Abstract
Allergies are a common health issue affecting many people around the world, especially in developed countries. They occur when the immune system overreacts to substances that are usually harmless. Some common allergic conditions include asthma, sinus infections, skin rashes, food allergies, hay fever, severe allergic reactions, eczema, swelling, and reactions to medications or insect stings. The causes of these allergies are complex and often linked to genetics, which can lead to heightened immune responses known as atopy. Throughout history, plant extracts have been used for various purposes, including medicine and food. In addition, their bioactive compounds show a wide range of beneficial effects, such as reducing allergic reactions, fighting oxidative stress, mast cell stabilizers, and lowering inflammation, highlighting their potential for treating various health conditions. Flavonoids and phenolic compounds are commonly used in anaphylaxis for their potent anti-inflammatory action. This review aims to promote the use of natural products as potential treatments for anaphylaxis. In addition, the discovery of new drugs derived from natural sources holds significant promise for the management of anaphylaxis.
Keywords: Natural products, Mast cells, Anaphylaxis, Allergy, Inflammatory mediators
Introduction
Anaphylaxis is a serious and potentially life-threatening allergic reaction that poses a significant challenge for individuals who are sensitive to allergens, as well as for their caregivers. Characterized by its sudden onset and rapid progression, anaphylaxis requires prompt recognition, swift intervention, and meticulous management to reduce its severe consequences (Karunarathna et al. 2024). Anaphylaxis can be triggered by a variety of allergens, including foods, insect stings, and medications (Worm et al. 2014). The pathophysiology of anaphylaxis primarily involves immunoglobulin E (IgE) antibodies that bind to specific allergens. This binding activates mast cells and basophils, leading to the release of inflammatory mediators such as histamine, leukotrienes, and cytokines. These mediators cause various physiological effects, including bronchoconstriction, vasodilation, increased vascular permeability, and ultimately result in symptoms such as difficulty breathing, skin flushing, hives, and a drop in blood pressure (Cingi et al. 2020).
Where the serious nature of anaphylaxis and its potential for rapid progression to life-threatening conditions are concerned, immediate recognition and intervention are crucial. Continued research into the mechanisms underlying anaphylaxis and the identification of effective treatments is essential for improving patient outcomes. Given the high prevalence and limitations of current treatments for anaphylaxis, there is an urgent need to discover new effective therapies. In recent decades, natural products have emerged as key resources for drug discovery due to their structural and chemical diversity, as well as their wide range of biological activities (Farhan et al. 2024; Liu et al. 2024). Numerous studies have demonstrated that natural products possess significant pharmacological properties, including immunomodulatory, anti-allergic, anti-inflammatory, and antioxidant effects (Chen et al. 2020; Farhan et al. 2024; Fonacier et al. 2024). These findings have spurred interest in the investigation and application of natural products for treating allergic diseases. However, there is a notable lack of reviews addressing the progress of natural products in anaphylaxis management. This review aims to highlight recent advancements in understanding the emerging pathogenesis of anaphylaxis. In addition, we provide a summary of the therapeutic effects and underlying mechanisms of natural phytochemicals, mainly polyphenolics, on anaphylaxis, along with pathophysiology and current treatments.
Causes and triggers of anaphylactic reaction
Anaphylaxis may be caused by several allergies, including specific foods, drugs, latex, and insect bites. The European Anaphylaxis Registry indicates that food, particularly eggs, cow’s milk, and nuts, is the primary trigger of anaphylaxis in children and adolescents (DuToit et al. 2024). Reactions to insect venom have also been observed in young adulthood. Although reactions to the venoms of snakes, other invertebrates, and cold-blooded vertebrates have also been documented, hymenopteran (bee and wasp) stings are the most common cause in North America and Europe (Stevens et al. 2023).
Important factors precipitating anaphylaxis involve not only allergen-related ones including its kind, amount, and physical and chemical stability, but also patient-related (endogenous) and environmental (exogenous) factors (Rossi et al. 2022; Cardona et al. 2020). Identifying the causes and risk factors for anaphylaxis is critical for preventing recurrence and educating patients on how to avoid known allergen exposures (Cimen et al. 2023).
According to European Registry research, the main risk factor is becoming older, and this seems to be linked to increased mast cell degranulation (Worm et al. 2018). Female sex has historically been thought to be a risk factor for food allergies and asthma as a result of enhanced mast cell activation and allergic sensitization caused by biological sex-specific hormones. Nevertheless, recent research has shown that anaphylaxis in both adults and children is more often caused by male sex (Pastorello et al. 2021).
Furthermore, the clinical progression and risk factors for anaphylaxis in children may differ from those in adults. Recent globalization has augmented the number of potential triggers, especially due to the worldwide distribution of food, the advancement of novel pharmaceutical substances, and the impact of climate change on regional insect populations. In adults, the most often reported triggering stimuli include medicines (35%), food (32%), insect venom (19%), and idiopathic (14%). In children, the predominant causes are associated with food (85%), followed by idiopathic (11%) and insect venom (4%) (Manchanda & Das 2023).
Pharmaceutical interventions such as antibiotics, monoclonal antibodies, non-steroidal anti-inflammatory medicines (NSAIDs), chemotherapeutic agents, and radiocontrast media are among the most often linked pharmaceuticals with anaphylaxis. Common perioperative triggers include neuromuscular blocking drugs, opioids, antibiotics, latex, and blood product transfusions (Regateiro et al. 2020).
Cofactors that exacerbate clinical symptoms and elevate the likelihood of poor consequences encompass infectious diseases, exercise, underlying mast cell disorders, stress, active allergic conditions such as asthma, advanced age, consumption of specific medications, delayed or omitted administration of adrenaline, and a history of prior anaphylaxis (DuToit et al. 2024).
Clinical presentation and symptoms
The presenting features of anaphylaxis can vary among patients, influenced by factors such as the triggering agent, timing of presentation, patient age, co-morbid conditions, and concurrent medication use (Dribin et al. 2021). Anaphylaxis signs and symptoms generally appear within minutes to 2 h following allergen exposure, food allergens frequently provoke symptoms within 30 min, while drug and insect stings induce more rapid reactions. Delayed anaphylaxis responses (occur within 3 to 5 h) following the consumption of red mead have recently been reported in patients with IgE specific for galactose alpha-1,3-galactose (alpha-gal) (Comberiati et al. 2024).
Anaphylaxis can impact multiple organ systems, primarily through the secretion of mediators from mast cells and basophils. This is particularly applicable to the tissues of the skin, pulmonary system, cardiac system, central nervous system, and gastrointestinal system. The circulatory system is significantly engaged during moderate anaphylactic reactions and is crucial in severe instances, where anaphylactic shock may occur (Martínez-Fernandez et al. 2019).
Approximately, 90% of the individuals are estimated to develop cutaneous symptoms, which may include urticaria, angioedema, flushing, and pus. Dermatological manifestations in a patient suspected of anaphylaxis serve as a useful, but not definitive, diagnostic indicator. Multiple organ systems may be concurrently involved. Atypical symptoms, including substernal chest pain, headache, or seizure, may infrequently manifest during anaphylaxis. The variability in anaphylaxis presentation can complicate diagnosis (Soucy and Michaud 2023). Common symptoms of anaphylaxis reaction are presented in Table 1 (Tomasiak-Łozowska et al. 2018).
Table 1.
Common symptoms of anaphylaxis reaction
| Skin (80–90%) | Itching, conjunctival congestion, maculopapular rash, angioedema, lip, tongue swelling, erythema, and urticaria |
|---|---|
| Respiratory system (70%) | Rhinorrhea, nasal itching, wheezing, stridor, sneezing, cough, dyspnea, throat tightness, tachypnea, and respiratory arrest |
| Gastrointestinal system (45%) | Pain in abdomen, difficulty swallowing, nausea, diarrhea, and vomiting |
| Central nervous system (15%) | Confusion, altered mental status, dizziness, tubular vision, and headache |
| Cardiovascular system (45%) | Arrhythmias, chest pain/tightness, cardiac arrest, hypotension, shock, and palpitations |
Pathophysiology
Anaphylaxis is generally a multiorgan event that engages various effector cells, such as mast cells, basophils, neutrophils, macrophages, and platelets. Anaphylaxis can be classified mechanistically into three categories: immunologic, nonimmunologic, and idiopathic (Bilò et al. 2021).
The idiopathic category is characterized by an unknown allergen or underlying mastocytosis, a clonal mast cell disorder. Approximately, 30–60% of the patients experiencing anaphylaxis may lack an obvious etiological trigger, leading to the classification of their condition as idiopathic anaphylaxis, which is ultimately a diagnosis of exclusion. Recently, expressions such as anaphylactoid and pseudo-allergic responses have gradually been supplanted with mechanism-based descriptions (Martínez-Fernandez et al. 2019; Nguyen et al. 2021).
Immunologic anaphylaxis can be classified into immunoglobulin E (IgE)-mediated forms, such as those triggered by food, drugs, and insect stings, and IgE-independent forms, which encompass immunoglobulin G (IgG)-dependent anaphylaxis, exemplified by high molecular weight iron dextran and the infusion of human monoclonal antibodies like infliximab, as well as complement-mediated anaphylaxis, including over sulfated chondroitin sulfate-contaminated heparin and polyethylene glycols. Chemotherapy can elicit mixed responses involving both IgE- and non-IgE-mediated mechanisms (Dribin et al. 2023).
Immunoglobulin E-mediated pathway
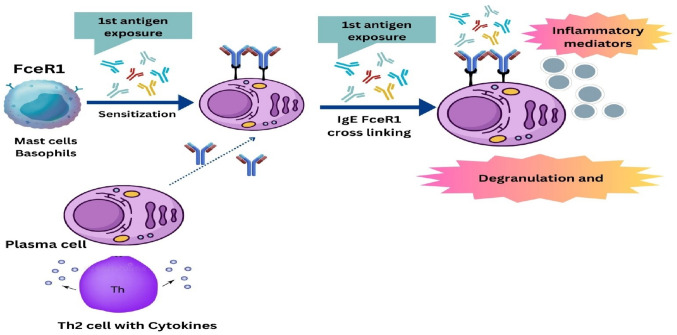
Reactions mediated by IgE to allergens are a recognized cause of anaphylaxis. These reactions entail the binding of IgE to the FceR1 receptor present on the surface of basophils and mast cells. Furthermore, exposure to a bi- or multivalent allergen leads to the cross-linking of Fcer1-bound IgE, which activates mast cells and basophils (Krishnaswamy 2021). This induces the immediate release of stored chemical mediators and also results in the de novo synthesis of additional inflammatory mediators as illustrated by Fig. 1. The chemical mediators include lipoxygenases, tryptase, carboxypeptidase A, proteoglycans, and histamine. Through the activation of cyclooxygenases, phospholipase A, and arachidonic acid, metabolites are produced which include leukotrienes, platelet-activating factors and prostaglandins (Jimenez-Rodriguez et al. 2018; Valenta et al. 2018).
Fig. 1.
IgE-mediated anaphylaxis
The inflammatory response is subsequently regulated by tumor necrosis factor (TNF-alpha), a late-phase reactant that has already been produced. Detailed explanations of the physiology of these chemical mediators are provided as follows (McLendon & Sternard 2024):
Histamine enhances vascular permeability and induces vasodilation, resulting in tissue hypoperfusion. The body reacts to these alterations by elevating heart rate and enhancing cardiac contraction.
Prostaglandin D acts as a bronchoconstrictor, concurrently restricting cardiac and pulmonary arteries. It also enhances peripheral vasodilation, leading to the hypoperfusion of essential organs.
Leukotrienes contribute to bronchoconstriction, increase vascular permeability, and promote airway remodeling.
The platelet activation factor functions as a bronchoconstrictor and enhances vascular permeability TNF-alpha stimulates neutrophils, contributing to stress-induced leukocytosis, and enhances chemokine production.
Nonetheless, IgE levels alone are insufficient to account for an individual’s risk of developing anaphylaxis. Individuals exhibiting nearly undetectable levels of specific IgE may still undergo near-fatal anaphylaxis. On the other hand, certain patients may not exhibit allergic reactions despite the presence of elevated levels of allergen-specific antibodies upon exposure to the allergen. Consequently, an alternative IgE-independent pathway for anaphylaxis must exist (Ebo et al. 2019) (Nuñez-Borque et al. 2022).
Immunoglobulin G-mediated pathway (IgE-independent pathway)
While allergies and anaphylaxis are traditionally attributed to IgE antibodies in humans, there is growing evidence that, in specific situations, IgG-dependent mechanisms may also play a role in these responses (Godon et al. 2021). Research conducted on humans and animal studies indicate that IgG, whether alone or in immune complexes, is capable of activating mast cells and either initiate or exacerbate the severity of anaphylactic reactions. IgG antibodies interact with Fc gamma receptors (FcgRs), which exhibit varying affinities and are present on various cell types (Cianferoni 2021).
Among numerous receptor categories implicated in activation signaling, FcgRI is the sole receptor that exhibits high-affinity binding to monomeric IgG, specifically IgG1 and IgG3. It is expressed on both mast cells and neutrophils. FcgRI binding to particular IgG1 may induce mast cell activation. Both IgG1 and IgG3 are capable of activating FcgRI present on macrophages and monocytes. While FcgRI receptors are often bound by monomeric IgG, this does not inhibit their activation by IgG immune complexes. The latter has greater binding affinity and can thus displace monomeric IgG, so eliciting hypersensitivity responses (Kow et al. 2019).
Mouse models’ researchers demonstrate that antigens can stimulate basophils, neutrophils, and macrophages to release platelet-activating factors (PAF) through the activation of FcgRIII or FcgRIV. These models indicate that IgG necessitates elevated levels of specific IgG antibodies and antigens, unlike IgE-mediated anaphylaxis. This is probably attributable to the reduced affinity of FcgRs in comparison to FcεRI (Fowler & Lieberman 2022).
Furthermore, murine studies have shown that blocking IgG response may be induced by low-dose antigens, which can prevent IgE-mediated anaphylaxis. Conversely, large concentrations of antigens have the potential to induce IgG-mediated anaphylaxis (Jimenez-Rodriguez et al. 2018). Possible instances of IgG-mediated anaphylaxis encompass reactions to the infusion of biological agents such as dextrans, infliximab, aprotinin, intravenous immunoglobulin in individuals with IgA deficiency, Von Willebrand factor infusions, along with various other medicines (Jimenez-Rodriguez et al. 2018; Jönsson et al. 2019).
Complement activation pathway
The ultimate pathway associated with the immunological mechanisms underlying anaphylaxis is the activation of complement. The start of the complement cascade is triggered by a variety of stimuli, leading to the production of C3a, C4a, and C5a. The mediators are known as anaphylatoxins (Fowler & Lieberman 2022). A multitude of evidence indicates the synthesis of C3a and C5a, alongside the reduction of complement levels in instances of human anaphylaxis. This indicates that these anaphylatoxins may contribute to the occurrence of anaphylaxis. Upon the activation of the complement system, the release of C3a and C5a occurs, which in turn initiates the activation of basophils, endothelial cells, mast cells, and smooth muscle cells through their specific receptors (Stevens et al. 2023).
How to manage anaphylaxis
Triage
Allergic reactions demand prompt triage due to the potential for rapid escalation to anaphylaxis, a life-threatening condition. Early identification and timely intervention are pivotal in mitigating severe consequences. Patients experiencing anaphylaxis must be urgently transported to a medical facility, where comprehensive care begins even during ambulance transit. Upon hospital admission, continuous monitoring of vital parameters, including electrocardiogram, pulse, and blood pressure, is essential to assess the severity of the reaction, detect shock, and evaluate the risk of biphasic anaphylactic responses (Bock et al. 2007; Carter et al. 2020).
Airway management
Establishing and maintaining a patent airway is the paramount priority in managing anaphylaxis. Clinical indicators of airway compromise are perioral edema, angioedema, and stridor. In cases of impending airway compromise, immediate definitive airway management, including endotracheal intubation, is critical to avert a failed intubation due to progressive edema. Delay increases the likelihood of requiring emergent surgical interventions, such as cricothyrotomy. In addition, administering 100% high-flow oxygen is imperative for patients presenting with cardiovascular or pulmonary symptoms to optimize tissue oxygenation (Sturm et al. 2018).
Source removal and decontamination
Following airway stabilization, identifying and eliminating exposure to the inciting allergen is essential to halt progression. While gastrointestinal decontamination methods like gastric lavage are generally contraindicated due to inefficacy and the potential to delay other critical interventions, the rapid removal of external allergens remains a cornerstone of management (Gunasena & Jayawardana 2024).
Hemodynamic support and fluid resuscitation
Early administration of intravenous fluids, in conjunction with the initial dose of adrenaline, is critical in patients experiencing cardiovascular compromise, as effective vasopressor activity may depend on adequate intravascular volume. Crystalloids are the preferred fluid choice, administered in boluses of 10 mL/kg (up to 500 mL) for children or 500 mL for adults, with repeated doses guided by clinical response. In severe cases with respiratory involvement, fluid resuscitation may be necessary after a second intramuscular dose of adrenaline (Muraro et al. 2022). Evidence, such as the findings from Ruiz-Garcia et al., highlights reversible cardiovascular dysfunction and associated gastrointestinal symptoms through early fluid administration. Intravenous fluids, therefore, play a vital role in stabilizing distributive shock and improving clinical outcomes (Ruiz-Garcia et al. 2021). In cases of anaphylaxis leading to distributive shock, fluid resuscitation is often effective. For adults, administer 1 to 2 L of isotonic saline, and for pediatric patients, deliver 10–20 mL/kg of isotonic fluids to address hypotension (Gunasena & Jayawardana 2024).
Pharmacological approaches to treat anaphylaxis
Adrenaline
Epinephrine is the cornerstone and the first-line treatment option in anaphylaxis management, exerting potent effects on cardiovascular and respiratory stabilization via vasoconstriction, bronchodilation, and reduction of mucosal edema. Umasunthar et al. (2013) reported that initial treatment of anaphylaxis is stronger than the evidence base for the use of antihistamines and glucocorticoids in anaphylaxis (Dodd et al. 2021). It should be administered promptly following allergen removal and patient assessment, per established emergency protocols (Bilò et al. 2020). The initial adult dose is 0.3–0.5 mL of 1:1,000 concentration, while the pediatric dose is 0.01 mg/kg (up to 0.15 mg). Doses may be repeated every 5–10 min with refractory patients by intravenous infusion under continuous hemodynamic monitoring (Gunasena and Jayawardana 2024).
Route of administration
Intramuscular administration into the vastus lateralis is preferred for its superior bioavailability and rapid systemic absorption (Dreborg and Kim 2021) compared to subcutaneous or deltoid injection sites (Simons et al. 2001). Evidence indicates that intramuscular adrenaline achieves higher plasma concentrations compared to administration via a metered-dose inhaler (Breuer et al. 2013). In addition, while studies comparing intramuscular and subcutaneous adrenaline are influenced by injection site variability, they consistently demonstrate superior plasma adrenaline levels with the intramuscular route (Simons et al. 2001).
Special considerations and training
Intramuscular adrenaline remains the preferred route due to its safety profile and effectiveness; however, intravenous adrenaline may be necessary in exceptional scenarios such as refractory respiratory or cardiovascular compromise (Muraro et al. 2022). Healthcare professionals must be adequately trained in the administration of intramuscular adrenaline and its timely use, especially in patients with a history of life-threatening reactions. Furthermore, patients on beta-blockers may exhibit diminished responsiveness to adrenaline, underscoring the need for vigilant monitoring and individualized management strategies. For optimal outcomes, adrenaline should be supplemented with intravenous fluids in severe presentations and administered without delay. The clinical environment, such as emergency or critical care settings, plays a crucial role in enabling safe and effective use of intravenous adrenaline (Turner et al. 2021).
