Abstract
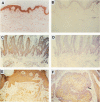
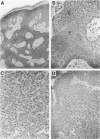
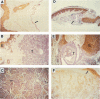
Transforming growth factor-alpha (TGF alpha) is a potent mitogen for epithelial cells that has been localized to normal human appendageal epithelia. To further understand the role of TGF alpha in human appendages, we examined TGF alpha expression immunohistochemically in 17 types of human appendageal tumors differentiating toward hair follicles, eccrine, apocrine, and sebaceous glands. In order of decreasing degrees of differentiation, tumors could be divided into hyperplasias, adenomas, benign epitheliomas, and primordial epitheliomas. Using an antibody that recognizes primarily the 6-kd and 13-kd forms of TGF alpha, TGF alpha immunostaining in 16 of 17 tumor types analyzed was found to follow a similar pattern, with expression in hyperplasias greater than adenomas greater than benign epitheliomas greater than primordial epitheliomas. Within a given tumor, TGF alpha expression also correlated well with the known differentiation state of the tumor cell types. The results suggest that TGF alpha expression is directly correlated with the differentiation state of hair follicle, eccrine, apocrine, and sebaceous tumors in human skin, and raises the possibility that TGF alpha may play a role in the differentiation of appendageal epithelia.
Full text
PDF










Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anklesaria P., Teixidó J., Laiho M., Pierce J. H., Greenberger J. S., Massagué J. Cell-cell adhesion mediated by binding of membrane-anchored transforming growth factor alpha to epidermal growth factor receptors promotes cell proliferation. Proc Natl Acad Sci U S A. 1990 May;87(9):3289–3293. doi: 10.1073/pnas.87.9.3289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bhawan J. Pilar sheath acanthoma. A new benign follicular tumor. J Cutan Pathol. 1979 Oct;6(5):438–440. doi: 10.1111/j.1600-0560.1979.tb01167.x. [DOI] [PubMed] [Google Scholar]
- Blecher S. R., Kapalanga J., Lalonde D. Induction of sweat glands by epidermal growth factor in murine X-linked anhidrotic ectodermal dysplasia. Nature. 1990 Jun 7;345(6275):542–544. doi: 10.1038/345542a0. [DOI] [PubMed] [Google Scholar]
- Brachmann R., Lindquist P. B., Nagashima M., Kohr W., Lipari T., Napier M., Derynck R. Transmembrane TGF-alpha precursors activate EGF/TGF-alpha receptors. Cell. 1989 Feb 24;56(4):691–700. doi: 10.1016/0092-8674(89)90591-6. [DOI] [PubMed] [Google Scholar]
- Brownstein M. H., Shapiro L. Trichilemmoma. Analysis of 40 new cases. Arch Dermatol. 1973 Jun;107(6):866–869. doi: 10.1001/archderm.107.6.866. [DOI] [PubMed] [Google Scholar]
- Carpenter G., Stoscheck C. M., Preston Y. A., DeLarco J. E. Antibodies to the epidermal growth factor receptor block the biological activities of sarcoma growth factor. Proc Natl Acad Sci U S A. 1983 Sep;80(18):5627–5630. doi: 10.1073/pnas.80.18.5627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castro C., Winkelmann R. K. Spiradenoma. Histochemical and electron microscopic study. Arch Dermatol. 1974 Jan;109(1):40–48. doi: 10.1001/archderm.109.1.40. [DOI] [PubMed] [Google Scholar]
