Abstract
Biological resilience, broadly defined as the ability to recover from an acute challenge and return to homeostasis, is of growing importance to the biology of aging. At the cellular level, there is variability across tissue types in resilience and these differences are likely to contribute to tissue aging rate disparities. However, there are challenges in addressing these cell-type differences at regional, tissue, and subject level. To address this question, we established primary cells from aged male and female baboons between 13.3 and 17.8 years spanning across different tissues, tissue regions, and cell types including (1) fibroblasts from skin and from the heart separated into the left ventricle (LV), right ventricle (RV), left atrium (LA), and right atrium (RA); (2) astrocytes from the prefrontal cortex and hippocampus; and (3) hepatocytes. Primary cells were characterized by their cell surface markers and their cellular respiration was assessed with Seahorse XFe96. Cellular resilience was assessed by modifying a live-cell imaging approach; we previously reported that monitors proliferation of dividing cells following response and recovery to oxidative (50 µM-H2O2), metabolic (1 mM-glucose), and proteostasis (0.1 µM-thapsigargin) stress. We noted significant differences even among similar cell types that are dependent on tissue source and the diversity in cellular response is stressor-specific. For example, astrocytes had a higher oxygen consumption rate and exhibited greater resilience to oxidative stress (OS) than both fibroblasts and hepatocytes. RV and RA fibroblasts were less resilient to OS compared with LV and LA, respectively. Skin fibroblasts were less impacted by proteostasis stress compared to astrocytes and cardiac fibroblasts. Future studies will test the functional relationship of these outcomes to the age and developmental status of donors as potential predictive markers.
Supplementary Information
The online version contains supplementary material available at 10.1007/s11357-024-01155-7.
Keywords: Baboons, Resilience, Bioenergetics, Astrocytes, Fibroblasts, Hepatocytes
Introduction
Healthy cells undergo functional decline over time, with variations in the extent or timing of these changes attributed to their distinct morphological and physiological properties [1, 2]. Similarly, tissues and organs age at different rates and this cellular diversity is likely to contribute to these outcomes [3, 4]. In response to cellular perturbations, the homeostatic network orchestrates a coordinated dynamic response to restore stability. Cellular resilience measures the ability of a cell to recover to homeostasis following a challenge and can be used as the functional readout of the homeostatic process. Central to the maintenance of cellular homeostasis is the pivotal role played by mitochondria, which provide the energy buffer required for appropriate stress responses and recovery through oxidative phosphorylation. A stabilized ATP supply enhances survival from stress, as well as the repair of damaged molecules to restore homeostasis [5]. Additionally, enhanced mitochondrial antioxidant capacity increases resilience to metabolic stress [6]. While energy demand and antioxidant defense capacity vary across different cell types, limited information exists concerning cellular resilience to stressors in different cell types.
Cell heterogeneity also increases with aging [7]. However, it is unclear whether these changes modify resilience or drive different resilience patterns across distinct cell types in an individual donor. That is, are cellular resilience properties conserved across the organ systems? This is important because understanding cell-specificity in homeostatic response may provide insights into the basis of differences in tissue and organ aging rates. One study demonstrated that neonatal rat primary cardiomyocytes are more vulnerable to oxidative damage than cardiac fibroblasts, exhibiting marked DNA fragmentation and reduced cell survival following exposure to hydrogen peroxide [8]. Leveraging an ongoing aging study in a clinically relevant nonhuman primate (NHP) model [9, 10], we here developed primary cell lines from aging baboons to address potential cellular resilience differences across multiple cell types from key organs of interest such as the liver (hepatocytes), brain (astrocytes), and heart (cardiac fibroblasts).
We prioritized hepatocytes for their primary role in liver metabolic function and selected two supporting cell types crucial for neuronal and cardiac health—astrocytes and cardiac fibroblasts—due to their ease of dispersion from tissues, in vitro proliferative capacity, long-term survival in culture, and relevance to our ongoing studies in the brain and cardiac aging, respectively [11, 12]. Astrocytes are glial cells that protect neurons against oxidative stress, regulate transmission of electrical signals between neurons, and receive damaged mitochondria from neurons for disposal and recycling [13, 14]. Fibroblasts provide structural support to the heart through extracellular matrix (ECM) formation and signal transduction. Cardiac fibroblasts also contribute to the electrical conductivity and rhythm of the heart [15, 16]. They sense and respond to mechanical stress by initiating tissue remodeling and repair [17]. Given the heterogenous nature of these cells, the intrinsic reserve that supports their specialized functions may modulate their resilience to stressors.
Assessing cellular resilience requires a robust approach that enables dynamic monitoring of cell responses to stressors. In an in vitro setting, markers that are intrinsic to cell physiology such as cell proliferation and viability can serve as a valuable tool for resilience measurement. Cell proliferation is associated with the metabolic program of a cell and can be a metabolic marker, given the interaction between cell cycle effectors and metabolic intermediates [18]. When cells are perturbed by an acute challenge, they enter a quiescent state or diminish their proliferative capacity, directing their activities towards cellular defense and repair. We propose that a compensatory restoration of cell proliferation following stress-induced cytostasis is representative of cellular resilience. We previously described a high throughput real-time, live-cell imaging approach that measures cellular proliferation response to acute stressors in proliferative cells [9].
In the present study, we assessed and compared the pattern of resilience across cell types in distinct organ regions and similar cell types within different anatomical locations in baboons. First, we derived and characterized resident cells from multiple regions of the heart, brain, and liver of adult male and female baboons. We assessed mitochondrial response to its electron transport chain modulators using a mitochondrial stress test and assessment of bioenergetic function. We also incorporated a secondary test of resilience to the mitochondrial stress test by exposing cells to low glucose to induce metabolic stress prior to the mitochondrial stress test. We also determined cellular proliferation response following short-term exposure to oxidative, proteostasis, and metabolic stress in the different cell types under the same stress intensity. This study lays the foundation for future investigations that will address the impact of age, sex, and developmental events on cellular resilience as well as other mechanistic studies within our baboon cohort.
Methods
Animals
All procedures involving animals were approved by the Institutional Animal Care and Use Committee of Texas Biomedical Research Institute (TX Biomed). The animal facilities at the Southwest National Primate Research Center (SNPRC), located within TX Biomed, are fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC), which adheres to the guidelines set forth by the National Institutes of Health (NIH) and the US Department of Agriculture.
Baboons (Papio species) were housed and maintained in outdoor social groups and fed ad libitum with normal monkey diet (Purina Monkey Diet 5LEO; 2.98 kcal/g). The welfare of the animals was enhanced by providing enrichment, such as toys, food treats, and music, which were offered daily under the supervision of the veterinary and behavioral staff at SNPRC. All baboons received veterinarian physical examinations at least twice per year and were healthy at the time of study.
The baboons were part of a cohort of animals studied throughout the life course to identify and establish effects of physiological aging [10–12]. All sample collections for primary cell cultures were taken opportunistically at prescribed euthanasia for animals in this larger study.
Sample collection
Healthy male and female adult baboons aged between 13.3 and 17.8 years (approximate human equivalent, 50–70 years), n = 10, were tranquilized with ketamine hydrochloride (10 mg/kg intramuscular injection) after an overnight fast, and then anesthetized with 1–2% isoflurane. While under general anesthesia, baboons were euthanized by exsanguination as approved by the American Veterinary Medical Association. We have shown that euthanasia by intravenous agents can alter tissue structure [19]. Following cardiac asystole, all tissues were collected according to a standardized protocol within < 1 h from the time of death, between 8:00 and 10:00 AM to minimize potential variation from circadian rhythms. Tissue collection was carried out under aseptic sterile conditions for isolation of resident primary cells. Skin biopsy was taken from behind the upper back portion of one ear of each baboon for isolation of epidermal fibroblasts. The prefrontal cortex was dissected from the left side of the brain. About 3-mm deep samples of gray matter from the prefrontal cortex, extending from the posterior part of the precentral sulcus to the intersection of the precentral sulcus and lateral sulcus, were collected. Area 10 of the Broca’s area of the prefrontal cortex was processed to obtain cortical astrocytes. The whole hippocampus was carefully dissected from the temporal lobe and cut coronally into three pieces comprising the anterior, middle, and posterior hippocampus, with about 2–3 mm of each piece combined for isolation of primary astrocytes. Cardiac tissues were collected separately from the left ventricle (LV), right ventricle (RV), left atrium (LA), and right atrium (RA) for cardiac fibroblast isolation. Both left and right liver lobes were collected separately for hepatocyte dispersion. All samples were collected on ice in sterile cell culture solution and processed within 1 h of collection.