Efficacy and challenges of adrenaline in biphasic reaction
A 2020 systematic review by the European Academy of Allergy and Clinical Immunology (EAACI) assessed observational studies on adrenaline’s efficacy in critical outcomes, including mortality (De Silva et al. 2020). The findings underscored that timely and sufficient adrenaline administration leads to symptom resolution (Grabenhenrich et al. 2018), while delays are linked to prolonged reactions, hypotension, and fatalities. Although fatalities from anaphylaxis are rare (Nassiri et al. 2015), severe reactions are unpredictable, necessitating the treatment of all anaphylaxis episodes as potentially life-threatening. In the UK, approximately, one-third of the food-induced anaphylaxis deaths occur despite prompt adrenaline administration, often due to severe reactions requiring multiple doses (Patel et al. 2021). Around 10% of the anaphylaxis cases show inadequate response to a single dose, with most resolving after additional doses. Notably, anaphylaxis may recur hours later without re-exposure to allergens (biphasic reaction). A meta-analysis of 27 studies (2,758 patients) reported a 5% biphasic reaction rate, with no significant impact of adrenaline on reducing this risk. These findings highlight the need for vigilance and repeated interventions in severe cases (Lee et al. 2015, p. 45).
Early adrenaline administration: best practices and risks
Early administration of adrenaline is crucial upon anaphylactic signs and symptoms appear (ASCIA 2024). It is generally recommended to administer adrenaline as soon as anaphylaxis symptoms become evident, as delays exceeding 30 min are associated with a higher risk of biphasic reactions (Liu 2020). The intramuscular (IM) route is the preferred method for initial adrenaline administration (Panesar et al. 2013) due to its favorable safety profile, including for patients with cardiovascular comorbidities (Cardona et al. 2020). Conversely, intravenous (IV) adrenaline should be reserved for perioperative settings and administered only by experienced clinicians as an infusion rather than a bolus (Simons et al. 2014). A systematic review revealed that IV bolus administration increased the risks of adrenaline overdose and cardiovascular events compared to IM administration (Campbell et al. 2015). Excessive adrenaline doses, particularly via the IV route, can lead to severe complications such as tachyarrhythmias, hypertension, myocardial infarction, and stroke. However, in cases of imminent cardiac arrest, a bolus dose of IV or intraosseous adrenaline may be warranted (McLure et al. 2021).
Optimal adrenaline dosing
Crossover randomized controlled trials (RCTs) have demonstrated that 300 µg of IM adrenaline for children and 500 µg for adolescents and adults exhibit favorable absorption profiles (Simons et al. 2002). Adrenaline auto-injector recommendations include 0.15 mg for children weighing 7.5 kg, 0.3 mg for those weighing 25 to 30 kg, and at least 0.3 mg for adolescents (Patel et al. 2020) and adults for more favorable absorption profile (Duvauchelle et al. 2018). Limited data exist for children under 15 kg, though healthcare settings commonly administer a dose of 0.01 mg/kg (Muraro et al. 2007). Auto-injectors designed for children as small as 7.5 kg are licensed in some countries, including Germany. However, concerns exist regarding the risk of the needle striking the underlying bone in smaller children. Challenges such as limited availability, high costs, and supply shortages of auto-injectors remain prevalent in many regions (Kim et al. 2014).
Adrenaline auto-injectors
In terms of IM administration, the EAACI 2020 systematic review identified one study in which untrained caregivers were more able to give adrenaline correctly using a prefilled syringe than when using an adrenaline auto-injector (AAI) (Suwan et al. 2018), which reduces administration time by an average of 70 s compared to drawing up manually from an ampoule and results in fewer administration errors. Most AAIs deliver a maximum of 300 µg of epinephrine, although the recommended dose in teenagers and adults is 500 µg, resulting in significant underdosing that may lead to deadly results (Marie 2020). A prior RCT in food-allergic teenagers revealed that a 500 µg dosage of AAI had a more favorable pharmacokinetic and pharmacodynamic profile than 300 µg, without causing a higher rate of systemic adverse events (Patel et al. 2020). As a result, while certain settings may prefer to employ an AAI to provide an initial dose of adrenaline (for speed and convenience), subsequent doses should be administered via needle/syringe to ensure an appropriate dose (Dodd et al. 2021).
Adrenaline auto-injectors are recommended as the primary tool for managing anaphylaxis due to their advantages over traditional needle-syringe methods. These devices are user-friendly, relatively safe, minimize errors, and enable rapid administration. Their utilization in healthcare settings allows patients to familiarize themselves with the process, either by observing or practicing, enhancing their confidence in managing anaphylactic emergencies. They are designed for storage at 20–25℃, and auto-injectors have a limited shelf life due to adrenaline degradation, necessitating regular replacement (Muraro et al. 2022). In severe cases of anaphylaxis, clinical settings may warrant higher doses, such as 0.5 mg or repeated 0.3 mg doses, particularly for older adolescents or adults. This approach ensures effective treatment tailored to the severity of the reaction (Greenhawt et al. 2019).
Inhaled adrenaline
Inhaled adrenaline by a nebulizer concomitantly with oxygen is highly recommended when laryngeal/pharyngeal edema is suspected. But due to the negligible systemic absorption of inhaled adrenaline, IM adrenaline shot should be taken (Hershko et al. 2023). Schlegel et al. (2009) evaluated the utility of including epinephrine inhalers in emergency kits for patients with anaphylaxis and concluded that there was insufficient evidence to add aerosolized epinephrine to emergency kits due to poor delivery to the lungs and low absorption, with the majority of inhaled epinephrine ending up in the oropharynx and being broken down by the GI tract. Furthermore, most children complained about the flavor and reported coughing and dizziness. The findings of this study imply that the potential advantage of inhaled epinephrine (Simons et al. 2000).
Intranasal adrenaline
Intranasal (IN) administration of adrenaline presents a promising alternative to intramuscular (IM) delivery, leveraging the highly vascularized nasal mucosa for rapid systemic absorption (Srisawat et al. 2022). This route may offer significant advantages in terms of patient acceptance, minimizing the psychological distress associated with needle-based administration. In addition, the nasal route is associated with minimal adverse effects and has limited contraindications, such as facial trauma and epistaxis (Bailey et al. 2017). Animal studies have demonstrated the systemic absorption of IN epinephrine with higher plasma concentrations observed for a 5 mg IN dose compared to a 0.3 mg IM dose within the first minute post-administration. These findings highlight its potential for faster onset with reduced risks of tachycardia (Dretchen et al. 2020). Furthermore, human trials have corroborated the efficacy of IN epinephrine in achieving plasma concentrations comparable to IM administration, without significant or sustained adverse effects. However, concerns remain regarding the impact of anaphylaxis-related histamine release, which can induce nasal mucosal edema, potentially impairing the absorption of IN epinephrine. Despite this limitation, the convenience and efficacy of IN administration make it a viable alternative, particularly in scenarios where IM delivery is not feasible or practical (Tuttle et al. 2020).
Sublingual adrenaline
Another conceivable route being studied is sublingual delivery of epinephrine, which must be absorbed by the buccal mucosa in order to enter the bloodstream. Compared to injectable epinephrine, a quickly disintegrating sublingual pill would have a longer shelf life of up to 7 years and enable smaller dosages (Rawas-Qalaji et al. 2013). It was hypothesized that instead of administering a 0.15 mg intramuscular dosage of epinephrine to treat pediatric anaphylaxis, they might be able to utilize a 30 mg sublingual tablet that dissolves quickly. Since oral and sublingual mucosal swelling in anaphylaxis may impact the rate and extent of epinephrine absorption, it will be crucial to observe how the sublingual route compares to intramuscular (IM) injection in human clinical trials and during anaphylactic episodes (Rachid 2013).
Novel approaches to the traditional IM device
Other studies have attempted to solve problems with epinephrine intramuscular delivery, such as patient convenience, mobility, and shelf life with innovative devices. For instance, ZENEO® is a needleless intramuscular device and the most recent experimental substitute for IM auto-injectors. The auto-injector is a prefilled single-use needle-free device currently under development (Alvarez-Perea et al. 2023). The drug is delivered intramuscularly via the device, which propels it at high enough pressure and speed to pass through skin. This portable, needle-free device might improve the outcome by mitigating needle anxiety in patients and parents and possibly making training easier (Sonoda et al. 2023).
Corticosteroids
Their precise effectiveness in anaphylaxis remains uncertain, and they show therapeutic benefit in conditions such as reactive airway diseases. For acute management, intravenous methylprednisolone (80–125 mg) or hydrocortisone (250–500 mg) is administered. Following the acute phase, oral prednisone (40–60 mg daily or divided into two doses) is recommended for 3–5 days. In situations involving unidentified allergens or delayed follow-up, a tapering regimen over 2 weeks may be appropriate. Methylprednisolone and dexamethasone are chosen since they have minimal mineralocorticoids like action abrogating the risk of fluid retention. Corticosteroids are recommended as a third-line treatment for underlying asthma or shock since their major action is to reduce the late-phase inflammatory response (Karunarathna et al. 2024). Given corticosteroids’ sluggish absorption kinetics and mode of action, it is theoretically improbable that they are useful in the acute treatment of anaphylaxis. In contrast, following systematic reviews have reaffirmed the paucity of evidence that corticosteroids lower reaction severity or prevent biphasic reactions (Alqurashi & Ellis 2017).
Corticosteroids are more frequently administered than adrenaline in the acute management of anaphylaxis which may inadvertently delay or distract from the timely administration of adrenaline (Dubus et al. 2019). Corticosteroids may offer therapeutic value in specific scenarios, such as refractory anaphylaxis and anaphylaxis occurring in the setting of poorly controlled asthma. Therefore, incorporating corticosteroids as an adjunctive treatment for refractory anaphylaxis is reasonable, but they should not replace adrenaline or other inotropic or vasopressor agents in the treatment hierarchy (Dodd et al. 2021).
Recent Cochrane reviews have assessed the efficacy and safety of glucocorticoids in acute anaphylaxis management, concluding that there is insufficient evidence to support their routine use in the emergency department or for the prevention of biphasic reactions (Choi et al. 2019). High-dose glucocorticoids (500–1,000 mg in adults) may be considered for patients presenting with generalized urticaria, coexisting asthma, airway edema, or stridor after stabilization (Böhm et al. 2018; Kim et al. 2014). In pediatric patients, prednisolone at a dose of 2 mg/kg can be administered via suppositories, enemas, or injectable formulations as appropriate (Quoc et al. 2021). Table 2 recapitulates the dosage and administration of three common parenteral glucocorticoids: hydrocortisone, methylprednisolone, and dexamethasone.
Table 2.
Dosage and administration of corticosteroid injections (Amir Rawa et al. 2022)
| Agent | Dose | Adults min. dose | Children max. dose | Notes |
|---|---|---|---|---|
|
Hydrocortisone IV or IM |
2–4 mg/kg | 200 mg | 100 mg | Rapid onset, but may contain alcohol, which may be hazardous in the management of anaphylaxis |
|
Methylprednisolone IV or IM |
1–2 mg/kg | 100 mg | 50 mg | Rapid onset, but may not be available at the primary care level |
|
Dexamethasone IV or IM |
0.1–0.4 mg/kg | 20 | 10 | Slow onset, inexpensive, widely available |
Antihistamines
Antihistamines, especially H1 receptor blockers, are commonly utilized to manage allergic reactions, including anaphylaxis. Diphenhydramine, the most widely used H1 blocker, is typically administered at 25–50 mg intravenously or intramuscularly. Besides its role in anaphylaxis, management remains uncertain, and it is effective in addressing milder allergic symptoms. In more severe reactions, combining H1 blockers with H2 receptor antagonists such as ranitidine (50 mg IV over 5 min) or cimetidine (300 mg IV) may provide additional benefit, as histamine affects multiple receptor subtypes. However, cimetidine requires careful use in patients with renal or hepatic dysfunction or those on beta-blockers due to potential drug interactions and side effects. Once the patient is stabilized, transitioning from intravenous to oral antihistamines is advisable if ongoing treatment is needed (Gunasena & Jayawardana 2024). Oral H1-antihistamines, such as dimetindene and clemastine, which are also available in IV formulations, can alleviate cutaneous symptoms of anaphylaxis and rhinoconjunctivitis (Simons et al. 2015).
Antihistamines limitations in allergic reactions including anaphylaxis
The ASCIA 2024 Guidelines reported that antihistamines should not be used as part of the primary emergency treatment for anaphylaxis as they have no role in treating respiratory or cardiovascular symptoms of anaphylaxis (ASCIA 2024). Their primary utility lies in managing cutaneous symptoms often associated with allergic reactions, including anaphylaxis. However, there is no robust evidence from randomized controlled trials (RCTs) to support their use in anaphylaxis, and they do not prevent biphasic reactions (Simons et al. 2015). In addition, H1-antihistamines, particularly when administered rapidly via intravenous bolus, may cause sedation, potentially masking anaphylaxis symptoms, and can precipitate hypotension (Nurmatov et al. 2014). Other reports recommend relegating antihistamines to second- or third-line intervention as a concern for delaying the administration of both initial and subsequent doses of adrenaline (Cardona et al. 2020).
Data from studies, including the Cross-Canada Anaphylaxis Registry (C-CARE), suggest that prehospital antihistamine use may be associated with lower administration rates of multiple adrenaline doses and delayed presentation to healthcare facilities, potentially increasing morbidity. In a large national prospective study, the Cross-Canada Anaphylaxis Registry (C-CARE) analyzed 3,498 cases of anaphylaxis over 6 years. Prehospital use of antihistamines was linked to hospitalization, intensive care admission, and intravenous fluid requirements (Gabrielli et al. 2019). Antihistamines are also ineffective in preventing biphasic reactions (Shaker et al. 2020). Furthermore, data from the European Anaphylaxis Register, which analyzed 9,171 anaphylaxis episodes, revealed a significant link between antihistamine use and a higher incidence of biphasic reactions (Kraft et al. 2020), likely due to the delay in administering adrenaline. Consequently, antihistamines are not recommended for the acute management of anaphylaxis (ASCIA 2024).
Antihistamines: supportive, not primary, in anaphylaxis management
The ASCIA 2020 guideline reported that antihistamines may be useful for persistent skin symptoms after anaphylaxis has been resolved but should only be administered once the acute reaction has been effectively treated with adrenaline and other first-line interventions (Cardona et al. 2020)). A recent meta-analysis suggested that antihistamines and glucocorticoids may help prevent reactions related to chemotherapy but not those triggered by radiocontrast media (Shaker et al. 2020). In addition, antihistamines are effective in reducing reactions during allergen immunotherapy (Roberts et al. 2018).
Misplaced priorities: overuse of corticosteroids and antihistamines
Research indicates that many physicians prioritize corticosteroids and antihistamines as first-line treatments for anaphylaxis, with this practice being even more prevalent among specialists. Studies reveal that these medications are administered more frequently than epinephrine, despite guidelines emphasizing epinephrine as the primary treatment. For instance, in a study involving 1,970 children with anaphylaxis, antihistamines and corticosteroids were used in 76% and 82% of cases, respectively, while epinephrine was utilized in only 28% (Grabenhenrich et al. 2016). Similarly, other studies reported antihistamine usage rates between 81.5% and 93% and corticosteroid administration in 55.5% to 73% of cases, compared to lower epinephrine use (46%–58.3%) (Sidhu et al. 2016). Although corticosteroids and antihistamines play a supportive role in managing refractory symptoms or preventing biphasic reactions, they are adjunctive treatments rather than first-line options (Muraro et al. 2014).
Bronchodilators
Bronchodilators play a vital supportive role in managing anaphylaxis accompanied by bronchospasm, especially in individuals with underlying respiratory conditions like asthma. Inhaled beta-agonists, such as albuterol or a combination of ipratropium bromide and albuterol, are the drugs of choice for wheezing (Gunasena and Jayawardana 2024) but caution that they are ineffective in addressing upper airway obstruction, hypotension, or shock. As a result, their role is strictly as adjunctive therapy rather than a primary treatment option (Cardona et al. 2020). For persistent bronchospasm unresponsive to standard therapy, intravenous magnesium sulfate may be used, adhering to dosing protocols similar to those for severe asthma exacerbations (Gunasena and Jayawardana 2024). In addition, Beta-2 agonists like salbutamol may serve as adjunctive therapy for addressing lower respiratory symptoms and significant bronchial obstruction during anaphylaxis. These should be administered following initial intramuscular adrenaline treatment, ideally using an oxygen-driven nebulizer or a metered-dose inhaler with a spacer for optimal delivery (Muraro et al. 2022). In cases of ongoing respiratory symptoms during anaphylaxis, beta-2 agonists (administered via inhalation or parenterally) should not replace additional parenteral adrenaline treatment. According to guidelines from RCUK (2008), WAO (2011/2020), EAACI (2014), and ASCIA (2024), bronchodilators are emphasized as supplementary therapies to support adrenaline, rather than as substitutes. But in case of mild or moderate respiratory symptoms, beta-2 agonists can be delivered using a metered-dose inhaler (MDI) with a large-volume spacer with no need for oxygen. However, there is limited evidence supporting the use of MDIs with spacers in cases of acute severe or life-threatening respiratory symptoms. In such situations, beta-2 agonists should be administered using an oxygen-driven nebulizer for optimal effectiveness (Payus et al. 2018).
International guidelines agreed that bronchodilators may be helpful for persisting wheeze, but caution that they do not prevent or relieve upper airway obstruction, hypotension or shock, and should, therefore, be used as adjunct treatments. Inhaled beta-2 agonists are commonly utilized as second-line treatments for anaphylaxis, although evidence supporting their effectiveness in this context is limited and largely derived from their use in acute asthma management (Simons et al. 2011). There are anecdotal reports of anaphylaxis first misdiagnosed as severe asthma, which did not respond to parenteral β-2 agonists therapy but did respond to adrenaline (Payus et al. 2018). For this reason, IV salbutamol must not be used in preference to adrenaline for acute anaphylaxis (Dodd et al. 2021).
Vasopressors and glucagon
Vasopressors may be considered for patients needing multiple doses of epinephrine but who develop significant side effects, such as arrhythmias or chest pain, during intravenous epinephrine infusion. Although no specific vasopressor is universally endorsed as a second-line option for anaphylaxis, their use typically aligns with established protocols for treating other types of hypotensive shock (Karunarathna et al. 2024). Parenteral glucagon can provide supportive benefits for patients who do not respond adequately to epinephrine or for those on β-blockers (Parish et al. 2019).
Omalizumab
Omalizumab, an anti-IgE monoclonal antibody, reduces the expression of high-affinity IgE receptors (FcεRI) on basophils, mast cells, and dendritic cells, thereby inhibiting the IgE-mediated signaling cascade (Benito-Villalvilla et al. 2023). It has been widely used in the management of moderate to severe asthma and chronic spontaneous or inducible urticaria (Kabashima et al. 2018). More recently, its applications have expanded to include severe food allergies, eosinophilic gastroenteritis, acute reactions during rush immunotherapy, mast cell disorders, and idiopathic anaphylaxis (Imakiire et al. 2020).
