Abstract
Purpose
Cowden syndrome (CS) is an autosomal dominant disorder characterized by benign and malignant tumors. One-quarter of patients who are diagnosed with CS have pathogenic germline PTEN mutations, which increase the risk of the development of breast, thyroid, uterine, renal, and other cancers. PTEN testing and regular, intensive cancer surveillance allow for early detection and treatment of these cancers for mutation-positive patients and their relatives. Individual CS-related features, however, occur commonly in the general population, making it challenging for clinicians to identify CS-like patients to offer PTEN testing.
Patients and Methods
We calculated the cost per mutation detected and analyzed the cost-effectiveness of performing selected PTEN testing among CS-like patients using a semi-quantitative score (the PTEN Cleveland Clinic [CC] score) compared with existing diagnostic criteria. In our model, first-degree relatives of the patients with detected PTEN mutations are offered PTEN testing. All individuals with detected PTEN mutations are offered cancer surveillance.
Results
CC score at a threshold of 15 (CC15) costs from $3,720 to $4,573 to detect one PTEN mutation, which is the most inexpensive among the different strategies. At base-case, CC10 is the most cost-effective strategy for female patients who are younger than 40 years, and CC15 is the most cost-effective strategy for female patients who are between 40 and 60 years of age and male patients of all ages. In sensitivity analyses, CC15 is robustly the most cost-effective strategy for probands who are younger than 60 years.
Conclusion
Use of the CC score as a clinical risk calculator is a cost-effective prescreening method to identify CS-like patients for PTEN germline testing.
INTRODUCTION
The goals of the Healthy People 2020 initiative of the US Department of Health and Human Services focus for the first time on genomic medicine among its list of priorities. The genomic objectives of Healthy People 2020 emphasize the importance of obtaining a family and genetic history as a powerful guide for clinical and public health initiatives.1 One of the most important applications of genetic testing is in individual risk assessment for the development of cancer, thus allowing for successful risk reduction strategies for the at-risk patient while avoiding unwarranted surveillance in unaffected family members. Currently, greater than 200 hereditary cancer susceptibility syndromes are described, accounting for 5% to 10% of all cancers. Given that there are 1.6 million new cancer cases each year in the United States,2 the challenge facing clinicians is to identify who among that group may be at risk of having an underlying hereditary cause. An even more critical challenge involves determining how to do so in a cost-effective manner.
Cowden syndrome (CS; Mendelian Inheritance in Man [MIM] No. 158350) is a difficult-to-recognize autosomal dominant, highly penetrant genetic disorder. More than 90% of CS individuals with germline (heritable) PTEN mutations are believed to manifest some feature of the syndrome (although rarely cancer) by the age of 20 years, and by the age of 30 years, nearly 100% of mutation carriers are believed to have developed at least some of the mucocutaneous signs. Affected individuals also have an increased risk of the development of several malignant diseases, including female breast cancer, thyroid cancer with an overrepresentation of follicular histology, and endometrial cancer,3–5 and are at a significantly increased risk (a seven-fold increased risk as compared with that in the general population) of experiencing a second primary malignant neoplasm.6
CS remains underdiagnosed because of its variable expression (often with only subtle skin signs); consequently, the current prevalence estimate of 1 in 200,000 is still likely to be an underestimate. Consensus diagnostic criteria for CS were first developed in 1996 by the International Cowden Consortium and form the basis for the National Comprehensive Cancer Network (NCCN) guidelines (Data Supplement).7 Relaxed criteria are defined as full criteria minus one criterion, and such individuals are referred to as CS-like. Most patients with CS are diagnosed only after a personal history of a second cancer.6 Many mucocutaneous features of CS predate the occurrence of cancer, and timely identification of patients with a germline PTEN mutation is important because it allows for gene-targeted surveillance for affected probands and predictive testing for relatives. To address this, more recently, the PTEN Cleveland Clinic (CC) score has been shown to provide a well-calibrated estimation of pretest probability of PTEN status.3 In this study, we examined the diagnostic utility and cost-effectiveness of using the CC score as a clinical predictive tool and compared it with existing clinical diagnostic criteria.
PATIENTS AND METHODS
Using data from the largest prospective cohort of over 3000 CS and CS-like (defined as International Cowden Consortium criteria minus one) patients enrolled globally,4,6,8 we created a decision model to simulate virtual clinical trials of different genetic screening strategies. The virtual trials included CS or CS-like patients who were older than 18 years and were referred to a clinical cancer genetics service. These patients were randomized to one of six genetic screening strategies: no germline PTEN mutation testing; germline PTEN mutation testing when CC scores are above the threshold of 5 (CC5); germline PTEN mutation testing when CC scores are above the threshold of 10 (CC10); germline PTEN mutation testing when CC scores are above the threshold of 15 (CC15); germline PTEN mutation testing if NCCN criteria are met; and germline PTEN mutation testing for all enrolled CS-like patients.
