Blood, Vol. 113, Issue 21, 5111-5120, May 21, 2009
Ectopic HOXB4 overcomes the inhibitory effect of tumor necrosis factor- on Fanconi anemia hematopoietic stem and progenitor cells
on Fanconi anemia hematopoietic stem and progenitor cells
Blood Milsom et al. 113: 5111
Supplemental materials for: Milsom et al
Mice
All animals were maintained in a specific pathogen-free environment and all experiments were approved by the Institutional Animal Care and Use Committee (IACUC) of Cincinnati Children’s Research Foundation. Both C57B6J (referred to as C57B6 hereafter) and B6.SJL-Ptprca Pepcb/BoyJ mice were obtained from Jackson Laboratories (Bar Harbor, ME). Fancc−∕− mice have been previously described1 and were backcrossed with C57B6 mice for 10 generations.
Generation of retroviral vectors
The SF-FAC-IG retroviral vector used in this study has been described previously (S11FCIEG).2 The SF-IG, SF-IV, SF-HOXB4-IG, SF-HOXB4-IV, and SF-HOXB4-ERT-IG retroviral vectors were based upon the SF91 backbone3 incorporating a version of the woodchuck hepatitis virus post-transcriptional regulatory element (Pre) which was completely devoid of X-protein coding sequences or other open reading frames greater than 25 amino acids in length.4 A cassette consisting of the encephalomyocarditis virus internal ribosome entry site (IRES) followed by either enhanced green fluorescent protein (eGFP) or Venus 5 was cloned directly downstream of the human HOXB4 cDNA. The resulting expression cassettes were then inserted into the SF91 backbone to generate SF-HOXB4-IG and SF-HOXB4-IV. The IRES-eGFP or IRES-Venus cassettes were directly cloned into the SF91 backbone in order to generate either the SF-IG or SF-IV control vectors, respectively. A mutant form of the ligand binding domain from murine estrogen receptor-α (kindly provided by Dr Tariq Enver, The Weatherall Institute of Molecular Medicine, Oxford, UK)6 was fused to the C-terminus of the HOXB4 cDNA. The resulting fusion construct was then subcloned into SF-IG upstream of the IRES-eGFP cassette to yield SF-HOXB4-ERT-IG.
Viral supernatants were generated by transient transfection of PhoenixGP cells (Nolan Laboratory, Stanford University Medical Center) using calcium phosphate precipitation (Invitrogen, Carlsbad, CA) as previously described,7 pseudotyped with either the ecotropic envelope protein from murine leukemia virus, or with the feline endogenous retrovirus (RD114) envelope protein. Retroviral particles were concentrated by ultracentrifugation8 following which they were re-suspended in Iscove’s Modified Dulbecco’s Media (IMDM) (Fischer Scientific, Pittsburg, PA). The infectious titer of concentrated supernatant was determined by transduction of either NIH-3T3 murine fibroblast cells (“ecotropic” pseudotype) or HT1080 human fibrosarcoma cells (RD114 pseudotype) at a limiting dilution.
