Fig. 6. Biotin Interference Assay of integrative recombination.
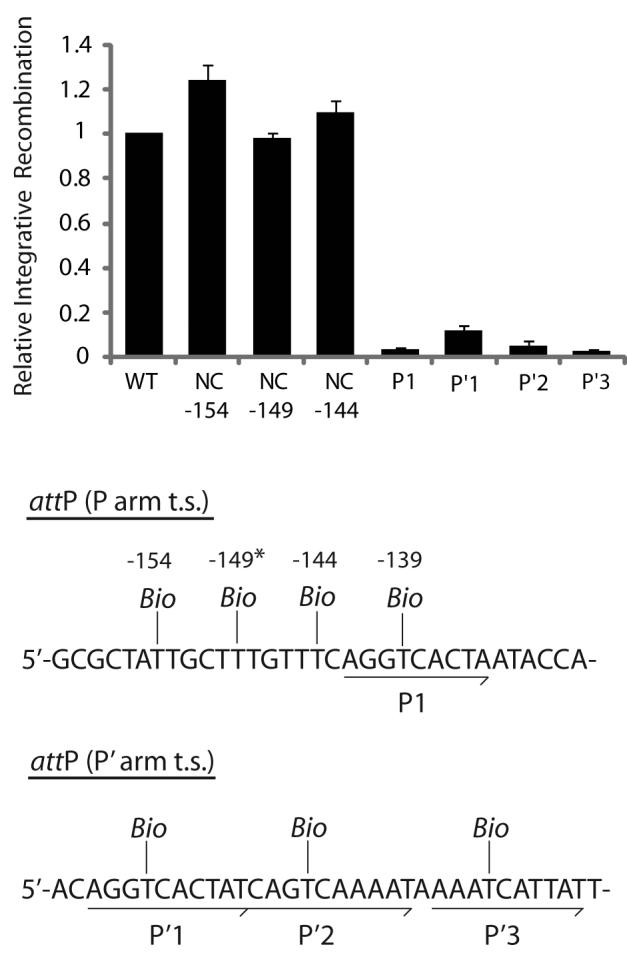
Supercoiled attP sites containing a single biotin dT substitution at one of the indicated positions were recombined with a radiolabeled linear attB partner and the recombination efficiency was normalized to that of a control reaction with no substitutions (10-20% recombination efficiency). Based on the X-ray crystal structure, the biotin at −149 (*) is predicted to interfere with the putative non-canonical Int binding in the same way that biotin at position −139 blocks Int binding at P1. Biotin dT substitutions at positions −154, −149, and −144 required changing the wild-type sequences from A, C, and A, respectively; in each case the unsubstituted control attP contained the identical changes.