If a patient was detected as being PTEN-mutation positive (PTENmut+), his or her first-degree relatives would be offered PTEN mutation testing as well. All detected PTENmut+ patients would be recommended for cancer surveillance (Data Supplement), while others (PTEN mutation negative or undetected PTENmut+) would receive the standard cancer screening recommended for the general population (ie, breast-cancer screening for women older than 40 years and no screening for men).
We first calculated the costs per mutation detected of each genetic screening strategy. We then followed all patients every year until death to calculate the cost-effectiveness ratio of each genetic screening strategy as compared with no genetic screening, and to compare the incremental cost-effectiveness ratios (ICERs) with the next-less-intensive genetic screening strategies.
Decision Model
We built a decision model to simulate the clinical trial. The model first simulates plausible outcomes of alternative genetic screening strategies in terms of genetic mutations that were detected or undetected in patients and their relatives (Fig 1 and Data Supplement) and then virtually followed the patients and their relatives until death (Data Supplement). The family structure of PTEN patients was assumed to be identical to that of the general population in the United States, as described in the model with the average number of children9 and average age when mothers give birth to first children in US families.10 Compliances with genetic testing of first-degree relatives were assumed to be 60% for parents and siblings and 70% for children.11 Compliance with cancer surveillance is assigned to be 77.9% for all types of cancers.12–16 The search process and derivation of model inputs are described in detail in the Data Supplement. Estimates for all model parameters are shown in the Data Supplement.
Fig 1.

Schematic diagram of genetic screening strategies. A Cowden syndrome (CS) -like patient is prescreened with a clinical calculator (National Comprehensive Cancer Network criteria or Cleveland Clinic score at different thresholds). If the individual meets the threshold, he or she will receive genetic testing for germline PTEN mutations. First-degree relatives of PTEN-mutation-positive individuals will be offered genetic testing for the specific mutations. All individuals who are positive for PTEN mutations will receive cancer surveillance according to recommendations.
Cost-Effectiveness Analysis
A societal perspective was used in the analysis. The model accounts for both direct medical costs (associated with consultation, tests, and interventions) and productivity costs in terms of loss of working days. All costs were adjusted to 2014 US dollar values with use of the medical care component of the Bureau of Labor Statistics Consumer Price Index. Health outcomes were measured in quality-adjusted life-years (QALYs), which is the product of health-state utility (with 0 denoting death, and 1 denoting perfect health) and the duration in that state. Costs and QALYs were discounted at 3% as recommended by the Panel on Cost-Effectiveness in Health and Medicine.17
We first calculated the cost per QALY gained for each genetic screening strategy compared with no screening. We then calculated the incremental cost-effectiveness ratio (ICER) of each strategy as compared with the next-less-intensive strategy after ruling out strategies by absolute and extended dominance.18,19 The optimal strategy was defined as the strategy with best outcome among strategies of which ICERs are less than the assumed willingness to pay US $100,000 per QALY.20,21
To account for uncertainty in estimates of parameters, we also performed one-way sensitivity analysis by changing one parameter at a time within relevant plausible ranges. Finally, we performed probabilistic sensitivity analysis by running the model 10,000 times and, within each run, randomly sampling values for all the parameters from their plausible ranges. We assumed beta, gamma, and log-normal distributions, respectively, for probability parameters, cost parameters, and relative risk parameters,22 except for those without sufficient data to estimate distribution parameters. We assigned triangular or uniform distributions to these parameters (Data Supplement). All the analyses were performed with use of TreeAge Pro 2014 (TreeAge Software, Williamstown, MA) and Microsoft Excel 2013 (Microsoft, Redmond, WA) in accordance with published recommendations.17,23
RESULTS
Cost per Mutation Detected
Using a CC score at a threshold of 15 (CC15) to prescreen patients for PTEN-gene testing costs $11,425 to detect one mutation, which was the most inexpensive when compared with no testing at all. (Fig 2, Table S4).
Fig 2.