Isolation of murine lineage negative (lin−), Sca-1+, c-Kit+ (LSK) cells
BM cells were isolated from the femora, tibiae, and iliac crests of sacrificed mice by crushing in IMDM using a mortar and pestle. The resulting cell suspension was filtered through a 40 µm cell strainer (BD Bioscience, San Jose, CA) and low density mononuclear (LDMN) BM cells were derived using Histopaque-1083 (Sigma-Aldrich, St. Louis, MO) according to the manufacturer’s instructions. LDMN BM cells were then incubated for 20 min at 4°C with gentle shaking in the following cocktail of biotin-conjugated rat anti-mouse antibodies (all BD Bioscience): 4.2 µg/ml CD5 (53-7.3), 2.8 µg/ml CD45R/B220 (RA3-6B2), 2.6 µg/ml CD11b/Mac1α (M1/70), 4.2 µg/ml CD8a (53-6.7), 2.4 µg/ml Ly-6G and Ly-6C/Gr1 (RB6-8C5), and 2.6 µg/ml TER-119 (TER-119), in phosphate buffered saline (PBS) (Fischer Scientific) containing 2% (v/v) fetal calf serum (FCS) (Omega Scientific, Tarzana, CA) at a density of 3.3 × 107 cells/ml. Labeled cells were then washed twice in ice-cold PBS and were incubated with Biotin Binder Dynabeads (Invitrogen) at a concentration of 4 beads per cell, for 20 min at 4°C with gentle shaking in PBS containing 2% (v/v) FCS. Lineage positive cells were then depleted using a Dynal MPC-L magnet (Invitrogen) as per the manufacturer’s instructions. The resulting lineage depleted cells were then incubated with 1.3 µg/ml phycoerythrin (PE)-conjugated streptavidin, 3.1 µg/ml fluorescein isothiocyanate (FITC)-conjugated rat anti-mouse Sca-1/Ly-6A/E (D7), 1.3 µg/ml allophycocyanin (APC)-conjugated rat anti-mouse c-Kit/CD117 (2B8) (all eBioscience, San Diego, CA), for 20 min at 4°C in PBS containing 2% (v/v) FCS at a density of 1 × 107 cells/ml. After washing in ice-cold PBS, cells which corresponded to the Sca-1+, c-Kit+, lin− immunophenotype were isolated by flow sorting (FACSVantage, BD Bioscience).
Transduction of murine LSK cells
Freshly isolated LSK cells were pre-stimulated in IMDM supplemented with 10% (v/v) FCS, along with 100 ng/ml rat recombinant stem cell factor (rrSCF), 100 ng/ml megakaryocyte growth and development factor (MGDF) and 100ng/ml granulocyte colony stimulating factor (G-CSF) (all Amgen, Thousand Oaks, CA), for 48 hrs at 37°C, 5% CO2. Retroviral transduction was performed in non-tissue culture coated multi-well plates (BD Biosciences) which had been pre-coated with fibronectin CH296 fragment (Takara Bio Inc., Otsu, Japan) at 4 µg/cm2. LSK cells were seeded at a density of 1–1.5 × 105 LSK cells per cm2 in the same media as used for pre-stimulation, along with concentrated retroviral supernatant, the volume of which was adjusted for each vector used to achieve a multiplicity of infection (MOI) of 4. Following 10–12 hrs exposure to retroviral vector particles, further concentrated viral supernatant was added to the cells, corresponding to a further MOI of 4. The total volume of viral supernatant added to cells did not exceed 1% of the culture volume. Thirty six hrs after the final exposure to viral particles, cells were harvested from the FN CH296 coated plates using cell dissociation buffer (Invitrogen) and washed once in PBS. Cells were then re-suspended in PBS, 2% FCS, 5 µg/ml 7-Amino-actinomycin D (7-AAD) (Invitrogen), and live GFP+/Venus+ cells were isolated by flow sorting (FACS Vantage). Transduction frequencies for LSK cells with individual vectors were in the range of 10–38% while the transduction frequencies of double transduced cells were in the range of 5–12%. Isolation and transduction of BM from mice pre-treated with 150 mg/kg 5-fluorouracil was performed as described in 9. Unless otherwise stated, In vitro expansion of HOXB4 transduced BM was performed in IMDM supplemented with 10% (v/v) FCS, along with 20 ng/ml murine IL3, 100 ng/ml IL6 (both from Peprotech, Rocky Hill, NJ), and 100 ng/ml rrSCF. BM was maintained in the range of 1 × 105 to 1 × 106 cells/ml.
