Abstract
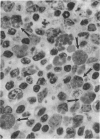
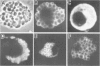
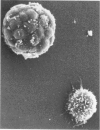
Mice homozygous for the autosomal recessive mutation "viable motheaten" (mev) are severely immunodeficient, show polyclonal B-cell activation, and express multiple autoantibodies over a maximum life span of 25 weeks. Lymphoid tissues from these mice contain large numbers of atypical plasma cells in which discrete glycoprotein inclusions are found within the endoplasmic reticulum. Such plasma cells are termed "Mott cells," and the inclusions are called "Russell bodies." Dense accumulations of Mott cells are present in the marginal zones of the spleen and in the lymph nodes of mev/mev mice. Russell bodies in Mott cells from mev/mev mice contain immunoglobulin (Ig) as shown by immunofluorescence microscopy at the light-microscopic level and by indirect protein A-immunogold localization at the electron-microscopic level. Ultrastructural analyses reveal the presence of amorphous, lamellar, and crystalline Russell bodies. These Ig crystals have a periodicity of 150-190 A. Lymph node cell preparations which were enriched in Mott cells by velocity sedimentation failed to secrete Ig in a polyclonal reverse plaque assay. An obligate role of the thymus in Mott cell development is evidenced by the absence of Mott cells in neonatally thymectomized mev/mev mice and in mice doubly homozygous for the nude (nu) and mev mutations. These data suggest that Mott cells in mev/mev mice are thymic-dependent plasmacytoid cells resulting from chronic B-cell activation accompanied by impaired Ig secretion.
Full text
PDF












Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Alanen A., Pira U., Lassila O., Roth J., Franklin R. M. Mott cells are plasma cells defective in immunoglobulin secretion. Eur J Immunol. 1985 Mar;15(3):235–242. doi: 10.1002/eji.1830150306. [DOI] [PubMed] [Google Scholar]
- Bendayan M., Zollinger M. Ultrastructural localization of antigenic sites on osmium-fixed tissues applying the protein A-gold technique. J Histochem Cytochem. 1983 Jan;31(1):101–109. doi: 10.1177/31.1.6187796. [DOI] [PubMed] [Google Scholar]
- Blom J., Mansa B., Wilk A. A study of Russell bodies in human monoclonal plasma cells by means of immunofluorescence and electron microscopy. Acta Pathol Microbiol Scand A. 1976 Jul;84(4):335–349. doi: 10.1111/j.1699-0463.1976.tb00124.x. [DOI] [PubMed] [Google Scholar]
- Cawley J. C., Smith J., Goldstone A. H., Emmines J., Hamblin J., Hough L. IgA and IgM cytoplastic inclusions in a series of cases of chronic lymphocytic leukaemia. Clin Exp Immunol. 1976 Jan;23(1):78–82. [PMC free article] [PubMed] [Google Scholar]
- Clark E. A., Shultz L. D., Pollack S. B. Mutations in mice that influence natural killer (NK) cell activity. Immunogenetics. 1981 Mar 1;12(5-6):601–613. doi: 10.1007/BF01561700. [DOI] [PubMed] [Google Scholar]
- Cooper M. D., Kearney J. F., Gathings W. E., Lawton A. R. Effects of anti-Ig antibodies on the development and differentiation of B cells. Immunol Rev. 1980;52:29–53. doi: 10.1111/j.1600-065x.1980.tb00329.x. [DOI] [PubMed] [Google Scholar]
- Davidson W. F., Morse H. C., 3rd, Sharrow S. O., Chused T. M. Phenotypic and functional effects of the motheaten gene on murine B and T lymphocytes. J Immunol. 1979 Mar;122(3):884–891. [PubMed] [Google Scholar]
- East J., de Sousa M. A., Parrott D. M. Immunopathology of New Zealand black (NZB) mice. Transplantation. 1965 Nov;3(6):711–729. doi: 10.1097/00007890-196511000-00003. [DOI] [PubMed] [Google Scholar]