Astrocyte cultures
The protocol for isolating baboon astrocytes was adapted from previously published procedures [20, 21]. Broca’s area 10 of the prefrontal cortex and hippocampus were minced in prewarmed 0.25% trypsin–EDTA (Gibco, Carlsbad, CA, USA) into small fragments. After 20 min of trypsinization in a standard cell culture incubator, an equal volume of DMEM-F12 Ham media (Sigma, St. Louis, MO, USA) supplemented with 10% FBS and 1% antibacterial and antimycotic solution was added to the tissue fragments. The tissue was mechanically disrupted by passing through a serological pipette and pelleted by centrifugation at 1000 × g for 2 min. Tissue fragments were further sheered by repeated pipetting. The processed mixture was then filtered through a 70-μm cell strainer, and the filtered cells were plated in a tissue culture flask (surface area of 75 cm2) in a fully supplemented DMEM-F12 Ham media.
Fibroblast cultures
Skin and cardiac fibroblasts were obtained using a similar cell culture isolation procedure. We previously published a detailed protocol for isolating primary fibroblasts from baboon skin samples [9] which was adapted for isolating cardiac fibroblasts. Briefly, tissues were minced and incubated with liberase (0.4 mg/ml, Sigma, St Louis, MO, USA) in a standard cell culture incubator for 3 h. Tissue mixture was resuspended in culture media, filtered with a 100-µm cell strainer, and centrifuged at 1000 × g for 5 min. Dispersed cells were cultured in Dulbecco’s modified eagle medium (DMEM) (Gibco, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS, Gibco) and 1% antibiotic and antimycotic solution. We leverage the adherent properties of fibroblasts to separate cardiac fibroblasts from other nonmyocytes [22] by pre-plating cells overnight, after which the media was changed to remove floating cells.
Hepatocyte cultures
The two-step EGTA/collagenase perfusion technique [23, 24] was adapted to isolate primary hepatocytes from baboon liver ex situ. Liver lobes were first separated from the entire liver piece and cut laterally closer to the caudal portion of the liver. Subsequently, two cannulae (16 g × 4 in) with a 3-mm smooth olive-shaped tip were positioned to target vascular channels in the liver for perfusion. We utilized a perfusion solution consisting of 1 × HBSS-HEPES (comprising 0.14 M NaCl, 50 mM KCL, 0.33 mM Na2HPO4, 0.44 mM KH2PO4, 10 mM Na-HEPES) buffered with 0.5 mM EGTA, 5 mM Glucose, and 4 mM NaHCO3 and adjusted to pH 7.2 (EGTA solution). The perfusion was facilitated using a Masterflex peristaltic pump (Cole-Palmer, Niles, IL, USA) set at a rate of 10 rpm, and this procedure continued for approximately 1 h at 37 °C. The EGTA solution was devoid of calcium, as the presence of calcium prevents dissociation of the extracellular matrix for effective isolation of live hepatocytes.
Following perfusion with the EGTA buffer, the solution was replaced with a collagenase solution which comprises 0.1% collagenase, 5 mM CaCl2, and 4 mM NaHCO3 in 1X HBSS solution, pH 7.5. The liver was then perfused at a rate of 8 rpm for a duration ranging from 45 min to 1 h depending on the liver size. The collagenase solution was collected and reused until visible signs of digestion were evident in the liver. A fully digested liver could be identified by its responsiveness to gentle pressure, resulting in an indentation, and the underlying cells are visible through Glisson’s capsule that overlays the liver. The digested liver was collected into a 10-mm culture dish containing chilled Gibco’s Williams media supplemented with 5% FBS, 1% glutamine, and antibiotics. The liver cells were dispersed into the ice-cold media under a sterile cell culture hood by gently removing the Glisson’s capsule and teasing apart the tissues with a 1-ml pipette tip. Cell suspensions were filtered through sterile folded gauze and centrifuged at 50 g, 4 °C for 5 min. The centrifugation step was repeated twice, and the resulting hepatocytes were plated on a collagen-coated plate containing prewarmed media overnight before performing further experiments the next day.
Characterization of primary cells with cell surface markers
Immunocytochemistry was used to validate cell type identity. Cells were fixed using 4% v/v paraformaldehyde and then rinsed with 1X HBSS and permeabilized with 0.1% Triton X-100. Cells were blocked with 5% bovine serum albumin in TBS with 0.1% Tween20 at room temperature for 1 h and then incubated in primary antibodies (detailed in Table 1) overnight at 4 °C. These primary antibodies were selectively targeted towards markers associated with fibroblasts (vimentin), hepatocytes (cytokeratin 8) and astrocytes (aquaporin 4, glial fibrillary acidic protein (GFAP), and excitatory amino acid transporter 2 (EAAT2). Markers of neurons (postsynaptic density protein 95; PSD95) and oligodendrocytes (myelin basic protein) were used to confirm whether isolated astrocyte populations are mixed with other brain cell types. Cultures were probed with secondary antibodies conjugated with FITC, counterstained using DAPI for nuclear identification, and imaged with a Zeiss LSM 880 confocal microscope using a 63 × oil immersion lens.
Table 1.
Primary antibodies specific for cell surface markers of fibroblasts, hepatocytes, and astrocytes
| Cell marker | Antibody | Dilution ratio | Company | Cat # |
|---|---|---|---|---|
| Fibroblasts | Vimentin | 1:200 | Santa Cruz | sc-6260 |
| Hepatocytes | Cytokeratin 8 | 1:200 | Santa Cruz | sc-8020 |
| Astrocytes |
Aquaporin 4 GFAP EAAT2 |
1:200 1:200 1:200 |
Millipore Millipore Santa Cruz |
AB2218 MAB360 sc-365634 |
| Neurons | PSD95 | 1:200 | Millipore | MABN1190 |
| Oligodendrocytes | MBP | 1:100 | Cell Signaling | 78896 |
Seahorse mitochondrial assay
Agilent Seahorse XFe96 Extracellular Flux Analyzer (North Billerica, MA, USA) was used to measure cellular respiration. Cells were plated in a 96-well seahorse plate at a density of 40,000 cells per well for all the different cell types. For hepatocytes requiring an adhesion material for cell attachment to the plate, collagen derived from rat tail (Sigma) was prepared in sterile double distilled H2O in 1:50 ratio and incubated in seahorse plates at 37 °C for 3 h before cell seeding. The XFe96 sensor cartridges were hydrated overnight with H2O at 37 °C and replaced with XF calibrant 1 h before the assay. Oxygen consumption rate (OCR), an index of cellular respiration, was measured under basal condition and in response to serial injection of mitochondrial inhibitors including 1.5 µM Oligomycin (Oligo) to inhibit ATP synthase, 0.5 µM FCCP (carbonyl cyanide-p-trifluoromethoxyphenylhydrazone; a mitochondrial uncoupler to measure maximum respiration), and 0.5 µM rotenone/antimycin A cocktail (Rot/AA) to inhibit electron flow through mitochondrial electron transport chain. To determine mitochondrial response to metabolic stress, a subset of cells was exposed to a low glucose media (1 mM glucose) for 2 h, before the mitochondrial stress test described above. After the assay, the OCR values were normalized to cell density measured by the IncuCyte live-cell imaging system. Data were analyzed by the Agilent WAVE software.
Cellular resilience assay
Cellular resilience was assessed in dividing primary cells (except hepatocytes which do not proliferate in vitro). Cell proliferation rate was determined in response to 50 µM hydrogen peroxide (H2O2), low glucose media (1 mM), or 0.1 µM thapsigargin to model oxidative, metabolic, and proteostasis stress, respectively, using a live-cell imaging system (IncuCyte S3). Cells were plated at a density of 2000 cells per well into a 96-well plate with cell growth captured every 6 h for 6 days. After 24 h of cell plating, cells were exposed to H2O2, low glucose, and thapsigargin for 2 h. Treatments were replaced with complete media and growth was monitored until the termination of the study. The duration and concentration of stressors used were previously validated in a prior study as appropriate to inhibit cell proliferation without inducing cell death [9], which we found applicable for the newly established cell lines in a pilot study. To determine cell proliferation rate, the IncuCyte analysis software quantifies cell surface area coverage to determine cell confluence values during the entire cell culture period. Each cell confluence data point (%) was plotted against its corresponding time (h) of data collection to generate a kinetic graph. The slope of the time-course changes in cell confluence during the exponential growth phase, after the stressor was removed, was used to determine the cell proliferation rate (% confluence per h) using GraphPad Prism 9 software.
Statistical analysis
Primary cell type from distinct tissue areas was categorized as an independent group. Data were analyzed by one-way analysis of variance (ANOVA) followed by Tukey post hoc test and unpaired t-test when comparing effects between two groups. The ANOVA was weighted using SEM to account for variability in technical replicates. Data were presented as mean ± SEM; p < 0.05 is considered statistically significant. Data from males and females were combined, n = 5–10 baboons per tissue cell type except where they are specifically separated by sex. The variability in animal number per tissue type was due to the failed culture of some cell types from a single donor. Bivariate correlation was used to determine the relationship between two variables. All analyses were carried out using GraphPad Prism 9.
