Abstract
The causative agent of Severe Acute Respiratory Syndrome (SARS) was identified as a coronavirus (CoV) following the outbreak of 2002–2003. There are currently no licensed vaccines or treatments for SARS-CoV infections. Potential prevention and control strategies that show promise in vitro must be evaluated in animal models. The aged BALB/c mouse model for SARS supports a high level of viral replication in association with clinical illness and disease that mimics SARS in the elderly. We tested two preventive strategies, vaccination and passive transfer of serum antibody, to determine the extent of protection achieved against SARS-CoV challenge in this model. These approaches were able to achieve or induce antibody titers sufficient to reduce viral load, protect from weight loss, and reduce or eliminate histopathologic changes in the lungs of aged mice. This study validates the utility of the aged BALB/c mouse model for evaluation of the efficacy of vaccines and immunoprophylaxis.
Keywords: SARS-CoV, Aged mouse model, Prophylaxis
Introduction
The agent that caused the 2002–2003 Severe Acute Respiratory Syndrome (SARS) outbreak was identified by sequence analysis and immunofluorescence as a coronavirus virus, SARS-CoV. SARS-CoV was a zoonosis and although closely related viruses have been identified in civet cats and Chinese horseshoe bats, the animal reservoir from which the virus was introduced into the human population has yet to be definitively identified [1,2]. Since July 2003, when the SARS outbreak ended, only a few cases of community–acquired and laboratory-acquired infection have occurred. The presence of an animal reservoir in nature suggests that the risk of re-introduction of a SARS-like CoV into humans remains, and efforts to develop prevention strategies continue. Animal models are needed to effectively test prevention strategies. Several animal species, including inbred strains of mice, have been found to support viral replication in the absence of clinical illness [3]. However, an animal model that mimics the natural course of disease affords a more stringent test of potential interventions. In several case series of SARS patients advanced age was a risk factor for severe disease, requiring intensive care and ventilatory support, as well as increased mortality [4,5,6]. We found that 12 to 14 month old BALB/c mice support high and prolonged levels of viral replication in the lungs, signs of clinical illness, and histopathological changes in the lungs including signs of acute and organized diffuse alveolar damage (DAD) [7]. Thus, this model reflected findings in elderly patients with SARS.
Active and passive immunization are two standard approaches for the prevention of viral infection; both have been considered for SARS and have been evaluated in young (4 to 6-week old) BALB/c mice [8,9]. Passive transfer of post-infection hyperimmune antiserum, derived from SARS-CoV infected mice, was shown to be effective in protecting immunologically naïve, young mice from subsequent challenge with intranasally administered SARS-CoV [8]. Attenuated, recombinant vesicular stomatitis virus (rVSV) is known to be a good vector for the expression of foreign proteins inducing both humoral (antibodies) and cell mediated immunity to the expressed proteins [10]. rVSV vaccines expressing HIV Gag and Env proteins, induced a strong and long lasting antibody recall [11, 12]. An attenuated VSV virus expressing the SARS-CoV spike (S) protein efficiently expressed SARS-CoV S protein and conferred long-lasting protection from viral challenge in young mice [9]. Here we selected two preventive strategies, that were effective in young mice, and evaluated them in old mice to determine if the immunoprophylactic measures that are effective in the young mouse are also effective in the more susceptible aged mouse. We tested the efficacy of passive transfer of post-infection murine SARS antiserum and active immunization with a rVSV vaccine encoding the SARS S protein (rVSV-S) in protection from challenge with intranasally administered SARS-CoV in this model.