- Coffey R. J., Jr, Derynck R., Wilcox J. N., Bringman T. S., Goustin A. S., Moses H. L., Pittelkow M. R. Production and auto-induction of transforming growth factor-alpha in human keratinocytes. 1987 Aug 27-Sep 2Nature. 328(6133):817–820. doi: 10.1038/328817a0. [DOI] [PubMed] [Google Scholar]
- Coffey R. J., Jr, Sipes N. J., Bascom C. C., Graves-Deal R., Pennington C. Y., Weissman B. E., Moses H. L. Growth modulation of mouse keratinocytes by transforming growth factors. Cancer Res. 1988 Mar 15;48(6):1596–1602. [PubMed] [Google Scholar]
- Derynck R., Goeddel D. V., Ullrich A., Gutterman J. U., Williams R. D., Bringman T. S., Berger W. H. Synthesis of messenger RNAs for transforming growth factors alpha and beta and the epidermal growth factor receptor by human tumors. Cancer Res. 1987 Feb 1;47(3):707–712. [PubMed] [Google Scholar]
- Elder J. T., Fisher G. J., Lindquist P. B., Bennett G. L., Pittelkow M. R., Coffey R. J., Jr, Ellingsworth L., Derynck R., Voorhees J. J. Overexpression of transforming growth factor alpha in psoriatic epidermis. Science. 1989 Feb 10;243(4892):811–814. doi: 10.1126/science.2916128. [DOI] [PubMed] [Google Scholar]
- Finzi E., Fleming T., Pierce J. H. Retroviral expression of transforming growth factor-alpha does not transform fibroblasts or keratinocytes. J Invest Dermatol. 1990 Oct;95(4):382–387. doi: 10.1111/1523-1747.ep12555464. [DOI] [PubMed] [Google Scholar]
- Finzi E., Fleming T., Segatto O., Pennington C. Y., Bringman T. S., Derynck R., Aaronson S. A. The human transforming growth factor type alpha coding sequence is not a direct-acting oncogene when overexpressed in NIH 3T3 cells. Proc Natl Acad Sci U S A. 1987 Jun;84(11):3733–3737. doi: 10.1073/pnas.84.11.3733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finzi E., Harkins R., Horn T. TGF-alpha is widely expressed in differentiated as well as hyperproliferative skin epithelium. J Invest Dermatol. 1991 Mar;96(3):328–332. doi: 10.1111/1523-1747.ep12465223. [DOI] [PubMed] [Google Scholar]
- Finzi E., Kilkenny A., Strickland J. E., Balaschak M., Bringman T., Derynck R., Aaronson S., Yuspa S. H. TGF alpha stimulates growth of skin papillomas by autocrine and paracrine mechanisms but does not cause neoplastic progression. Mol Carcinog. 1988;1(1):7–12. doi: 10.1002/mc.2940010105. [DOI] [PubMed] [Google Scholar]
- Gottlieb A. B., Chang C. K., Posnett D. N., Fanelli B., Tam J. P. Detection of transforming growth factor alpha in normal, malignant, and hyperproliferative human keratinocytes. J Exp Med. 1988 Feb 1;167(2):670–675. doi: 10.1084/jem.167.2.670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HASHIMOTO K., LEVER W. F. ECCRINE POROMA; HISTOCHEMICAL AND ELECTRON MICROSCOPIC STUDIES. J Invest Dermatol. 1964 Oct;43:237–247. [PubMed] [Google Scholar]
- Hashimoto K., DiBella R. J., Borsuk G. M., Lever W. F. Eruptive hidradenoma and syringoma. Histological, histochemical, and electron microscopic studies. Arch Dermatol. 1967 Nov;96(5):500–519. [PubMed] [Google Scholar]
- Hashimoto K., DiBella R. J., Lever W. F. Clear cell hidradenoma. Histological, histochemical, and electron microscopic studies. Arch Dermatol. 1967 Jul;96(1):18–38. doi: 10.1001/archderm.96.1.18. [DOI] [PubMed] [Google Scholar]
- Hashimoto K., Gross B. G., Lever W. F. Syringoma. Histochemical and electron microscopic studies. J Invest Dermatol. 1966 Feb;46(2):150–166. [PubMed] [Google Scholar]