Anaphylaxis management involves both acute interventions to alleviate symptoms and long-term strategies to prevent recurrence. Long-term approaches include avoiding known triggers through specific immunotherapy or modulating the IgE response. In cases of food-related anaphylaxis, anti-IgE therapy has emerged as a promising option. Numerous case series have demonstrated its clinical efficacy when used either as a standalone treatment or in combination with oral immunotherapy (Quoc et al. 2021).
Allergen immunotherapy and drug desensitization
Immunomodulatory approaches should be considered in venom immunotherapy and drug desensitization (Kim et al. 2014). Indeed, patients with VIA show the best response to subcutaneous venom immunotherapy for preventing anaphylaxis in both children and adults (Monsieurs et al. 2015). For drug desensitization, administration of drugs (with increasing doses) can achieve a tolerant state to targeted drug doses. Desensitization can be successful for patients with anaphylaxis induced by chemotherapy, biologics, NSAIDs, and antibiotics (Maris et al. 2021).
Investigational therapeutic agent: sirtuin 6
Sirtuin 6 (SIRT6), a NAD-dependent deacetylase, is being explored as a potential therapeutic target for anaphylaxis. Its mechanism involves inhibiting the transcription of protein tyrosine phosphatase receptor type C, which in turn downregulates the FcERI signaling pathway in mast cells. By suppressing mast cell activation, SIRT6 activation could help mitigate the severity of anaphylactic reactions, offering a promising new approach under investigation for anaphylaxis treatment (Karunarathna et al. 2024).
Emergency response to anaphylaxis
When anaphylaxis is suspected, prompt actions is crucial as shown in Fig. 2. The patient’s airway, breathing, circulation, and mental status should be assessed immediately. If the allergen is identifiable, efforts should be made to remove it. Assistance should be sought from nearby individuals or caregivers, and an emergency call should be placed without delay. Alternatively, the patient should be taken directly to an emergency department for medical care (Amir Rawa et al. 2022). While awaiting the arrival of emergency responders, any suspected allergen should be removed if feasible.
Fig. 2.
Systematic representation for preliminary management of anaphylaxis
In cases of anaphylaxis, individuals should be positioned in supine position to enhance venous return to the heart or seated upright if experiencing respiratory distress. If vomiting occurs, the head should be turned slightly downward, and any material obstructing the airway should be promptly cleared to prevent aspiration. Epinephrine auto-injector should be used if available according to the instructions provided on the packaging (Alsabri et al. 2024). Infants should not be held upright; however, if breathing is compromised, the patient may be allowed to sit to ease respiratory effort (Alsabri et al. 2024). For pregnant patients, positioning on the left side with the bed tilted in a head-down position is recommended to optimize venous return and reduce pressure on the inferior vena cava. Sudden changes to an upright posture should be avoided (Tan et al. 2022).
Epinephrine should be administered immediately after removing the trigger, as it plays a critical role in stabilizing cardiovascular and respiratory functions by enhancing vasoconstriction, peripheral vascular resistance, bronchodilation, and reducing mucosal edema (Böhm et al. 2018). Following epinephrine administration, the patient should be evaluated in accordance with established emergency protocols (Cardona et al. 2020). Timely administration of adrenaline has been shown to decrease the likelihood of biphasic reactions (Liu et al. 2020). Upon hospital arrival, continuous monitoring of vital signs including electrocardiogram (ECG), pulse, and blood pressure is essential to assess the severity of the reaction, detect signs of shock, and evaluate the risk of biphasic reactions (Monsieurs et al. 2015). Patients exhibiting cardiovascular or respiratory involvement should receive high-flow oxygen (100%) to support critical functions (Takazawa et al. 2021). In cases of snake envenomation, administration of antivenom should be promptly initiated as per clinical guidelines (Muraro et al. 2022).
Supportive measures for anaphylaxis
Supportive care is integral to the management of anaphylaxis and achieving favorable patient outcomes. This involves continuous monitoring of vital parameters, including pulse, blood pressure, electrocardiogram (ECG), and oxygen saturation. High-flow oxygen supplementation should be provided to maintain oxygen saturation between 94 and 98% (Arroabarren et al. 2011). In cases of hypotension or inadequate response to initial therapy, intravenous (IV) access should be established, and a rapid fluid bolus of 10 mL/kg administered (Simons 2009). For the symptomatic management of vomiting, diphenhydramine may be given intramuscularly or intravenously at a dose of 1 mg/kg, not exceeding 50 mg per dose (Arroabarren et al. 2011). In addition, ranitidine can be administered orally or intravenously at the same dose (1 mg/kg), with a maximum of 50 mg per dose (Clark et al. 2023).
In cases of respiratory failure or significant respiratory distress caused by airway edema or bronchospasm, advanced airway management should be initiated promptly, either through endotracheal intubation or the insertion of a supraglottic airway device. A tracheotomy may be warranted if intubation proves unsuccessful (Amir Rawa et al. 2022). Concurrently, IM epinephrine should be administered promptly in cases of GRADE II anaphylaxis or higher, using the recommended dosages (Amir Rawa et al. 2022; Kim et al. 2018). If there is no clinical improvement, the dose may be repeated every 5–15 min as needed. For patients classified as GRADE IV, experiencing or at imminent risk of cardio-respiratory arrest, IV bolus epinephrine is indicated (Hammad et al. 2022).
For GRADE II and GRADE III anaphylaxis patients with established venous access and under close monitoring, such as in an ICU or perioperative setting, IV bolus epinephrine may be considered as outlined in Table 3. If there is no clinical improvement within 3–5 min for GRADE IV patients or 1–2 min for GRADE II and III patients, an additional dose of IV bolus epinephrine should be administered (Pouessel et al. 2024).
Table 3.
Recommended doses of IV bolus of epinephrine in anaphylaxis
| Grade | Dose recommended |
|---|---|
| GRADE II | 0.01–0.05 mg for patients ≥ 14 years old; 0.001–0.002 mg/kg (1–2 μg/kg) for patients < 14 years old |
| GRADE III | 0.1–0.2 mg for patients ≥ 14 years old; 0.002–0.01 mg/kg (2–10 μg/kg) for patients < 14 years old |
| GRADE IV | 1 mg for patients ≥ 14 years old; 0.01–0.02 mg/kg for patients < 14 years old |
For those with GRADE IV anaphylaxis, IV epinephrine infusion can commence once stabilization begins, even if cardiopulmonary symptoms persist incompletely resolved. The dose of epinephrine IV infusion should be 3–30 μg/kg/h. During IV epinephrine administration, continuous monitoring of electrocardiogram (ECG), blood pressure (BP), respiratory rate, and oxygen saturation is essential. In cases where bronchospasm persists or stridor develops following epinephrine administration, high-dose nebulized budesonide may be considered as an adjunct treatment. For patients exhibiting circulatory compromise, fluid resuscitation is advised, starting with an initial bolus of 20 mL/kg, with subsequent adjustments based on clinical response. Following appropriate intervention, those patients should undergo hospital monitoring for a minimum of 12 h. Key parameters to observe include heart rate, blood pressure, respiratory rate, oxygen saturation, and urine output (Amir Rawa et al. 2022).
Natural products with anti-anaphylaxis potential
German chamomile extract
The methanolic extract of Matricaria recutita L., German chamomile was found to effectively inhibit mast cell histamine release in a mast cell mediated allergy model in vitro, although more research is required to define its mechanism of action (Chandrashekhar et al. 2011). The pharmacokinetic parameters of quercetin, luteolin, and apigenin after oral administration of M. chamomilla extract in rats were determined, where the results showed that all three compounds were rapidly absorbed, with Tmax values of 0.79 h, 0.42 h, and 0.51 h for quercetin, luteolin, and apigenin, respectively. Luteolin exhibited the highest systemic exposure (Cmax = 3.04 μg/ml, AUC0–∞ = 19.89 μg*h/ml), followed by apigenin (Cmax = 0.42 μg/ml, AUC0–∞ = 5.03 μg*h/ml) and quercetin (Cmax = 0.29 μg/ml, AUC0–∞ = 3.88 μg*h/ml). Despite their rapid absorption, elimination was relatively slow, with half-lives (T1/2) of 13.60 h for quercetin, 4.43 h for luteolin, and 8.82 h for apigenin, suggesting prolonged systemic retention (Dong et al. 2017). However, till the time of this review, the pharmacokinetics of chamomile in humans is not well described (Kimura et al. 2024), requiring further studies in humans to establish clinical relevance.
Impatiens textori extract
The 35% ethanol extract of the flowers of Impatiens textori MIQ. exhibited anti-anaphylactic and anti-pruritic effects. Its key bioactive compounds, including apigenin, apigenin 7-glucoside, and luteolin, were shown to significantly inhibit blood pressure decreases in both IgE-dependent and IgE-independent anaphylaxis models. The extract of the flowers of I. textori also mitigated the reduction in blood flow caused by antigen-induced anaphylaxis in sensitized mice. Beyond anaphylaxis, the extract effectively reduced scratching behavior induced by platelet-activating factor (PAF), serotonin (5-HT), and proteases without central depressant effects (Ueda et al. 2005). However, to date, a literature search for pharmacokinetic studies on I. textori yielded no relevant publications in animal or human models. Thus, future research should focus on elucidating the pharmacokinetics of I. textori to support its safe and effective use in medicinal applications.
Kincung flower extract
The ethanol extract of Etlingera elatior—Kincung flower—was found to suppress allergic reactions by inhibiting mast cell degranulation and lowering serum IL-4 and IgE levels in sensitized mice. Its potential effectiveness in controlling type I hypersensitivity and active cutaneous anaphylaxis is also shown by its reduction of basophil and eosinophil infiltration at inflammatory sites (Elidahanum et al. 2020). However, further studies on the pharmacokinetics, bioavailability, and molecular mechanism of E. elatior are needed (Juwita et al. 2018).
Litsea cubeba essential oil
Litsea cubeba, also known as Makauy, is a traditional herb with diverse therapeutic uses, including anti-anaphylaxis and anti-inflammatory effects. The essential oil of L. cubeba (LCEO), rich in citral (neral and geranial), demonstrated immunosuppressive effects by reducing TNF-α and IL-12 production in dendritic cells (DCs). LCEO also inhibited contact hypersensitivity (CHS) responses and decreased T-cell infiltration in inflammatory sites (Chen et al. 2016; Qian et al. 1980).
Patchouli oil from Pogostemon cablin
Patchouli oil, the essential oil of Pogostemon cablin (Blanco) Benth. (Lamiaceae), and its main component, patchouli alcohol, were found to show anti-allergic action by lowering mast cell degranulation, reducing cytokine generation, and thus modifying dendritic cell responses. Supported by delay in hypersensitivity models and inhibition of passive cutaneous anaphylaxis, their therapeutic effects reach to reducing systemic inflammation and hypersensitivity (Chen et al. 2013; He et al. 2013; Su et al. 2015). The pharmacokinetics of patchouli alcohol, a tricyclic sesquiterpene and a critical bioactive ingredient of patchouli oil, have been extensively studied in several reports, demonstrating a two-compartment distribution with linear elimination kinetics. However, oral bioavailability remains limited due to poor solubility and first-pass metabolism (Hu et al. 2017; Zhang et al. 2016; Zhu et al. 2024a, b).
Clinacanthus nutans extract
Clinacanthus nutans (Burm. f.) Lindau water leaf extract was found to downregulate lipid metabolism and improve propanoate and amino acid pathways in an in-vivo ovalbumin-induced active systemic anaphylaxis model, displaying anti-anaphylactic effects. Supported by 1H-NMR metabolomics approach, these results draw attention to the extract’s overall systemic regulating action on allergic mechanisms (Elgendy 2017). The in-vitro effect of C. nutans leaves methanol extract on the activity of CYP3A4 and CYP2E1 in human liver microsomes was evaluated, showing significant inhibition (Quah et al. 2017).
Isoquercitrin and hyperin flavonoids from persimmon peel
Isoquercitrin and hyperin, isolated from the peel of persimmon, showed anti-anaphylactic action by inhibiting mast cell degranulation, mainly by lowering the intracellular calcium (Ca2⁺) elevation, which is a necessary trigger for histamine release. Their mechanism is direct scavenging of reactive oxygen species (ROS) generated by NADPH oxidase (NOX), so reducing the oxidative stress related with allergic reactions (Itoh et al. 2011). Isoquercetin was also found to have a significant inhibitory effect on histamine release from rat mast cells (Park 2020). These results place isoquercitrin and hyperin as potential candidates for reducing type I allergic reactions by means of their combined antioxidant and anti-inflammatory action. The bioavailability of isoquercitrin was found to be better than quercetin by 2.35 times, and first concentration peak of quercetin conjugates appear 60–90 min after the intake of isoquercitrin, followed by the second, higher peak at 2.5–4 h due to enterohepatic circulation (Valentová et al. 2014).
Spinacetin flavonoid from Inula japonica
Derived from Inula japonica Thunb., spinacetin reduced mast cell activation and passive cutaneous anaphylaxis (PCA) in mice. Spinacetin also reduced the synthesis of inflammatory mediators including leukotriene C4 (LTC4) and interleukin-6 (IL-6) as well as histamine release. Spinacetin was found to interact with signaling pathways including Syk, LAT, PLCγ, MAPKs, and NF-κB. Spinacetin’s oral administration dose-dependently attenuated IgE/Ag-mediated PCA reaction in mouse model (Ji et al. 2018). The computational pharmacokinetic profile of spinacetin showed it can be well absorbed by oral intake, without passing the blood brain barrier or becoming a P-glycoprotein substrate (Jose et al. 2022).
Naringenin
Naringenin, a citrus-derived flavonoid, exhibited anti-anaphylactic activity by targeting immune and inflammatory pathways. Studies have shown that naringenin suppresses the activation and migration of macrophages, key players in allergic inflammation. In addition, it inhibits the production of pro-inflammatory mediators such as MCP-1, NO, and TNF-α in LPS-stimulated RAW 264.7 macrophages (Fang et al. 2010; Hirai et al. 2007). Naringenin exhibited dose-proportional pharmacokinetics in humans, and Tmax occurring at 2.41–3.17 h, while the half-life (t1/2) ranged from 2.65 to 3.0 h, indicating rapid clearance. No adverse effects were observed with a suggested 300 mg twice daily dosing (Rebello et al. 2020).
Luteolin
Flavonoids are widely known for their anti-inflammatory and anti-anaphylactic properties, including luteolin and quercetin derivatives (Middleton et al. 2000). Luteolin, from Folium perillae and Flos lonicerae, was found to reduce phosphodiesterase 4 (PDE-4), thus lowering cyclic AMP (cAMP) hydrolysis and preventing neutrophil adhesion and cytokine release (Jiang et al. 2015). Luteolin is rapidly absorbed following oral intake in rats through the intestine in its aglycone form following hydrolysis of luteolin glucoside by the sodium glucose co-transporter 1 (SGLT1). However, further research is necessary to fully characterize the pharmacokinetic profile of luteolin in humans (Wang et al. 2021a, b).
Wogonin
Wogonin, a flavonoid isolated from Radix scutellariae, exhibits significant anti-anaphylactic effects alongside a broad range of pharmacological activities, including anti-inflammatory and antiviral properties. Screening using a two-dimensional online cell membrane chromatography method revealed that wogonin had specific activity on the epidermal growth factor receptor (EGFR). In-vitro studies demonstrated its dose-dependent inhibition of high EGFR-expressing cell viability, suggesting immunomodulatory properties (Shalaby et al. 2023). However, wogonin displays very low oral bioavailability (1.10%), likely resulting from poor solubility and extensive first-pass metabolism (Hassanin et al. 2019).
Rosmarinic acid
Found in many herbs, rosmarinic acid was found to lower inflammatory markers and to exert anti-anaphylaxis activity in atopic dermatitis, asthma, allergic rhinitis, and ovalbumin (OVA)-induced intestinal allergies. It was found to significantly lower IgE, histamine, and mast cell proteinase levels in serum of OVA challenged mice while raising antioxidant enzyme activity. The ability of rosmarinic acid to balance pro- and anti-inflammatory cytokines emphasizes its therapeutic value in control of food allergies and anaphylaxis (Jia et al. 2023). Upon oral intake, rosmarinic acid is mainly metabolized by the gut microflora into simple phenolic units. Its elimination is mainly through renal excretion, with no serious adverse effects of herbal remedies containing rosmarinic acid reported (Hitl et al. 2020).
Isoxazolines from Xanthoceras sorbifolia
Two isoxazolines isolated from Xanthoceras sorbifolia Bunge fruit husks, 1-oxa-2-azaspiro[4.5]dec-2-ene-8β-ol and 1-oxa-2-azaspiro[4.5]dec-2-ene-8α-ol, showed notable in-silico anti-anaphylaxis potential by phosphodiesterase IV (PDE-4) inhibition, modulating inflammatory pathways involved in anaphylaxis. The binding affinity and stability of these molecules revealed by computational modeling and molecular docking studies exceeded the clinical PDE-4 inhibitor Rolipram (Ge et al. 2016).
Lipoic acid
Lipoic acid was evaluated for its effects on soybean β-conglycinin-induced hypersensitivity in a rat model. Administration of 25 mg/kg lipoic acid significantly reduced β-conglycinin-specific IgE and histamine levels in serum and the jejunum. In addition, lipoic acid supplementation did not affect interferon-γ or interleukin-4 levels, indicating its effects might be independent of typical Th1/Th2 cytokine modulation. These findings highlight its potential as an immunomodulator for managing food-induced hypersensitivity reactions, such as soybean allergies (Han et al. 2010).
Fungal immunomodulatory protein from Lignosus rhinocerotis
A novel fungal immunomodulatory protein (FIP-Lrh) from Lignosus rhinocerotis, commonly known as Tiger Milk mushroom, exhibited promising anti-anaphylactic and immunomodulatory properties. FIP-Lrh was found to strongly bind to glycoproteins such as N-acetylgalactosamine and N-acetylglucosamine, which are crucial for immune cell interactions. Functional studies have confirmed its potential to modulate immune responses, making it a candidate for both preventive and therapeutic applications in managing anaphylaxis and other immune-related disorders (Pushparajah et al. 2016).
Phenanthrenes and stilbenes from Gymnadenia conopsea tubers
The methanolic extracts of the tubers of Gymnadenia conopsea containing stilbenes and phenanthrenes were found to inhibit antigen-induced degranulation by 65.5 to 99.4% at 100 μM in RBL-2H3 cells. Furthermore, the methanolic extract significantly inhibited allergic reactions of ear passive cutaneous anaphylaxis in mice (Matsuda et al. 2004).
Picroliv ididoid glycosides from Picrorhiza kurroa
Picroliv, a standardized iridoid glycoside fraction derived from the root and rhizomes of Picrorhiza kurroa, inhibited passive cutaneous anaphylaxis (PCA) in mice and rats at 25 mg/kg. Though it was not found to directly act as a histamine receptor antagonist, it lowered mast cell degranulation and prevented Schultz–Dale reactions in sensitized guinea pig ileum preparations (Baruah et al. 1998).
Alginic acid
Made from seaweed, alginic acid showed strong anti-anaphylactic action by blocking histamine release and nuclear factor-κB (NF-κB) activation suppression. In mice, experimental studies found that it significantly lowered both systemic and passive cutaneous anaphylaxis. It also reduced the expression of inflammatory cytokines TNF-α and IL-1β in mast cells, so positioning it as a potential natural treatment for anaphylaxis and associated inflammatory diseases (Jeong et al. 2006). Table 4 categorizes the discussed natural products by their chemical nature and summarizes their mechanisms of action and experimental evidence.