Material and methods
Post-infection hyperimmune SARS antiserum was generated in BALB/c mice following both intranasal (i.n.) and intraperitoneal (i.p.) injections of SARS-CoV (Urbani). The efficacy of passive transfer was tested in twelve-month-old mice, in groups of 12, that received antiserum by i.p. injection. The negative control group received normal (non-immune) BALB/c mouse sera (Harlan, Indianapolis, Indiana); the experimental groups received either undiluted post-infection hyperimmune SARS antiserum or a 1:4 dilution (in PBS) of the antiserum. Sera from the recipient mice were collected 24 hours after i.p. injection, to determine the neutralizing antibody level achieved. Neutralizing antibody titers were determined by a micro-neutralization assay in Vero cell monolayers [8]. Mice were then challenged i.n. with 105 TCID50 SARS-CoV (Urbani). On day 2 post-infection (p.i.), when virus titer in the lungs is expected to peak, four mice per group were sacrificed and lungs were harvested to determine levels of virus present [7]. Briefly, lungs were homogenized as a 10% w/v suspension; clarified by low speed centrifugation; and titered on Vero monolayers in 24-well and 96-well plates [8]. Because infectious virus can be neutralized ex vivo in lung homogenates and this can affect virus titers, we confirmed the presence of virus in lung homogenates by quantitative PCR. Total RNA was isolated and purified from 10% w/v lung homogenates using the RNeasy Mini Kit (Qiagen, Valencia, CA) with an on-column DNase digestion, as per manufacturer’s protocol. RNA concentration was determined and 1μg was reverse transcribed using random primers included in the Stratascript First-Strand Synthesis System (Stratagene, La Jolla, CA). PCR reactions were performed at 59°C for a total of 40 cycles using the Brilliant SYBR Green QPCR Master Mix (Stratagene) on an Mx4000 Multiplex Quantitative PCR System (Stratagene). SARS genomic RNA was amplified using forward primer 28411F (GGTGACGGCAAAATGAAAGAGC) and reverse primer 28725R (GGAGAATTTCCCCTACTG). 18S rRNA was amplified from each sample as an internal control using 18SF (GGTACAGTGAAACTGCGAAT) and 18SR (CAGTTATCCAAGTAGGAGAG). CT is the cycle number when double stranded DNA was detected above background levels. Four mice per group were sacrificed on days 3 and 8 p.i. for evaluation of histopathologic changes. These time points were selected because at day 3 p.i., viral antigen and focal interstitial inflammation should be present and clearance of antigen accompanied by histopathologic changes indicative of lung repair should be visible by day 8 post-infection [7]. The lungs were inflated with 10% formalin and processed for histopathological examination. Pulmonary histopathology was evaluated and the distribution of SARS-CoV antigens was determined by immunohistochemical (IHC) staining [8].
In the vaccine study, mice received one of the three following interventions by i.n. inoculation: 1.4 x104 pfu rVSV-S (the vaccine), 1.6x104 pfu rVSV (vector without a foreign gene, as a negative control) [9] or 104 TCID50 SARS-CoV (Urbani) as a positive control for protection from subsequent challenge. Sera were collected on day 30 post-vaccination or post-infection to determine neutralizing antibody titers achieved. Mice were then challenged 3 days later with 105 TCID50 SARS-CoV (Urbani) to determine the efficacy of the vaccine. Four mice per vaccine group were weighed daily for a week. On day 2 post-challenge, 4 mice per group were sacrificed and the lungs harvested to determine titers of infectious virus.
Results
Passive Immunization
Passive transfer of undiluted or diluted post-infection SARS-CoV hyperimmune murine antisera achieved significant neutralizing antibody titers in recipient mice (Figure 1). Mice that received undiluted hyperimmune sera had a mean neutralizing antibody titer of 1:109 ± 11.5, while mice that received a 1:4 dilution of SARS hyperimmune antisera had a mean neutralizing titer of 1:18 ± 1.7. . In contrast, mice that received the normal (non-immune) mouse serum did not have detectable neutralizing antibodies (titers were below the limit of detection, <1:8). These differences were statistically significant, p ≤ 0.0001, Mann Whitney U test.
Figure 1. Passive transfer of SARS hyperimmune antisera protects mice from challenge with SARS-CoV.