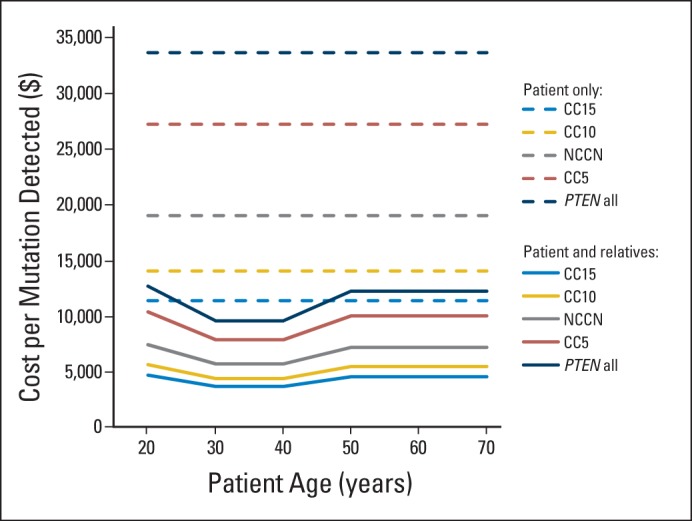
Cost per mutation by patient age, with or without relatives tested. Graph illustrates the cost per mutation detected and patient age in Cowden syndrome (CS) -like patients. Costs per mutation detected decrease as first-degree relatives are included in the testing. CC, PTEN Cleveland Clinic score; CC5, individuals whose CC scores are above the threshold of 5; CC10, individuals whose CC scores are above the threshold of 10; CC15, individuals whose CC scores are above the threshold of 15; NCCN, National Comprehensive Cancer Network guidelines; PTEN all, germline PTEN mutation testing for all enrolled.
When genetic testing was offered for first-degree relatives of PTENmut+ probands, the cost per mutation detected decreased by almost two thirds, even at a compliance rate of 60% to 70%. For example, for CC15, the cost per mutation detected drops from $11,425 to between $3,720 and 4,573 when first-degree relatives are included. Genetic screening costs less per mutation detected for patients who are 30 to 40 years old than it does for patients at the two extremes of the age spectrum. CC15 was the strategy with the lowest cost per mutation detected, irrespective of the patient's age.
Cost-Utility Ratio
Compared with not conducting genetic screening, selective genetic screening with CC15 consistently incurred the lowest cost per QALY gained, ranging from $58,884 to $107,390 per QALY for male probands and from $50,569 to $155,367 per QALY for female probands. The ranking of cost-effectiveness ratio (from lowest to highest) is CC15, CC10, NCCN, CC5, and universal PTEN testing in all age and sex groups (Fig 3; Data Supplement).
Fig 3.

Cost-effectiveness ratio of competing genetic screening strategies by age and sex. Diagram illustrates the cost per quality-adjusted life-year (QALY) gained for each genetic screening strategy in different age and sex groups. The upper panel (A) and lower panel (B) show cost-effectiveness ratios for female and male patients, respectively. The different colors of the bar denote different genetic screening strategies. PTEN Cleveland Clinic score 15 (CC15) has the lowest cost-effectiveness ratios in all age and sex groups. CC5, individuals whose CC scores are above the threshold of 5; CC10, individuals whose CC scores are above the threshold of 10; CC15, individuals whose CC scores are above the threshold of 15; NCCN, National Comprehensive Cancer Network guidelines; PTEN all, germline PTEN mutation testing for all enrolled.
Incremental Cost-Effectiveness Ratio
CC15, CC10, CC5, and universal genetic screening produced greater amounts of QALY improvements among the cohort of patients. NCCN is not included in the calculation of ICER due to its absolute dominance by CC10 among patients younger than 70 years and extended dominance among the group of patients who were 70 years old. (Fig 4; Data Supplement). At a cost-effectiveness threshold of $100,000 per QALY, CC10 is the most effective strategy with a lower ICER for female patients younger than 40 years and CC15 is the most effective strategy with a lower ICER for female patients between 40 and 60 years and male patients of all ages.
Fig 4.
Cost, effectiveness, and incremental cost-effectiveness ratio (ICER) of competing genetic screening strategies by age and sex. This graph demonstrates the cost, quality-adjusted life-years (QALYs) gained, and ICERs of competing genetic screening strategies. Results for female and male patients are presented in the left (A) and right (B) panels, respectively. Patient ages are indicated by different colors. Each data point represents the cost and QALYs gained of a particular screening strategy. Different strategies are indicated by various shapes. Different filling patterns represent ICERs of different ranges. Not conducting PTEN testing is denoted by a solid circle. CC, PTEN Cleveland Clinic score; CC5, offering germline PTEN mutation testing when CC scores are above the threshold of 5 ; CC10, offering germline PTEN mutation testing when CC scores are above the threshold of 10; CC15, offering germline PTEN mutation testing when CC scores are above the threshold of 15; NCCN, offering germline PTEN mutation testing if National Comprehensive Cancer Network criteria are met; PTEN all, germline PTEN mutation testing for all enrolled.