Competitive repopulation assay
The indicated number of transduced and sorted LSK cells were injected into the tail vein of lethally irradiated recipient mice 11.75 Gy, 56 cGy/min, 135Cs source Mark I Model 68A Irradiator (J.L. Shepherd and Associates, San Fernando, CA), split dose), along with 1 × 106 freshly isolated whole BM competitor cells. Following red cell lysis (Pharm Lyse, Becton Dickinson), peripheral blood (PB), and BM cells were either directly assessed for fluorescence (FACS Canto, Beckton Dickinson) or were stained with fluorescent labeled monoclonal antibodies (anti-mouse CD3ε (145-2C11), B220 (RA3-6B2), GR1 (RB6-8C5), and MAC1 (M1/70), all E-Bioscience) following the manufacturer’s instructions, prior to flow analysis. Viable cells were identified by exclusion of 7-AAD. PB cell counts were obtained using a Hemavet 850 FS Hematology Analyzer (Drew Scientific, Oxford, CT). For in vivo activation of the HOXB4-ERT fusion construct, tamoxifen citrate (Sigma-Aldrich) was dissolved at 10 mg/ml in ethanol. Tamoxifen citrate was then diluted to a final concentration of 100 µg/ml in acidified drinking water which was administered to mice starting 1 week pre-transplant.
Colony forming assays
Primary murine BM cells were plated in triplicate in methylcellulose (1 × MethoCult, Stem Cell Technologies, Vancouver, Canada) supplemented with 30% (v/v) FCS, 2 mM L-Glutamine (Hyclone), 200 U Penicillin/Streptomycin (Hyclone), 100 µM β-mercaptoethanol (Thermo Fisher), 1% BSA (Roche, Indianapolis, IN), 100 ng/ml rrSCF, 100 ng/ml mIL3 (Peprotech, Rocky Hill, NJ), and 4 U/ml Epogen (Amgen). Plates were additionally supplemented with murine tumor necrosis factor-α (TNFα) or murine interferon-γ (IFNγ) (both Peprotech) at the concentrations indicated in the text. Plates were then incubated in a humidified atmosphere at 37°C, 5% CO2 for 7 days, following which colonies comprising 50 or more cells were scored.
Immunophenotypic analysis of TNFα receptor expression levels
Sorted transduced LSK cells were expanded for 7 days in IMDM supplemented with 10% (v/v) FCS, along with 100 ng/ml rrSCF, 100 ng/ml MGDF, and 100ng/ml G-CSF, at 37°C, 5% CO2. Cells were then blocked with rat anti-mouse CD16/32 (2.4G2) (BD Biosciences) and subsequently stained with the following antibodies according to the manufacturer’s instructions: rat anti-mouse CD5 (53-7.3), rat anti-mouse CD45R/B220 (RA3-6B2), rat anti-mouse CD11b/Mac1α (M1/70), rat anti-mouse CD8a (53-6.7), rat anti-mouse Ly-6G and Ly-6C/Gr1 (RB6-8C5), and rat anti-mouse TER-119 (TER-119) (all BD Bioscience, all biotinylated), along with PE-Cy7–conjugated rat anti-mouse Sca-1/Ly-6A/E (D7), PE-Cy5–conjugated rat anti-mouse c-Kit/CD117 (2B8) (both E-Bioscience) and either Alexa Fluor 647-conjugated hamster anti-mouse CD120a (55R-286) or Alexa Fluor 647-conjugated hamster anti-mouse CD120b (TR75-89) (both Serotec, Raleigh, NC). After washing twice in ice cold PBS, cells were stained with APC-Cy7–conjugated streptavidin (E-Bioscience) as per the manufacturer’s instructions. Cells were then immediately analyzed by flow cytometry (FACSCanto, BD Biosciences).
Flow analysis of apoptosis and reactive oxygen species (ROS) levels
Transduced LSK cells were expanded for 2 days as described above. For ROS analysis, BM were labeled with CM-H2DCFDA (Molecular Probes, Eugene, OR) for 15 min at 37°C according to the manufacturer’s instructions, prior to staining for immunophenotypic markers. Staining for lineage specific markers, c-Kit, and Sca-1 was performed as described above, with the exception that APC-conjugated rat anti-mouse c-Kit/CD117 (2B8) (E-Bioscience)was substituted for staining of c-Kit. For analysis of apoptosis, stained BM cells were subsequently incubated with FITC-conjugated Annexin V (BD Biosciences) as per manufacturer’s instructions. Cells were then analyzed by flow cytometry (FACSCanto).