Results
Characterization of baboon primary astrocytes, fibroblasts, and hepatocytes
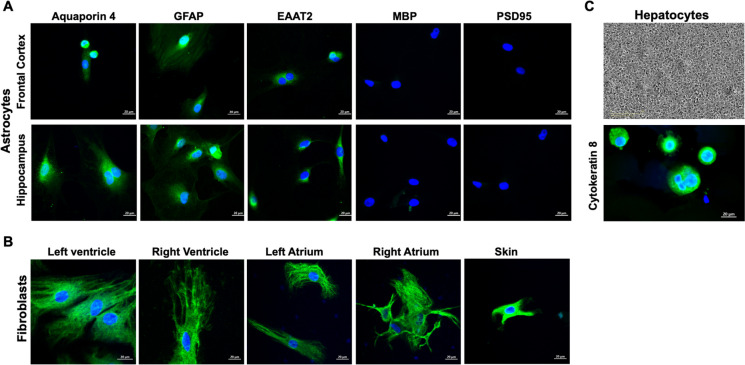
Astrocytes derived from the prefrontal cortex and hippocampus show strong immunoreactivity for astrocyte markers; aquaporin 4, glial fibrillary acidic protein (GFAP), and excitatory amino acid transporter 2 (EAAT2). The absence of immunoreactive signals for myelin basic protein and postsynaptic density protein 95 (PSD95) indicates that astrocyte cultures do not contain oligodendrocytes and neurons, respectively. Both skin and cardiac fibroblasts showed strong immunoreactive signal to vimentin, a marker of fibroblasts. Phase contrast image showed hepatocytes as polygonal cells with distinct cell borders. The hepatocytes were immunopositive for cytokeratin 8 (Fig. 1).
Fig. 1.
Representative photomicrographs of baboon astrocyte, fibroblast, and hepatocyte cultures. A Astrocytes derived from the prefrontal cortex and hippocampus show immunoreactivity for astrocyte markers: aquaporin 4, glial fibrillary acidic protein (GFAP), and excitatory amino acid transporter 2 (EAAT2). The absence of immunoreactive signals for myelin basic protein and postsynaptic density protein 95 (PSD95) indicates that astrocyte cultures do not contain oligodendrocytes and neurons, respectively. B Expression of fibroblast marker, vimentin in fibroblasts from different heart regions such as the left ventricle, right ventricle, left atrium, and right atrium, and skin epidermal fibroblasts express fibroblast marker, vimentin. C Phase contrast microscopy of hepatocyte cultures and immunoreactive signals of epithelial cell marker, cytokeratin 8. Scale 20 µm. Images were acquired with a Zeiss LSM 880 confocal microscope using a 63 × oil immersion lens. Donor animal: male, 16.4 years old
Mitochondrial bioenergetics differ among astrocytes, fibroblasts, and hepatocytes
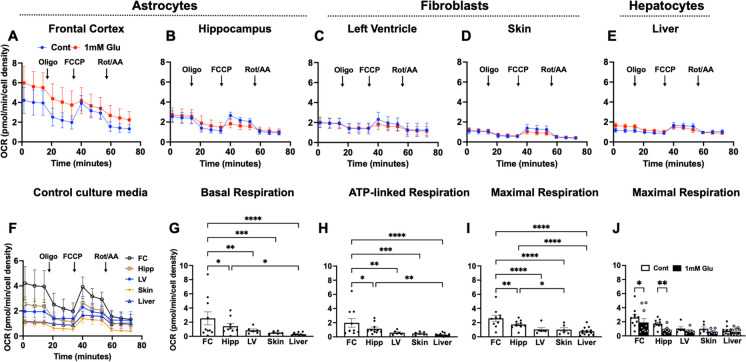
In Fig. 2A–E, we present OCR kinetics in astrocytes, fibroblasts, and hepatocytes under normal culture conditions (designated as control) and in response to acute metabolic stress induced by exposure to low glucose (1 mM glucose for 2 h). These were combined data for male and female. With the exception of astrocytes, the OCR of other cell types examined was comparable under both control culture conditions and those under metabolic stress prior to testing. However, by testing all under the same conditions, we show the differential respiratory patterns, both basal and under mitochondrial stress during the assay, among the different cell types from aging baboon cells. Figure 2F shows the cellular respiration pattern of these 5 different cell types under control conditions. Further, Fig. 2G–I show the integrated data and significant differences among these cell types in their basal, ATP-linked, and maximal respiration properties.
Fig. 2.
Oxygen consumption rate in baboon astrocytes, fibroblasts, and hepatocytes. Oxygen consumption rate (OCR) was measured under basal condition and in response to serial injection of mitochondrial inhibitors including Oligomycin (Oligo; 1.5 µM), carbonyl cyanide-p-trifluoromethoxyphenylhydrazone (FCCP; 0.5 µM), and rotenone and antimycin A cocktail (Rot/AA; 0.5 µM). OCR kinetics in A astrocytes derived from the Broca’s area of the prefrontal cortex (FC), B astrocytes from the hippocampus, C fibroblasts from the left ventricle of the heart, D skin fibroblasts, and E hepatocytes under standard culture condition and in response to low glucose (1 mM glucose for 2 h). F Time-course OCR of cortical astrocytes, hippocampal astrocytes, LV fibroblasts, skin fibroblasts, and hepatocytes under standard culture conditions. G Basal respiration, H ATP-linked respiration, and I maximal respiration in astrocytes derived from the FC and hippocampus (hipp) as well as left ventricle fibroblasts (LV), skin fibroblasts (skin), and hepatocytes (liver). Data from individual animal were expressed as mean ± standard error of mean, with male and female data combined. Data from left and right lobe hepatocytes were pooled for comparison with other cell types. For each primary cell line, respiration rates were from 4 to 6 replicate samples measured using the Seahorse XFe96 flux analyzer. Donor age, 13.3–17.8 years, sample size (cortical astrocytes, n = 10; hippocampal astrocytes, n = 10; LV and skin fibroblasts, n = 6, respectively; hepatocytes, n = 7). *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001
Cortical astrocytes exhibited the highest overall OCR among the cell types, followed by hippocampal astrocytes, while OCR in LV and skin fibroblasts as well as hepatocytes were comparable. There was a significantly higher basal, ATP-linked, and maximal respiration in cortical astrocytes relative to hippocampal astrocytes (p < 0.05), LV fibroblasts (p < 0.01), skin fibroblasts (p < 0.001), and hepatocytes (p < 0.0001). Hippocampal astrocytes exhibited significantly higher maximal respiration compared to skin fibroblasts (p < 0.05) and hepatocytes (p < 0.0001) but not with LV fibroblasts (Fig. 2G–H). Interestingly, astrocytes were sensitive to acute exposure to low glucose, evidenced by reduced maximal respiration in both cortical (p < 0.05) and hippocampal astrocytes (p < 0.01), whereas other cell types did not exhibit changes.
While we found no effect of metabolic stress (low glucose) on respiratory outcomes in fibroblasts and hepatocytes, we did see the astrocytes from both cortex and hippocampus show reduced maximal respiration when cultured under metabolic challenge (Fig. 2J). We used LV cardiac fibroblasts as representative of this tissue/cell type because we found no significant difference in mitochondrial respiration among cell lines isolated from the heart (discussed below). No significant difference in bioenergetics was observed between left lobe and right lobe hepatocytes (data not shown); thus, data from both lobes were pooled for comparison with other cell types.
We next asked whether donor sex affected these cellular and mitochondrial phenotypes. Data from both males and females were relatively consistent with the data analyzed as combined sexes above (Supplemental Fig. 1). For example, cortical astrocytes exhibited higher maximal OCR compared to skin fibroblasts and hepatocytes when data are analyzed for only male and for only female donors. This observation is similar to those obtained from combined data from both sexes. We compared differences between male and female in each cell type using Student’s t-test, and found that only female hepatocytes had higher maximal OCR compared to males (p < 0.05) while other cell types exhibited similar OCR between male and female donors (Supplemental Fig. 1 C).
Comparison of astrocyte and fibroblast resilience to oxidative, metabolic, and proteostasis stress
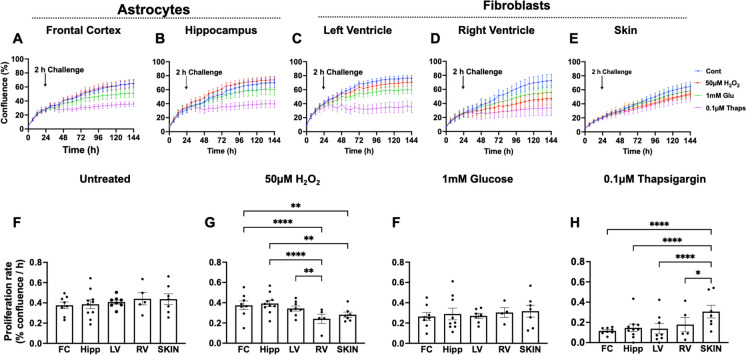
Using a protocol we previously developed for epidermal-derived fibroblasts [9], we measured cellular resilience in response to different stressors such as H2O2 (50 µM), low glucose (1 mM), and thapsigargin (0.1 µM) to model oxidative, metabolic, and proteostasis stress, respectively. For these studies, hepatocytes were excluded due to their lack of proliferation under standard culturing conditions. Baboon astrocytes and fibroblasts from different anatomical regions exhibited similar proliferation rate when cultured in standard culture media or exposed to low glucose media but they responded quite differently to oxidative or proteostasis stress (Fig. 3). The proliferation of astrocytes from both the prefrontal cortex and hippocampus was not altered by short-term exposure to 50 µM H2O2 whereas RV and skin fibroblasts had reduced proliferation (p < 0.01) in response to a 2 h H2O2 challenge suggesting increased cellular resilience to challenge in astrocytes. Interestingly, the impact of oxidative stress on LV fibroblasts was similar to that of astrocytes. The exposure of astrocytes and fibroblasts from different anatomical regions to low glucose media for 2 h inhibited cell growth in a comparable pattern, resulting in no discernible difference between the different cell types. However, with the thapsigargin challenge, the resulting reduction in proliferation rate was significantly greater in astrocytes (p < 0.0001), LV (p < 0.0001), and RV (p < 0.05) fibroblasts compared to skin fibroblasts suggesting low resilience to the proteostatic stress in these cell types (Fig. 3).