- Gebbers J. O., Otto H. F. Plasma cell alterations in ulcerative colitis. An electron microscopic study. Pathol Eur. 1976;11(4):271–279. [PubMed] [Google Scholar]
- Gordon J. Molecular aspects of immunoglobulin expression by human B cell leukemias and lymphomas. Adv Cancer Res. 1984;41:71–154. doi: 10.1016/s0065-230x(08)60015-9. [DOI] [PubMed] [Google Scholar]
- Green M. C., Shultz L. D. Motheaten, an immunodeficient mutant of the mouse. I. Genetics and pathology. J Hered. 1975 Sep-Oct;66(5):250–258. doi: 10.1093/oxfordjournals.jhered.a108625. [DOI] [PubMed] [Google Scholar]
- Griffith A. L., Catsimpoolas N., Dewanjee M. K., Wortis H. H. Electrophoretic separation of radioisotopically labeled mouse lymphocytes. J Immunol. 1976 Nov;117(5 PT2):1949–1952. [PubMed] [Google Scholar]
- Gronowicz E., Coutinho A., Melchers F. A plaque assay for all cells secreting Ig of a given type or class. Eur J Immunol. 1976 Aug;6(8):588–590. doi: 10.1002/eji.1830060812. [DOI] [PubMed] [Google Scholar]
- Hayzer D. J., Jaton J. C. Immunoglobulin M (IgM). Methods Enzymol. 1985;116:26–36. doi: 10.1016/s0076-6879(85)16004-0. [DOI] [PubMed] [Google Scholar]
- Hearn S. A., Silver M. M., Sholdice J. A. Immunoelectron microscopic labeling of immunoglobulin in plasma cells after osmium fixation and epoxy embedding. J Histochem Cytochem. 1985 Dec;33(12):1212–1218. doi: 10.1177/33.12.3934258. [DOI] [PubMed] [Google Scholar]
- Hsu S. M., Hsu P. L., McMillan P. N., Fanger H. Russell bodies: a light and electron microscopic immunoperoxidase study. Am J Clin Pathol. 1982 Jan;77(1):26–31. doi: 10.1093/ajcp/77.1.26. [DOI] [PubMed] [Google Scholar]
- Hsu S. M. Phenotypic expression of B lymphocytes. III. Marginal zone B cells in the spleen are characterized by the expression of Tac and alkaline phosphatase. J Immunol. 1985 Jul;135(1):123–130. [PubMed] [Google Scholar]
- Humphrey J. H., Grennan D. Different macrophage populations distinguished by means of fluorescent polysaccharides. Recognition and properties of marginal-zone macrophages. Eur J Immunol. 1981 Mar;11(3):221–228. doi: 10.1002/eji.1830110311. [DOI] [PubMed] [Google Scholar]
- Kumararatne D. S., Bazin H., MacLennan I. C. Marginal zones: the major B cell compartment of rat spleens. Eur J Immunol. 1981 Nov;11(11):858–864. doi: 10.1002/eji.1830111103. [DOI] [PubMed] [Google Scholar]
- Lawton A. R., 3rd, Asofsky R., Hylton M. B., Cooper M. D. Suppression of immunoglobulin class synthesis in mice. I. Effects of treatment with antibody to -chain. J Exp Med. 1972 Feb 1;135(2):277–297. doi: 10.1084/jem.135.2.277. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manning D. D., Jutila J. W. Immunosuppression of mice injected with heterologous anti-immunoglobulin heavy chain antisera. J Exp Med. 1972 Jun 1;135(6):1316–1333. doi: 10.1084/jem.135.6.1316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matthews J. B. The immunoglobulin nature of Russell bodies. Br J Exp Pathol. 1983 Jun;64(3):331–335. [PMC free article] [PubMed] [Google Scholar]
- Miller R. G., Phillips R. A. Separation of cells by velocity sedimentation. J Cell Physiol. 1969 Jun;73(3):191–201. doi: 10.1002/jcp.1040730305. [DOI] [PubMed] [Google Scholar]
