Figure 1.
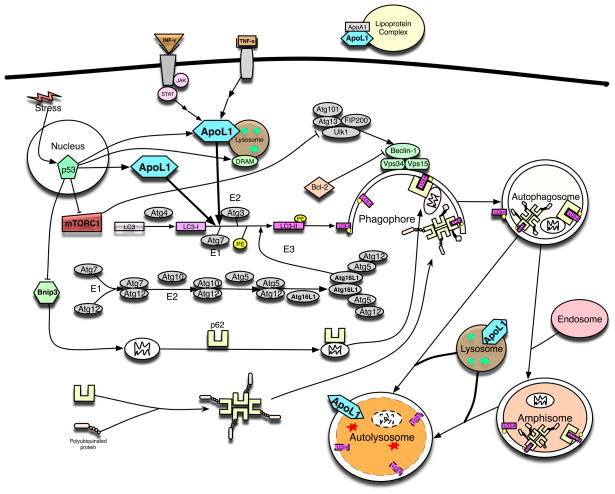
The role and subcellular localization of ApoL1 in autophagy in human cells. A wealth of literature has shown that expression of APOL1 can be induced by the signaling pathways of IFN-γ, TNF-α and p53. Intracellularly, ApoL1 is lysosomal and cytosolic (see Figure 2A), and can induce (macro)autophagy and autophagy-associated cell death when overexpressed. Interestingly, another lysosomal protein, DRAM, also a p53 downstream target, can induce autophagy and apoptosis. Autophagy, a lysosome-dependent, self-eating, and catabolic mechanism, can be initiated by various stresses (for example, starvation, inflammatory cytokines, and pathogens). Upon autophagy induction, mTORC1 is inactivated leading to the activation of ULK1 complex (including ULK1, Atg13, FIP200, and Atg101), that in turn activates the class III phosphatidylinositol 3-kinase complex, including at least Vps34, Vps15, and Beclin 1. The elongation of phagophore membranes into autophagosomes is regulated by two ubiquitination-like reactions. First, the ubiquitin-like molecule Atg12 is conjugated to Atg5 by Atg7, which acts like an E1 ubiquitin-activating enzyme, and by Atg10, which is similar to an E2 ubiquitin-conjugating enzyme. The Atg5-Atg12 complex then interacts non-covalently with Atg16L1 and this resulting the Atg12-Atg5-Atg16L1 complex associates with phagophores but dissociates from mature autophagosomes. The second ubiquitin-like reaction involves the conjugation of mammalian LC3-II, a homologue of yeast Atg8, to phosphatidylethanolamine (PE) by Atg7 and Atg3 (another E2-like), resulting in autophagosome-associated LC3-II. The Atg12-Atg5 complex may be able to enhance LC3-II conjugation to PE by acting in an E3-like fashion. In this way, the Atg12-Atg5-Atg16L1 complex may determine the sites of autophagosome synthesis by regulating the targeting of LC3-II to Atg12-Atg5-associated membranes. Interestingly, LC3-II remains associated with autophagosomes until after their fusion with lysosomes. The LC3-II inside the autolysosomes is degraded, while the LC3-II on the cytoplasmic surface can be delipidated and recycled. Subsequently, the autophagosome fuses with endosomes and lysosomes to form two other types of autophagic vesicles, amphisomes and autolysosomes in human cells. This process results in the degradation of the sequestered materials by lysosomal hydrolases in the autolysosome. P62, an ubiquitously expressed protein, is one of the best characterized substrates of selective autophagy and mitophagy. p62 has been shown to bind polyubiquinated proteins and altered mitochondria and directly interact with LC3II on the phagophore through the LC3-interacting region. Subsequently, p62-cargo complex is incorporated into the autophagosome and then degraded. Mitophagy has been shown to be regulated by Bnip3, a BH3-only protein and a p53 downstream target. Extracellularly, ApoL1 has been shown to be an interacting protein of ApoA1 in lipoprotein complexes.