In vitro treatment of transduced BM with TNFα
LSK cells were pre-stimulated and transduced as described above, with the exception that the described media was substituted with StemSpan serum free expansion medium (StemCell Technologies, Vancouver, Canada) supplemented with 100 ng/ml rrSCF, 100 ng/ml MGDF, and 100ng/ml G-CSF. 36 hrs post-transduction, TNFα was added to the media at the stated concentration. Following a further 24 hr incubation at 37°C, 5% CO2, BM were stained for lineage specific markers, c-Kit, and Sca-1 as described previously and analyzed by flow cytometry (FACSCanto).
Analysis of G2/M arrest and FANCD2 monubiquitination in human FA lymphoblast cell lines (LCLs)
The derivation of Epstein-Barr virus-transformed LCLs from FA patient samples, and details of their subsequent culture conditions have been previously described.2 LCLs were transduced with RD114 pseudotyped retroviral vector essentially as described in 2, following which eGFP+ cells were isolated by flow sorting (FACSVantage). Analysis of cell cycle arrest in response to treatment with 0.5 µg/ml melphalan (Sigma-Aldrich, St. Louis, MO) was performed as described.2 Analysis of FANCD2 monoubiquitination in response to treatment with hydroxyurea (Sigma-Aldrich) was performed as described in 10.
Phospho-specific immunoblots
32D cells were transduced with either the control vector (SF-IV) or the HOXB4-expressing vector (SF-HOXB4-IV), and Venus+ cells were isolated by flow sorting 2 days later. After a further 4 days expansion in vitro, transduced cells were harvested by centrifugation and re-suspended in fresh growth media at a density of 1 × 107 cells per ml. Cells were then incubated at 37°C for 30 min, following which cells were treated with either recombinant murine TNFα (Peprotech) at a final concentration of 20 ng/ml or with an equivalent volume of PBS. After a 5 min incubation at 37°C, cells were harvested and snap frozen in liquid nitrogen. Following cell lysis, the lysate from 2 × 105 cells was probed by immunoblot using antibodies specific for either NF-κB p65 phosphorylated at the Ser536 residue (Rabbit anti–phospho-NFκB p65 (Ser536) clone 93H1, Cell Signaling Technology) or SAPK/JNK phosphorylated at Thr183/Tyr185 (Mouse anti–phospho-SAPK/JNK (Thr183/Tyr185) clone G9, Cell Signaling Technology). Membranes were then probed with the appropriate secondary antibodies (goat anti-rabbit igG-HRP conjugate or horse anti-mouse IgG-HRP conjugate, both Cell Signaling Technology) and bands were visualized using LumiGLO reagent (Cell Signaling Technology) followed by autoradiography. Membranes were then stripped of antibody by washing with Restore PLUS Western Blot Stripping Buffer (Thermo Scientific) at room temperature for 15 min, following which they were re-probed with antibodies against either total NF-κB p65 (Rabbit polyclonal anti–NF-κB p65, Cell Signaling Technology) or total SAPK/JNK (Rabbit polyclonal anti-SAPK/JNK, Cell Signaling Technology). Following visualization of protein bands as described above, the membranes were once again stripped and then re-probed with an antibody directed against β-actin (Mouse anti–β-actin, clone AC-15, Sigma-Aldrich).
REFERENCES
1. Chen M, Tomkins DJ, Auerbach W, et al. Inactivation of Fac in mice produces inducible chromosomal instability and reduced fertility reminiscent of Fanconi anaemia. Nature Genetics. 1996;12:448–451.
2. Chandra S, Levran O, Jurickova I, et al. A rapid method for retrovirus-mediated identification of complementation groups in Fanconi anemia patients. Mol Ther. 2005;12:976–984.
3. Hildinger M, Abel KL, Ostertag W, Baum C. Design of 5′ untranslated sequences in retroviral vectors developed for medical use. J Virol. 1999;73:4083–4089.