Sensitivity Analysis
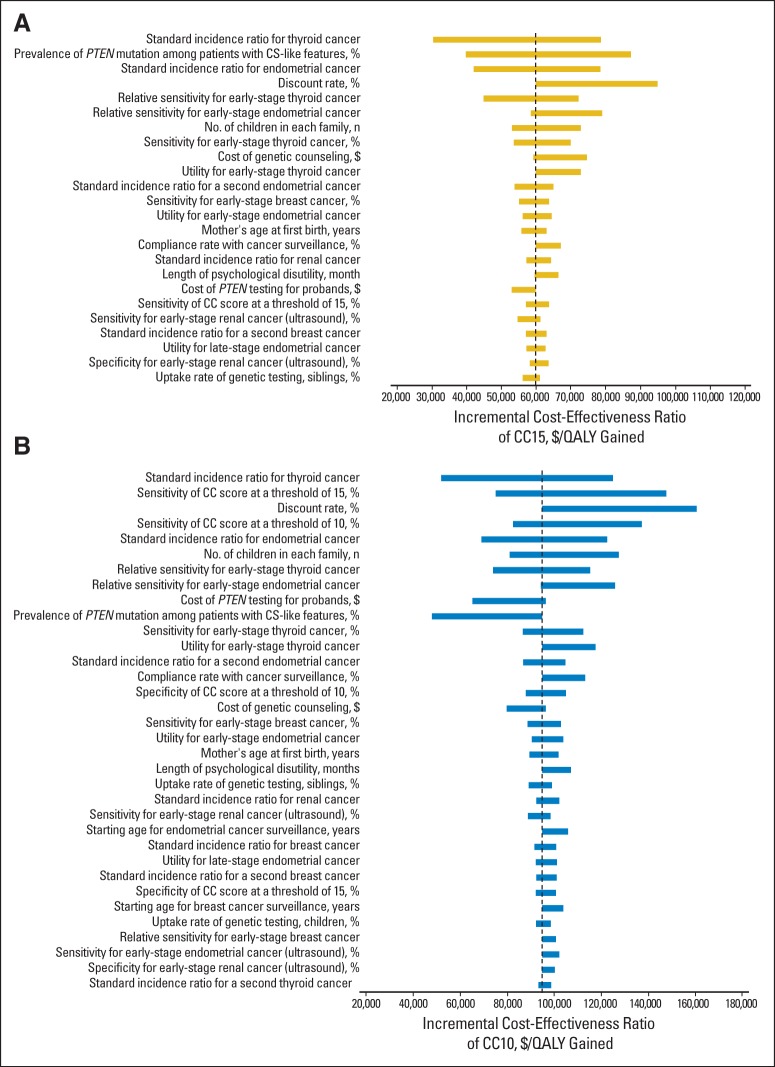
Since CC15 and CC10 were chosen as the optimal strategies based on best estimates of the parameters, we performed one-way sensitivity analysis for the ICER of CC15 compared with no genetic screening and for the ICER of CC10 compared with CC15. Analyses were conducted for all sex and age groups, and it was found that the cost-effectiveness of CC10 and CC15 were sensitive to changes in the effectiveness of cancer surveillance, cancer incidences, and cost of genetic screening. However, even in the analysis of parameters that the model is most sensitive to, the ICERs for CC15 and CC10 hardly exceeded $100,000 per QALY and $150,000 per QALY, respectively. The sensitivity analysis results for probands who were 40 years of age, which is the median age of CS-like patients, were shown in Figure 5, Data Supplement.
Fig 5.
One-way sensitivity analysis in the cohort of 40-year-old female Cowden syndrome (CS) -like patients. Tornado diagrams illustrate the range of permutation in incremental incremental cost-effectiveness ratios (ICERs) of PTEN Cleveland Clinic score (CC) 15 compared with no genetic screening (A) and CC10 compared with CC15 (B) as each parameter varies within its plausible ranges. The results are sorted by the range of permutation in ICERs. The vertical dashed lines represent the ICERs derived with use of the base values of these parameters. The results presented in the figure are for 40-year-old female patients; the other groups have a similar pattern. Only parameters that are associated with permutations of > 5,000 $/QALY are shown in this figure. Full results of one-way sensitivity analyses are documented in the Data Supplement. $/QALY, cost per quality-adjusted life-year.
We tested the robustness of the results further by changing the base-case inputs of some parameters simultaneously in the direction that is against genetic screening. We examined different scenarios, such as reducing the base-case incidences of all four types of cancers by 20% (Data Supplement), increasing the progression rate of detected (and treated) early stage cancer by 1.5 times (Data Supplement) or decreasing the mortality rate of late-stage cancers by half (Data Supplement); even so, we still found that CC15 is consistently the most cost-effective strategy for all probands who were younger than 60 years of age.
Finally, we ran the model 10,000 times and randomly sampled values for all the parameters from their plausible distributions (Data Supplement) within each run. At a cost-effectiveness threshold of $100,000 per QALY, genetic screening with CC15, CC10, and no genetic screening were the optimal strategies in 48.6%, 30.8%, and 18.4% of the iterations, respectively (Fig 6).