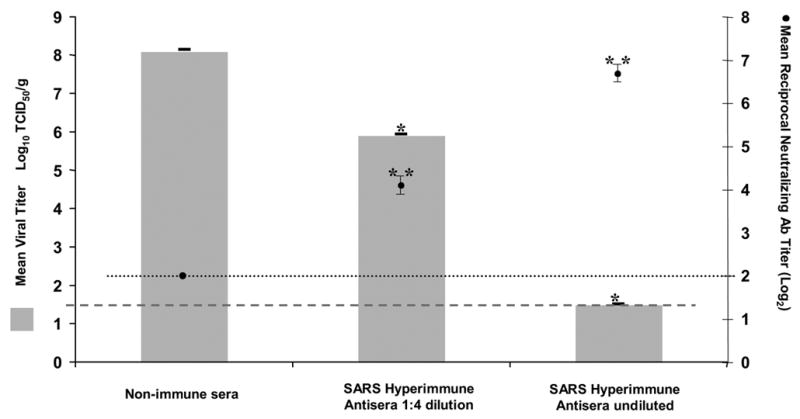
Left-hand y-axis: Mean viral titer in lungs is expressed as log10TCID50/g tissue with a lower limit of detection of 1.5 log10TCID50/g, error bars represent the SE, *p = 0.02 pairwise comparison of test groups to control group that received non-immune mouse serum
Right-hand y-axis: Reciprocal mean antibody titer, expressed as log2, is the dilution of serum antibody that neutralized 100 TCID50 of SARS-CoV. The lower limit of detection was ≤ 2, error bars represent SE, **p ≤ 0.0001 pairwise comparison of test groups to control group that received non-immune mouse serum.
Mice that received murine SARS hyperimmune antisera were protected from pulmonary virus replication on challenge with SARS-CoV. Virus was not detected in the lungs of mice that received undiluted SARS hyperimmune antiserum. Mice that received diluted SARS hyperimmune antiserum had a >100-fold reduction in viral titer as compared to mice that received non-immune mouse serum (105.9 ± 0.3 TCID50/g and 108.1 ± 0.5 TCID50/g, respectively, p = 0.02, Mann Whitney U test). Reductions in titers of infectious virus were confirmed using real-time PCR. Data is presented as the mean cycle number (CT ) at which18S rRNA and N-gene SARS-CoV-specific RNA, respectively were amplified above background. The CT values for mice that received hyperimmune antisera were 15.9±0.5 and 33.0±0.8, for mice that received a 1:4 dilution of hyperimmune antisera were 16.5±0.1 and 20.8±0.2, and for mice that received non-immune sera were 15.2±0.3 and 15.3±0.1. There was no significant difference in the CT value for amplification of 18S rRNA in the three groups (Kruskal-Wallis p=0.29) but there was significant difference among the three groups for amplification of SARS-CoV RNA (Kruskal-Wallis p=0.007). SARS-CoV RNA was not amplified from the lungs of mice that received undiluted hyperimmune serum and there was a reduction in viral load in the lungs of mice that received diluted hyperimmune serum compared to mice that received non-immune serum. On histologic examination of the lungs, mice that received non-immune mouse sera had multiple perivascular foci of mononuclear inflammatory infiltrates on both days 3 and 8 post-challenge. SARS-CoV antigens were present in the lungs on day 3 post-challenge and were cleared by day 8 (Figures 2a, 3a and b). In contrast, mice that received undiluted hyperimmune SARS antiserum had no significant inflammation in the lungs on either days 3 or 8 post-challenge and IHC staining revealed no SARS-CoV antigens on either day (Figures 2c, 3e and f). Mice that received diluted hyperimmune SARS antiserum had rare perivascular foci of mononuclear inflammatory infiltrates with some SARS antigen in epithelial cells lining the airways on day 3 post-challenge. These mice had no significant inflammation and SARS-CoV antigens were cleared by day 8 post-challenge (Figures 2b, 3c and d).
Figure 2. Lower resolution histopathologic features of mouse lungs 8 days following infection with SARS-CoV (strain Urbani).
