Blood, Vol. 114, Issue 10, 2077-2086, September 3, 2009
AAV-1–mediated gene transfer to skeletal muscle in humans results in dose-dependent activation of capsid-specific T cells
Blood Mingozzi et al. 114: 2077
Supplemental materials for: Mingozzi et al
Files in this Data Supplement:
- Table S1. High-resolution HLA typing of subjects enrolled in the study (PDF, 15.3 KB)
- Table S2. Baseline anti-AAV-1 neutralizing antibody titer (PDF, 12.8 KB)
- Figure S1. Serum levels of CPK are shown for individual subjects from the low-dose, left panel, and high-dose, right panel, cohorts (JPG, 208 KB) -
ULN, upper limit of normal for CPK (190 IU/L). *Subject with positive IFN-γ response to capsid measured by ELISpot.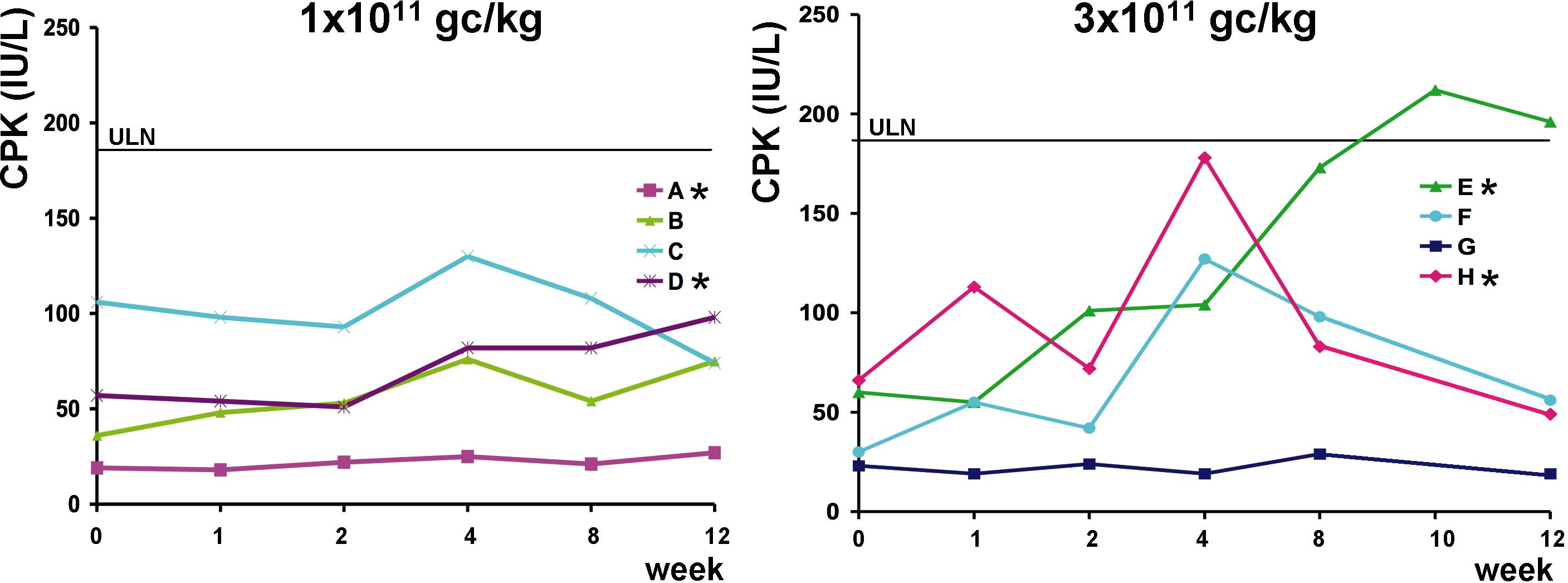
- Figure S2. Capsid-specific IFN-γ ELISpot on PBMC from Subjects A, B, C, D, F, G, and H; results are expressed in SFU/106 PBMC (average ± standard deviation) (JPG, 378 KB) -
P 1 to P 24, AAV peptide matrix pools. PMA, positive control (>1000 SFU/106 PBMC for all time points); M, medium only; AAV, AAV-1 empty capsids; CEF, CMV EBV Flu peptide pool.
- Figure S3. Polyfunctional analysis of T-cell responses to the AAV-1 capsid in subject E (JPG, 364 KB) -
The histograms represent the frequency of CD4+ (upper graph) or CD8+ (lower graph) T cells expressing 0 to 5 activation markers (IL-2, CD107a, IFN-γ, perforin, and TNF-α) simultaneously in response to AAV capsid-derived peptides over time (baseline, week 4, week 6, and week 12). The pie charts summarize the results of the polyfunctional analysis by reporting the fraction of capsid-specific CD4+ (upper charts) or CD8+ (lower charts) expressing 0 to 5 activation markers simultaneously over time.