Table 4.
Summary of natural products with anti-anaphylaxis potential
| Extracts | References | ||
|---|---|---|---|
| Natural product | Source | Experimental findings | |
| German chamomile extract | Matricaria recutita |
Inhibition of mast cell degranulation Suppressed histamine release in vitro |
(Chandrashekhar et al. 2011) |
| Impatiens textori extract | Impatiens textori MIQ |
Stabilization of mast cells, inhibition of PAF and serotonin effects Reduced BP decrease and scratching behavior in mice |
(Ueda et al. 2005) |
| Kincung flower extract | Etlingera elatior |
IL-4 and IgE suppression, reduction in eosinophil and basophil infiltration Lowered serum IL-4 and IgE in sensitized mice |
(Elidahanum et al. 2020) |
| Litsea cubeba essential oil | Litsea cubeba |
TNF-α and IL-12 suppression, inhibition of T-cell infiltration Reduced CHS responses and dendritic cell activity in mice |
(Chen et al. 2016; Qian et al. 1980) |
| Clinacanthus nutans extract | Clinacanthus nutans |
Downregulation of lipid metabolism, improvement of amino acid pathways Alleviated OVA-induced anaphylaxis |
(Elgendy 2017) |
| Phenolics | |||
|---|---|---|---|
| Natural product | Source | Experimental findings | Reference |
| Isoquercitrin, hyperin | Persimmon peel |
ROS scavenging, inhibition of Ca2⁺ elevation, mast cell degranulation Reduced histamine release in rat mast cells |
(Itoh et al. 2011; J. Park 2020) |
| Spinacetin | Inula japonica |
Inhibition of Syk, LAT, PLCγ, MAPKs, NF-κB, and histamine release Reduced IgE-mediated PCA in mice |
(Ji et al. 2018) |
| Naringenin | Citrus fruits |
Suppression of macrophage activation, cytokine inhibition (MCP-1, NO, TNF-α) Reduced inflammation in LPS-stimulated macrophages |
(Fang et al. 2010; Hirai et al. 2007) |
| Luteolin | Folium perillae, Flos lonicerae |
Inhibition of PDE-4, suppression of cAMP hydrolysis Prevented neutrophil adhesion and cytokine release |
(Jiang et al. 2015; Middleton et al. 2000) |
| Rosmarinic acid | Various herbs |
Balancing pro-/anti-inflammatory cytokines, lowering IgE and histamine Reduced inflammatory markers in OVA-induced allergic mice |
(Jia et al. 2023) |
| Alkaloids | Reference | ||
|---|---|---|---|
| Natural product | Source | Experimental Findings | |
| Isoxazolines | Xanthoceras sorbifolia |
PDE-4 inhibition, modulation of inflammatory pathways Computational modeling showed higher binding affinity than Rolipram |
(Ge et al. 2016) |
| Other natural products | |||
| Natural product | Source | Experimental findings | Referrences |
| Patchouli oil | Pogostemon cablin |
Mast cell degranulation suppression, cytokine inhibition Reduced PCA and hypersensitivity in animal models |
(Chen et al. 2013; He et al. 2013; Su et al. 2016) |
| Lipoic acid | Organosulfur compound |
IgE and histamine suppression, villus height increase Reduced β-conglycinin-specific IgE and histamine levels |
(Han et al. 2010) |
| FIP-Lrh protein | Lignosus rhinocerotis |
Glycoprotein binding, immune modulation Functional recombinant protein produced in E. coli cells |
(Pushparajah et al. 2016) |
| Phenanthrenes, stilbenes | Gymnadenia conopsea |
Inhibition of antigen-induced degranulation Reduced PCA in RBL-2H3 cells and mouse ear models |
(Matsuda et al. 2004) |
| Picroliv | Picrorhiza kurroa |
Mast cell stabilization, Schultz–Dale response inhibition Reduced PCA in sensitized rodents |
(Baruah et al. 1998) |
| Alginic acid | Seaweed |
Histamine release inhibition, NF-κB suppression Reduced systemic and passive cutaneous anaphylaxis in mice |
(Jeong et al. 2006) |
Anaphylaxis: a complex immune system underlying multiple cellular pathways
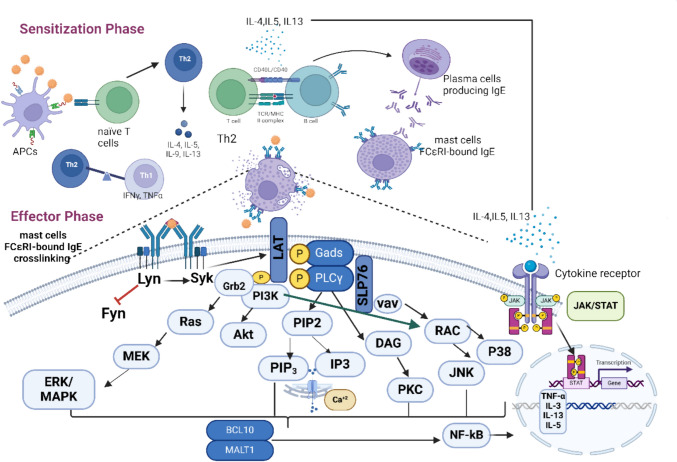
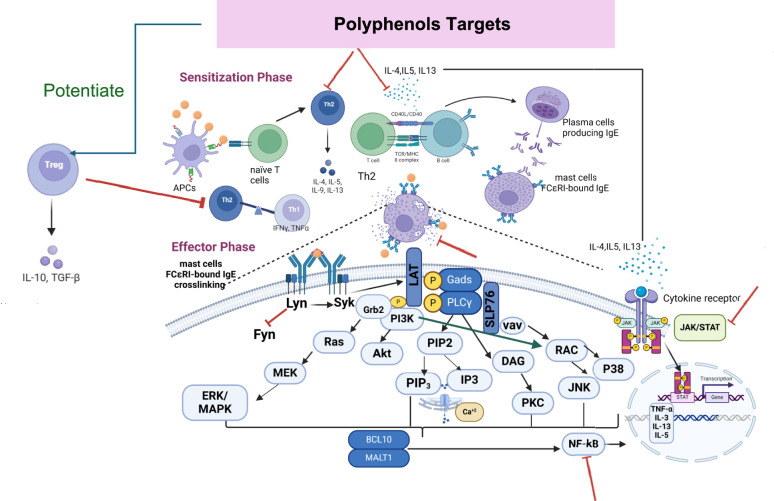
Anaphylaxis is classified as a type I hypersensitivity allergic reaction. Two major phases are involved in this kind of allergic reaction. Initially, during the sensitization or induction phase when an allergen is first encountered, antigen-presenting cells (APCs) or macrophages identify, engulf, and deliver it to naïve T cells, which will subsequently differentiate into T helper 2 (Th2) cells (Dera et al. 2020). Through the production of pro-inflammatory cytokines such as interleukin-4 (IL-4), IL-5, or IL-13, these Th2 cells transform B cells into cells that generate IgE, or plasma cells. The generated IgE attaches itself to the α-subunit of mast and basophil cells’ high-affinity IgE receptors (FcεRIs) (Chang et al. 2015; Park et al. 2020), which are the predominant effector cells in type I allergy (Li et al. 2022).
The effector phase starts when the same allergen recurs (Dera et al. 2020). The allergen will crosslink two nearby FcεRI-bound IgE, which will set off a series of downstream signaling cascades, including those involving tyrosine kinase, protein kinase C (PKC), mitogen-activated protein kinase (MAPK) (Chang et al. 2015; Yan et al. 2024; Yoo et al. 2017), Janus kinase-signal transducer and activator of transcription (JAK/STAT), and nuclear factor κB (NF-κB) ( Liu et al. 2023). In addition, calcium influx (Yan et al. 2024) and cytoskeleton remodeling will draw in secondary cells, including neutrophils (Dera et al. 2020), which will cause degranulation (i.e., the release of histamine and β-hexosaminidase), the generation of reactive oxygen species (ROS) (Vo et al. 2020), as well as the production of various pro-inflammatory cytokines and chemokines (Barbosa et al. 2018; Chang et al. 2015; Kobayashi et al. 2015; Mwakalukwa et al. 2019; Yoo et al. 2017; Yoshioka et al. 2020).
The most prevalent inflammatory and immunomodulatory mechanisms implicated in anaphylaxis
FcεRI signaling pathway
An allergen’s cross-linking of IgE activates the heterotetrameric (one α, one β, and two γ subunits) FcεRI receptors, which in turn activates two protein tyrosine kinases (PTKs) belonging to the Src family, Lyn and Fyn. By recruiting spleen tyrosine kinase (Syk) to FcεRIβ immunoreceptor tyrosine-based activation motifs (ITAMs), these Lyn and Fyn activate FcεRIβ-ITAMs (Athari 2019; Li et al. 2022). PKC, rat sarcoma (Ras), phosphoinositide 3-kinase (PI3K), protein kinase B (Akt), guanosine triphosphatase (GTPase), and phospholipase Cγ (PLCγ) are among the additional signaling cascades that are activated by the Syk (Jiao et al. 2017; Lee et al. 2020; Min et al. 2021; Park et al. 2020; Yoo et al. 2017; Yoshioka et al. 2020). Activated Syk additionally activates linkers for T-cell activation (LAT) and leukocyte-specific phosphoproteins (SLP-76) that include the src homology 2 (SH2) domains.
Upon this, cytosolic adaptor molecules as glutamic acid decarboxylase 2 (Gad2), growth factor receptor bound protein 2 (Grb2), PLCγ1, and guanine exchange factors (VAV and SOS) attach to LAT, further activating the PI3K and MAPK signaling pathways (Athari 2019; Li et al. 2022).
PLCγ further lowers intracellular Ca2 + by converting phosphatidylinositol 4,5-bisphosphate (PIP2) into diacylglycerol (DAG) and inositol triphosphate (IP3) (Jo et al. 2022; Lim et al. 2023). B cell lymphoma/leukemia 10 (BCL10), mucosa-associated lymphoid tissue lymphoma translocation protein 1 (MALT1), and p38 MAPK are all activated by the activation of PKC via Ca2 + and DAG (Jo et al. 2022; Li et al. 2022). PIP2 gets transformed into phosphatidylinositol 3,4,5-triphosphate (PIP3) through the active PI3K, and PIP3 then activates ERK1/2 and JNK (via RAC and MAPK4).
Phospholipase A2 (PLA2) is activated by ERK1/2, which raises prostaglandins and leukotrienes (Jo et al. 2022). High antigen-specific IgE levels are maintained, immune cells like eosinophils are drawn to inflammatory sites, mucus production is increased, and chronic allergic inflammation that causes tissue damage and remodeling is started during the later effector phase due to the overexpression of Th2-related immune response and increased production of Th2 cytokines, such as IL-4, IL-5, and IL-13 (Barnes 2011).
MAPK and JAK/STAT signaling pathway
These pathways are the most crucial allergy signaling pathways (Lee et al. 2020; Liu et al. 2023). A sequential activation of five protein kinases controls the MAPK signaling cascade including MAP4K, MAP3K, MAPKK, MAPK, and MAPK-activated protein kinases (MAPKAPK). Studies typically provide an explanation for MAP3K, MAP2K, and MAPK (Guo et al. 2020; Soares-Silva et al. 2016). According to their structure and function, MAPK can be divided into four groups: p38 MAPK, c-Jun N terminal kinase 1/2 (JNK1/2), extracellular signal-regulated kinase 1/2 (ERK1/2), and ERK5 (Li et al. 2022; Liu et al. 2023; Soares-Silva et al. 2016; Yan et al. 2024). All types of MAPK are activated by pro-inflammatory stimuli, ERK1/2 is activated by growth factors and hormones, and p38 MAPK and JNK 1/2 are activated by cellular and environmental stressors (Soares-Silva et al. 2016).
A G-protein called Ras is activated when a ligand first attaches to a receptor tyrosine kinase (RTK) in the classic activation of the ERK1/2 MAPK cascade. MAPK/ERK kinases (MEK1/2), a MAP2K sometimes referred to as MKK1/2, are activated by the serine/threonine protein kinase Raf (a MAP3K), which is recruited and activated by the Ras. These MEKs then activate ERK1/2 (a MAPK) (Cui et al. 2007; Guo et al. 2020; Soares-Silva et al. 2016; Yuan et al. 2020). The proteins upstream of the signaling cascades, such as SOS protein, Raf-1, and MEKs, are likewise given negative feedback by ERK1/2 (Guo et al. 2020). The ERK controls several transcription factors, including c-Fos, c-Jun, c-Myc, Elk-1, and ATF2, once it has been translocated into the nucleus (Guo et al. 2020). Th2 cells produce IL-4, -5, -9, and -13 via regulating the synthesis of IL-10, which is regulated by the ERK (Soares-Silva et al. 2016). In addition, Syk triggers ERK1/2, which in turn triggers the arachidonic signaling pathways and mast cell production of TNF-α, IL-2, IL-5, and IL-13. In reaction to stress or cytokines, Rho protein or tumor necrosis factor receptor-associated factor 2/3/6 (TRAF) stimulates MEKK1, SAK1, or TAK1 (a MAPK3K) They trigger the activation of MKK3 or MKK6 (a MAP2K), which in turn triggers the activation of p38 MAPK (Cui et al. 2007; Davis 2000; Soares-Silva et al. 2016; Zarubin & Han 2005).
The transcription factors ATF, NFAT, Elk-1, and HBP1 are further regulated by p38, which in turn controls the synthesis of cytokines (Zarubin & Han 2005). Th cell differentiation into Th2 cells, which generate IL-2, IFN-γ, and TNF-α/β, is boosted by the regulation of IL-12 production by the p38 MAPK (Soares-Silva et al. 2016). Numerous stressors, including cytokines (e.g., TNF and IL-1), trigger the JNK pathway via different receptors including as TNFR, GPCR, TGFBR, and TLR (Zeke et al. 2016). Rac1/Cdc42 is activated by a variety of signals, which in turn activates downstream proteins such as MLK, ASK, DLK, MEKK, and TAK. They also trigger MKK4 or MKK7, which triggers JNK (Cui et al. 2007; Davis 2000).
In addition, DLK which is an upstream protein was provided by JNK negative feedback (Zeke et al. 2016). The JNK also activates AP-1, which controls the production of cytokines, by impacting a number of transcription factors, including c-Fos, ATF, Jun B, Jun D, and c-Jun (Cui et al. 2007; Davis 2000; Zeke et al. 2016). Elk-1, NFAT, and P53 are likewise regulated by the JNK [32]. TNF-α, IL-2, and IL-6 are activated by the JNK, while p38 MAPK stimulates IL-4 (Lee et al. 2020; Liu et al. 2023).
JAK/STAT signaling pathway
Through the JAK/STAT signaling pathways, IL-4 and IL-13 increase the expression of pro-inflammatory genes in allergic disorders (Shankar et al. 2022). When these cytokines attach to receptors, Janus kinase (JAK) is drawn (recruited) in and the receptor dimerizes. The activated JAKs activate the receptors and attract STATs to the receptors. The active STATs separate from the receptors as homo or heterodimers, go into the nucleus, bind to DNA, and control gene expression (Hu et al. 2021; Shankar et al. 2022).
NF-κB can be activated by MAPK (Jo et al. 2022). Trimeric forms of inactive NF-kB and an NF-kB inhibitor (IkB) are present in the cytoplasm. MEKK1 signals trigger the IkB kinase complex (IKK) (Schulze-Osthoff et al. 1997), which in turn triggers IkB (at Ser 32 and Ser 36 residues). This releases NF-kB into the nucleus, where it attaches to the promotor regions' kB binding site and triggers the activation of mediators and gene expression, including COX-2, TNF-α, and IL-1β, -6, and -8 (Chu et al. 2016; Dasiman et al. 2022; Wang et al. 2020). When the 26S proteasome ubiquitinates the activated IkB, the active NF-kB manifests as a heterodimer of the p65 and p50 subunits (Chu et al. 2016) (Fig. 3).
Fig. 3.
Anaphylaxis immunomodulatory–inflammatory multi-axis via sensitization and effector phases
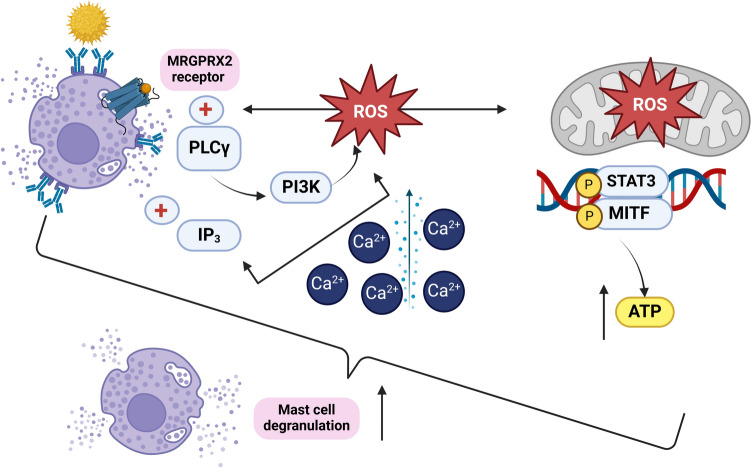
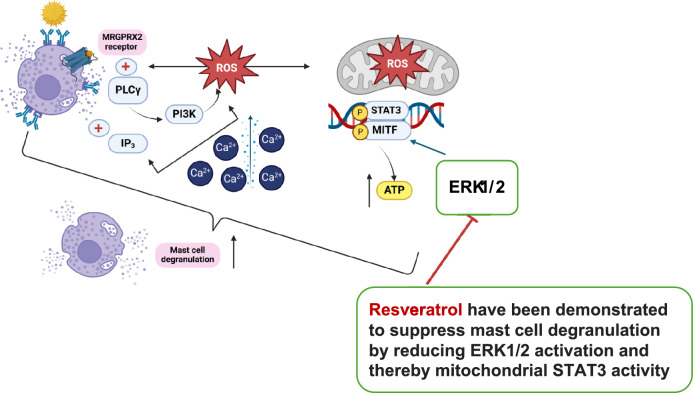
Mast cell degranulation signaling pathway
Mast cells are granulocytes (Breedveld et al. 2017) that circulate around microvessels in the skin and visceral mucosa. They include heparin, histamine, and 5-hydroxytryptamine. Because of interaction between IgE antibodies and antigens linked to mast cells, the cells largely disintegrate, releasing particles and chemicals that might induce fast allergic responses in tissues. At the same time, they release a range of cytokines and help regulate the immune system. It is primarily caused by antigen-induced aggregation of FcεRI receptor molecules on the surface of mast cells, which triggers the release of inflammatory mediators from mast cells. Mast cell activation triggers the release of inflammatory mediators, which launch a signal transduction cascade. The first factor of Ca2+ influx in mast cells is the cross-linking of allergen and the IgE-FcRI complex, which activates phospholipase C (PLC) and produces phosphatidylinositol 4,5-diphosphate (PIP2). PIP2 produces secondary messengers such IP3 (inositol 1,4,5-triphosphate) and DAG (diacylglycerol). On the endoplasmic reticulum membrane, IP3 reacts with the IP3 receptor (IP3R) to release Ca2+ from the endoplasmic reticulum Ca2+ storage (Taylor et al. 2009). Ca2+ influx is predominantly induced by endoplasmic reticulum Ca2+ storage depletion (Michell 1975; Putney 1986). Its molecular mechanism is that the endoplasmic reticulum contains proteins STIM1 and STIM2 with EF-hand domains that may detect Ca2+ depletion in the calcium storage (Liou et al. 2005). Then, it migrates to the plasma membrane, where it interacts with the Orai-protein and opens the cell membrane's Ca2+ channel, allowing external Ca2+ to enter (Putney 2009). Ca2 + plays a role in the mast cell activation signaling pathway.