Fig 6.

Probabilistic sensitivity analysis, performed by running the model 10,000 times and within each run, randomly sampling values for all the parameters from their plausible ranges. This diagram illustrates the proportion of iterations in which each strategy is the optimal strategy as a function of willingness to pay. At a cost-effectiveness threshold of $100,000 per quality-adjusted life-year (QALY), offering germline PTEN mutation CC15, CC10, and no genetic screening were the optimal strategies in 48.6%, 30.8 and 18.4% of the iterations, respectively. At a cost-effectiveness threshold of $150,000 per QALY, CC10, CC15, CC5, and no genetic screening were the optimal strategies in 53.2%, 34.1%, 9.5%, and 2.2% of the iterations, respectively. CC5, offering germline PTEN mutation testing when Cleveland Clinic (CC) scores are above the threshold of 5 ; CC10, offering germline PTEN mutation testing when CC scores are above the threshold of 10; CC15, offering germline PTEN mutation testing when CC scores are above the threshold of 15; NCCN, offering germline PTEN mutation testing if National Comprehensive Cancer Network criteria are met; PTEN all, germline PTEN mutation testing for all enrolled.
DISCUSSION
This study provides the first comprehensive cost-effectiveness analysis of multiple strategies to test for germline PTEN mutations among patients who present with CS-like features in the United States, and may serve as a useful model for many inherited cancer syndromes. Notably, we have shown that sex as well as age of the proband impacts overall cost-effectiveness, given the sex- and age-specific differences in terms of cancer risk for PTENmut+ patients.
Our results suggest that the systematic application of strategies to detect germline PTEN mutations among individuals suspected of having CS could provide substantial clinical benefits at acceptable costs. We had previously recommended that clinicians consider germline PTEN testing for individuals who had a CC score threshold of 10, as that corresponded to a 3% clinically significant risk of detecting a germline PTEN mutation.3 Our results confirmed that the use of clinical risk calculators such as the CC score based purely on clinical history is a cost-effective prescreening method in the selection of patients for PTEN germline mutation when CS is suspected. Here, we demonstrate that CC15, which corresponds to a 10% a priori risk of germline PTEN mutation, has the lowest cost per PTEN mutation carrier detected among all the strategies across sex and age-at-presentation. Our baseline estimates of family size (ie, between five and six first-degree relatives per proband undergoing predictive testing) is comparable, if not conservative, to what happens in clinical practice.24,25 Despite this, by taking into account predictive testing in first-degree relatives, all strategies were more cost-effective, decreasing the cost per mutation detected by more than two-thirds. This result is consistent with the literature on Lynch syndrome, in which it has been reported that the cost-effectiveness of testing also is strongly dependent on the participation rate among relatives who are at risk of the development of Lynch syndrome.26
When we examined the lifetime cost-effectiveness of competing genetic screening strategies, for patients of both sexes, ICERs of CC15 decrease and then increase as patient age increases from 20 to 70 years, which reflects changes in the ages and sexes of relatives who are eligible for genetic screening and increases in competing risks of death from noncancer causes with increasing age. At a cost-effectiveness threshold of $100,000 per QALY, CC15 is cost-effective for female probands who are younger than 60 years and male probands of all ages. For female probands younger than 40 years, CC10, which will benefit more patients than CC15, is also cost-effective. These results are comparable to ICERs for clinical preventive services in practice, which range from negative to more than $200,000 per QALY.27,28
Our model was sensitive to the cost of genetic counseling and the cost of genetic testing in the proband. Although the latter has a much wider range of variation than the former, altering the former had more influence on cost-effectiveness because every CS-like patient receives genetic counseling while only a subgroup of these patients who have positive clinical scores go on to do genetic screening. Intuitively, genetic screening becomes more cost-effective as prevalence of PTEN carriers among CS-like patients increases, as reflected by our current data. The results of our probability sensitivity analysis confirm that the CC score is robustly cost-effective at a cost-effectiveness threshold of $100,000 per QALY. Our data suggest that altering the starting age for cancer surveillance for individual CS-related cancers affects overall cost-effectiveness and could be used to guide discussions on how best to balance the cost and benefit of screening programs.