- Hashimoto K., Gross B. G., Nelson R. G., Lever W. F. Eccrine spiradenoma. Histochemical and electron microscopic studies. J Invest Dermatol. 1966 Apr;46(4):347–365. [PubMed] [Google Scholar]
- Hashimoto K., Lever W. F. Histogenesis of skin appendage tumors. Arch Dermatol. 1969 Sep;100(3):356–369. [PubMed] [Google Scholar]
- Hashimoto K., Lever W. F. Histogenesis of skin appendage tumors. Arch Dermatol. 1969 Sep;100(3):356–369. [PubMed] [Google Scholar]
- Hashimoto K. Syringocystadenoma papilliferum. An electron microscopic study. Arch Dermatol Forsch. 1972;245(4):353–369. [PubMed] [Google Scholar]
- Jhappan C., Stahle C., Harkins R. N., Fausto N., Smith G. H., Merlino G. T. TGF alpha overexpression in transgenic mice induces liver neoplasia and abnormal development of the mammary gland and pancreas. Cell. 1990 Jun 15;61(6):1137–1146. doi: 10.1016/0092-8674(90)90076-q. [DOI] [PubMed] [Google Scholar]
- KLIGMAN A. M., PINKUS H. The histogenesis of nevoid tumors of the skin. The folliculoma--a hair-follicle tumor. Arch Dermatol. 1960 Jun;81:922–930. doi: 10.1001/archderm.1960.03730060038007. [DOI] [PubMed] [Google Scholar]
- Landry M., Winkelmann R. K. An unusual tubular apocrine adenoma. Arch Dermatol. 1972 Jun;105(6):869–879. [PubMed] [Google Scholar]
- Marquardt H., Hunkapiller M. W., Hood L. E., Twardzik D. R., De Larco J. E., Stephenson J. R., Todaro G. J. Transforming growth factors produced by retrovirus-transformed rodent fibroblasts and human melanoma cells: amino acid sequence homology with epidermal growth factor. Proc Natl Acad Sci U S A. 1983 Aug;80(15):4684–4688. doi: 10.1073/pnas.80.15.4684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Massagué J. Epidermal growth factor-like transforming growth factor. II. Interaction with epidermal growth factor receptors in human placenta membranes and A431 cells. J Biol Chem. 1983 Nov 25;258(22):13614–13620. [PubMed] [Google Scholar]
- Matias J. R., Orentreich N. Stimulation of hamster sebaceous glands by epidermal growth factor. J Invest Dermatol. 1983 Jun;80(6):516–519. doi: 10.1111/1523-1747.ep12535112. [DOI] [PubMed] [Google Scholar]
- Matsui Y., Halter S. A., Holt J. T., Hogan B. L., Coffey R. J. Development of mammary hyperplasia and neoplasia in MMTV-TGF alpha transgenic mice. Cell. 1990 Jun 15;61(6):1147–1155. doi: 10.1016/0092-8674(90)90077-r. [DOI] [PubMed] [Google Scholar]
- Moore G. P., Panaretto B. A., Robertson D. Epidermal growth factor delays the development of the epidermis and hair follicles of mice during growth of the first coat. Anat Rec. 1983 Jan;205(1):47–55. doi: 10.1002/ar.1092050107. [DOI] [PubMed] [Google Scholar]
- Moore G. P., Panaretto B. A., Robertson D. Inhibition of wool growth in merino sheep following administration of mouse epidermal growth factor and a derivative. Aust J Biol Sci. 1982;35(2):163–172. doi: 10.1071/bi9820163. [DOI] [PubMed] [Google Scholar]
- Nanney L. B., Magid M., Stoscheck C. M., King L. E., Jr Comparison of epidermal growth factor binding and receptor distribution in normal human epidermis and epidermal appendages. J Invest Dermatol. 1984 Nov;83(5):385–393. doi: 10.1111/1523-1747.ep12264708. [DOI] [PubMed] [Google Scholar]
- Niizuma K. Syringocystadenoma papilliferum: light and electron microscopic studies. Acta Derm Venereol. 1976;56(5):327–336. [PubMed] [Google Scholar]