Ca2+ contributes to the mast cell activation signaling pathway. Intracellular Ca2+ controls cell granule movement, membrane fusion, and degranulation (Hartzell et al. 2016). FcεRI (high-affinity IgE receptor) on inactivated mast cells activates FcεRI cross-linking, leading to T-cell activation. LAT is phosphorylated in a manner determined by tyrosine-protein kinases Lyn and Syk (Zhang et al. 2002). Molecular mechanism of mast cell degranulation mainly focuses on tyrosine kinase Lyn and Fyn-dependent signal transduction pathway and increase of intracellular Ca2+ level (Takata et al. 1994).
Degranulation led to activation of signal transduction phospholipases PLCγ, protein kinase C, and increased calcium ions. TRPC1 channels also promoted Ca2+ inflow (Svetlov et al. 1996). The activation of the RAS-RAF-MAPK pathway produces eicosanoids (including leukotrienes C4 and prostaglandin D2) as well as cytokines. InsP3R (Inositol Triphosphate Receptor), a membrane glycoprotein complex triggered by InsP3, functions as a calcium channel (Hagar et al. 1998), while STIM (Stromal interaction molecules) is a calcium receptor on the endoplasmic reticulum (Wang et al. 2021a, b). Therefore, blocking the binding of FcRI and IgE effectively inhibits allergic reactions.
Oxidative stress and mast cell degranulation
Recent research has demonstrated immediate hypersensitivity responses caused by the activation of Mas-related G protein coupled receptor member X2 (MRGPRX2) expressed in mast cells. The binding of these chemicals to the MRGPRX2 receptor activates cytosolic G proteins, elevates intracellular calcium concentrations, and eventually causes the exocytosis of anaphylactic mediators (Subramanian et al. 2016). There is a relationship between mast cell degranulation and mitochondrial respiratory complex chain activity, which causes oxidative phosphorylation. Usually, mast cell activation is linked with increased mitochondrial respiration. Thus, IgE-mediated activation increased oxygen consumption rate by 60% (Buttgereit et al. 2022).
As a result, inhibiting mitochondrial respiratory complex chain lowered both IgE- and non-IgE-mediated mast cell degranulation. Inhibiting complexes, I and III inhibited IgE-mediated mast cell degranulation (Suzuki et al. 2005; Takekawa et al. 2012). Enzymatic reactions occur in cells and involve the transfer of energy, electrons, or protons. Some molecules, such as oxygen or nitrogen, can acquire or transfer electrons, causing them to become unpaired. This new molecule is known as a “free radical” because it may interact with other, more stable molecules. Reactive oxygen species (ROS) and reactive nitrogen species (RNS) contribute to physiological activities required for appropriate cellular function, as well as pathological processes that affect energy generation, apoptosis, or mitophagy. Oxidative stress is characterized as an imbalance in ROS generation and neutralization by antioxidant mechanisms. IgE activation of mast cells resulted in elevated intracellular ROS levels and degranulation (Collaco et al. 2006; Tagen et al. 2009).
Following allergen stimulation, intracellular ROS generation in mast cells increased in a time and allergen dose-dependent manner before plateau (Yasui et al. 2007). Allergen stimulation via IgE receptor FcεRI activates Lyn, Fyn, and Syk kinases, leading to activation of PLCγ and PI3K. PLCγ creates DAG and IP3, resulting in calcium-dependent degranulation. IP3 interacts to its receptors IP3R on the endoplasmic reticulum, causing Ca2 + release. PI3K stimulates ROS generation via NOX action. MRGPRX2 activation enhances PI3K and PLCγ activation. Increased intracellular Ca2 + causes granule exocytosis. ROS modulates mast cell degranulation via Ca2 + efflux from the endoplasmic reticulum and extracellular influx.
mtROS controls PKC phosphorylation, resulting in degranulation. Mast cell activation causes mitochondrial fragmentation, morphological alterations, and the relocation of mitochondria from the perinuclear area to the cell surface. Phosphorylation of the two transcriptional factors STAT3 and MITF causes mitochondrial respiration, ATP generation, and mitochondrial ROS, which promotes mast cell degranulation.
Thus, ROS play an important role in mast cell control, including activation and degranulation. In conclusion, a recent study found that antigen activation of mast cells causes enhanced ROS generation from NADPH oxidases and the mitochondrial respiratory chain. In-vitro antioxidant therapy of mast cells seems to prevent degranulation by lowering ROS levels, but further research is needed to validate this. Exposure to pro-oxidant environmental circumstances, such as air pollution, appears to promote mast cell degranulation via boosting ROS production (Piotin et al. 2024) (Fig. 4).
Fig. 4.
Relation between oxidative stress and mast cell degranulation
Some natural compounds, such as the polyphenol and resveratrol, have been demonstrated to suppress mast cell degranulation by reducing ERK1/2 activation and thereby mitochondrial STAT3 activity (Paradis et al. 2024). Furthermore, after mast cell activation, PIAS3, the major endogenous inhibitor of STAT3 found in mitochondria, rose and worked as a regulator of the mitochondrial allergic response (Erlich et al. 2014) (Fig. 5).
Fig. 5.
Polyphenols defeat oxidative stress action towards mast cell degranulation
Mechanisms of action of natural products in treatment of anaphylaxis
Through a variety of molecular pathways, it has recently been shown that a wide range of natural compounds, such as flavonoids, terpenes, phenols, saponins, alkaloids, coumarins, lignans, and quinones, might reduce the symptoms of hypersensitivity type I reactions, including anaphylactic reactions.
One of the most well-known natural bioactive substances with well-established anti-inflammatory, antioxidant, and immunomodulatory qualities is dietary polyphenols, which have been the subject of numerous studies. Polyphenols are a valuable and promising dietary intervention in the prevention and treatment of allergic diseases because of their safety profile, broad distribution in plants, frequent inclusion in the daily diet, and a wide range of bioactivity, including immunomodulatory and anti-inflammatory qualities (Eseberri et al. 2022; Rakha et al. 2022; Rana et al. 2022). Thus, in response to the increasing need for the creation of novel therapeutic and preventative options based on natural products, polyphenols have attracted a lot of scientific attention and been the focus of in-depth study in recent years (Kumari et al. 2023; Wu et al. 2023; Zeng et al. 2023a, b). Quercetin, curcumin, resveratrol, catechins, and many other polyphenols may reduce allergic inflammation, relieve food allergy, asthma, and allergic rhinitis symptoms, and stop the onset of an allergic immunological response. It is yet unknown and requires clarification of what precise molecular and cellular processes polyphenols may use to prevent and treat allergy disorders. Yet, it is hypothesized that polyphenols’ advantageous anti-allergic action is linked to their impact on primary targets such as interaction with allergic proteins and decrease in their allergenicity, modification of the systemic and local immune response (Fig. 6).
Fig. 6.
Summary of the polyphenols most common anti-anaphylactic effect among anaphylaxis pathways
Interaction with allergic proteins and decrease in their allergenicity
Allergic protein modification
In fact, it has been discovered that certain polyphenols can reduce allergenicity by changing the secondary and tertiary structures of proteins, which alters the conformational epitopes of the allergen, and by masking the linear epitopes of the allergen by conjugation with nucleophilic amino acids (Liu et al. 2021a, b; Yan et al. 2023; Zhang et al. 2021; Zhou et al. 2024). Numerous investigations concerning the phytochemical alteration of a main allergen in cow’s milk β-lactoglobulin (β-LG) have shown that covalent conjugation with different polyphenols, such as rutin, ferulic acid, caffeic acid, epigallocatechin (EGCG), and chlorogenic acid, results in conformational changes that cause proteins to become more unfolded, which is correlated with a decreased ability of IgG/IgE to bind (Deng et al. 2023; Liu et al. 2021a, b; Wang et al. 2023; Wu et al. 2018; Xu et al. 2019; Xue et al. 2023). Moreover, a number of flavonoids, including EGCG, naringenin, myricetin, kaempferol, and quercetin, can also reduce the allergenicity of β-lactoglobulin through non-covalent interactions. EGCG demonstrated the strongest inhibitory potency on β-LG antigenicity, resulting in a 73% decrease in IgE binding ability (Pu et al. 2021).
The ovalbumin allergenicity was evaluated in vitro as the ability to cause effector cell degranulation and in vivo as the severity of the allergic immune response and symptom score (T. Zhang et al. 2020). Likewise, the covalent conjugation between quercetin and ovalbumin altered the secondary and tertiary conformation of the protein, resulting in a less folded structure and decreased allergen stability. Furthermore, following covalent and non-covalent binding with quercetin, the spectrometric structural analysis of allergens belonging to the profilin family revealed a 40% reduction in the α-helical structures in the conjugates. This, in conjunction with the antigenic epitopes being obscured, led to a significantly reduced level of allergenicity (Zhou et al. 2023).
According to recent reports, covalent interactions with CA, EGCG, and polyphenols isolated from the algae Sargassum fusiforme can cause conformational changes in shrimp tropomyosin's structure. These alterations can significantly reduce allergenicity, which in turn reduces shrimp-induced allergic symptoms in vivo (Lv et al. 2021; Zhao et al. 2023).
Boosting the allergen’s digestibility
For example, experimental investigations have shown that covalent attachment of EGCG and CA to peanut proteins reduces allergenicity dramatically, not only by modifying linear and conformational epitopes, but also by enhancing peanut allergen digestion.
In vitro and in a food allergy mouse model, peanut protein was found to have lesser allergenicity, resulting in fewer symptoms, mast cell frequency, and intestinal damage (Bansode et al. 2019; He et al. 2023, 2020).
Five major apple polyphenols (epicatechin, phlorizin, rutin, chlorogenic acid, and catechin) showed similar effects on simulated gastric digestion and the spatial structure of Ara h 1 peanut protein. Epicatechin was found to have the strongest inhibitory effect on peanut allergy (Sun et al. 2023).
Furthermore, the IgE/IgG binding capacity was impacted by the covalent binding of wheat gliadin with luteolin and chlorogenic acid, which impacted the protein conformation and gave it a more ordered structure. This also greatly enhanced the thermal stability and in-vitro digestibility of allergic proteins (Yang et al. 2023a, b; Zhang et al. 2022a, b).
Polyphenols may reduce allergen load by causing protein aggregation and cross-linking
Possibly due to the loss of reactive allergens and reduced accessibility of reactive epitopes (Bessa et al. 2021; Zhang et al. 2021; Zhou et al. 2024). Meanwhile, Polyphenols’ ability to form cross-linked protein polymers allows allergen binding to be efficient even when the number of polyphenol molecules is less than the number of allergen reaction sites, and the resulting polyphenol–allergen complexes are more stable and thus more effective (Bessa et al. 2021; Pi et al. 2023; Zhou et al. 2024). This effect was clearly demonstrated in a large number of studies examining the structural and functional features of several soybean globulins following covalent interaction with polyphenols such as EGCG, chlorogenic acid, caffeic acid, gallic acid, and tannic acid (Li et al. 2023; Lin et al. 2022; Pi et al. 2023; Zhou et al. 2020). In all cases, the formation of polyphenol-soybean globulin conjugate and cross-linking of soybean proteins resulted in structural changes that concealed or destroyed allergen epitopes, as well as increased UV absorption and protein digestibility, resulting in reduced IgE binding activity and histamine release in vitro (Li et al. 2023; Lin et al. 2022; Pi et al. 2023; Zhou et al. 2020).
It is interesting to note that studies using the mouse model of allergy showed that covalently conjugating the soy 11S protein with EGCG and chlorogenic acid can effectively induce the development of oral tolerance to soy allergen in addition to lowering the protein’s allergenicity and easing allergy symptoms (Li et al. 2023). Combining all the aforementioned information, dietary polyphenols have a significant potential to decrease food allergenicity; as a result, they may be helpful in creating hypoallergenic meals that may lessen the symptoms of food allergies or even stop them from occurring by fostering tolerance.
Immunomodulatory effect
Polyphenols have been shown to have both stimulatory and inhibitory effects at two crucial stages, during the sensitization and effector phases of allergic disease, according to evidence gathered from in-vitro and in-vivo studies (del Cornò et al. 2016; Haftcheshmeh et al. 2022; Kumari et al. 2023; Magrone et al. 2020; Mlcek et al. 2016; Mwakalukwa et al. 2019; Rakha et al. 2022; Shaik et al. 2018; Shakoor et al. 2021; Wu et al. 2023; Zeng et al. 2023a, b; Zhang et al. 2022a, b).
Sensitization phase
Dendritic cells (DCs) present the entering allergen to naïve CD4 + T cells in draining lymph nodes as the initial step in the sensitization phase. This causes the naïve CD4 + T cells to differentiate into allergen-specific Th2 cells that produce proallergic cytokines (IL-4, IL-5, IL-9, and IL-13) (Humeniuk et al. 2017). By influencing DC differentiation, maturation, and the ability to trigger T-cell differentiation into allergy type Th2 cells, some polyphenol groups have been shown to obstruct the antigen presentation pathway (del Cornò et al. 2016). Resveratrol affect how human DC differentiate from monocytes and prevent DC maturation, which results in the production of an immature phenotype (Buttari et al. 2013; Švajger et al. 2010). Several polyphenols, including quercetin, curcumin, fisetin, silibinin, isoflavones, and blackberry polyphenols, have been shown to inhibit the phenotypic and functional development of DC generated from murine bone marrow. Furthermore, these substances suppress the expression of major histocompatibility complex (MHC) class II and co-stimulatory molecules (CD83, CD80, and CD86) on the surface of DCs, which impedes effective antigen presentation (Dai et al. 2007; Huang et al. 2010; Liu et al. 2010; Yoneyama et al. 2008; Yoon et al. 2006). In addition to influencing DC differentiation and reducing antigen absorption activity, other polyphenols, EGCG, and apigenin have also been shown to induce apoptosis in DC-precursors and immature DCs (Kang et al. 2009; Yoneyama et al. 2008).
In addition, the next significant event in the sensitization phase, naïve CD4 + T cell priming, can be regulated by polyphenols. Indeed, it has been demonstrated that kaempferol and lycoricidine disrupt TCR-mediated signaling cascades, therefore, preventing naïve CD4 + T cells from activating and differentiating into Th2 effector cells (Lee & Jeong 2021; Lee et al. 2021). Apart from DCs presenting allergens, cytokines such as TSLP, IL-25, and IL-33, which are released by epithelial cells lining barrier sites in response to food and aero-allergens, are crucial in the allergic sensitization phase because they activate DCs and innate lymphoid cells type 2 (ILC2) and stimulate the growth of Th2 cells (Hammad & Lambrecht 2015; Pasha et al. 2019).
By generating IL-4, IL-13, and IL-5 during the early stages of antigen sensitization, ILC2 are also crucial for promoting the Th2 immune response (Zheng et al. 2021). In human keratinocyte models of atopic dermatitis (AD) as well as AD-like mice models, many polyphenols, including quercetin, curcumin, and baicalin, have been found to inhibit the production and secretion of TSLP and IL-33 (Beken et al. 2020; Sharma et al. 2020; Wang et al. 2022). Resveratrol and naringenin, two other polyphenols have the ability suppress TSLP synthesis and mRNA expression in human mast cell lines (Moon et al. 2011, 2021). In experimental models of allergic airway inflammation, quercetin was also shown to have a modulatory influence on epithelium-derived cytokines, as evidenced by its considerable reduction of TSLP expression in lung tissue and IL-25, IL-33, and TSLP levels in BAL (Caglayan Sozmen et al. 2016).
In the mouse model of allergic rhinitis and asthma, it was recently reported that Fallopia japonica, also known as Asian knotweed, a traditional medicinal herb rich in polyphenols such as resveratrol and flavones, targets the IL-33/TSLP signaling pathway and significantly lowers these cytokine levels in both nasal and bronchoalveolar lavage fluid (Jin et al. 2023).
Although polyphenols may have an impact on B cell recruitment, maturation, and function, this effect has not yet been fully explored and documented (Bessa et al. 2021; Shakoor et al. 2021; Singh et al. 2011). However, several polyphenols, including curcumin, rosmarinic acid, quercetin, ferulic acid, tea catechins (EGCG, ellagitannins, and gallic acid), and red grape polyphenols, have been shown in in-vitro and in-vivo studies to have the ability to inhibit the production of antigen-specific IgE in a dose- and time-dependent manner (Acar et al. 2016; Liang et al. 2020; Magrone et al. 2020).
Dihydromyricetin, a naturally occurring flavonoid, was used to demonstrate the modulatory effect of polyphenols. It successfully suppressed the sensitization phase by lowering the number of B cells and their production of antigen-specific IgE and by blocking the FcεRI–IgE interaction (Zhang et al. 2019). Likewise, tea catechins and phlorotannins (such as dieckol and eckol) may interact with FcεRI by directly binding to the α chain, preventing antigen-specific IgE from binding to FcεRI and reducing the mast cell sensitization phase (Kim et al. 2020; Li et al. 2021; Sugiura et al. 2021).
By hampering the expression of the FcεRI receptor, which is essential for the long-term sensitization of mast cells and their subsequent degranulation during the effector phase, it has also been demonstrated that phlorotannins, saponins, catechins, quercetin, kaempferol, and resveratrol may help to attenuate the allergic reaction (Li et al. 2021; Nagata et al. 2023; Zhang et al. 2022a, b).
Effector phase
Whenever the same allergen is encountered again during the effector phase, IgE bound to FcεRI on the surface of mast cells and basophils cross-links, activating and degranulating them and releasing reactive mediators that cause an acute systemic allergic response (Pasha et al. 2019). The ways in which polyphenols may modulate mast cells, which are important effector cells of the allergic reaction, have been the subject of several recent in-vitro and in-vivo investigations (Rakha et al. 2022; Wu et al. 2023; Zhang et al. 2022a, b). Apart from the previously mentioned effects on FcεRI receptor expression and FcεRI–IgE binding, various polyphenols, resveratrol, quercetin, and procyanidins from apple extract or cinnamon, can inhibit mast cell activation by preventing allergens from cross-linking IgE on the cell surface (Civelek et al. 2022; Kandhare et al. 2017; Shaik et al. 2018). Furthermore, some polyphenols, such as quercetin, phlorotannins, luteolin, and myricetin, have been shown to downregulate the expression of calcium channel proteins and inhibit calcium influx and intracellular calcium elevation, which are required for mast cell degranulation (Mwakalukwa et al. 2019; Park 2020; Sugiura et al. 2021; Vo et al. 2020). This has been shown to stabilize mast cell membranes and thereby suppress their degranulation. The release of histamine and β-hexosaminidase, which are indicators to assess the degree of mast cell degranulation, was in fact markedly inhibited by these phenolic substances, curcumin, EGCG, rosmarinic acid, and resveratrol (Barbosa et al. 2019; Civelek et al. 2022; Kong et al. 2020; Magrone et al. 2020; Mwakalukwa et al. 2019; Vo et al. 2020).