This analysis was based on generally conservative assumptions and some simplification. First, although we assumed that single-site testing was only offered to first-degree relatives of probands, in clinical practice, efforts are commonly made to offer single-site tests to second-degree relatives as well. Based on the comparison of screening patients alone and screening patients along with relatives, we would anticipate improved cost-effectiveness if second-degree relatives are included. Second, as is the case with all modeling exercises, we made a number of assumptions in our model. However, while most of our assumptions introduce bias in the direction against genetic screening (Data Supplement), our results indicate that genetic screening with a CC score of 15 remains cost-effective for most groups of patients, which adds to our confidence in our results. Third, we did not explicitly account for psychological impact of false-positive surveillance tests. However, in the field itself, there is an absence of high-quality data.29 Existing evidence is mixed and the methodologies are not consistent,30–34 making it challenging to distill the “best evidence” to include in our calculation. Finally, we have included in this study current cancer surveillance recommendations for patients with germline PTEN mutations, but we did not account for the health utility implications of neurocognitive35–37 and other nonmalignant38 implications. However, it can be postulated that finding a germline PTEN mutation early can also lead to prevention or mitigation of neurocognitive deficits and diabetes. If the latter can be achieved, then we may assume that competing risks of noncancer morbidity and mortality would be decreased, allowing for cancer surveillance across all ages to become cost-effective.
Future developments in technology could influence the strategy of choice and almost certainly would affect cost-effectiveness. Increasingly, patients at risk for a diverse range of diseases can be offered multigene disease-predisposition testing, likely reducing the costs of genetic testing and making our strategy of choice more cost-effective. In conclusion, use of the CC score as a clinical risk calculator is an accurate and cost-effective prescreening method in the selection of patients who are candidates for PTEN germline analysis when CS is suspected.
Glossary Terms
- cost-effectiveness analysis:
an economic evaluation in which the costs and consequences of alternative interventions are expressed as a cost per unit of health outcome. Cost-effectiveness analysis is used to determine technical efficiency (ie, comparison of costs and consequences of competing interventions for a given patient group within a given budget).
- PTEN (phophatase and tensin homolog):
a tumor suppressor gene with a gamut of regulatory activities. The gene product is a multifunctional molecule. The predominant activity identified for PTEN is its lipid phosphatase activity that converts inositol trisphosphates into inositol bisphosphates, thus inhibiting survival and proliferative pathways that are activated by inositol trisphosphates. PTEN acts to maintain arrest in the G1 phase of the cell cycle and enable apoptosis through an AKT-dependent mechanism.
Footnotes
Funded, in part, by National Cancer Institute Grant No. P01CA124570 and the Breast Cancer Research Foundation (both to C.E.). J.N. was the Ambrose Monell Foundation Cancer Genomic Medicine Fellow at the Genomic Medicine Institute, Cleveland Clinic, and is a Transition Award recipient of the Singapore Ministry of Health's National Medical Research Council. C.E. is the Sondra J. and Stephen R. Hardis Chair of Cancer Genomic Medicine at the Cleveland Clinic and is an American Cancer Society Clinical Research Professor, generously funded in part, by the F.M. Kirby Foundation. D.M. is a STaR Investigator Awardee of the Singapore Ministry of Health's National Medical Research Council and the Agency for Science, Technology and Research.
Terms in blue are defined in the glossary, found at the end of this article and online at www.jco.org.
Authors' disclosures of potential conflicts of interest are found in the article online at www.jco.org. Author contributions are found at the end of this article.
AUTHORS' DISCLOSURES OF POTENTIAL CONFLICTS OF INTEREST
Disclosures provided by the authors are available with this article at www.jco.org.
AUTHOR CONTRIBUTIONS
Conception and design: Joanne Ngeow, Chang Liu, Charis Eng
Financial support: Charis Eng
Administrative support: Charis Eng
Provision of study materials or patients: Charis Eng
Collection and assembly of data: Joanne Ngeow, Chang Liu, Ke Zhou
Data analysis and interpretation: All authors
Manuscript writing: All authors
Final approval of manuscript: All authors
AUTHORS' DISCLOSURES OF POTENTIAL CONFLICTS OF INTEREST
Detecting Germline PTEN Mutations Among At-Risk Patients With Cancer: An Age- and Sex-Specific Cost-Effectiveness Analysis
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to www.asco.org/rwc or jco.ascopubs.org/site/ifc.
Joanne Ngeow
Consulting or Advisory Role: Astra Zeneca
Chang Liu
No relationship to disclose
Ke Zhou
No relationship to disclose
Kevin D. Frick
No relationship to disclose
David B. Matchar
No relationship to disclose
Charis Eng
No relationship to disclose
REFERENCES
- 1.US Department of Health and Human Services. Washington, DC: Office of Disease Prevention and Health Promotion; Healthy People 2020. www.healthypeople.gov/2020/topics-objectives/topic/genomics. [Google Scholar]
- 2.American Cancer Society. Cancer Facts & Figures 2014. http://www.cancer.org/Research/CancerFactsStatistics/CancerFactsFigures2014/cancer-facts-and-figures-2014.pdf.