4. Schambach A, Bohne J, Baum C, et al. Woodchuck hepatitis virus post-transcriptional regulatory element deleted from X protein and promoter sequences enhances retroviral vector titer and expression. Gene Ther. 2006;13:641–645.
5. Nagai T, Ibata K, Park ES, Kubota M, Mikoshiba K, Miyawaki A. A variant of yellow fluorescent protein with fast and efficient maturation for cell-biological applications. Nat Biotechnol. 2002;20:87–90.
6. Heyworth C, Gale K, Dexter M, May G, Enver T. A GATA-2/estrogen receptor chimera functions as a ligand-dependent negative regulator of self-renewal. Genes Dev. 1999;13:1847–1860.
7. Thomas EK, Cancelas JA, Chae HD, et al. Rac guanosine triphosphatases represent integrating molecular therapeutic targets for BCR-ABL–induced myeloproliferative disease. Cancer Cell. 2007;12:467–478.
8. Schambach A, Galla M, Modlich U, et al. Lentiviral vectors pseudotyped with murine ecotropic envelope: increased biosafety and convenience in preclinical research. Exp Hematol. 2006;34:588–592.
9. Milsom MD, Woolford LB, Margison GP, Humphries RK, Fairbairn LJ. Enhanced In Vivo Selection of Bone Marrow Cells by Retroviral-Mediated Co-exprssion of Mutant O6-Methylguanine-DNA-Methytransferase and HOXB4. Molecular Therapy. 2004;10:862–873.
10. Meetei AR, Medhurst AL, Ling C, et al. A human ortholog of archaeal DNA repair protein Hef is defective in Fanconi anemia complementation group M. Nat Genet. 2005;37:958–963.
Files in this Data Supplement:
- Figure S1. Peripheral blood cells which express eGFP and/or Venus are easily discerned by flow cytometry (JPG, 165 KB) -
Mice were transplanted with sorted transduced BM cells as described in methods. Mice received cells that were (A) transduced with a vector which expresses Venus; (B) transduced with a vector which expresses eGFP; (C) separately transduced with either a vector which expresses eGFP or a vector which expresses Venus; (D) co-transduced with a vector which expresses eGFP and a vector which expresses Venus. Following the co-transduction procedure, cells which were double positive for eGFP and Venus were purified by flow sorting prior to injection. At ≥ 6 months post transplant, peripheral blood was subject to flow analysis following red cell lysis. Flow analysis was performed using a FACSCanto (Becton Dickinson) flow cytometer which had been equipped with a 510/20 band pass filter coupled with a 500 long pass dichroic filter and a 545/35 band pass filter coupled with a 525 long pass dichroic filter.
- Figure S2. Survival of transduced progenitor cells with increasing concentrations of IFNγ (JPG, 91.7 KB) -
Murine LSK cells were transduced with the indicated retroviral vectors and sorted transduced cells were then plated in methylcellulose supplemented with either 1 ng/ml, 10 ng/ml or no (NTX) IFNγ. The frequency of surviving day 7 colonies, expressed as a percentage of non-treated controls, is shown. Data represent the mean ± SEM from 3 independent experiments. **pFancc−∕− tranduced with SF-IG, paired Student’s t-test.
- Figure S3. Competitive repopulation assay using lineage depleted Fancc−∕− murine BM cells (JPG, 40.8 KB) -
(A) Schematic representation of competitive transplant assay used to determine the relative engraftment potential of lineage depleted Fancc−∕− BM cells transduced with different retroviral vectors. (B) Fancc−∕− BM was depleted of lineage positive cells as described in methods. Isolated cells were subsequently transduced with the indicated retroviral vectors and the resulting eGFP+ cells were then isolated by flow sorting, as per the transduction and isolation of transduced LSK cells described in methods. The proportion of eGFP+ cells present in the peripheral blood was determined by flow analysis at the time points indicated. Data represent the mean ± SEM of two independent experiments comprising 6 mice per group. **p
- Figure S4. Lineage analysis of transduced peripheral blood cells (JPG, 69 KB) -
Peripheral blood was obtained after 6 months engraftment from mice which had received transduced BM cells. The proportion of transduced cells corresponding to neutrophils, T and B cells in the peripheral blood was determined by flow analysis. Data represent the mean ± SEM of two independent experiments incorporating 4–10 mice per experimental group. **p
- Figure S5. G2/M arrest of transduced LCLs following treatment with melphalan (JPG, 42.3 KB) -
The human FANCC−∕− lymphoblast cell line HSC2911-FAC was transduced with the indicated retroviral vectors. The transduced cells were then treated with melphalan as described in methods. The proportion of eGFP+ cells in G2/M following melphalan treatment is indicated. NTX = non-treated.