Furthermore, polyphenols have been shown to be a strong suppressor of the MAPK and NF-κB signaling pathways, as well as the FcεRI-mediated protein kinases (Syk, Lyn, PLCγ, and PKC) signaling cascade, which are essential for the allergic reaction. This has been shown to attenuate the production of lipid mediators (prostaglandin D2, leukotrienes) and pro-inflammatory cytokines (IL-4, TNF-α) (Civelek et al. 2022; Crozier et al. 2023; Hwang et al. 2019; Vo et al. 2020; Yousef et al. 2020; Zeng et al. 2023a, b). The anti-allergic potential of stingless bee honey (Kelulut honey) was interestingly confirmed via the mechanism of mast cell activation and degranulation. However, the inhibitory effect was solely dependent on the botanical source of honey, as it was only indicated in the case of honey that was rich in polyphenols and derived from rubber and bamboo trees, while honey that was inadequate in polyphenols and derived from noni and mango did not exhibit such anti-allergic action (Yong et al. 2023).
The immunomodulatory effects of polyphenols at several critical stages of the effector phase, such as inhibition of Th2 differentiation, downregulation of Th2-related cytokine production, reduction of inflammatory cell infiltration, and consequently, suppression of allergic inflammation, have been confirmed by numerous experimental studies employing cellular and animal models.
Most significantly, polyphenols were discovered to successfully resolve the Th1/Th2 imbalance by inhibiting the overexpression of Th2-mediated immune responses and upregulating the Th1 pathways (Rakha et al. 2022; Shakoor et al. 2021; Singh et al. 2011). Curcumin has demonstrated anti-allergic effects in a number of models of allergic diseases, including lowering Th2 cell activity and proliferation, lowering IL-4, IL-5, and IL-13 secretion, preventing macrophage, monocyte, neutrophil, and eosinophil activation and infiltration into inflammatory sites, and shifting the Th1/Th2 response toward the Th1 phenotype (Chauhan et al. 2018; Shahgordi et al. 2020; Shahid et al. 2019; Sharma et al. 2020). By reducing the synthesis of IL-4, Il-5, and Il-13 in the serum and bronchoalveolar lavage fluid (BALF), kaempferol and rosmarinic acid, with particular consideration paid to asthma models, attenuated airway inflammation. They also successfully decreased the recruitment of eosinophils into lung tissues, airway hyperresponsiveness, and mucus production (Liang et al. 2020; Molitorisova et al. 2021; Shakeri et al. 2019). Flavonoids such as quercetin, isoquercetin, myricetin, and luteolin have been shown in studies in a mouse model of allergic rhinitis to reduce inflammation of the nasal mucosa by promoting the Th1 pathway and preserving the Th1/TH2 balance in addition to inhibiting Th2 cell differentiation and cytokine secretion (Dong et al. 2021; Feng et al. 2020; Hwang et al. 2018; Ke et al. 2023; Shi et al. 2023). Furthermore, in mouse models of food allergy, quercetin and tea catechins (gallic acid and ellagitannins) have been demonstrated to be potent inhibitors of the ovalbumin (OVA)-induced allergic response, promoting immune tolerance through Th1/Th2 modulation and induction of regulatory T cells (Treg) (Feng et al. 2020; Mine et al. 2020). Indeed, disruption of the equilibrium between Th17/Treg cells, in addition to Th1/Th2 dysregulation, plays an essential role in the development and progression of chronic allergic inflammation by contributing to the breakdown of immunological tolerance (Boonpiyathad et al. 2020; Tortola et al. 2020). By increasing the amount of Tregs and reestablishing the balance between Th17/Treg, several flavonoids, including quercetin, luteolin, cyanidin, and baicalin, have been shown in recent experimental trials to have an anti-allergic impact (Ke et al. 2023; Li et al. 2020; Liu et al. 2017; Yang et al. 2023a, b).
In a murine model of asthma, curcumin also had a modulatory impact on the Th17/Treg imbalance, considerably boosting the frequency of Treg subtypes while successfully decreasing Th17 cell differentiation (Chen et al. 2018; Ma et al. 2013; Wu et al. 2020). In conclusion, a wealth of evidence from animal models and in-vitro experiments suggests that polyphenols may be able to prevent the onset of allergic diseases by modifying the process of allergic sensitization. In addition, their effects on allergy effector cells upon re-exposure may represent a novel therapeutic approach.
Conclusions and future directions
This article offers a thorough examination of the molecular mechanisms through which natural compounds exert therapeutic effects on anaphylaxis. In general, these natural compounds demonstrate distinct benefits in managing allergic inflammation, and most are safe and easily accessible. However, it is essential that these natural compounds undergo further evaluation through rigorous and extensive clinical studies. There is a need for greater emphasis on clinical translational research to improve the quality of evidence from clinical studies. There is a notable lack of clinical trials specifically evaluating natural products for their efficacy in managing anaphylaxis. Although many natural compounds have anti-inflammatory and anti-allergic effects and have been investigated in preclinical settings, animal models and in-vitro studies, their direct use in anaphylaxis has not been thoroughly tested in human clinical trials. The complexity and urgency of anaphylactic reactions, which require rapid intervention, making it challenging to design controlled studies, contributing in part to this disparity. Furthermore, the field lacks standardized protocols for testing natural products in severe allergic conditions, and ethical concerns further limit such trials. Rather than acute anaphylaxis, current data are mostly derived from studies on related disorders including allergic rhinitis, asthma, and atopic dermatitis. For natural products to be integrated into anaphylaxis management, rigorous, large-scale clinical trials are essential to validate their safety, efficacy, and mechanism of action in this context. In addition, the specific targets of natural products in treating anaphylaxis remain unclear, and multi-omics approaches may help identify these functional targets. Many natural products exhibit poor absorption, distribution, metabolism, excretion (ADME) properties, and low water solubility. This highlights the need for chemical modifications or the use of nano-based drug delivery systems to enhance their effectiveness. Overall, addressing these limitations through focused research could significantly advance the therapeutic application of natural compounds in managing allergic conditions.
Acknowledgements
Not applicable.
Funding
Open access funding provided by The Science, Technology & Innovation Funding Authority (STDF) in cooperation with The Egyptian Knowledge Bank (EKB). This research received no external funding.
Data availability
No datasets were generated or analyzed during the current study.
Declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
Not applicable.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- Acar M, Muluk NB, Yigitaslan S, Cengiz BP, Shojaolsadati P, Karimkhani H, Cingi C (2016) Can curcumin modulate allergic rhinitis in rats? J Laryngol Otol 130(12):1103–1109. 10.1017/S0022215116008999 [DOI] [PubMed] [Google Scholar]
- Alqurashi W, Ellis AK (2017) Do corticosteroids prevent biphasic anaphylaxis? J Allergy Clin Immunol 5(5):1194–1205. 10.1016/j.jaip.2017.05.022 [DOI] [PubMed] [Google Scholar]
- Alsabri M, Mourid MR, Alaswad M, Faheem YAM (2024) Management of pediatric anaphylaxis in the emergency department in low-resource settings: a narrative review. Curr Emerg Hospital Med Rep 12:169–178 [Google Scholar]
- Alvarez-Perea A, Cabrera-Freitag P, Fuentes-Aparicio V, Infante SJCPD (2023) Advancements in anaphylaxis. Management 29(3):185–195 [DOI] [PubMed] [Google Scholar]
- Amir Rawa MS, Mazlan MKN, Ahmad R, Nogawa T, Wahab HA (2022) Roles of Syzygium in anti-cholinesterase, anti-diabetic, anti-inflammatory, and antioxidant: from Alzheimer’s perspective. Plants 11(11):1476 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arroabarren E, Lasa E, Olaciregui I, Sarasqueta C, Munoz J, Pérez-Yarza EJPA (2011) Improving anaphylaxis management in a pediatric emergency department. Pediatr Allergy Immunol 22(7):708–714 [DOI] [PubMed] [Google Scholar]
- ASCIA (2024). Guidelines_Acute_Management_Anaphylaxis. ASCIA Guidelines.
- Athari SS (2019) Targeting cell signaling in allergic asthma. Signal Transduct Target Ther 4(1):45. 10.1038/s41392-019-0079-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bailey AM, Baum RA, Horn K, Lewis T, Morizio K, Schultz A (2017) Review of intranasally administered medications for use in the emergency department. J Emerg Med 53(1):38–48 [DOI] [PubMed] [Google Scholar]
- Bansode RR, Randolph PD, Plundrich NJ, Lila MA, Williams LL (2019) Peanut protein-polyphenol aggregate complexation suppresses allergic sensitization to peanut by reducing peanut-specific IgE in C3H/HeJ mice. Food Chem 299:125025. 10.1016/j.foodchem.2019.125025 [DOI] [PubMed] [Google Scholar]
- Barbosa M, Lopes G, Valentão P, Ferreres F, Gil-Izquierdo Á, Pereira DM, Andrade PB (2018) Edible seaweeds’ phlorotannins in allergy: A natural multi-target approach. Food Chem 265:233–241. 10.1016/j.foodchem.2018.05.074 [DOI] [PubMed] [Google Scholar]
- Barbosa M, Lopes G, Andrade PB, Valentão P (2019) Bioprospecting of brown seaweeds for biotechnological applications: Phlorotannin actions in inflammation and allergy network. Trends Food Sci Technol 86:153–171. 10.1016/j.tifs.2019.02.037 [Google Scholar]
- Barnes PJ (2011) Pathophysiology of allergic inflammation. Immunol Rev 242(1):31–50. 10.1111/j.1600-065X.2011.01020.x [DOI] [PubMed] [Google Scholar]
- Baruah CC, Gupta PP, Nath A, Patnaik LGK, Dhawan BN (1998) Anti-allergic and anti-anaphylactic activity of picroliv—a standardised iridoid glycoside fraction ofpicrorhiza kurroa. Pharmacol Res 38(6):487–492. 10.1006/phrs.1998.0397 [DOI] [PubMed] [Google Scholar]
- Beken B, Serttas R, Yazicioglu M, Turkekul K, Erdogan S (2020) Quercetin improves inflammation, oxidative stress, and impaired wound healing in atopic dermatitis model of human keratinocytes. Pediatr Allergy Immunol Pulmonol 33(2):69–79. 10.1089/ped.2019.1137 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benito-Villalvilla C, de la Rocha-Muñoz A, López-Abente J, Eggel A, Bottoli I, Severin T, Palomares OJA (2023) Ligelizumab impairs IgE-binding to plasmacytoid dendritic cells more potently than omalizumab and restores IFN-α production and FOXP3+ Treg generation. Allergy 78(4):1060–1072 [DOI] [PubMed] [Google Scholar]
- Bessa C, Francisco T, Dias R, Mateus N, Freitas VD, Pérez-Gregorio R (2021) Use of polyphenols as modulators of food allergies from chemistry to biological implications. Front Sustain Food Syst 5:623611 [Google Scholar]
- Bilò MB, Corsi A, Martini M, Penza E, Grippo F, Bignardi DJA (2020) Fatal anaphylaxis in Italy: Analysis of cause-of-death national data, 2004–2016. Allergy 75(10):2644–2652 [DOI] [PubMed] [Google Scholar]
- Bilò MB, Martini M, Tontini C, Corsi A, Antonicelli L (2021) Anaphylaxis. Eur Ann Allergy Clin Immunol 53(1):4–17. 10.23822/EurAnnACI.1764-1489.158 [DOI] [PubMed] [Google Scholar]
- Bock SA, Muñoz-Furlong A, Sampson HAJJ (2007) Further fatalities caused by anaphylactic reactions to food, 2001–2006. Allergy 119(4):1016–1018 [DOI] [PubMed] [Google Scholar]
- Böhm R, Proksch E, Schwarz T, Cascorbi IJ (2018) Drug hypersensitivity: diagnosis, genetics, and prevention. Deutsches Ärzteblatt Int 115(29–30):501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boonpiyathad T, Sözener ZC, Akdis M, Akdis CA (2020) The role of Treg cell subsets in allergic disease. Asian Pac J Allergy Immunol 38(3):139–149 [DOI] [PubMed] [Google Scholar]
- Breedveld A, Groot Kormelink T, van Egmond M, de Jong EC (2017) Granulocytes as modulators of dendritic cell function. J Leukoc Biol 102(4):1003–1016. 10.1189/jlb.4MR0217-048RR [DOI] [PubMed] [Google Scholar]
- Breuer C, Wachall B, Gerbeth K, Abdel-Tawab M, Fuhr UJE (2013) Pharmacokinetics and pharmacodynamics of moist inhalation epinephrine using a mobile inhaler. Eur J Clin Pharmacol 69:1303–1310 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buttari B, Profumo E, Facchiano F, Ozturk EI, Segoni L, Saso L, Riganò R (2013) Resveratrol prevents dendritic cell maturation in response to advanced glycation end products. Oxid Med Cell Longev 2013(1):574029. 10.1155/2013/574029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buttgereit T, Pfeiffenberger M, Frischbutter S, Krauß P-L, Chen Y, Maurer M, Gaber T (2022) Inhibition of Complex I of the Respiratory Chain, but Not Complex III, Attenuates Degranulation and Cytokine Secretion in Human Skin Mast Cells. Int J Mol Sci 23(19):11591 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Caglayan Sozmen S, Karaman M, Cilaker Micili S, Isik S, Bagriyanik A, Arikan Ayyildiz Z, Karaman O (2016) Effects of quercetin treatment on epithelium-derived cytokines and epithelial cell apoptosis in allergic airway inflammation mice model. Iran J Allergy Asthma Immunol 15(6):487–497 [PubMed] [Google Scholar]
- Campbell RL, Bellolio MF, Knutson BD, Bellamkonda VR, Fedko MG, Nestler DM, Hess EP (2015) Epinephrine in anaphylaxis: higher risk of cardiovascular complications and overdose after administration of intravenous bolus epinephrine compared with intramuscular epinephrine. J Allergy Clin Immunol 3(1):76–80. 10.1016/j.jaip.2014.06.007 [DOI] [PubMed] [Google Scholar]
- Cardona V, Ansotegui IJ, Ebisawa M, El-Gamal Y, Rivas MF, Fineman S, Borges MS (2020) World allergy organization anaphylaxis guidance 2020. World Allergy Organ J 13(10):100472 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carter MC, Akin C, Castells MC, Scott EP, Lieberman PJA, o. A., Asthma, & Immunology. (2020) Idiopathic anaphylaxis yardstick: practical recommendations for clinical practice. Ann Allergy Asthma Immunol 124(1):16–27 [DOI] [PubMed] [Google Scholar]
- Chandrashekhar VM, Halagali KS, Nidavani RB, Shalavadi MH, Biradar BS, Biswas D, Muchchandi IS (2011) Anti-allergic activity of German chamomile (Matricaria recutita L) in mast cell mediated allergy model. J Ethnopharmacol 137(1):336–340. 10.1016/j.jep.2011.05.029 [DOI] [PubMed] [Google Scholar]
- Chang Y-Y, Hsu W-H, Pan T-M (2015) Monascus secondary metabolites monascin and ankaflavin inhibit activation of RBL-2H3 Cells. J Agric Food Chem 63(1):192–199. 10.1021/jf504013n [DOI] [PubMed] [Google Scholar]
- Chauhan PS, Jaiswal A (2018) Combination therapy with curcumin alone plus piperine ameliorates ovalbumin-induced chronic asthma in mice. Inflammation 41(5):1922–1933. 10.1007/s10753-018-0836-1 [DOI] [PubMed] [Google Scholar]
- Chen M, Zhang J, Lai Y, Wang S, Li P, Xiao J, Wang Y (2013) Analysis of Pogostemon cablin from pharmaceutical research to market performances. Expert Opinion Investigational Drugs 22(2):245–257. 10.1517/13543784.2013.754882 [DOI] [PubMed] [Google Scholar]
- Chen HC, Chang WT, Hseu YC, Chen HY, Chuang CH, Lin CC, Lin MK (2016) Immunosuppressive effect of Litsea cubeba L. essential oil on dendritic cell and contact hypersensitivity responses. Int J Mol Sci. 10.3390/ijms17081319 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen BL, Chen YQ, Ma BH, Yu SF, Li LY, Zeng QX, Li CW (2018) Tetrahydrocurcumin, a major metabolite of curcumin, ameliorates allergic airway inflammation by attenuating Th2 response and suppressing the IL-4Rα-Jak1-STAT6 and Jagged1/Jagged2 -Notch1/Notch2 pathways in asthmatic mice. Clin Exp Allergy 48(11):1494–1508. 10.1111/cea.13258 [DOI] [PubMed] [Google Scholar]
- Chen H, He Y, Chen S, Qi S, Shen J (2020) Therapeutic targets of oxidative/nitrosative stress and neuroinflammation in ischemic stroke: Applications for natural product efficacy with omics and systemic biology. Pharmacol Res 158:104877 [DOI] [PubMed] [Google Scholar]
- Choi YJ, Kim J, Jung JY, Kwon H, Park JWJA, Asthma, (2019) Underuse of epinephrine for pediatric anaphylaxis victims in the emergency department: a population-based study. Allergy Asthuma Immune 11(4):529–537 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chu H, Tang Q, Huang H, Hao W, Wei X (2016) Grape-seed proanthocyanidins inhibit the lipopolysaccharide-induced inflammatory mediator expression in RAW264.7 macrophages by suppressing MAPK and NF-κb signal pathways. Environ Toxicol Pharmacol 41:159–166. 10.1016/j.etap.2015.11.018 [DOI] [PubMed] [Google Scholar]
- Cianferoni A (2021) Non–IgE-mediated anaphylaxis. Journal of Allergy and Clinical Immunology 147(4):1123–1131. 10.1016/j.jaci.2021.02.012 [DOI] [PubMed] [Google Scholar]
- Cimen SS, Suleyman A, Yucel E, Guler N, Tamay Z (2023) Evaluation of the triggers and the treatment models of anaphylaxis in pediatric patients. Northern Clin Istanbul 10(5):609 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cingi, C., Bayar Muluk, N., Cingi, C., & Bayar Muluk, N. (2020). Pathophysiology of Anaphylaxis. Quick Guide to Anaphylaxis, 163–182.