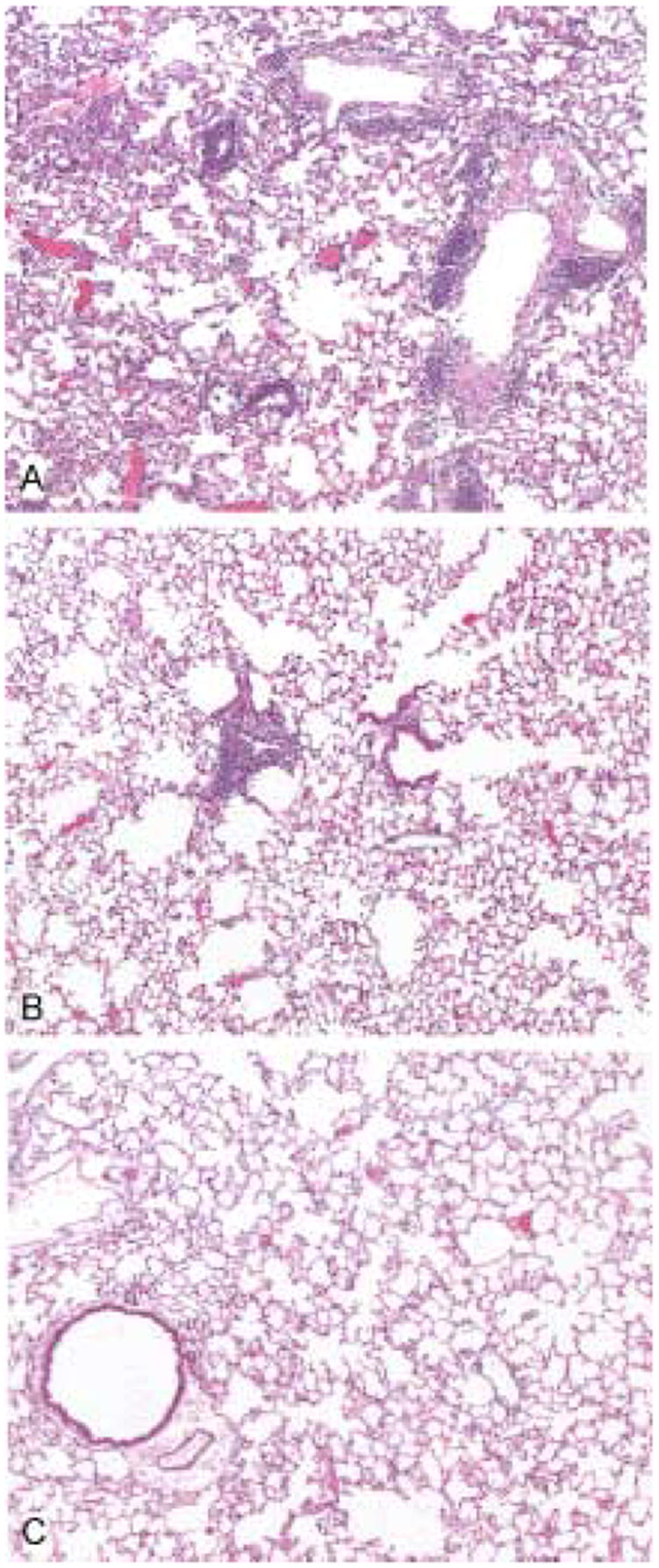
The lungs of mice receiving normal mouse serum show multifocal and extensive perivascular and interstitial inflammatory cell infiltrates (A). In contrast, mice receiving a 1:4 dilution of hyperimmune SARS-CoV antiserum show only occasional small foci of perivascular infiltrates (B) and the mice that received undiluted hyperimmune antiserum showed no significant pulmonary inflammation (C). Hematoxylin and eosin stain. Original magnifications × 25.
Figure 3. Higher resolution histopathology and immunohistochemical (IHC) staining of mouse lungs infected with SARS-CoV (strain Urbani) 3 days postinfection.