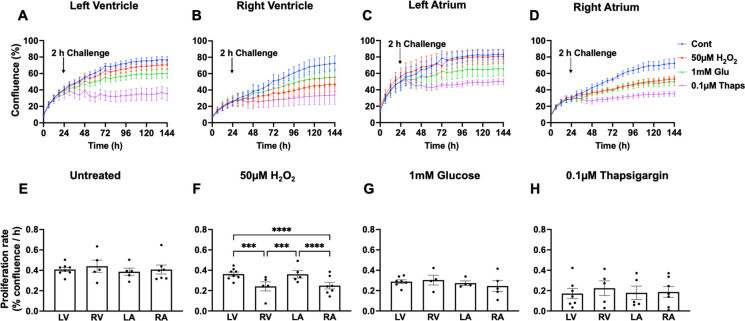
Fig. 3.
Effects of oxidative, metabolic, and proteostasis stress on proliferation of baboon astrocytes and fibroblasts. Astrocytes derived from prefrontal cortex Broca’s area 10 (FC) or the entire hippocampus (hipp) encompassing anterior, middle, and posterior hippocampus and fibroblasts from the left ventricle (LV), right ventricle (RV), and ear skin (skin) were exposed to 50 µM H2O2, 1 mm glucose, and 0.1 µM thapsigargin for 2 h to model oxidative, metabolic, and proteostasis stress, respectively. Time-course changes in cell confluence in response to challenge were monitored in real time with the IncuCyte live-cell imaging system housed within a cell culture incubator (3% O2, 5% CO2 at 37 °C). The blue line represents untreated cells (designated as control); the red line, 50 µM H2O2; the green, 1 mM glucose; and the purple, 0.1 µM thapsigargin. Proliferation rate (% confluence/h) calculated from the slope of the kinetic graph between 30 and 144 h was shown in response to untreated and challenged cells. Proliferation rate was analyzed using two-way ANOVA. Data expressed as mean ± standard error of mean; each data point represents 3 replicate wells for each animal; cell seeding density was 2000 cells/well; donor age was between 13.3 and 17.8 years, with male and female data combined. Sample size: FC, n = 8; hipp, n = 10; LV, n = 8; RV, n = 5; skin, n = 7. *p < 0.05, **p < 0.01, ****p < 0.0001
Similar to our mitochondria assays above, we found only small effects of donor sex on these outcomes in these data sets. The response of the different cell types to stressors was similar in male and female subjects except the slight variation in the response of cortical astrocytes and RV fibroblasts to oxidative and proteostasis stress, respectively, between males and females (Supplemental Fig. 2).
Given the higher metabolism of astrocytes and their resilience to 50 µM H2O2 challenge, we reasoned that there may be a relationship between bioenergetics and resilience of primary astrocytes. Indeed, astrocyte maximal respiration significantly correlates (p < 0.05) with their proliferation response to H2O2 challenge (Fig. 4), which suggests a relationship between cellular metabolism and resilience to oxidative stress.
Fig. 4.
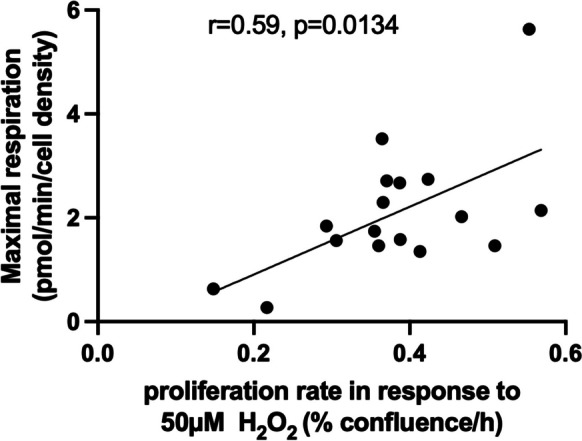
Correlation between astrocyte maximal respiration and proliferation response to oxidative stress. The maximal oxygen consumption rates (OCR) in baboon astrocytes, induced by the mitochondrial uncoupler FCCP, show a positive correlation with their proliferation response to 50 µM H2O2. OCR was determined with the Seahorse XFe96 flux analyzer. Cellular proliferation was monitored with a live-cell imaging system (IncuCyte) with the proliferation rate calculated from the slope of cell kinetics over 5-day period. The correlation between maximal respiration and proliferation rate in response to H2O2 was determined using Pearson’s correlation coefficient. Data are from primary astrocytes obtained from the baboon prefrontal cortex and hippocampus encompassing both male and female animals, with ages ranging from 13.3 to 17.8 years (n = 17)
We also examined whether cells that present high resilience to oxidative stress are also highly resilient to either metabolic or proteostatic stress. Data from similar cell type were combined for the correlative analysis to enhance the robustness of the analysis. In both astrocytes and cardiac fibroblasts, resilience to oxidative stress (H2O2) positively correlates with resilience to metabolic stress (low glucose). There was also a relationship between cardiac fibroblasts’ resilience to metabolic stress and proteostasis stress (thapsigargin) but not in astrocytes. Resilience to oxidative stress did not correlate with proteostasis stress in both cell types (Table 2).
Table 2.
Correlation of resilience to oxidative, proteostatic, and metabolic stress in astrocytes and cardiac fibroblasts
| Cells | Correlation to H2O2 | Correlation to thapsigargin | |
|---|---|---|---|
| Glucose | Thapsigargin | Glucose | |
| Astrocytes | r = 0.75, p = 0.001 | r = 0.22, p = 0.396 | r = 0.06, p = 0.837 |
| Cardiac fibroblasts | r = 0.47, p = 0.030 | r = 0.21, p = 0.341 | r = 0.69, p = 0.001 |
Pearson’s correlation coefficient: r. n = 18 for astrocytes and 21 for cardiac fibroblasts. Values in bold font represent correlations that are significantly different.
Mitochondrial bioenergetics in cardiac fibroblasts from different heart regions
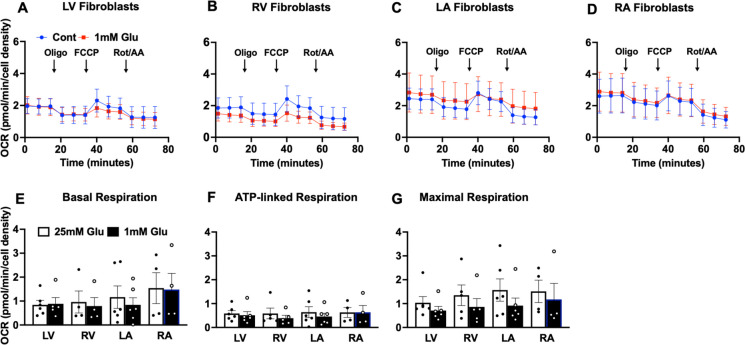
Even within a single tissue, cells are not homogenous in cell type, distribution, location, or niche. Here, we addressed whether cells isolated from different regions of the same tissue (heart) might differ in patterns of resilience. We made a comparison between primary fibroblasts from heart chambers to determine whether site-specific differences exist in cell bioenergetics. Cardiac fibroblasts from different heart regions (LV, RV, LA, and RA) exhibited similar OCR for all measures of seahorse mitochondrial stress test under standard cell culture conditions (25 mM glucose in culture media) or following 2-h exposure to low glucose media to model acute metabolic stress. Figure 5A–D show OCR kinetics in LV, RV, LA, and RA fibroblasts from which basal, ATP-dependent, and maximal respiration were derived (Fig. 5E–F). These data then suggest, at least in the heart, that anatomical location has little impact on the respiratory function of primary cells.
Fig. 5.