- Civelek M, Bilotta S, Lorentz A (2022) Resveratrol attenuates mast cell mediated allergic reactions: potential for use as a nutraceutical in allergic diseases? Mol Nutr Food Res 66(15):2200170. 10.1002/mnfr.202200170 [DOI] [PubMed] [Google Scholar]
- Clark E, Kase Tanno L, Vo T, Blanc B, Demoly P, Caimmi D (2023) Anaphylaxis management in a French pediatric emergency department: Lessons from the ANA-PED study. Clin Transl Allergy 13(8):e12289. 10.1002/clt2.12289 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Collaco CR, Hochman DJ, Goldblum RM, Brooks EG (2006) Effect of sodium sulfite on mast cell degranulation and oxidant stress. Ann Allergy Asthma Immunol 96(4):550–556. 10.1016/S1081-1206(10)63549-1 [DOI] [PubMed] [Google Scholar]
- Comberiati P, Innocenti F, Bernardini R, Landi M, Martelli A, Peroni D (2024) Diagnosis and management of anaphylaxis in children. Global Pediatrics 7:100106. 10.1016/j.gpeds.2023.100106 [Google Scholar]
- Crozier RWE, Yousef M, Coish JM, Fajardo VA, Tsiani E, MacNeil AJ (2023) Carnosic acid inhibits secretion of allergic inflammatory mediators in IgE-activated mast cells via direct regulation of Syk activation. J Biol Chem 299(4):102867. 10.1016/j.jbc.2022.102867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui J, Zhang M, Zhang Y-Q, Xu Z-H (2007) JNK pathway: diseases and therapeutic potential. Acta Pharmacol Sin 28(5):601–608. 10.1111/j.1745-7254.2007.00579.x [DOI] [PubMed] [Google Scholar]
- Dai J, Patel JD, Mumper RJ (2007) Characterization of blackberry extract and its antiproliferative and anti-inflammatory properties. J Med Food 10(2):258–265. 10.1089/jmf.2006.238 [DOI] [PubMed] [Google Scholar]
- Dasiman R, Nor NM, Eshak Z, Mutalip SSM, Suwandi NR, Bidin H (2022) A review of procyanidin: Updates on current bioactivities and potential health benefits. Biointerface Res Appl Chem 12(5):5918–5940 [Google Scholar]
- Davis RJ (2000) Signal transduction by the JNK group of MAP kinases. Cell 103(2):239–252 [DOI] [PubMed] [Google Scholar]
- del Cornò M, Scazzocchio B, Masella R, Gessani S (2016) Regulation of dendritic cell function by dietary polyphenols. Crit Rev Food Sci Nutr 56(5):737–747 [DOI] [PubMed] [Google Scholar]
- Deng Z, Xia Y, Chen L, Zhao Y, Wang R, Liang G (2023) Insight into covalent conjugates of β-lactoglobulin with rutin: Characterizing allergenicity, digestibility, and antioxidant properties in vitro. Food Res Int 173:113401. 10.1016/j.foodres.2023.113401 [DOI] [PubMed] [Google Scholar]
- Dera A, Rajagopalan P, Ahmed I, Alfhili M, Alsughayyir J, Chandramoorthy HC (2020) Thymoquinone attenuates IgE-mediated allergic response via pi3k-Akt-NFκB pathway and upregulation of the Nrf2-HO1 axis. J Food Biochem 44(6):e13216. 10.1111/jfbc.13216 [DOI] [PubMed] [Google Scholar]
- Dodd A, Hughes A, Sargant N, Whyte AF, Soar J, Turner PJ (2021) Evidence update for the treatment of anaphylaxis. Resuscitation 163:86–96. 10.1016/j.resuscitation.2021.04.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong X, Lan W, Yin X, Yang C, Wang W, Ni J (2017) Simultaneous determination and pharmacokinetic study of quercetin, luteolin, and apigenin in rat plasma after oral administration of matricaria chamomilla L extract by HPLC-UV. Evidence-Based Complemen Alternative Med 2017(1):8370584. 10.1155/2017/8370584 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong J, Xu O, Wang J, Shan C, Ren X (2021) Luteolin ameliorates inflammation and Th1/Th2 imbalance via regulating the TLR4/NF-κB pathway in allergic rhinitis rats. Immunopharmacol Immunotoxicol 43(3):319–327 [DOI] [PubMed] [Google Scholar]
- Dreborg S, Kim H (2021) The pharmacokinetics of epinephrine/adrenaline autoinjectors. Allergy Asthma Clin Immunol 17:1–11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dretchen KL, Mesa Z, Robben M, Slade D, Hill S, Croutch C, Mesa M (2020) Intranasal epinephrine in dogs: Pharmacokinetic and heart rate effects. Pharmacol Res Perspect 8(2):e00587. 10.1002/prp2.587 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dribin TE, Schnadower D, Spergel JM, Campbell RL, Shaker M, Neuman MI, Brousseau DC (2021) Severity grading system for acute allergic reactions: a multidisciplinary Delphi study. J Allergy Clin Immunol 148(1):173–181 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dribin TE, Motosue MS, Campbell RL (2023) Overview of allergy and anaphylaxis. Immunol Allergy Clin 43(3):435–451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dubus J-C, Lê M-S, Vitte J, Minodier P, Boutin A, Carsin A (2019) Use of epinephrine in emergency department depends on anaphylaxis severity in children. Eur J Pediatr 178:69–75 [DOI] [PubMed] [Google Scholar]
- DuToit G, Smith P, Muraro A, Fox AT, Roberts G, Ring J, Worm M (2024) Identifying patients at risk of anaphylaxis. World Allergy Organ J 17(6):100904 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duvauchelle, T., Robert, P., Donazzolo, Y., Loyau, S., Orlandini, B., Lehert, P., . . . Schwartz, J.-C. (2018). Bioavailability and Cardiovascular Effects of Adrenaline Administered by Anapen Autoinjector in Healthy Volunteers. The Journal of Allergy and Clinical Immunology: In Practice, 6(4), 1257–1263. 10.1016/j.jaip.2017.09.021 [DOI] [PubMed]
- Ebo DG, Clarke RC, Mertes P-M, Platt PR, Sabato V, Sadleir PHM (2019) Molecular mechanisms and pathophysiology of perioperative hypersensitivity and anaphylaxis: a narrative review. Br J Anaesth 123(1):e38–e49. 10.1016/j.bja.2019.01.031 [DOI] [PubMed] [Google Scholar]
- Elgendy, A. A. M. (2017). Phytoconstituents, LC-ESI-MS profile, antioxidant and antimicrobial activities of Citrus x limon L. Burm. f. cultivar variegated pink lemon. Journal of Pharmaceutical Sciences and Research (2017), 9 (4): 375–391.
- Elidahanum, H., Relin, Y., & Yufri, A. (2020). The Extract of Kincung Flower (Etlingera elatior (Jack) R.M.Sm.) Activity to Decrease IL-4 and IgE Levels in Type I Hypersensitivity White Male Mice. Pharmacognosy Journal, 12(4).
- Erlich TH, Yagil Z, Kay G, Peretz A, Migalovich-Sheikhet H, Tshori S, Razin E (2014) Mitochondrial STAT3 plays a major role in IgE-antigen–mediated mast cell exocytosis. Journal of Allergy and Clinical Immunology 134(2):460-469.e410. 10.1016/j.jaci.2013.12.1075 [DOI] [PubMed] [Google Scholar]
- Eseberri I, Trepiana J, Léniz A, Gómez-García I, Carr-Ugarte H, González M, Portillo MP (2022) Variability in the Beneficial Effects of Phenolic Compounds: A Review. Nutrients 14(9):1925 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fang F, Tang Y, Gao Z, Xu Q (2010) A novel regulatory mechanism of naringenin through inhibition of T lymphocyte function in contact hypersensitivity suppression. Biochem Biophys Res Commun 397(2):163–169. 10.1016/j.bbrc.2010.05.065 [DOI] [PubMed] [Google Scholar]
- Farhan M, Rizvi A, Aatif M, Muteeb G, Khan K, Siddiqui FA (2024) Dietary Polyphenols, Plant Metabolites, and Allergic Disorders: A Comprehensive Review. Pharmaceuticals 17(6):670 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng H, Xiong X, Xu Q, Zhang Z, Feng J, Wu Y (2020) Study on the immunomodulatory effect of quercetin nanoparticles loaded with chitosan on a mouse model of ovalbumin-induced food allergy. Nanosci Nanotechnol Lett 12(7):915–920 [Google Scholar]
- Fonacier, L., Uter, W., & Johansen, J. D. (2024). Recognizing and managing allergic contact dermatitis: focus on major allergens. The Journal of Allergy and Clinical Immunology: In Practice, 12(9), 2227–2241. [DOI] [PubMed]
- Fowler J, Lieberman P (2022) Pathophysiology of Immunologic and Nonimmunologic Systemic Reactions Including Anaphylaxis. Immunol Allergy Clin North Am 42(1):27–43. 10.1016/j.iac.2021.09.011 [DOI] [PubMed] [Google Scholar]
- Gabrielli, S., Clarke, A., Morris, J., Eisman, H., Gravel, J., Enarson, P., . . . Ben-Shoshan, M. (2019). Evaluation of Prehospital Management in a Canadian Emergency Department Anaphylaxis Cohort. The Journal of Allergy and Clinical Immunology: In Practice, 7(7), 2232–2238.e2233. 10.1016/j.jaip.2019.04.018 [DOI] [PubMed]
- Ge H-Q, Wan G-S, Wang D, Wu J-M, Sun B-H, Wu L-J, Gao H-Y (2016) Two new isoxazolines from the husks of Xanthoceras sorbifolia Bunge. J Asian Nat Prod Res 18(8):744–751. 10.1080/10286020.2016.1167045 [DOI] [PubMed] [Google Scholar]
- Godon O, Hechler B, Jönsson F (2021) The role of IgG subclasses and platelets in experimental anaphylaxis. Journal of Allergy and Clinical Immunology 147(4):1209–1211 [DOI] [PubMed] [Google Scholar]
- Grabenhenrich LB, Dölle S, Moneret-Vautrin A, Köhli A, Lange L, Spindler T, Ruëff F, Nemat K, Maris I, Roumpedaki E, Scherer K, Ott H, Reese T, Mustakov T, Lang R, Fernandez-Rivas M, Kowalski ML, Bilò MB, Hourihane JO, Papadopoulos NG, Beyer K, Muraro A, Worm M (2016) Anaphylaxis in children and adolescents: The European Anaphylaxis Registry. J Allergy Clin Immunol 137(4):1128–1137.e1 [DOI] [PubMed]
- Grabenhenrich LB, Dölle S, Ruëff F, Renaudin JM, Scherer K, Pföhler C, Treudler R, Koehli A, Mahler V, Spindler T, Lange L, Bilò MB, Papadopoulos NG, Hourihane JOB, Lang R, Fernández-Rivas M, Christoff G, Cichocka-Jarosz E, Worm M (2018) Epinephrine in Severe Allergic Reactions: The European Anaphylaxis Register. J Allergy Clin Immunol Pract 6(6):1898-1906.e1 [DOI] [PubMed]
- Greenhawt M, Gupta RS, Meadows JA, Pistiner M, Spergel JM, Camargo CA Jr, Simons FER, Lieberman PL (2019) Guiding Principles for the Recognition, Diagnosis, and Management of Infants with Anaphylaxis: An Expert Panel Consensus. J Allergy Clin Immunol Pract 7(4):1148-1156.e5 [DOI] [PubMed]
- Gunasena, P., & Jayawardana, A. (2024). Differentiating anaphylaxis from other allergic reactions: A clinical guide. In: ResearchGate. https://www.researchgate.net/publication
- Guo YJ, Pan WW, Liu SB, Shen ZF, Xu Y, Hu LL (2020) ERK/MAPK signalling pathway and tumorigenesis (Review). Exp Ther Med 19(3):1997–2007. 10.3892/etm.2020.8454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haftcheshmeh SM, Mirhafez SR, Abedi M, Heydarlou H, Shakeri A, Mohammadi A, Sahebkar A (2022) Therapeutic potency of curcumin for allergic diseases: A focus on immunomodulatory actions. Biomed Pharmacother 154:113646. 10.1016/j.biopha.2022.113646 [DOI] [PubMed] [Google Scholar]
- Hagar RE, Burgstahler AD, Nathanson MH, Ehrlich BE (1998) Type III InsP3 receptor channel stays open in the presence of increased calcium. Nature 396(6706):81–84. 10.1038/23954 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammad M, Adam N, Sarfaraz K, Adam M, Mansoor NJC (2022) A Case report of lifesaving intravenous bolus epinephrine administration in a case of severe refractory anaphylactic shock 14(8):e28249 [DOI] [PMC free article] [PubMed]
- Hammad H, Lambrecht BN (2015) Barrier Epithelial Cells and the Control of Type 2 Immunity. Immunity 43(1):29–40. 10.1016/j.immuni.2015.07.007 [DOI] [PubMed] [Google Scholar]
- Han P, Ma X, Yin J (2010) The effects of lipoic acid on soybean ?-conglycinin-induced anaphylactic reactions in a rat model. Arch Anim Nutr 64(3):254–264. 10.1080/17450391003625003 [DOI] [PubMed] [Google Scholar]
- Hartzell, C. A., Jankowska, K. I., Burkhardt, J. K., & Lewis, R. S. (2016). Calcium influx through CRAC channels controls actin organization and dynamics at the immune synapse. eLife, 5, e14850. 10.7554/eLife.14850 [DOI] [PMC free article] [PubMed]
- Hassanin M, Tolba M, Tadros M, Elmazar M, Singab AN (2019) Wogonin a Promising Component of Scutellaria baicalensis: A Review on its Chemistry, Pharmacokinetics and Biological Activities. Archives of Pharmaceutical Sciences Ain Shams University 3:170–179. 10.21608/aps.2019.18854.1016 [Google Scholar]
- He J-J, Chen H-M, Li C-W, Wu D-W, Wu X-L, Shi S-J, Lai X-P (2013) Experimental study on antinociceptive and anti-allergy effects of patchouli oil. J Essent Oil Res 25(6):488–496. 10.1080/10412905.2013.809319 [Google Scholar]
- He W, Zhang T, Velickovic TC, Li S, Lyu Y, Wang L, Wu X (2020) Covalent conjugation with (−)-epigallo-catechin 3-gallate and chlorogenic acid changes allergenicity and functional properties of Ara h1 from peanut. Food Chem 331:127355. 10.1016/j.foodchem.2020.127355 [DOI] [PubMed] [Google Scholar]
- He W, He K, Liu X, Ye L, Lin X, Ma L, Wu X (2023) Modulating the allergenicity and functional properties of peanut protein by covalent conjugation with polyphenols. Food Chem 415:135733. 10.1016/j.foodchem.2023.135733 [DOI] [PubMed] [Google Scholar]
- He J-J, Chen H-M, Li C-W, Wu D-W, Wu X-L, Shi S-J, Lai X-P (2013) Experimental study on antinociceptive and anti-allergy effects of patchouli oil. J Essent Oil Res 25(6):488–496 10.1080/10412905.2013.809319
- Tal Y, Ribak Y, Rubin L, Talmon A, Shamriz O, Hershko AY, Blotnick S, Bouhajib M, Krayz GT, Abrutzky C, Megiddo D, Lapidot T, Caraco Y (2023) Fast acting, dry powder, needle-free, intranasal epinephrine spray: A promising future treatment for anaphylaxis. J Allergy Clin Immunol Pract 11(10):3047–3054 [DOI] [PubMed]
- Hirai S, Kim Y II, Goto T, Kang M-S, Yoshimura M, Obata A, Kawada T (2007) Inhibitory effect of naringenin chalcone on inflammatory changes in the interaction between adipocytes and macrophages. Life Sci 81(16):1272–1279. 10.1016/j.lfs.2007.09.001 [DOI] [PubMed] [Google Scholar]
- Hitl M, Kladar N, Gavarić N, Božin B (2020) Rosmarinic Acid-Human Pharmacokinetics and Health Benefits. Planta Med 87(04):273–282. 10.1055/a-1301-8648 [DOI] [PubMed] [Google Scholar]
- Hu X, li, J., Fu, M., Zhao, X., & Wang, W. (2021) The JAK/STAT signaling pathway: from bench to clinic. Signal Transduct Target Ther 6(1):402. 10.1038/s41392-021-00791-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu G, Peng C, Xie X, Zhang S, Cao X (2017) Availability, pharmaceutics, security, pharmacokinetics, and pharmacological activities of patchouli alcohol. Evid Based Complement Alternat Med 2017(1):4850612 10.1155/2017/4850612 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang R-Y, Yu Y-L, Cheng W-C, OuYang C-N, Fu E, Chu C-L (2010) Immunosuppressive Effect of Quercetin on Dendritic Cell Activation and Function. J Immunol 184(12):6815–6821. 10.4049/jimmunol.0903991 [DOI] [PubMed] [Google Scholar]
- Humeniuk P, Dubiela P, Hoffmann-Sommergruber K (2017) Dendritic Cells and Their Role in Allergy: Uptake, Proteolytic Processing and Presentation of Allergens. Int J Mol Sci 18(7):1491 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hwang K-A, Hwang Y-J, Song J (2018) Anti-allergic effect of Aster yomena on ovalbumin-sensitized mouse and RHL-2H3 cells via Th1/Th2 cytokine balance. Journal of Functional Foods 44:1–8. 10.1016/j.jff.2018.02.026 [Google Scholar]
- Hwang, D., Kang, M. J., Kang, C. W., & Kim, G. D. (2019). Kaempferol‑3‑O‑β‑rutinoside suppresses the inflammatory responses in lipopolysaccharide‑stimulated RAW264.7 cells via the NF‑κB and MAPK pathways. Int J Mol Med, 44(6), 2321–2328. 10.3892/ijmm.2019.4381 [DOI] [PubMed]
- Imakiire R, Fujisawa T, Nagao M, Tokuda R, Hattori T, Kainuma K, Kawano Y. Basophil Activation Test Based on CD203c Expression in the Diagnosis of Fish Allergy (2020) Allergy Asthma Immunol Res 12(4):641–652 [DOI] [PMC free article] [PubMed]
- Itoh T, Ohguchi K, Nakajima C, Oyama M, Iinuma M, Nozawa Y, Ito M (2011) Inhibitory effects of flavonoid glycosides isolated from the peel of Japanese persimmon (Diospyros kaki Fuyu) on antigen-stimulated degranulation in rat basophilic leukaemia RBL-2H3 cells. Food Chem 126(1):289–294. 10.1016/j.foodchem.2010.10.058 [DOI] [PubMed] [Google Scholar]
- Jeong HJ, Lee SA, Moon PD, Na HJ, Park RK, Um JY, Hong SH (2006) Alginic acid has anti-anaphylactic effects and inhibits inflammatory cytokine expression via suppression of nuclear factor-kappaB activation. Clin Exp Allergy 36(6):785–794. 10.1111/j.1365-2222.2006.02508.x [DOI] [PubMed] [Google Scholar]
- Ji N, Pan S, Shao C, Chen Y, Zhang Z, Wang R, Kong D (2018) Spinacetin Suppresses the Mast Cell Activation and Passive Cutaneous Anaphylaxis in Mouse Model. Front Pharmacol 9:824. 10.3389/fphar.2018.00824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jia B, Shang J, Zeng H, Wang X, Fang M, Xu L, Yang Q (2023) Hepatoprotective Effects of Rosmarinic Acid on Ovalbumin-Induced Intestinal Food Allergy Mouse Model. Molecules 28(2):788 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang D-X, Liu S-R, Zhang M-H, Zhang T, Ma W-J, Mu X, Chen W (2015) Luteolin prevents fMLP-induced neutrophils adhesion via suppression of LFA-1 and phosphodiesterase 4 activity. J Integr Agric 14(1):140–147. 10.1016/S2095-3119(14)60904-7 [Google Scholar]
