. doi: 10.1172/jci80445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Robert, et al. The immunoreceptor TIGIT regulates antitumor and antiviral CD8 + T cell effector function. Cancer Cell. 2014;26:923–937. doi: 10.1016/j.ccell.2014.10.018. [DOI] [PubMed] [Google Scholar]
- 5.Oyama R, et al. CD155 expression and its clinical significance in non-small cell lung cancer. Oncol. Lett. 2022 doi: 10.3892/ol.2022.13286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lee BR, et al. Combination of PD-L1 and PVR determines sensitivity to PD-1 blockade. JCI Insight. 2020 doi: 10.1172/jci.insight.128633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lee JB, et al. Overexpression of PVR and PD-L1 and its association with prognosis in surgically resected squamous cell lung carcinoma. Sci. Rep. 2021 doi: 10.1038/s41598-021-87624-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Müller S, Mayer S, Möller P, Barth TFE, Marienfeld R. Spatial distribution of immune checkpoint proteins in histological subtypes of lung adenocarcinoma. Neoplasia. 2021;23:584–593. doi: 10.1016/j.neo.2021.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Horvath L, Pircher A. ASCO 2020 non-small lung cancer (NSCLC) personal highlights. Memo. 2021 doi: 10.1007/s12254-020-00673-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cho BC, et al. Tiragolumab plus atezolizumab versus placebo plus atezolizumab as a first-line treatment for PD-L1-selected non-small-cell lung cancer (CITYSCAPE): Primary and follow-up analyses of a randomised, double-blind, phase 2 study. Lancet Oncol. 2022;23:781–792. doi: 10.1016/s1470-2045(22)00226-1. [DOI] [PubMed] [Google Scholar]
- 11.Recondo G, Mezquita L. Tiragolumab and atezolizumab in patients with PD-L1 positive non-small-cell lung cancer. Lancet Oncol. 2022;23:695–697. doi: 10.1016/s1470-2045(22)00261-3. [DOI] [PubMed] [Google Scholar]
- 12.Roche, [Ad hoc announcement pursuant to Art. 53 LR] Roche reports interim results for phase III SKYSCRAPER-01 study in PD-L1-high metastatic non-small cell lung cancer, 2023. https://www.roche.com//media/releases/med-cor-2022-05-11.
- 13.Govindan R, Bogart J, Vokes EE. Locally advanced non-small cell lung cancer: The past, present, and future. J. Thorac. Oncol. 2008;3:917–928. doi: 10.1097/jto.0b013e318180270b. [DOI] [PubMed] [Google Scholar]
- 14.Aupérin A, et al. Meta-analysis of concomitant versus sequential radiochemotherapy in locally advanced non–small-cell lung cancer. J. Clin. Oncol. 2010;28:2181–2190. doi: 10.1200/jco.2009.26.2543. [DOI] [PubMed] [Google Scholar]
- 15.Antonia SJ, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N. Engl. J. Med. 2018;379:2342–2350. doi: 10.1056/nejmoa1809697. [DOI] [PubMed] [Google Scholar]
- 16.Antonia SJ, et al. Durvalumab after chemoradiotherapy in stage III non–small-cell lung cancer. N. Engl. J. Med. 2017;377:1919–1929. doi: 10.1056/nejmoa1709937. [DOI] [PubMed] [Google Scholar]
- 17.Brahmer JR, et al. The Society for Immunotherapy of cancer consensus statement on immunotherapy for the treatment of non-small cell lung cancer (NSCLC) J. Immunother. Cancer. 2018;6:75. doi: 10.1186/s40425-018-0382-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Faivre-Finn C, et al. Four-year survival with durvalumab after chemoradiotherapy in stage III NSCLC-an update from the PACIFIC trial. J. Thorac. Oncol. 2021;16:860–867. doi: 10.1016/j.jtho.2020.12.015. [DOI] [PubMed] [Google Scholar]