- 3.Tan MH, Mester J, Peterson C, et al. A clinical scoring system for selection of patients for PTEN mutation testing is proposed on the basis of a prospective study of 3042 probands. Am J Hum Genet. 2011;88:42–56. doi: 10.1016/j.ajhg.2010.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ngeow J, Mester J, Rybicki LA, et al. Incidence and clinical characteristics of thyroid cancer in prospective series of individuals with Cowden and Cowden-like syndrome characterized by germline PTEN, SDH, or KLLN alterations. J Clin Endocrinol Metab. 2011;96:E2063–E2071. doi: 10.1210/jc.2011-1616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tan MH, Mester JL, Ngeow J, et al. Lifetime cancer risks in individuals with germline PTEN mutations. Clin Cancer Res. 2012;18:400–407. doi: 10.1158/1078-0432.CCR-11-2283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ngeow J, Stanuch K, Mester JL, et al. Second malignant neoplasms in patients with Cowden syndrome with underlying germline PTEN mutations. J Clin Oncol. 2014;32:1818–1824. doi: 10.1200/JCO.2013.53.6656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Eng C. Will the real Cowden syndrome please stand up: Revised diagnostic criteria. J Med Genet. 2000;37:828–830. doi: 10.1136/jmg.37.11.828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Foca F, Mancini S, Bucchi L, et al. Decreasing incidence of late-stage breast cancer after the introduction of organized mammography screening in Italy. Cancer. 2013;119:2022–2028. doi: 10.1002/cncr.28014. [DOI] [PubMed] [Google Scholar]
- 9.Chandra A, Martinez GM, Mosher WD, et al. Fertility, family planning, and reproductive health of US women: Data from the 2002 National Survey of Family Growth. Vital Health Stat. 2005;23:40. [PubMed] [Google Scholar]
- 10.Mathews T, Hamilton BE. Mean age of mother, 1970-2000. Natl Vital Stat Rep. 2002;51:1–14. [PubMed] [Google Scholar]
- 11.Dinh TA, Rosner BI, Atwood JC, et al. Health benefits and cost-effectiveness of primary genetic screening for Lynch syndrome in the general population. Cancer Prevention Research. 2011;4:9–22. doi: 10.1158/1940-6207.CAPR-10-0262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang H, Kåresen R, Hervik A, et al. Mammography screening in Norway: Results from the first screening round in four counties and cost-effectiveness of a modeled nationwide screening. Cancer Causes Control. 2001;12:39–45. doi: 10.1023/a:1008999403069. [DOI] [PubMed] [Google Scholar]
- 13.Arveux P, Wait S, Schaffer P. Building a model to determine the cost-effectiveness of breast cancer screening in France. Eur J Cancer Care. 2003;12:143–153. doi: 10.1046/j.1365-2354.2003.00373.x. [DOI] [PubMed] [Google Scholar]
- 14.Okonkwo QL, Draisma G, der Kinderen A, et al. Breast cancer screening policies in developing countries: A cost-effectiveness analysis for India. J Natl Cancer Inst. 2008;100:1290–1300. doi: 10.1093/jnci/djn292. [DOI] [PubMed] [Google Scholar]
- 15.de Gelder R, Bulliard JL, de Wolf C, et al. Cost-effectiveness of opportunistic versus organised mammography screening in Switzerland. Eur J Cancer. 2009;45:127–138. doi: 10.1016/j.ejca.2008.09.015. [DOI] [PubMed] [Google Scholar]
- 16.Woo PP, Kim JJ, Leung GM. What is the most cost-effective population-based cancer screening program for Chinese women? J Clin Oncol. 2007;25:617–624. doi: 10.1200/JCO.2006.06.0210. [DOI] [PubMed] [Google Scholar]
- 17.Siegel JE, Weinstein MC, Russell LB, et al. Recommendations for reporting cost-effectiveness analyses. Panel on Cost-Effectiveness in Health and Medicine. JAMA. 1996;276:1339–1341. doi: 10.1001/jama.276.16.1339. [DOI] [PubMed] [Google Scholar]
- 18.Torrance GW, Siegel JE, Luce BR. Framing and designing the cost-effectiveness analysis. In: Gold MR, Siegel JE, Russell LB, et al., editors. Cost-Effectiveness in Health and Medicine. New York, NY: Oxford University Press; 1996. p. 65. [Google Scholar]
- 19.Siegel JE, Weinstein MC, Torrance GW. Reporting cost-effectiveness studies and results. In: Gold MR, Siegel JE, Russell LB, et al., editors. Cost-Effectiveness in Health and Medicine. New York, NY: Oxford University Press; 1996. pp. 285–287. [Google Scholar]
