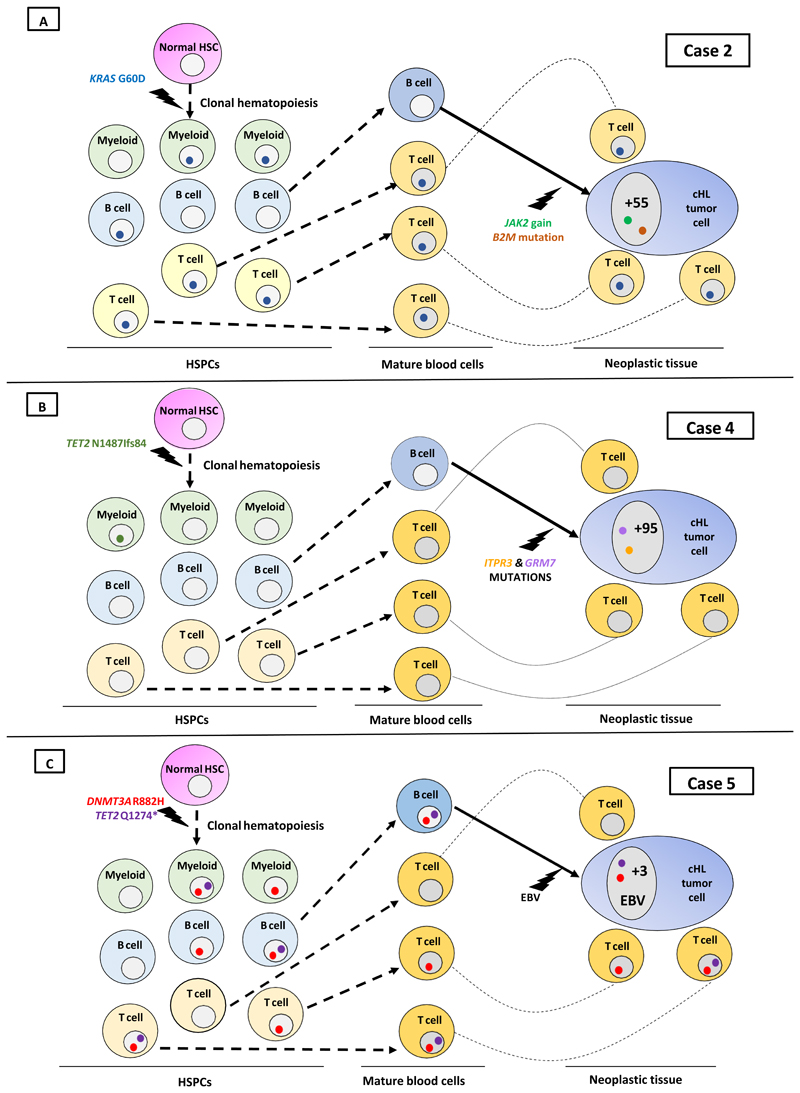
Figure 1. Tissue involvement by clonal hematopoiesis in cHL.
A. In this EBV-negative mixed cellularity cHL (case 2), the reactive microenvironment (mostly represented by T cells - not shown) belonged to clonal hematopoiesis almost entirely, as it featured a KRAS G60D mutation at a VAF of 45.9%. However, the HRS cell clone developed from a B cell not belonging to clonal hematopoiesis, after acquiring a JAK2 gain and a B2M mutation (genetic lesions typical of cHL) along with 55 additional somatic protein-changing mutations detected by WES in the HRS cell clone (ref. 15; “+55” in the HRS cell nucleus). Myeloid involvement by the clonal hematopoiesis is presumptive because we did not have a blood or bone marrow sample from this patient where to formally confirm it. HSC, hematopoietic stem cell.
B. In EBV-negative nodular-sclerosis case4, clonal hematopoiesis (carrying TET2 N1487Ifs84), which was detected in whole-blood leukocytes and likely involved the myeloid lineage (only or mainly), did not spread through either the lymphoid HRS cell clone or its reactive tissue microenvironment. Also indicated are mutations of two genes (ITPR3 and GRM7, recurrently targeted in cHL: ref. 15) that were found in HRS cells but not in blood leukocytes of this cHL case, along with additional 95 somatic protein-changing mutations detected by WES in the HRS cell clone (ref. 15; “+95” in the HRS cell nucleus).
C. Tissue distribution of clonal hematopoiesis in case 5, showing a double mutant DNMT3A R882H and TET2 Q1274* clone with likely multilineage involvement of B, T and myeloid cells, which spread through both the microenvironmental and HRS cell components of cHL. The latter component was infected by EBV and lacked almost any other somatic mutations that could have caused the neoplastic transformation of its mature B-cell of origin (only three protein-changing mutations being present exome-wide in the HRS cell clone but not in reactive lymphoid cells, indicated as “+3” within the HRS cell nucleus; and none of them targeted a gene recurrently mutated in cHL; ref. 15). Involvement of myeloid cells by clonal hematopoiesis is presumptive because we did not have a blood or bone marrow sample from this patient where to formally confirm it.