- Rosenthal A., Lindquist P. B., Bringman T. S., Goeddel D. V., Derynck R. Expression in rat fibroblasts of a human transforming growth factor-alpha cDNA results in transformation. Cell. 1986 Jul 18;46(2):301–309. doi: 10.1016/0092-8674(86)90747-6. [DOI] [PubMed] [Google Scholar]
- SANDERSON K. V., RYAN E. A. The histochemistry of eccrine poroma. Br J Dermatol. 1963 Feb;75:86–88. doi: 10.1111/j.1365-2133.1963.tb13943.x. [DOI] [PubMed] [Google Scholar]
- Sandgren E. P., Luetteke N. C., Palmiter R. D., Brinster R. L., Lee D. C. Overexpression of TGF alpha in transgenic mice: induction of epithelial hyperplasia, pancreatic metaplasia, and carcinoma of the breast. Cell. 1990 Jun 15;61(6):1121–1135. doi: 10.1016/0092-8674(90)90075-p. [DOI] [PubMed] [Google Scholar]
- Tam J. P. Physiological effects of transforming growth factor in the newborn mouse. Science. 1985 Aug 16;229(4714):673–675. doi: 10.1126/science.3860952. [DOI] [PubMed] [Google Scholar]
- Taub M., Wang Y., Szczesny T. M., Kleinman H. K. Epidermal growth factor or transforming growth factor alpha is required for kidney tubulogenesis in matrigel cultures in serum-free medium. Proc Natl Acad Sci U S A. 1990 May;87(10):4002–4006. doi: 10.1073/pnas.87.10.4002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tepass U., Theres C., Knust E. crumbs encodes an EGF-like protein expressed on apical membranes of Drosophila epithelial cells and required for organization of epithelia. Cell. 1990 Jun 1;61(5):787–799. doi: 10.1016/0092-8674(90)90189-l. [DOI] [PubMed] [Google Scholar]
- Todaro G. J., De Larco J. E., Cohen S. Transformation by murine and feline sarcoma viruses specifically blocks binding of epidermal growth factor to cells. Nature. 1976 Nov 4;264(5581):26–31. doi: 10.1038/264026a0. [DOI] [PubMed] [Google Scholar]
- Toribio J., Zulaica A., Peteiro C. Tubular apocrine adenoma. J Cutan Pathol. 1987 Apr;14(2):114–117. doi: 10.1111/j.1600-0560.1987.tb00482.x. [DOI] [PubMed] [Google Scholar]
- Tsambaos D., Greither A., Orfanos C. E. Multiple malignant Spiegler tumors with brachydactyly and racket-nails. Light and electron microscopic study. J Cutan Pathol. 1979 Feb;6(1):31–41. doi: 10.1111/j.1600-0560.1979.tb00303.x. [DOI] [PubMed] [Google Scholar]
- WINKELMANN R. K., MULLER S. A. SWEAT GLAND TUMORS. I. HISTOCHEMICAL STUDIES. Arch Dermatol. 1964 Jun;89:827–831. doi: 10.1001/archderm.1964.01590300055017. [DOI] [PubMed] [Google Scholar]
- Winkelmann R. K., McLeod W. A. The dermal duct tumor. Arch Dermatol. 1966 Jul;94(1):50–55. [PubMed] [Google Scholar]
- Winkelmann R. K., Wolff K. Histochemistry of hidradenoma and eccrine spiradenoma. J Invest Dermatol. 1967 Aug;49(2):173–180. doi: 10.1038/jid.1967.121. [DOI] [PubMed] [Google Scholar]
- Wong S. T., Winchell L. F., McCune B. K., Earp H. S., Teixidó J., Massagué J., Herman B., Lee D. C. The TGF-alpha precursor expressed on the cell surface binds to the EGF receptor on adjacent cells, leading to signal transduction. Cell. 1989 Feb 10;56(3):495–506. doi: 10.1016/0092-8674(89)90252-3. [DOI] [PubMed] [Google Scholar]
- Yochem J., Greenwald I. glp-1 and lin-12, genes implicated in distinct cell-cell interactions in C. elegans, encode similar transmembrane proteins. Cell. 1989 Aug 11;58(3):553–563. doi: 10.1016/0092-8674(89)90436-4. [DOI] [PubMed] [Google Scholar]