Oxygen consumption rate of baboon cardiac fibroblasts. Time-course graph of oxygen consumption rates (OCR) measured with a Seahorse XFe96 flux analyzer using baboon cardiac fibroblasts derived from A left ventricle, LV; B right ventricle, RV; C left atrium, LA; and D right atrium, RA, under standard culture condition (designated as control) and in response to low glucose media (1 mM glucose). E Basal respiration, F ATP-linked respiration, and G maximal respiration were derived from the kinetic graph following initial measurement, in the presence of oligomycin and in response to carbonyl cyanide-p-trifluoromethoxyphenylhydrazone (FCCP), respectively. Data, expressed as mean ± standard error of mean, are from individual animal, with male and female data combined. For each primary cell line, respiration rates from 4 to 6 replicate samples were measured using the Seahorse XFe96 flux analyzer. Donor age, 13.3–17.8 years, n = 5–6 per cardiac fibroblasts from different heart regions
Resilience of cardiac fibroblasts to oxidative, metabolic, and proteostasis stress
On the other hand, we did find evidence that resilience to challenge among cells within a tissue may be impacted by the anatomical location of development. Figure 6 shows the kinetics and proliferation rate of cardiac fibroblast in normal growth media and in response to challenge compounds. Cardiac fibroblasts from different heart regions exhibited similar proliferation rates under normal culture conditions (Fig. 6A–E). However, upon short-term exposure to H2O2, the proliferation of both RV and RA fibroblasts reduced significantly compared to the effect of H2O2 on LV and LA fibroblasts (p < 0.001), suggesting that fibroblasts from the right heart region are less resilient to oxidative stress (Fig. 6A–D and F). When subjected to either metabolic or proteostasis stress, (1 mM glucose or 0.1 µM thapsigargin, respectively) cardiac fibroblasts demonstrated impaired proliferation in a similar manner across different heart regions (Fig. 6A–D, G, and H).
Fig. 6.
Effects of oxidative, metabolic, and proteostasis stress on proliferation of cardiac fibroblasts from different heart regions. Fibroblasts derived from the four chambers of the baboon heart encompassing the left ventricle (LV), right ventricle (RV), left atrium (LA), and right atrium (RA) were exposed to 50 µM H2O2, 1 mm glucose, and 0.1 µM thapsigargin for 2 h to model oxidative, metabolic, and proteostasis stress, respectively. Time-course changes in cell confluence of cardiac fibroblasts in response to challenge compounds were monitored in real time with the IncuCyte live-cell imaging system housed within a cell culture incubator (3% O2, 5% CO2 at 37 °C). The blue line represents untreated cells (designated as control); red line, 50 µM H2O2; green, 1 mM glucose; and purple, 0.1 µM thapsigargin. Proliferation rate (% confluence/h) calculated from the slope of the kinetic graph between 30 and 144 h was shown in response to untreated and challenged cells. Proliferation rate was analyzed using two-way ANOVA. Data expressed as mean ± standard error of mean; each data point represents 3 replicate wells for each animal; cell seeding density was 2000 cells/well; donor age was between 13.3 and 17.8 years, male and female data combined; n = 8 for LV, 5 for RV, 5 for LA and 7 for RA. ***p < 0.001, ****p < 0.0001
Discussion
The newly established cell lines described in this study represent a valuable resource for mechanistic studies that can be coupled with in vivo approaches to bridge some translational gaps between basic and clinical studies in aging research and other allied fields. For example, retention of in vivo phenotype in an in vitro system opens avenues for validation of potential in vivo mechanisms. Experiments can be designed to test aging mechanisms at the cellular level which may inform novel therapeutic development or personalized precision medicine. These data can also contribute to building network modules that encompass cellular, tissue, and systemic phenotypes for a comprehensive understanding of human physiology and pathophysiology. This work is part of an ongoing initiative to generate different primary cell types from an aging baboon cohort across the life course. The data presented herein pertain specifically to adult baboons aged between 13.3 and 17.8 years and thus do not ask specifically whether age changes these patterns of resilience; primary cells from younger baboons, including various cell types beyond skin fibroblasts, will be tested in a similar manner in the near future. We characterized the identity of multiple tissue cultures in baboons and determined their bioenergetic and resilient properties under similar experimental conditions. In most instances, the sex of the donor animal did not significantly alter the observed differences between cell types and the combination of male and female added more statistical power. As a result, we premised data interpretation primarily on combined data from both sexes, which aligns with the results obtained from individual sex-based analyses for most variables. Significant findings here include the following: (i) Prefrontal cortex astrocytes exhibit higher mitochondrial respiration than hippocampal astrocytes. (ii) Astrocytes display higher cellular respiration and exhibit greater resilience to oxidative stress than both fibroblasts and hepatocytes. (iii) Metabolic stress alters maximal respiration only in astrocytes but changes in fibroblasts and hepatocytes were not statistically significant. (iv) Fibroblasts obtained from the right heart region (RV and RA) were less resilient to oxidative stress compared to fibroblasts from the left heart region (LV and LA). (v) Skin fibroblasts were less impacted by proteostasis stress relative to astrocytes and cardiac fibroblasts. (vi) Higher maximal respiration may serve as a predictive indicator of astrocyte resilience to oxidative stress.
We characterized the identity of primary cells with their cell surface markers using immunocytochemistry. Astrocyte cultures exhibited strong reactivity to key astrocyte markers such as GFAP, aquaporin 4, and EAAT2, and importantly, they did not contain mixed populations of oligodendrocytes and neurons. Similarly, both skin and cardiac fibroblasts demonstrated robust immunoreactivity for vimentin, a recognized marker of fibroblasts, while hepatocytes were immunopositive to cytokeratin 8, a marker for epithelial cells. It is worth noting that cardiac fibroblasts are larger in size and morphologically different than skin fibroblasts (Fig. 1). Whether these differences translate to any functional difference is not clear. The choice of these cell types is premised on other parallel studies on developmental programming and aging interactions in our baboon cohort where we observed accelerated aging of the brain, cardiac remodeling, and metabolic dysfunction due to developmental exposure to maternal undernutrition [25–27]. Although neuronal and cardiomyocyte cultures are desirable, there are technical limitations of generating viable neurons or cardiomyocytes in aging NHP. Besides fibroblast cultures, there are few studies that have established primary cultures of key organs from baboons [28], and to our knowledge, this study is the first to establish primary cells from multiple regions of the heart, brain, and liver in aging baboons (13.3–17.8 years). Baboon lifespan at SNPRC, where our baboons are maintained, has been reported as 11 or 21 years [29, 30], and thus, these animals represent the transition period from mid- to late-life.
As a key determinant of cellular health, the mitochondria play a significant role in energy metabolism, metabolite production, and cellular signaling [31]. However, their contribution to various cellular processes varies across different cell types, depending on the cell’s specific demands, differences in mitochondrial protein composition, and internal mitochondrial structures such as matrix and cristae [32]. In heart and brain cells, the mitochondria are predominantly oriented towards energy metabolism, converting substrates such as glucose to ATP to sustain high-energy-demanding tasks of the heart and brain [33, 34]. The liver in contrast plays a more significant role in metabolite biosynthesis and its mitochondria have a lower metabolic dynamic range for energy conversion compared to those of the heart [32, 35]. Thus, we expect that the specialized functions and variable energy demand of the cells examined in the current study reflect the physiological diversity of mitochondrial oxidative phosphorylation in the heart, brain, and liver [36].
Using cellular OCR as a proxy for mitochondrial respiration, we observed similar mitochondrial respiration levels between cardiac fibroblasts and hepatocytes. Given that myocardial contraction, an energy-dependent process, is determined by cardiomyocytes, it is plausible that the differences between heart and liver mitochondria may be closely related to the cardiomyocytes than cardiac fibroblasts. Astrocytes exhibited higher mitochondrial respiration than fibroblasts (both cardiac and skin) and hepatocytes. This finding may be attributed to the abundance of mitochondria in astrocytes and the overall high-energy metabolism observed in the brain [37]. Although neurons consume about 80% of energy in the brain, most of which are expended on synaptic signaling, astrocytes play a significant role in ensuring energy delivery from capillaries to synapses, as such, astrocytes are considered key cells in coupling synaptic activity with energy metabolism [38]. Given the role that astrocytes play in the energetic requirement of neurons, it is unsurprising that astrocytes exhibit higher mitochondrial respiration compared to other examined cell types. When cells were exposed to acute low glucose challenge prior to assessing mitochondrial function, only astrocytes showed higher sensitivity to the challenge, which demonstrates greater glucose utilization and demand in astrocytes compared to fibroblasts and hepatocytes. This aligns with higher glucose metabolism in astrocytes compared to neurons [39] and parallels the essence for brain glycogen content storage mostly in astrocytes [40]. In fibroblasts and hepatocytes, the similarity in mitochondrial OCR between normal and low glucose-exposed cells may be attributed to a preferential switch to alternative fuel source such as fatty acids or the high metabolic flexibility of these cell types to a low glucose challenge. In particular, cardiac fibroblasts exhibit greater dependence on fatty acid oxidation over glucose oxidation to meet their metabolic demands [41], and in relation to this study, this underscores their adaptability to low glucose.