- 20.van Hees F, Habbema JD, Meester RG, et al. Should colorectal cancer screening be considered in elderly persons without previous screening? A cost-effectiveness analysis. Ann Intern Med. 2014;160:750–759. doi: 10.7326/M13-2263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Neumann PJ, Cohen JT, Weinstein MC. Updating cost-effectiveness—the curious resilience of the $50,000-per-QALY threshold. N Engl J Med. 2014;371:796–797. doi: 10.1056/NEJMp1405158. [DOI] [PubMed] [Google Scholar]
- 22.Briggs AH, Goeree R, Blackhouse G, et al. Probabilistic analysis of cost-effectiveness models: Choosing between treatment strategies for gastroesophageal reflux disease. Med Decis Making. 2002;22:290–308. doi: 10.1177/0272989X0202200408. [DOI] [PubMed] [Google Scholar]
- 23.Weinstein MC, Siegel JE, Gold MR, et al. Recommendations of the Panel on Cost-effectiveness in Health and Medicine. JAMA. 1996;276:1253–1258. [PubMed] [Google Scholar]
- 24.de la Chapelle A, Palomaki G, Hampel H. Identifying Lynch syndrome. Int J Cancer. 2009;125:1492–1493. doi: 10.1002/ijc.24491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Umans-Eckenhausen MA, Defesche JC, Sijbrands EJ, et al. Review of first 5 years of screening for familial hypercholesterolaemia in the Netherlands. Lancet. 2001;357:165–168. doi: 10.1016/S0140-6736(00)03587-X. [DOI] [PubMed] [Google Scholar]
- 26.Ladabaum U, Wang G, Terdiman J, et al. Strategies to identify the Lynch syndrome among patients with colorectal cancer: A cost-effectiveness analysis. Ann Intern Med. 2011;155:69–79. doi: 10.7326/0003-4819-155-2-201107190-00002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Maciosek MV, Coffield AB, Edwards NM, et al. Priorities among effective clinical preventive services: Results of a systematic review and analysis. Am J Prev Med. 2006;31:52–61. doi: 10.1016/j.amepre.2006.03.012. [DOI] [PubMed] [Google Scholar]
- 28.Grosse SD, Teutsch SM, Haddix AC. Lessons from cost-effectiveness research for United States public health policy. Annu Rev Public Health. 2007;28:365–391. doi: 10.1146/annurev.publhealth.28.021406.144046. [DOI] [PubMed] [Google Scholar]
- 29.Bond M, Pavey T, Welch K, et al. Systematic review of the psychological consequences of false-positive screening mammograms. Health Technol Assess. 2013;17:1–170. v–vi. doi: 10.3310/hta17130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bull AR, Cambell MJ. Assessment of the physiological impact of a breast screening programme. Br J Radiol. 1991;64:510–515. doi: 10.1259/0007-1285-64-762-510. [DOI] [PubMed] [Google Scholar]
- 31.Ellman R, Angeli N, Christians A, et al. Psychiatric morbidity associated with screening for breast cancer. Br J Cancer. 1989;60:781–784. doi: 10.1038/bjc.1989.359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Ong G, Austoker J, Brett J. Breast screening: Adverse psychological consequences one month after placing women on early recall because of a diagnostic uncertainty. A multicentre study. J Med Screen. 1997;4:158–168. doi: 10.1177/096914139700400309. [DOI] [PubMed] [Google Scholar]
- 33.Ekeberg Ø, Skjauff H, Kåresen R. Screening for breast cancer is associated with a low degree of psychological distress. The Breast. 2001;10:20–24. doi: 10.1054/brst.2000.0177. [DOI] [PubMed] [Google Scholar]
- 34.Gram IT, Lund E, Slenker SE. Quality of life following a false positive mammogram. Br J Cancer. 1990;62:1018–1022. doi: 10.1038/bjc.1990.430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Hobert JA, Embacher R, Mester JL, et al. Biochemical screening and PTEN mutation analysis in individuals with autism spectrum disorders and macrocephaly. Eur J Hum Genet. 2014;22:273–276. doi: 10.1038/ejhg.2013.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Butler MG, Dasouki MJ, Zhou XP, et al. Subset of individuals with autism spectrum disorders and extreme macrocephaly associated with germline PTEN tumour suppressor gene mutations. J Med Genet. 2005;42:318–321. doi: 10.1136/jmg.2004.024646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Mester JL, Tilot AK, Rybicki LA, et al. Analysis of prevalence and degree of macrocephaly in patients with germline PTEN mutations and of brain weight in Pten knock-in murine model. Eur J Hum Genet. 2011;19:763–768. doi: 10.1038/ejhg.2011.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Pal A, Barber TM, Van de Bunt M, et al. PTEN mutations as a cause of constitutive insulin sensitivity and obesity. N Engl J Med. 2012;367:1002–1011. doi: 10.1056/NEJMoa1113966. [DOI] [PMC free article] [PubMed] [Google Scholar]