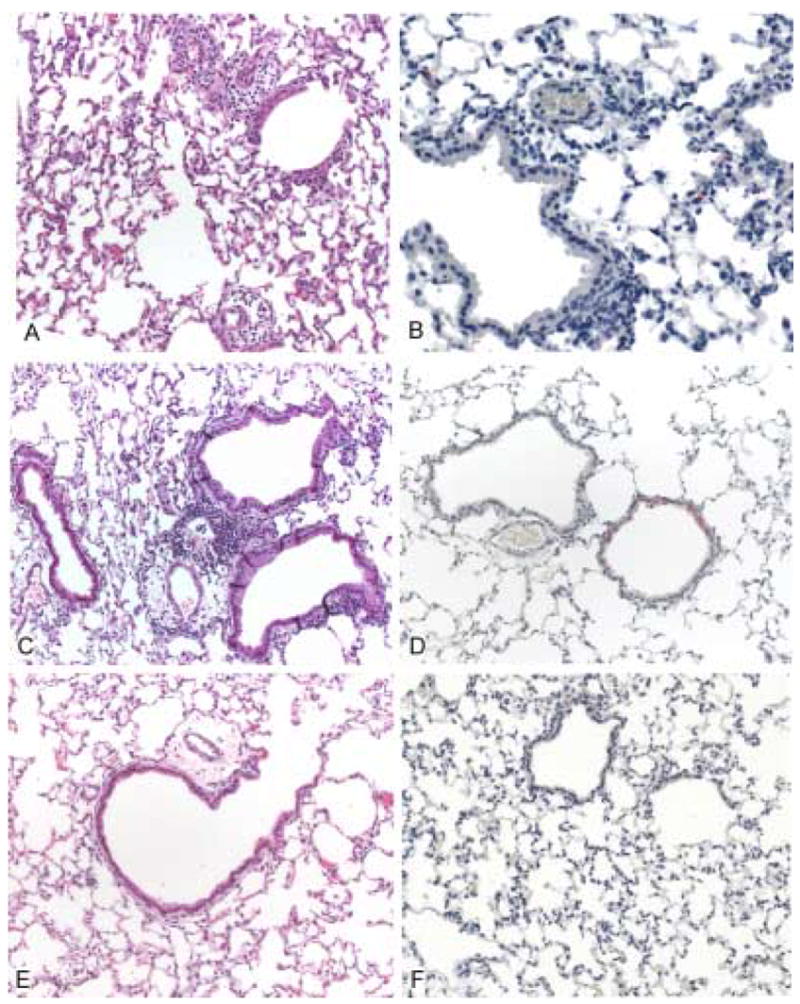
In the lungs of a mouse treated with normal mouse serum, predominantly mononuclear inflammatory cell infiltrates are identified around small blood vessels and in the alveolar capillaries (A), and are associated with presence of SARS-CoV antigens (red) in alveolar pneumocytes (B). The lungs of infected mice that received a 1:4 dilution of SARS-CoV hyperimmune antiserum show focal mild perivascular infiltrates (C), and occasional IHC staining of SARS-CoV antigens, localized predominantly in bronchiolar epithelium (D). Mice that were treated with undiluted hyperimmune mouse serum showed no significant pulmonary inflammation (E) or IHC evidence of infection with SARS-CoV (F). Hematoxylin and eosin stain (A, C, E). Rabbit anti-SARS-CoV antibody, immunoalkaline phosphatase with naphthol fast-red and hematoxylin counterstain (B, D, F). Original magnifications × 50.
Active Immunization
On day 30 post-vaccination, three of four mice that received rVSV-S vaccination achieved a detectable neutralizing antibody titer that was at the lower limit of detection (1:8). The fourth mouse did not have a detectable neutralizing antibody response (≤1:8) (Table 1). Mice that received vector alone (rVSV) did not have a detectable neutralizing antibody response (≤1:8). The control group of mice infected with SARS-CoV achieved a significant mean neutralizing antibody titer on day 30 post-infection of 1:89 ± 34.8.