- 19.Grapin M, et al. Optimized fractionated radiotherapy with anti-PD-L1 and anti-TIGIT: A promising new combination. J. Immunother. Cancer. 2019;7:160. doi: 10.1186/s40425-019-0634-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yoneda K, et al. Alteration in tumoural PD-L1 expression and stromal CD8-positive tumour-infiltrating lymphocytes after concurrent chemo-radiotherapy for non-small cell lung cancer. Br. J. Cancer. 2019;121:490–496. doi: 10.1038/s41416-019-0541-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Herter-Sprie GS, et al. Synergy of radiotherapy and PD-1 blockade in Kras-mutant lung cancer. JCI Insight. 2016 doi: 10.1172/jci.insight.87415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kim K-J, et al. Radiation improves antitumor effect of immune checkpoint inhibitor in murine hepatocellular carcinoma model. Oncotarget. 2017;8:41242–41255. doi: 10.18632/oncotarget.17168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Verbrugge I, et al. Radiotherapy increases the permissiveness of established mammary tumors to rejection by immunomodulatory antibodies. Cancer Res. 2012;72:3163–3174. doi: 10.1158/0008-5472.can-12-0210. [DOI] [PubMed] [Google Scholar]
- 24.Dovedi SJ, et al. Acquired resistance to fractionated radiotherapy can be overcome by concurrent PD-L1 blockade. Cancer Res. 2014;74:5458–5468. doi: 10.1158/0008-5472.can-14-1258. [DOI] [PubMed] [Google Scholar]
- 25.Park SS, et al. PD-1 restrains radiotherapy-induced abscopal effect. Cancer Immunol. Res. 2015;3:610–619. doi: 10.1158/2326-6066.cir-14-0138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rodriguez-Ruiz ME, et al. Abscopal effects of radiotherapy are enhanced by combined immunostimulatory mabs and are dependent on CD8 T cells and crosspriming. Cancer Res. 2016;76:5994–6005. doi: 10.1158/0008-5472.can-16-0549. [DOI] [PubMed] [Google Scholar]
- 27.Cha YJ, Kim HR, Lee CY, Cho BC, Shim HS. Clinicopathological and prognostic significance of programmed cell death ligand-1 expression in lung adenocarcinoma and its relationship with p53 status. Lung Cancer. 2016;97:73–80. doi: 10.1016/j.lungcan.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 28.Ji M, et al. PD-1/PD-L1 expression in non-small-cell lung cancer and its correlation with EGFR/KRAS mutations. Cancer Biol. Ther. 2016;17:407–413. doi: 10.1080/15384047.2016.1156256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Shimoji M, et al. Clinical and pathologic features of lung cancer expressing programmed cell death ligand 1 (PD-L1) Lung Cancer. 2016;98:69–75. doi: 10.1016/j.lungcan.2016.04.021. [DOI] [PubMed] [Google Scholar]
- 30.Song Z, Yu X, Cheng G, Zhang Y. Programmed death-ligand 1 expression associated with molecular characteristics in surgically resected lung adenocarcinoma. J. Transl. Med. 2016 doi: 10.1186/s12967-016-0943-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhang Y, et al. Protein expression of programmed death 1 ligand 1 and ligand 2 independently predict poor prognosis in surgically resected lung adenocarcinoma. Onco Targets Ther. 2014 doi: 10.2147/ott.s59959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Schmidt LH, et al. PD-1 and PD-L1 expression in NSCLC indicate a favorable prognosis in defined subgroups. PLoS ONE. 2015;10:e0136023. doi: 10.1371/journal.pone.0136023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Yang C-Y, Lin M-W, Chang Y-L, Wu C-T, Yang P-C. Programmed cell death-ligand 1 expression in surgically resected stage I pulmonary adenocarcinoma and its correlation with driver mutations and clinical outcomes. Eur. J. Cancer. 2014;50:1361–1369. doi: 10.1016/j.ejca.2014.01.018. [DOI] [PubMed] [Google Scholar]
- 34.Chen L, Han X. Anti–PD-1/PD-L1 therapy of human cancer: Past, present, and future. J. Clin. Invest. 2015;125:3384–3391. doi: 10.1172/jci80011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dong H, et al. Tumor-associated B7–H1 promotes T-cell apoptosis: A potential mechanism of immune evasion. Nat. Med. 2002;8:793–800. doi: 10.1038/nm730. [DOI] [PubMed] [Google Scholar]
- 36.Zhang Q, et al. Blockade of the checkpoint receptor TIGIT prevents NK cell exhaustion and elicits potent anti-tumor immunity. Nat. Immunol. 2018;19:723–732. doi: 10.1038/s41590-018-0132-0. [DOI] [PubMed] [Google Scholar]
- 37.Hirai A, et al. Prognostic impact of programmed death-ligand 1 expression in correlation with human leukocyte antigen class I expression status in stage I adenocarcinoma of the lung. J. Thorac. Cardiovasc. Surg. 2018;155:382–392.e381. doi: 10.1016/j.jtcvs.2017.05.106. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.