- Figure S6. Determination of TNFα receptor expression levels in transduced LSK BM by flow (JPG, 90.3 KB) -
Transduced murine BM cells were isolated by flow sort and were cultured for 7 days. Cells were then stained with antibodies directed against lineage markers, c-Kit, Sca-1, and either TNFR1 or TNFR2. (A) Flow gating used to define LSK population analyzed for TNFR1 and TNFR2 expression. Panels show sequential gating upon (i) live cells; (ii) eGFP+ cells; (iii) Lineage negative cells; and (iv) Sca-1+ c-Kit+ cells. (B) Histogram of TNFR1 expression level in transduced LSK cells from a representative experiment. The percentage of TNFR1+ cells and the mean fluorescent intensity (MFI) of staining for TNFR1 in this population is indicated for (i) WT LSK cells transduced with SF-IG; and Fancc−∕− LSK cells transduced with (ii) SF-IG; (iii) SF-FAC-IG; and (iv) SF-HOXB4-IG.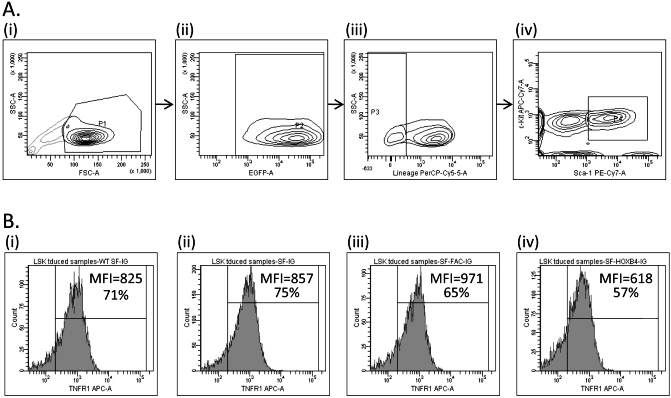
- Figure S7. Ectopic HOXB4 suppresses expression of TNFα receptors in WT C57B6 LSK BM cells (JPG, 116 KB) -
Transduced murine WT BM cells were isolated by flow sort and were cultured for 7 days. Cells were then stained with antibodies directed against lineage markers, c-Kit, Sca-1, and either TNFR1 or TNFR2. (A) The mean percentage of TNFR1+ or TNFR2+ LSK cells and (B) the mean fluorescent intensity of staining for TNFR1 or TNFR2 within different retroviral transduced populations. Data represent the mean ± SEM from 3 independent experiments. *p
- Figure S8. Ectopic HOXB4 protects 32D cells against apoptosis induced by TNFα (JPG, 43.5 KB) -
The 32D myeloid progenitor cell line was transduced separately with either the control vector SF-IV or the HOXB4-expressing vector SF-HOXB4-IV. At 72 hrs post-transduction, cells were harvested by centrifugation and then seeded at a density of 5 × 105 cells per ml in either pre-warmed normal growth media (NTX) or pre-warmed growth media supplemented with either 20 ng/ml or 100 ng/ml murine recombinant TNFα. After a further 24 hrs incubation at 37 °C, 5% CO2, cells were harvested and apoptotic cells were stained using Annexin V-APC as described in methods. Stained cells were then assessed by flow cytometry. The mean percentage of Venus+ cells that stained positive for Annexin V is shown ± SD. **p