Table 1.
Vaccination with live attenuated rVSV-S vaccine protects mice from challenge with SARS-CoV
| Intervention group | Mouse number | Pre-challenge neutralizing antibody titer in serum (log2)a | Virus titer in lungsb | Mean maximum change in weight (on day 7)c |
|---|---|---|---|---|
| SARS-CoV | 1 | 5.5 | ≤ 1.5d | |
| 2 | 7.0 | ≤ 1.5 | ||
| 3 | 7.7 | ≤ 1.5 | ||
| 4 | 5.3 | ≤ 1.5 | ||
| Mean ≤ 1.5 | + 0.5% | |||
| rVSV-S | 5 | ≤ 2e | 4.2 | |
| 6 | 3 | 3.7 | ||
| 7 | 3 | 3.4 | ||
| 8 | 3 | ≤ 1.5 | ||
| Mean 3.2 ± 0.60 | +0.5% | |||
| rVSV | 9 | ≤ 2 | 7.7 | |
| 10 | ≤ 2 | 7.4 | ||
| 11 | ≤ 2 | 7.9 | ||
| 12 | ≤ 2 | 7.9 | ||
| Mean 7.8 ± 0.12 | −10% |
Log transformed reciprocal mean SARS-CoV specific neutralizing antibody titer
Viral titers expressed as log10TCID50/g tissue, p ≤ 0.0001 SARS-CoV compared to rVSV control and p = 0.0003 for rVSV-S compared to rVSV control, day 2 post challenge
Expressed as percent change in weight from weight on day of challenge (d 0)
Virus not detected, this value is the lower limit of virus detection
Neutralizing antibody not detected, this value is at the lower limit of detection
Although mice vaccinated with rVSV-S had low neutralizing titers on day 30 post-vaccination, they were partially protected from weight loss as well as pulmonary virus replication upon SARS-CoV challenge. On days 1 through 3 post-challenge, mice that were immunized with rVSV lost 7% of their pre-challenge weight while rVSV-S vaccinated mice lost less than 3% of pre-challenge body weight. Maximum mean change in weight from day of challenge occurred on day 7 post challenge. rVSV vaccinated mice lost 10% of pre-challenge weight on day 7 post-challenge while SARS-CoV and rVSV-S treated groups began to regain weight (5% and 3%, respectively) (Table 1). rVSV-S vaccinated mice had a mean pulmonary viral titer on day 4 post-challenge of 103.2 ± 0.6 TCID50/g. One mouse in this group was completely protected and, in the remaining three mice, viral replication was reduced by 10,000-fold as compared to the mice that received vector alone. Mice that were previously infected with SARS-CoV and subsequently challenged on day 30 were completely protected from pulmonary viral replication. Both groups had statistically significant reductions in mean pulmonary viral titers as compared to the rVSV control group, 107.80 ± 0.12TCID50/g (p<0.04) (Table 1).
Discussion
Severe acute respiratory syndrome, caused by a novel coronavirus, (SARS-CoV) spread through a naïve human population in over 30 countries in a matter of months. Control of the outbreak was achieved by the use of stringent public health measures. Although the disease has not reappeared in epidemic form since 2003, related viruses have been identified in animals from which they may be re-introduced into humans. In the event that the virus reemerges in the human population specific preventive strategies should be available to be implemented along with public health measures.
A number of animal models have been evaluated for the study of SARS-CoV prevention and control. The golden Syrian hamster model was used to study the control of infection when monoclonal antibodies (MAbs) were administered for prophylaxis or treatment [13,14]. Used post-exposure, MAbs were able to alter the course and severity of SARS-CoV infection in hamsters. The hamster model, like the aged mouse model, supports replication of virus in the presence of significant pathological changes to the lungs. In this study we demonstrate the utility of the aged BALB/c mouse model for the evaluation of vaccines and immunoprophylaxis.