When we considered brain region-specific variation in astrocytic mitochondrial function, cortical astrocytes exhibited higher basal, ATP-linked, and maximal respiration compared to hippocampal astrocytes. These functional disparities align with inherent morphological differences between the two astrocyte populations. In a morphometric analysis of differentiated cortical and hippocampal astrocytes derived from neural progenitor cells, cortical astrocytes displayed numerous short, well-defined projections with multiple branches per projection and smaller soma areas. In contrast, hippocampal astrocytes possess larger soma areas with few but long projections with almost no branching [42]. The distinctions in astrocytic projections, which are crucial for energy processing in support of neuronal metabolism through delivery of substrate from capillaries to synapse, may underlie the heightened energetic profile of cortical astrocytes compared to hippocampal astrocytes. Determining mitochondrial function in astrocytic branches will be important to demonstrate the bioenergetics of these astrocytic projections.
Cardiac fibroblasts play a crucial role as supporting cells for cardiomyocytes. As part of the myocardial wall, cardiac fibroblasts are involved in synthesis and maintenance of extracellular matrix (ECM) network which is essential for electrical conductivity and heartbeat rhythm [16]. We did not observe any region-specific differences in mitochondrial respiration of cardiac fibroblasts, which is consistent with other reports that have demonstrated that the processes of energy metabolism including mitochondrial protein contents in the left and right heart are identical [43, 44]. Interestingly, fibroblasts from the right side of the heart (RV and RA) were less resilient to H2O2 (inducer of oxidative stress) compared to those from the left region (LV and RA). Single-nuclear transcriptome profiling of the human heart identified chamber specificity in cardiac fibroblasts gene expression which demonstrates the molecular diversity of cardiac fibroblasts [45]. Structural and physiological differences in heart chambers may also contribute to variability in stress resiliency. The left heart region is nearly constantly aerated with oxygenated blood relative to the right but whether this influences stress response is not clear. The small muscle mass of the RV is associated with its low oxygen demand and poor tolerance to increased afterload when pulmonary pressure rises [46]. One possibility may be that the region-specific response of cardiac fibroblasts to oxidative stress may be related to stress adaptation in the left side of the heart, possibly influenced by the differential pressure loads between the heart chambers. Pressure overload is a form of mechanical stress and is linked to increased formation of superoxide anions [47]. Our results suggest that LV fibroblasts may have developed a reserve capacity for stress resilience compared to the RV. Indeed, others have reported that ROS-induced damage occurs earlier in RV than LV due to lower antioxidant capacity in RV tissues [48]. Thus, at the cellular level, the differential response to acute H2O2 challenge between the right and left heart region resonates with the intrinsic difference in their functional capacity. This differential response of cardiac fibroblasts from different heart regions is however specific to oxidative stress as there were no significant differences in proliferation response of cardiac fibroblasts to metabolic and proteostasis stress. We did not test this resilience assay in hepatocytes since they are non-dividing cells in vitro.
When we compared the effect of H2O2 on proliferation of astrocyte and fibroblasts from different regions, H2O2 significantly decreased the proliferation of RV and skin fibroblasts compared to both cortical and hippocampal astrocytes. The effect of acute exposure to H2O2 was similar in astrocytes and LV fibroblasts. In contrast, skin fibroblasts were less impacted by thapsigargin exposure compared to astrocytes, LV, and RV fibroblasts. This suggests opposing sensitivity to oxidative and proteostasis stress in different cell types, an important consideration for future investigations aimed at between defining the molecular factors regulating resilience across tissue types. The relationship we observed between astrocyte bioenergetics and proliferation response to oxidative stress propels the idea that we can model interconnected hallmarks of resilience that would serve as predictive markers for clinical applications. The hallmarks may encompass variables such as mitochondrial bioenergetics, proteostasis, and transcriptional response. In astrocytes, a higher maximal respiration correlates with resilience to oxidative stress.
One key question we attempted to address in the present study was whether a cell that presents high resilience to a particular stressor also demonstrates a similar level of resilience to another. In both astrocytes and cardiac fibroblasts, cells that show high resilience to H2O2-induced oxidative stress were also resilient to metabolic inhibition induced by low glucose concentrations. This might be expected since both H2O2 and glucose deprivation can drive ROS generation [49, 50], and suggest that a common cellular element may be targeted by the individual cell machinery. We previously demonstrated a similar relationship in mouse fibroblasts which display correlated resistance to H2O2 and glucose deprivation [51]. Potential factors that modulate resilience capacity include the ability to mobilize defensive molecules such as antioxidants, the presence of a robust transcriptional program, and the induction of repair mechanism [52]. Cells with adequate reserve of these factors might be relatively resilient to both of H2O2 and low glucose-induced stress. Further investigations into the molecular circuits that connect the metabolic effects of low glucose to oxidative stress are warranted. Further, unlike astrocytes, we observed that cardiac fibroblasts that were resilient to low glucose were equally resilient to thapsigargin. Thapsigargin is an intracellular Ca2+ pump inhibitor that induces endoplasmic reticulum (ER) stress by altering calcium homeostasis and causing protein aggregation [53, 54]. One of the cellular responses to the ER stress is the induction of ER chaperone proteins to restore homeostasis [55]. Glucose deprivation can as well induce ER stress [56] which might be a potential link between resilience to low glucose and thapsigargin challenge. The basis for specificity to cardiac fibroblasts is however unclear.
A major limitation of this study is the relatively small sample size. We could not ascertain whether an animal that exhibits high resilience in one cell type (e.g., cortical astrocytes) exhibits the same in another (e.g., left ventricle fibroblasts or skin fibroblasts). In a correlative analysis with a sample size of 8 animals (data not shown), we observed that cortical astrocyte’s resilience to metabolic stress parallels that of the hippocampus in the same animal (r = 0.71, p = 0.051). The positive relationship between cortical astrocytes and LV fibroblasts was not significant (r = 0.61, p = 0.276, n = 5). Although inconclusive, this analysis suggests the possibility of synchronized resilience pattern across specific organ systems in an individual donor. We were also limited in confirming whether age influences the resilience of the different cell types and the clear impact of donor sex on the variable examined. We also did not examine the molecular basis of the differential response to stressors and only speculated on the potential factors driving cellular heterogeneity in resilience.
In summary, we provide evidence of successful establishment of astrocytes, fibroblasts, and hepatocyte cultures from our aging baboon cohort and that these cells display differential responses to multiple forms of stress and mitochondrial bioenergetics. This cellular heterogeneity implies differential mechanisms underly stress resiliency across cell types which may impact their aging rates. Our future studies will determine the role of sex in larger sample size, age, and developmental exposures on resilience across different cell types as we increase animal sampling.
Supplementary Information
Below is the link to the electronic supplementary material.
Acknowledgements
The authors acknowledge the administrative and technical support of Karen Moore, Wenbo Qi, Benjamin Morr, Raechel Camones, and Jonathan Gelfond. We also acknowledge support from the SNPRC which is funded by P51 OD011133.
Funding
This research was funded in part by R01 AG057431, I01BX004167 (ABS), the San Antonio Nathan Shock Center (P30 AG 013319), and the Geriatric Research, Education and Clinical Center of the South Texas Veterans Health Care System. This material is the result of work supported with resources and the use of facilities at South Texas Veterans Health Care System, San Antonio, Texas. Baboons in this study were maintained under 1U19AG057758-01A1 (PWN, LAC).
Data availability
The data presented in the work are available from the corresponding author upon request.
Declarations
Competing interests
The authors declare no competing interest.