- Mond J. J., Scher I., Cossman J., Kessler S., Mongini P. K., Hansen C., Finkelman F. D., Paul W. E. Role of the thymus in directing the development of a subset of B lymphocytes. J Exp Med. 1982 Mar 1;155(3):924–936. doi: 10.1084/jem.155.3.924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ORTEGA L. G., MELLORS R. C. Cellular sites of formation of gamma globulin. J Exp Med. 1957 Nov 1;106(5):627–640. doi: 10.1084/jem.106.5.627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ralph P., Jeong G., Welte K., Mertelsmann R., Rabin H., Henderson L. E., Souza L. M., Boone T. C., Robb R. J. Stimulation of immunoglobulin secretion in human B lymphocytes as a direct effect of high concentrations of IL 2. J Immunol. 1984 Nov;133(5):2442–2445. [PubMed] [Google Scholar]
- SJODIN K., DALMASSO A. P., SMITH J. M., MARTINEZ C. THYMECTOMY IN NEWBORN AND ADULT MICE. Transplantation. 1963 Oct;1:521–525. doi: 10.1097/00007890-196301040-00011. [DOI] [PubMed] [Google Scholar]
- Shultz L. D., Coman D. R., Bailey C. L., Beamer W. G., Sidman C. L. "Viable motheaten," a new allele at the motheaten locus. I. Pathology. Am J Pathol. 1984 Aug;116(2):179–192. [PMC free article] [PubMed] [Google Scholar]
- Shultz L. D., Green M. C. Motheaten, an immunodeficient mutant of the mouse. II. Depressed immune competence and elevated serum immunoglobulins. J Immunol. 1976 Apr;116(4):936–943. [PubMed] [Google Scholar]
- Sidman C. L., Marshall J. D., Masiello N. C., Roths J. B., Shultz L. D. Novel B-cell maturation factor from spontaneously autoimmune viable motheaten mice. Proc Natl Acad Sci U S A. 1984 Nov;81(22):7199–7202. doi: 10.1073/pnas.81.22.7199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sidman C. L., Shultz L. D., Evans R. A serum-derived molecule from autoimmune viable motheaten mice potentiates the action of a B cell maturation factor. J Immunol. 1985 Aug;135(2):870–872. [PubMed] [Google Scholar]
- Sidman C. L., Shultz L. D., Hardy R. R., Hayakawa K., Herzenberg L. A. Production of immunoglobulin isotypes by Ly-1+ B cells in viable motheaten and normal mice. Science. 1986 Jun 13;232(4756):1423–1425. doi: 10.1126/science.3487115. [DOI] [PubMed] [Google Scholar]
- Sidman C. L., Shultz L. D., Unanue E. R. The mouse mutant "motheaten." II. Functional studies of the immune system. J Immunol. 1978 Dec;121(6):2399–2404. [PubMed] [Google Scholar]
- Theofilopoulos A. N., Dixon F. J. Murine models of systemic lupus erythematosus. Adv Immunol. 1985;37:269–390. doi: 10.1016/s0065-2776(08)60342-9. [DOI] [PubMed] [Google Scholar]
- WHITE R. G. Observations on the formation and nature of Russell bodies. Br J Exp Pathol. 1954 Aug;35(4):365–376. [PMC free article] [PubMed] [Google Scholar]
- Waldmann T. A., Goldman C. K., Robb R. J., Depper J. M., Leonard W. J., Sharrow S. O., Bongiovanni K. F., Korsmeyer S. J., Greene W. C. Expression of interleukin 2 receptors on activated human B cells. J Exp Med. 1984 Nov 1;160(5):1450–1466. doi: 10.1084/jem.160.5.1450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss S., Burrows P. D., Meyer J., Wabl M. R. A Mott cell hybridoma. Eur J Immunol. 1984 Aug;14(8):744–748. doi: 10.1002/eji.1830140814. [DOI] [PubMed] [Google Scholar]
- Wortis H. H., Burkly L., Hughes D., Roschelle S., Waneck G. Lack of mature B cells in nude mice with X-linked immune deficiency. J Exp Med. 1982 Mar 1;155(3):903–913. doi: 10.1084/jem.155.3.903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ZLOTNICK A., GERICHTER C. B., NIR I. Experimental production of grape cells and their relation to the serum gamma globulin and seromucoids. Blood. 1959 May;14(5):564–570. [PubMed] [Google Scholar]
- van Vliet E., Melis M., van Ewijk W. Marginal zone macrophages in the mouse spleen identified by a monoclonal antibody. Anatomical correlation with a B cell subpopulation. J Histochem Cytochem. 1985 Jan;33(1):40–44. doi: 10.1177/33.1.3880783. [DOI] [PubMed] [Google Scholar]