The aged mouse model differs from the young mouse model in which the only measure of efficacy is quantitative virology. Viral replication in the lungs of young mice occurs in the absence of clinical illness, extrapulmonary spread, or elevated pro-inflammatory cytokines. Peak pulmonary viral titers of around 107 TCID50/g are seen on day 1 post infection, with clearance by days 5 through 7. Histopathological exam two days post infection reveals only mild focal peribronchiolar mononuclear inflammatory infiltrates with clearance of viral antigen by day 9 [8]. Aged mice allow analysis of four outcome measures: quantitative virology, clinical symptoms, histopathological changes, and immunohistochemistry in the lungs. High titer viral replication is accompanied by significant weight loss on days 3–6 post infection, with a nadir of 8% on day 4 post infection and extra-pulmonary spread to the liver on days 2 and 5. Peak pulmonary viral titers of about 108 TCID50/g are seen on day 2 post-infection, with evidence of viral replication at a high titer through day 5. Multifocal interstitial lymphocytic infiltrates are seen by histopathology on day 5, with gradual resolution and repair on days 9 and 13 post-infection [7]. Complete protection from SARS-CoV infection in aged mice is demonstrated when animals achieve a high level of neutralizing antibody. This was seen when aged mice were passively immunized using murine SARS hyperimmune antisera. Even with low levels of SARS-CoV specific neutralizing antibody after passive immunization, mice were partially protected from viral infection. A greater than 100-fold reduction in viral titer was observed in mice that received a 1:4 dilution of hyperimmune antisera compared to mice that received non-immune mouse serum prior to challenge. Reductions in extent and duration of lung inflammation were seen when viral load was reduced by this level. No significant inflammation was found in mice that achieved sterilizing immunity. Thus, prior administration of SARS hyperimmune anitsera prevents viral replication and associated disease in aged BALB/c mice.
Although rVSV vaccines induce high and prolonged antibody responses in young mice we did not see a similar robust level of antibody response in old mice [9]. There are two possible explanations for the poor antibody response to SARS-CoV following immunization of aged mice with rVSV-S; either rVSV did not replicate efficiently or the response of old mice to the SARS-CoV spike protein is poor. Antibody titers to VSV in these mice were robust (data not shown, ≥ 1:1280), suggesting that the latter explanation is likely. A 10,000-fold reduction in viral load and protection from weight loss and histopathological changes were seen in mice that were immunized with rVSV-S vaccine compared to mice that received rVSV. A surprising finding was that this protection occurred in the absence of high neutralizing antibody levels. Protection from rVSV vectored vaccines is mediated by strong humoral and cytotoxic T lymphocyte (CTL) responses. A CTL response is in part responsible for the rapid control and clearance of many viral infections [10,11,12]. Although antibodies may have contributed to protection, we speculate that the efficacy of rVSV-S in old mice results from a strong CTL response. This speculation is further supported by the observation that although neutralizing antibody titers were lower, the reduction in pulmonary virus replication was greater in mice immunized with rVSV-S than in mice that were passively immunized with a 1:4 dilution of hyperimmune SARS antiserum.
The aged mouse model, which mimics findings in elderly SARS patients, has proven to be a useful tool in the evaluation of strategies to prevent SARS-CoV infection. In this study, both passive antibody transfer and vaccination with a live virus vaccine were effective strategies to control viral replication and associated disease.