Disclaimer
The contents do not represent the views of the US Department of Veterans Affairs or the US Government.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Tuttle CSL, Waaijer MEC, Slee-Valentijn MS, Stijnen T, Westendorp R, Maier AB. Cellular senescence and chronological age in various human tissues: a systematic review and meta-analysis. Aging Cell. 2020;19(2):e13083. 10.1111/acel.13083. 10.1111/acel.13083 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Tran NM, Shekhar K, Whitney IE, Jacobi A, Benhar I, Hong G, Yan W, Adiconis X, Arnold ME, Lee JM, Levin JZ, Lin D, Wang C, Lieber CM, Regev A, He Z, Sanes JR. Single-cell profiles of retinal ganglion cells differing in resilience to injury reveal neuroprotective genes. Neuron. 2019;104(6):1039-1055.e12. 10.1016/j.neuron. 10.1016/j.neuron [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nie C, Li Y, Li R, Yan Y, Zhang D, Li T, Li Z, Sun Y, Zhen H, Ding J, Wan Z, Gong J, Shi Y, Huang Z, Wu Y, Cai K, Zong Y, Wang Z, Wang R, Jian M, Jin X, Wang J, Yang H, Han JJ, Zhang X, Franceschi C, Kennedy BK, Xu X. Distinct biological ages of organs and systems identified from a multi-omics study. Cell Rep. 2022;38(10):110459. 10.1016/j.celrep.2022.110459. 10.1016/j.celrep.2022.110459 [DOI] [PubMed] [Google Scholar]
- 4.Zhan M, Yamaza H, Sun Y, Sinclair J, Li H, Zou S. Temporal and spatial transcriptional profiles of aging in Drosophila melanogaster. Genome Res. 2007;17(8):1236–43. 10.1101/gr.6216607. 10.1101/gr.6216607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Picard M, McEwen BS, Epel ES, Sandi C. An energetic view of stress: focus on mitochondria. Front Neuroendocrinol. 2018;49:72–85. 10.1016/j.yfrne.2018.01.001. 10.1016/j.yfrne.2018.01.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lee HY, Choi CS, Birkenfeld AL, Alves TC, Jornayvaz FR, Jurczak MJ, Zhang D, Woo DK, Shadel GS, Ladiges W, Rabinovitch PS, Santos JH, Petersen KF, Samuel VT, Shulman GI. Targeted expression of catalase to mitochondria prevents age-associated reductions in mitochondrial function and insulin resistance. Cell Metab. 2010;12(6):668–74. 10.1016/j.cmet.2010.11.004. 10.1016/j.cmet.2010.11.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kimmel JC, Penland L, Rubinstein ND, Hendrickson DG, Kelley DR, Rosenthal AZ. Murine single-cell RNA-seq reveals cell-identity- and tissue-specific trajectories of aging. Genome Res. 2019;29(12):2088–103. 10.1101/gr.253880.119. 10.1101/gr.253880.119 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhang X, Azhar G, Nagano K, Wei JY. Differential vulnerability to oxidative stress in rat cardiac myocytes versus fibroblasts. J Am Coll Cardiol. 2001;38(7):2055–62. 10.1016/s0735-1097(01)01665-5. 10.1016/s0735-1097(01)01665-5 [DOI] [PubMed] [Google Scholar]
- 9.Adekunbi DA, Li C, Nathanielsz PW, Salmon AB. Age and sex modify cellular proliferation responses to oxidative stress and glucocorticoid challenges in baboon cells. Geroscience. 2021;43(4):2067–85. 10.1007/s11357-021-00395-1. 10.1007/s11357-021-00395-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Huber HF, Gerow KG, Li C, Nathanielsz PW. Walking speed declines with age in male and female baboons (Papio sp.): Confirmation of findings with sex as a biological variable. J Med Primatol. 2021;50(5):273–5. 10.1111/jmp.12538. 10.1111/jmp.12538 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kuo AH, Li C, Huber HF, Nathanielsz PW, Clarke GD. Ageing changes in biventricular cardiac function in male and female baboons (Papio spp.). J Physiol. 2018;596(21):5083–98. 10.1113/JP276338. 10.1113/JP276338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cox LA, Puppala S, Chan J, Zimmerman KD, Hamid Z, Ampong I, Huber HF, Li G, Jadhav AYL, Wang B, Li C, Baxter MG, Shively C, Clarke GD, Register TC, Nathanielsz PW, Olivier M. Integrated multi-omics analysis of brain aging in female nonhuman primates reveals altered signaling pathways relevant to age-related disorders. Neurobiol Aging. 2023;132:109–19. 10.1016/j.neurobiolaging.2023.08.009. 10.1016/j.neurobiolaging.2023.08.009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rosenberg PA, Aizenman E. Hundred-fold increase in neuronal vulnerability to glutamate toxicity in astrocyte-poor cultures of rat cerebral cortex. Neurosci Lett. 1989;103(2):162–8. 10.1016/0304-3940(89)90569-7 [DOI] [PubMed] [Google Scholar]
- 14.Davis CH, Kim KY, Bushong EA, Mills EA, Boassa D, Shih T, Kinebuchi M, Phan S, Zhou Y, Bihlmeyer NA, Nguyen JV, Jin Y, Ellisman MH, Marsh-Armstrong N. Transcellular degradation of axonal mitochondria. Proc Natl Acad Sci U S A. 2014;111(26):9633–8. 10.1073/pnas.1404651111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kanekar S, Hirozanne T, Terracio L, Borg TK. Cardiac fibroblasts form and function. Cardiovasc Pathol. 1998;7(3):127–33. 10.1016/s1054-8807(97)00119-1. 10.1016/s1054-8807(97)00119-1 [DOI] [PubMed] [Google Scholar]
- 16.Plikus MV, Wang X, Sinha S, Forte E, Thompson SM, Herzog EL, Driskell RR, Rosenthal N, Biernaskie J, Horsley V. Fibroblasts: origins, definitions, and functions in health and disease. Cell. 2021;184(15):3852–72. 10.1016/j.cell.2021.06.024. 10.1016/j.cell.2021.06.024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tian G, Ren T. Mechanical stress regulates the mechanotransduction and metabolism of cardiac fibroblasts in fibrotic cardiac diseases. Eur J Cell Biol. 2023;102(2):151288. 10.1016/j.ejcb.2023.151288. 10.1016/j.ejcb.2023.151288 [DOI] [PubMed] [Google Scholar]
- 18.Buchakjian MR, Kornbluth S. The engine driving the ship: metabolic steering of cell proliferation and death. Nat Rev Mol Cell Biol. 2010;11(10):715–27. 10.1038/nrm2972. 10.1038/nrm2972 [DOI] [PubMed] [Google Scholar]
- 19.Grieves JL, Dick EJ Jr, Schlabritz-Loutsevich NE, Butler SD, Leland MM, Price SE, Schmidt CR, Nathanielsz PW, Hubbard GB. Barbiturate euthanasia solution-induced tissue artifact in nonhuman primates. J Med Primatol. 2008;37(3):154–61. 10.1111/j.1600-0684.2007.00271.x. 10.1111/j.1600-0684.2007.00271.x [DOI] [PubMed] [Google Scholar]
- 20.Noble M, Mayer-Proschel M. Culture of astrocytes, oligodendrocytes, and O-2A progenitor cells. G. Banker, K. Goslin (Eds.), Culturing Nerve Cells, MIT Press, Cambridge, Massachusetts;1998, pp.499–54
- 21.Lin DT, Wu J, Holstein D, Upadhyay G, Rourk W, Muller E, Lechleiter JD. Ca2+ signaling, mitochondria and sensitivity to oxidative stress in aging astrocytes. Neurobiol Aging. 2007;28(1):99–111. 10.1016/j.neurobiolaging. 10.1016/j.neurobiolaging [DOI] [PubMed] [Google Scholar]
- 22.Blondel B, Roijen I, Cheneval JP. Heart cells in culture: a simple method for increasing the proportion of myoblasts. Experientia. 1971;27(3):356–8. 10.1007/BF02138197. 10.1007/BF02138197 [DOI] [PubMed] [Google Scholar]
- 23.Seglen PO. Preparation of isolated rat liver cells. Methods Cell Biol. 1976;13:29–83. 10.1016/s0091-679x(08)61797-5. 10.1016/s0091-679x(08)61797-5 [DOI] [PubMed] [Google Scholar]
- 24.Lee SM, Schelcher C, Demmel M, Hauner M, Thasler WE. Isolation of human hepatocytes by a two-step collagenase perfusion procedure. J Vis Exp. 2013;79:50615. 10.3791/50615. 10.3791/50615 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Franke K, Clarke GD, Dahnke R, Gaser C, Kuo AH, Li C, Schwab M, Nathanielsz PW. Premature brain aging in baboons resulting from moderate fetal undernutrition. Front Aging Neurosci. 2017;9:92. 10.3389/fnagi.2017.00092. 10.3389/fnagi.2017.00092 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Kuo AH, Li C, Li J, Huber HF, Nathanielsz PW, Clarke GD. Cardiac remodelling in a baboon model of intrauterine growth restriction mimics accelerated ageing. J Physiol. 2017;595(4):1093–110. 10.1113/JP272908. 10.1113/JP272908 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Choi J, Li C, McDonald TJ, Comuzzie A, Mattern V, Nathanielsz PW. Emergence of insulin resistance in juvenile baboon offspring of mothers exposed to moderate maternal nutrient reduction. Am J Physiol Regul Integr Comp Physiol. 2011;301(3):R757–62. 10.1152/ajpregu.00051.2011. 10.1152/ajpregu.00051.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Eugster AK, Kalter SS. Viral susceptibility of some in vitro cultured tissues from baboons (Papio sp.). Arch Gesamte Virusforsch. 1969;26(3):249–59. 10.1007/BF01242377. 10.1007/BF01242377 [DOI] [PubMed] [Google Scholar]
