Fig. (1).
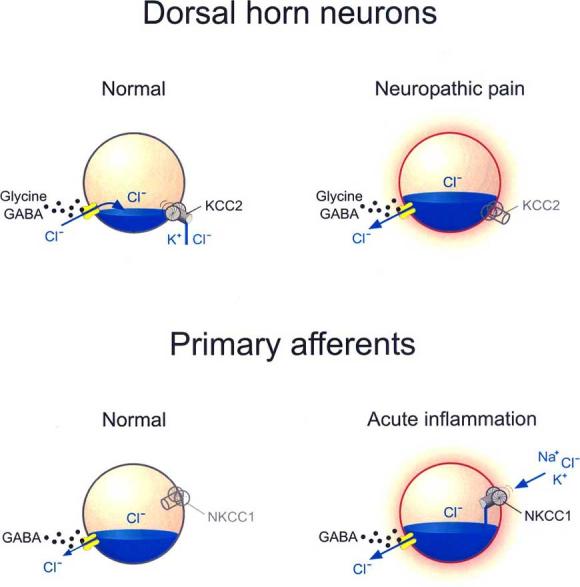
Role of Cation Chloride Cotransporters in regulation of Cl− gradients and flux. Cation-chloride cotransporters are responsible for setting the reversal potential for GABA-A or glycine receptor channels. In adult dorsal horn neurons, the anion reversal potential is maintained hyperpolarizing due to Cl− extrusion via KCC2. Trans-synaptic loss of KCC2 expression following peripheral nerve injury causes an intracellular Cl− accumulation which inverts the anion flux upon GABA-A or glycine receptor activation, and thus reverses their action. In primary afferents, GABA-A receptor activation is already depolarizing due to a slightly depolarizing Cl− reversal potential, maintained by the lack of KCC2 expression and by a weak NKCC1 expression. This depolarization remains inhibitory by causing Na+ channel inactivation and decreasing Ca++ influx upon action potential invasion in the terminal. Following acute inflammation, upregulation or enhanced activity of NKCC1 causes further accumulation of intracellular Cl− which produces an enhanced GABA-A receptor-mediated depolarization that can trigger action potentials effectively converting presynaptic inhibition into cross excitation between afferents.