Acknowledgments
Funding for this research was supported in part by the Intramural Research Program of the NIH, NIAID and NIH grant AI057158. We would also like to thank Jadon Jackson for his assistance in all animal work.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Guan Y, Zheng BJ, He YQ, Liu XL, Zhuang ZX, Cheung CL, et al. Isolation and characterization of viruses related to the SARS coronavirus from animals in southern China. Science. 2003;302(5643):276–8. doi: 10.1126/science.1087139. [DOI] [PubMed] [Google Scholar]
- 2.Lau SK, Woo PC, Li KS, Huang Y, Tsoi HW, Wong BH, et al. Severe acute respiratory syndrome coronavirus-like virus in Chinese horseshoe bats. Proc Natl Acad Sci USA. 2005;102(39):14040–5. doi: 10.1073/pnas.0506735102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Subbarao K, Roberts A. Is there an ideal animal model for SARS? Trends Microbiol. 2006;14(7):299–303. doi: 10.1016/j.tim.2006.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Peiris JS, Lai ST, Poon LL, Guan Y, Yam LY, Lim W, et al. Coronavirus as a possible cause of severe acute respiratory syndrome. Lancet. 2003;361:1767–1772. doi: 10.1016/S0140-6736(03)13077-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Chan JW, Ng CK, Chan YH, Mok TY, Lee S, Chu SY, et al. Short term outcome and risk factors for adverse clinical outcomes in adults with severe acute respiratory syndrome (SARS) Thorax. 2003;58:686–689. doi: 10.1136/thorax.58.8.686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Tsui PT, Kwok ML, Yuen H, Lai ST. Severe acute respiratory syndrome: clinical outcome and prognostic correlates. Emerg Infect Dis. 2003;9:1064–1069. doi: 10.3201/eid0909.030362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Roberts A, Paddock C, Vogel L, Butler E, Zaki S, Subbarao K. Aged BALB/c mice as a model for increased severity of severe acute respiratory syndrome in elderly humans. J Virol. 2005;79(9):5833–5838. doi: 10.1128/JVI.79.9.5833-5838.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Subbarao K, McAuliffe J, Vogel L, Fahle G, Fischer S, Tatti K, et al. Prior infection and passive transfer of neutralizing antibody prevent replication of severe acute respiratory syndrome coronavirus in the respiratory tract of mice. J Virol. 2004;78(7):3572–3577. doi: 10.1128/JVI.78.7.3572-3577.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kapadia SU, Rose JK, Lamirande E, Vogel L, Subbarao K, Roberts A. Long-term protection from SARS coronavirus infection conferred by a single immunization with an attenuated VSV-based vaccine. Virology. 2005;340(2):174–82. doi: 10.1016/j.virol.2005.06.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Hangartner L, Zinkernagel RM, Hengartner H. Antiviral antibody responses: the two extremes of a wide spectrum. Nat Rev Immunol. 2006;6(3):231–43. doi: 10.1038/nri1783. [DOI] [PubMed] [Google Scholar]
- 11.Haglund K, Leiner I, Kerksiek K, Buonocore L, Pamer E, Rose JK. High-level primary CD8(+) T-cell response to human immunodeficiency virus type 1 gag and env generated by vaccination with recombinant vesicular stomatitis viruses. J Virol. 2002;76(6):2730–8. doi: 10.1128/JVI.76.6.2730-2738.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Haglund K, Leiner I, Kerksiek K, Buonocore L, Pamer E, Rose JK. Robust recall and long-term memory T-cell responses induced by prime-boost regimens with heterologous live viral vectors expressing human immunodeficiency virus type 1 Gag and Env proteins. J Virol. 2002;76(15):7506–17. doi: 10.1128/JVI.76.15.7506-7517.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Greenough TC, Babcock GJ, Roberts A, Hernandez HJ, Thomas WD, Jr, Coccia JA, et al. Development and characterization of a severe acute respiratory syndrome-associated coronavirus-neutralizing human monoclonal antibody that provides effective immunoprophylaxis in mice. J Infect Dis. 2005;191(4):507–14. doi: 10.1086/427242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Roberts A, Thomas WD, Guarner J, Lamirande EW, Babcock GJ, Greenough TC, et al. Therapy with a severe acute respiratory syndrome-associated coronavirus-neutralizing human monoclonal antibody reduces disease severity and viral burden in golden Syrian hamsters. J Infect Dis. 2006;193(5):685–92. doi: 10.1086/500143. [DOI] [PMC free article] [PubMed] [Google Scholar]