- 29.Martin LJ, Mahaney MC, Bronikowski AM, Carey KD, Dyke B, Comuzzie AG. Lifespan in captive baboons is heritable. Mech Ageing Dev. 2002;123(11):1461–7. 10.1016/s0047-6374(02)00083-0. 10.1016/s0047-6374(02)00083-0 [DOI] [PubMed] [Google Scholar]
- 30.Bronikowski AM, Alberts SC, Altmann J, Packer C, Carey KD, Tatar M. The aging baboon: comparative demography in a non-human primate. Proc Natl Acad Sci U S A. 2002;99(14):9591–5. 10.1073/pnas.142675599. 10.1073/pnas.142675599 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Vakifahmetoglu-Norberg H, Ouchida AT, Norberg E. The role of mitochondria in metabolism and cell death. Biochem Biophys Res Commun. 2017;482(3):426–31. 10.1016/j.bbrc.2016.11.088. 10.1016/j.bbrc.2016.11.088 [DOI] [PubMed] [Google Scholar]
- 32.Glancy B, Kim Y, Katti P, Willingham TB. The functional impact of mitochondrial structure across subcellular scales. Front Physiol. 2020;11:541040. 10.3389/fphys.2020.541040. 10.3389/fphys.2020.541040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Nguyen BY, Ruiz-Velasco A, Bui T, Collins L, Wang X, Liu W. Mitochondrial function in the heart: the insight into mechanisms and therapeutic potentials. Br J Pharmacol. 2019;176(22):4302–18. 10.1111/bph.14431. 10.1111/bph.14431 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yin F, Sancheti H, Patil I, Cadenas E. Energy metabolism and inflammation in brain aging and Alzheimer’s disease. Free Radic Biol Med. 2016;100:108–22. 10.1016/j.freeradbiomed.2016.04.200. 10.1016/j.freeradbiomed.2016.04.200 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Phillips D, Covian R, Aponte AM, Glancy B, Taylor JF, Chess D, Balaban RS. Regulation of oxidative phosphorylation complex activity: effects of tissue-specific metabolic stress within an allometric series and acute changes in workload. Am J Physiol Regul Integr Comp Physiol. 2012;302(9):R1034–48. 10.1152/ajpregu.00596.2011. 10.1152/ajpregu.00596.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Benard G, Faustin B, Passerieux E, Galinier A, Rocher C, Bellance N, Delage JP, Casteilla L, Letellier T, Rossignol R. Physiological diversity of mitochondrial oxidative phosphorylation. Am J Physiol Cell Physiol. 2006;291(6):C1172–82. 10.1152/ajpcell.00195.2006. 10.1152/ajpcell.00195.2006 [DOI] [PubMed] [Google Scholar]
- 37.Gollihue JL, Norris CM. Astrocyte mitochondria: central players and potential therapeutic targets for neurodegenerative diseases and injury. Ageing Res Rev. 2020;59:101039. 10.1016/j.arr.2020.101039. 10.1016/j.arr.2020.101039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Magistretti PJ, Allaman I. A cellular perspective on brain energy metabolism and functional imaging. Neuron. 2015;86(4):883–901. 10.1016/j.neuron.2015.03.035. 10.1016/j.neuron.2015.03.035 [DOI] [PubMed] [Google Scholar]
- 39.Jakoby P, Schmidt E, Ruminot I, Gutiérrez R, Barros LF, Deitmer JW. Higher transport and metabolism of glucose in astrocytes compared with neurons: a multiphoton study of hippocampal and cerebellar tissue slices. Cereb Cortex. 2014;24(1):222–31. 10.1093/cercor/bhs309. 10.1093/cercor/bhs309 [DOI] [PubMed] [Google Scholar]
- 40.Öz G, DiNuzzo M, Kumar A, Moheet A, Seaquist ER. Revisiting glycogen content in the human brain. Neurochem Res. 2015;40(12):2473–81. 10.1007/s11064-015-1664-4. 10.1007/s11064-015-1664-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Gorski DJ, Petz A, Reichert C, Twarock S, Grandoch M, Fischer JW. Cardiac fibroblast activation and hyaluronan synthesis in response to hyperglycemia and diet-induced insulin resistance. Sci Rep. 2019;9(1):1827. 10.1038/s41598-018-36140-6. 10.1038/s41598-018-36140-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sathe S, Chan XQ, Jin J, Bernitt E, Döbereiner HG, Yim EKF. Correlation and comparison of cortical and hippocampal neural progenitor morphology and differentiation through the use of micro- and nano-topographies. J Funct Biomater. 2017;8(3):35. 10.3390/jfb8030035. 10.3390/jfb8030035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Singh S, White FC, Bloor CM. Myocardial morphometric characteristics in swine. Circ Res. 1981;49(2):434–41. 10.1161/01.res.49.2.434. 10.1161/01.res.49.2.434 [DOI] [PubMed] [Google Scholar]
- 44.Phillips D, Aponte AM, Covian R, Neufeld E, Yu ZX, Balaban RS. Homogenous protein programming in the mammalian left and right ventricle free walls. Physiol Genomics. 2011;43(21):1198–206. 10.1152/physiolgenomics.00121.2011. 10.1152/physiolgenomics.00121.2011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Tucker NR, Chaffin M, Fleming SJ, Hall AW, Parsons VA, Bedi KC Jr, Akkad AD, Herndon CN, Arduini A, Papangeli I, Roselli C, Aguet F, Choi SH, Ardlie KG, Babadi M, Margulies KB, Stegmann CM, Ellinor PT. Transcriptional and cellular diversity of the human heart. Circulation. 2020;142(5):466–82. 10.1161/CIRCULATIONAHA.119.045401. 10.1161/CIRCULATIONAHA.119.045401 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bernal-Ramirez J, Díaz-Vesga MC, Talamilla M, Méndez A, Quiroga C, Garza-Cervantes JA, Lázaro-Alfaro A, Jerjes-Sanchez C, Henríquez M, García-Rivas G, Pedrozo Z. Exploring functional differences between the right and left ventricles to better understand right ventricular dysfunction. Oxid Med Cell Longev. 2021;28(2021):9993060. 10.1155/2021/9993060. 10.1155/2021/9993060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Dos Santos Lacerda D, Türck P, Gazzi de Lima-Seolin B, Colombo R, Duarte Ortiz V, PolettoBonetto JH, Campos-Carraro C, Bianchi SE, Belló-Klein A, Linck Bassani V, da Rosa Sander, Araujo A. Pterostilbene reduces oxidative stress, prevents hypertrophy and preserves systolic function of right ventricle in cor pulmonale model. Br J Pharmacol. 2017;174(19):3302–14. 10.1111/bph.13948. 10.1111/bph.13948 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Schlüter KD, Kutsche HS, Hirschhäuser C, Schreckenberg R, Schulz R. Review on chamber-specific differences in right and left heart reactive oxygen species handling. Front Physiol. 2018;9:1799. 10.3389/fphys.2018.01799. 10.3389/fphys.2018.01799 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Park WH. The effects of exogenous H2O2 on cell death, reactive oxygen species and glutathione levels in calf pulmonary artery and human umbilical vein endothelial cells. Int J Mol Med. 2013;31(2):471–6. 10.3892/ijmm.2012.1215. 10.3892/ijmm.2012.1215 [DOI] [PubMed] [Google Scholar]
- 50.Graham NA, Tahmasian M, Kohli B, Komisopoulou E, Zhu M, Vivanco I, Teitell MA, Wu H, Ribas A, Lo RS, Mellinghoff IK, Mischel PS, Graeber TG. Glucose deprivation activates a metabolic and signaling amplification loop leading to cell death. Mol Syst Biol. 2012;26(8):589. 10.1038/msb.2012.20. 10.1038/msb.2012.20 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Leiser SF, Salmon AB, Miller RA. Correlated resistance to glucose deprivation and cytotoxic agents in fibroblast cell lines from long-lived pituitary dwarf mice. Mech Ageing Dev. 2006;127(11):821–9. 10.1016/j.mad.2006.08.003. 10.1016/j.mad.2006.08.003 [DOI] [PubMed] [Google Scholar]
- 52.Smirnova L, Harris G, Leist M, Hartung T. Cellular resilience. ALTEX - Altern Animal Experimentation. 2015;32(4):247–60. [DOI] [PubMed] [Google Scholar]
- 53.Lytton J, Westlin M, Hanley MR. Thapsigargin inhibits the sarcoplasmic or endoplasmic reticulum Ca-ATPase family of calcium pumps. J Biol Chem. 1991;266(26):17067–71. 10.1016/S0021-9258(19)47340-7 [DOI] [PubMed] [Google Scholar]
- 54.Szegezdi E, Logue SE, Gorman AM, Samali A. Mediators of endoplasmic reticulum stress-induced apoptosis. EMBO Rep. 2006;7(9):880–5. 10.1038/sj.embor.7400779. 10.1038/sj.embor.7400779 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Bravo R, Parra V, Gatica D, Rodriguez AE, Torrealba N, Paredes F, Wang ZV, Zorzano A, Hill JA, Jaimovich E, Quest AF, Lavandero S. Endoplasmic reticulum and the unfolded protein response: dynamics and metabolic integration. Int Rev Cell Mol Biol. 2013;301:215–90. 10.1016/B978-0-12-407704-1.00005-1. 10.1016/B978-0-12-407704-1.00005-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.de la Cadena SG, Hernández-Fonseca K, Camacho-Arroyo I, Massieu L. Glucose deprivation induces reticulum stress by the PERK pathway and caspase-7- and calpain-mediated caspase-12 activation. Apoptosis. 2014;19(3):414–27. 10.1007/s10495-013-0930-7. 10.1007/s10495-013-0930-7 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data presented in the work are available from the corresponding author upon request.