- Jiao W-H, Cheng B-H, Shi G-H, Chen G-D, Gu B-B, Zhou Y-J, Lin H-W (2017) Dysivillosins A-D, Unusual Anti-allergic Meroterpenoids from the Marine Sponge Dysidea villosa. Sci Rep 7(1):8947. 10.1038/s41598-017-04021-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jimenez-Rodriguez TW, Garcia-Neuer M, Alenazy LA, Castells M (2018) Anaphylaxis in the 21st century: phenotypes, endotypes, and biomarkers. J Asthma Allergy 11:121–142. 10.2147/jaa.S159411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin J, Fan YJ, Nguyen TV, Yu ZN, Song CH, Lee S-Y, Chai OH (2023) Fallopia japonica Root Extract Ameliorates Ovalbumin-Induced Airway Inflammation in a CARAS Mouse Model by Modulating the IL-33/TSLP/NF-κB Signaling Pathway. Int J Mol Sci 24(15):12514 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jo H, Shim K, Jeoung D (2022) The Crosstalk between FcεRI and Sphingosine Signaling in Allergic Inflammation. Int J Mol Sci 23(22):13892 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jönsson F, de Chaisemartin L, Granger V, Gouel-Chéron A, Gillis CM, Zhu Q, Dib F, Nicaise-Roland P, Ganneau C, Hurtado-Nedelec M, Paugam-Burtz C, Necib S, Keita-Meyer H, Le Dorze M, Cholley B, Langeron O, Jacob L, Plaud B, Fischler M, Sauvan C, Guinnepain MT, Montravers P, Aubier M, Bay S, Neukirch C, Tubach F, Longrois D, Chollet-Martin S, Bruhns P (2019) An IgG-induced neutrophil activation pathway contributes to human drug-induced anaphylaxis. Sci Transl Med 10:11(500):eaat1479 [DOI] [PubMed]
- Jose S, Devi SS, P, S., & Al-Khafaji, K. (2022) Phytochemical constituents of Inula britannica as potential inhibitors of dihydrofolate reductase: A strategic approach against shigellosis. J Biomol Struct Dyn 40(22):11932–11947. 10.1080/07391102.2021.1966508 [DOI] [PubMed] [Google Scholar]
- Juwita T, Puspitasari IM, Levita J (2018) Torch Ginger (Etlingera elatior): A Review on its Botanical Aspects, Phytoconstituents and Pharmacological Activities. Pak J Biol Sci 21:151–165. 10.3923/pjbs.2018.151.165 [DOI] [PubMed] [Google Scholar]
- Kabashima, K., Nakashima, C., Nonomura, Y., Otsuka, A., Cardamone, C., Parente, R., . . . Triggiani, M. J. I. r. (2018). Biomarkers for evaluation of mast cell and basophil activation. 282(1), 114–120. [DOI] [PubMed]
- Kandhare AD, Aswar UM, Mohan V, Thakurdesai PA (2017) Ameliorative effects of type-A procyanidins polyphenols from cinnamon bark in compound 48/80-induced mast cell degranulation. Anatomy & Cell Biology 50(4):275–283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kang H-K, Ecklund D, Liu M, Datta SK (2009) Apigenin, a non-mutagenic dietary flavonoid, suppresses lupus by inhibiting autoantigen presentation for expansion of autoreactive Th1 and Th17 cells. Arthritis Res Ther 11(2):R59. 10.1186/ar2682 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karunarathna I, Kusumarathna K, Jayathilaka P, Rathnayake B, Bandara S, Abeyak M, Priyalath N, Gunarathna I, Disanayake D, Kurukulasooriya P, Samarasinghe A, Ratnayake C, Withanagama C (2024) How to recognize, respond to, and prevent anaphylaxis: A comprehensive guide. Uva Clinical Lab. https://www.researchgate.net/publication/380096299
- Karunarathna I, Alvis K, Gunasena P, Hapuarachchi T, Ekanayake U, Rajapaksha S, Gunawardana K, Aluthge P, Gunathilake S, Bandara S, Jayawardana A (2024) Differentiating anaphylaxis from other allergic reactions: a clinical guide. 10.13140/RG.2.2.15115.30244
- Ke X, Chen Z, Wang X, Kang H, Hong S (2023) Quercetin improves the imbalance of Th1/Th2 cells and Treg/Th17 cells to attenuate allergic rhinitis. Autoimmunity 56(1):2189133 [DOI] [PubMed] [Google Scholar]
- Kim TH, Yoon SH, Lee SY, Choi YH, Park CM, Kang HR, Cho SH (2018) Biphasic and protracted anaphylaxis to iodinated contrast media. Eur Radiol 28(3):1242–1252. 10.1007/s00330-017-5052-0 [DOI] [PubMed] [Google Scholar]
- Kim, L., Nevis, I. F., Tsai, G., Dominic, A., Potts, R., Chiu, J., . . . Immunology, C. (2014). Children under 15 kg with food allergy may be at risk of having epinephrine auto-injectors administered into bone. 10, 1–6. [DOI] [PMC free article] [PubMed]
- Kim, M.-J., Je, I.-G., Song, J., Fei, X., Lee, S., Yang, H., . . . Kim, S.-H. (2020). SG-SP1 Suppresses Mast Cell-Mediated Allergic Inflammation via Inhibition of FcεRI Signaling. Frontiers in Immunology, 11. 10.3389/fimmu.2020.00050 [DOI] [PMC free article] [PubMed]
- Kimura R, Schwartz JA, Romeiser JL, Senzel L, Galanakis D, Halper D, Bennett-Guerrero E (2024) The Acute Effect of Chamomile Intake on Blood Coagulation Tests in Healthy Volunteers: A Randomized Trial. The Journal of Applied Laboratory Medicine 9(3):468–476. 10.1093/jalm/jfad120 [DOI] [PubMed] [Google Scholar]
- Kobayashi S, Kato T, Azuma T, Kikuzaki H, Abe K (2015) Anti-allergenic activity of polymethoxyflavones from Kaempferia parviflora. Journal of Functional Foods 13:100–107. 10.1016/j.jff.2014.12.029 [Google Scholar]
- Kong Z-L, Sudirman S, Lin H-J, Chen W-N (2020) In vitro anti-inflammatory effects of curcumin on mast cell-mediated allergic responses via inhibiting FcεRI protein expression and protein kinase C delta translocation. Cytotechnology 72(1):81–95. 10.1007/s10616-019-00359-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kow ASF, Chik A, Soo K-M, Khoo LW, Abas F, Tham CL (2019) Identification of soluble mediators in IgG-mediated anaphylaxis via Fcγ receptor: a meta-analysis. Front Immunol 10:190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kraft, M., Hofmeier, K. S., Ruëff, F., Pföhler, C., Renaudin, J.-M., Bilò, M. B., . . . Practice, C. I. I. (2020). Risk factors and characteristics of biphasic anaphylaxis. 8(10):3388–3395. e3386. [DOI] [PubMed]
- Krishnaswamy G (2021) Critical care management of the patient with anaphylaxis: a Concise definitive review. Crit Care Med 49(5):838–857 [DOI] [PubMed] [Google Scholar]
- Kumari, M., Siddiqui, M. A., & Gupta, A. (2023). Recent Advancement and Novel Application of Natural Polyphenols for the Treatment of Allergy Asthma: From Phytochemistry to Biological Implications. Critical Reviews™ in Immunology, 43(4). [DOI] [PubMed]
- Lee H-S, Jeong G-S (2021) Therapeutic effect of kaempferol on atopic dermatitis by attenuation of T cell activity via interaction with multidrug resistance-associated protein 1. Br J Pharmacol 178(8):1772–1788. 10.1111/bph.15396 [DOI] [PubMed] [Google Scholar]
- Lee J-Y, Park S-H, Jhee K-H, Yang S-A (2020) Tricin Isolated from Enzyme-Treated Zizania latifolia Extract Inhibits IgE-Mediated Allergic Reactions in RBL-2H3 Cells by Targeting the Lyn/Syk Pathway. Molecules 25(9):2084 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee H-S, Kim J, Choi HG, Kim E-K, Jun C-D (2021) Licoricidin Abrogates T-Cell Activation by Modulating PTPN1 Activity and Attenuates Atopic Dermatitis In Vivo. J Investig Dermatol 141(10):2490-2498.e2496. 10.1016/j.jid.2021.02.759 [DOI] [PubMed] [Google Scholar]
- Lee, S., Bellolio, M. F., Hess, E. P., Erwin, P., Murad, M. H., Campbell, R. L. J. T. J. o. A., & Practice, C. I. I. (2015). Time of onset and predictors of biphasic anaphylactic reactions: a systematic review and meta-analysis. 3(3), 408–416. e402. [DOI] [PubMed]
- Li J, Lin X, Liu X, Ma Z, Li Y (2020) Baicalin regulates Treg/Th17 cell imbalance by inhibiting autophagy in allergic rhinitis. Mol Immunol 125:162–171. 10.1016/j.molimm.2020.07.008 [DOI] [PubMed] [Google Scholar]
- Li Q-S, Wang Y-Q, Liang Y-R, Lu J-L (2021) The anti-allergic potential of tea: a review of its components, mechanisms and risks. Food Funct 12(1):57–69. 10.1039/D0FO02091E [DOI] [PubMed] [Google Scholar]
- Li Y, Leung PSC, Gershwin ME, Song J (2022) New Mechanistic Advances in FcεRI-Mast Cell-Mediated Allergic Signaling. Clin Rev Allergy Immunol 63(3):431–446. 10.1007/s12016-022-08955-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li L, Chai W, Ma L, Zhang T, Chen J, Zhang J, Wu X (2023) Covalent polyphenol with soybean 11S protein to develop hypoallergenic conjugates for potential immunotherapy. Journal of Functional Foods 104:105518. 10.1016/j.jff.2023.105518 [Google Scholar]
- Liang Z, Wu L, Deng X, Liang Q, Xu Y, Deng R, He J (2020) The Antioxidant Rosmarinic Acid Ameliorates Oxidative Lung Damage in Experimental Allergic Asthma via Modulation of NADPH Oxidases and Antioxidant Enzymes. Inflammation 43(5):1902–1912. 10.1007/s10753-020-01264-3 [DOI] [PubMed] [Google Scholar]
- Lim S, Oh S, Nguyen QTN, Kim M, Zheng S, Fang M, Yi T-H (2023) Rosa davurica Inhibited Allergic Mediators by Regulating Calcium and Histamine Signaling Pathways. Plants 12(7):1572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin X, Ye L, He K, Zhang T, Sun F, Mei T, Wu X (2022) A new method to reduce allergenicity by improving the functional properties of soybean 7S protein through covalent modification with polyphenols. Food Chem 373:131589. 10.1016/j.foodchem.2021.131589 [DOI] [PubMed] [Google Scholar]
- Liou J, Kim ML, Do Heo W, Jones JT, Myers JW, Ferrell JE Jr, Meyer T (2005) STIM Is a Ca<sup>2+</sup> Sensor Essential for Ca<sup>2+</sup>-Store-Depletion-Triggered Ca<sup>2+</sup> Influx. Curr Biol 15(13):1235–1241. 10.1016/j.cub.2005.05.055 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S-H, Lin C-H, Hung S-K, Chou J-H, Chi C-W, Fu S-L (2010) Fisetin Inhibits Lipopolysaccharide-Induced Macrophage Activation and Dendritic Cell Maturation. J Agric Food Chem 58(20):10831–10839. 10.1021/jf1017093 [DOI] [PubMed] [Google Scholar]
- Liu J, Wang Y, Tu Z-C, Chen W-M, Yuan T (2021a) Bovine β-Lactoglobulin Covalent Modification by Flavonoids: Effect on the Allergenicity and Human Intestinal Microbiota. J Agric Food Chem 69(24):6820–6828. 10.1021/acs.jafc.1c02482 [DOI] [PubMed] [Google Scholar]
- Liu X, Song Q, Li X, Chen Y, Liu C, Zhu X, Huang J (2021b) Effects of different dietary polyphenols on conformational changes and functional properties of protein–polyphenol covalent complexes. Food Chem 361:130071. 10.1016/j.foodchem.2021.130071 [DOI] [PubMed] [Google Scholar]
- Liu Y, Shu Z, Li Y, Chen H, Liu H, Yang X, Liu Q (2023) Deep-sea-derived viridicatol relieves allergic response by suppressing MAPK and JAK-STAT signalling pathways of RBL-2H3 cells. Food Hydrocolloids 34(1):2207791. 10.1080/09540105.2023.2207791 [Google Scholar]
- Liu T, Zhang R, Jiang L, Zhou L, Zhang H, Liang F, Shen X (2024) The potential application and molecular mechanisms of natural products in the treatment of allergic rhinitis: A review. Phytomedicine 129:155663 [DOI] [PubMed] [Google Scholar]
- Liu, C., Zhu, L., Fukuda, K., Ouyang, S., Chen, X., Wang, C., . . . Li, X. (2017). The flavonoid cyanidin blocks binding of the cytokine interleukin-17A to the IL-17RA subunit to alleviate inflammation in vivo. Science Signaling, 10(467), eaaf8823. 10.1126/scisignal.aaf8823 [DOI] [PMC free article] [PubMed]
- Liu, X., Lee, S., Lohse, C. M., Hardy, C. T., Campbell, R. L. J. T. J. o. A., & Practice, C. I. I. (2020). Biphasic reactions in emergency department anaphylaxis patients: a prospective cohort study. 8(4), 1230–1238. [DOI] [PubMed]
- Liu, X. (2020). Biphasic Reactions in Emergency Department Anaphylaxis Patients: A Prospective Cohort Study. The Journal of Allergy and Clinical Immunology: In Practice, 8(4), 1230–1238. 10.1016/j.jaip.2019.10.027 [DOI] [PubMed]
- Lv L, Qu X, Yang N, Liu Z, Wu X (2021) Changes in structure and allergenicity of shrimp tropomyosin by dietary polyphenols treatment. Food Res Int 140:109997. 10.1016/j.foodres.2020.109997 [DOI] [PubMed] [Google Scholar]
- Ma C, Ma Z, Fu Q, Ma S (2013) Curcumin attenuates allergic airway inflammation by regulation of CD4+CD25+ regulatory T cells (Tregs)/Th17 balance in ovalbumin-sensitized mice. Fitoterapia 87:57–64. 10.1016/j.fitote.2013.02.014 [DOI] [PubMed] [Google Scholar]
- Magrone, T., Magrone, M., Russo, M. A., & Jirillo, E. (2020). Recent Advances on the Anti-Inflammatory and Antioxidant Properties of Red Grape Polyphenols: In Vitro and In Vivo Studies. Antioxidants, 9(1), 35. [DOI] [PMC free article] [PubMed]
- Manchanda Y, Das S (2023) Anaphylaxis–A must know for all. Indian Journal of Skin Allergy 2(1):8–13 [Google Scholar]
- Marie, S. A. (2020). Regulation 28: Prevention of Future Deaths report. www.judiciary.uk/wp-content/uploads/2020/08/Shant-Turay Thomas-2020–0124_Redacted.pdf.
- Maris I, Dölle-Bierke S, Renaudin JM, Lange L, Koehli A, Spindler T, Kemen CJA (2021) Peanut-Induced Anaphylaxis in Children and Adolescents: Data from the European Anaphylaxis Registry. 76(5):1517–1527 [DOI] [PubMed] [Google Scholar]
- Martínez-Fernandez P, Vallejo-de-Torres G, Sánchez-de-León-Robles MS, Navarro-Escayola E, Moro-Moro M, Alberti-Masgrau N, Tejedor-Alonso MA (2019) Medical and pathologic characteristics of fatal anaphylaxis: a Spanish nationwide 17-year series. Forensic Sci Med Pathol 15:369–381 [DOI] [PubMed] [Google Scholar]
- Matsuda H, Morikawa T, Xie H, Yoshikawa M (2004) Antiallergic phenanthrenes and stilbenes from the tubers of Gymnadenia conopsea. Planta Med 70(9):847–855. 10.1055/s-2004-827234 [DOI] [PubMed] [Google Scholar]
- McLendon K, Sternard BT (2024) Anaphylaxis. In StatPearls. StatPearls Publishing, Treasure Island (FL) [PubMed] [Google Scholar]
- McLure M, Eastwood K, Parr M, Bray J (2021) A rapid review of advanced life support guidelines for cardiac arrest associated with anaphylaxis. Resuscitation 159:137–149. 10.1016/j.resuscitation.2020.10.001 [DOI] [PubMed] [Google Scholar]
- Michell, R. H. (1975). Inositol phospholipids and cell surface receptor function. Biochimica et Biophysica Acta (BBA) - Reviews on Biomembranes, 415(1), 81–147. 10.1016/0304-4157(75)90017-9 [DOI] [PubMed]
- Middleton E, Kandaswami C, Theoharides TC (2000) The Effects of Plant Flavonoids on Mammalian Cells: Implications for Inflammation, Heart Disease, and Cancer. Pharmacol Rev 52(4):673 [PubMed] [Google Scholar]
- Min, S.-Y., Park, C.-H., Yu, H.-W., & Park, Y.-J. (2021). Anti-Inflammatory and Anti-Allergic Effects of Saponarin and Its Impact on Signaling Pathways of RAW 264.7, RBL-2H3, and HaCaT Cells. International Journal of Molecular Sciences, 22(16), 8431. [DOI] [PMC free article] [PubMed]
- Mine Y, Majumder K, Jin Y, Zeng Y (2020) Chinese sweet tea (Rubus suavissimus) polyphenols attenuate the allergic responses in a Balb/c mouse model of egg allergy. Journal of Functional Foods 67:103827. 10.1016/j.jff.2020.103827 [Google Scholar]
- Mlcek J, Jurikova T, Skrovankova S, Sochor J (2016) Quercetin and Its Anti-Allergic Immune Response. Molecules 21(5):623 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molitorisova M, Sutovska M, Kazimierova I, Barborikova J, Joskova M, Novakova E, Franova S (2021) The anti-asthmatic potential of flavonol kaempferol in an experimental model of allergic airway inflammation. Eur J Pharmacol 891:173698. 10.1016/j.ejphar.2020.173698 [DOI] [PubMed] [Google Scholar]
- Monsieurs KG, Nolan JP, Bossaert LL, Greif R, Maconochie IK, Nikolaou NI, Wyllie JJR (2015) European resuscitation council guidelines for resuscitation 2015: section “Triage.” Executive Summary 95:1–80 [DOI] [PubMed] [Google Scholar]
- Moon P-D, Choi I-H, Kim H-M (2011) Berberine inhibits the production of thymic stromal lymphopoietin by the blockade of caspase-1/NF-κB pathway in mast cells. Int Immunopharmacol 11(11):1954–1959. 10.1016/j.intimp.2011.08.004 [DOI] [PubMed] [Google Scholar]
- Moon P-D, Han N-R, Lee JS, Jee H-W, Kim J-H, Kim H-M, Jeong H-J (2021) Effects of Resveratrol on Thymic Stromal Lymphopoietin Expression in Mast Cells. Medicina 57(1):21 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muraro A, Worm M, Alviani C, Cardona V, DunnGalvin A, Garvey LH, Clinical Immunology FAAGG (2022) EAACI guidelines: Anaphylaxis (2021 update). Allergy 77(2):357–377. 10.1111/all.15032 [DOI] [PubMed] [Google Scholar]
- Muraro, A., Roberts, G., Clark, A., Eigenmann, P. A., Halken, S., Lack, G., . . . Allergy, E. T. F. o. A. i. C. J. (2007). The management of anaphylaxis in childhood: position paper of the European academy of allergology and clinical immunology. 62(8), 857–871. [DOI] [PubMed]
- Muraro, A., Roberts, G., Worm, M., Bilò, M., Brockow, K., Fernández Rivas, M., . . . Beyer, K. J. A. (2014). Anaphylaxis: guidelines from the E uropean A cademy of A llergy and C linical I mmunology. 69(8), 1026–1045. [DOI] [PubMed]
- Mwakalukwa R, Ashour A, Amen Y, Niwa Y, Tamrakar S, Miyamoto T, Shimizu K (2019) Anti-allergic activity of polyphenolic compounds isolated from olive mill wastes. Journal of Functional Foods 58:207–217. 10.1016/j.jff.2019.04.058 [Google Scholar]
- Nagata K, Araumi S, Ando D, Ito N, Ando M, Ikeda Y, Nishiyama C (2023) Kaempferol Suppresses the Activation of Mast Cells by Modulating the Expression of FcεRI and SHIP1. Int J Mol Sci 24(6):5997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nassiri M, Babina M, Dölle S, Edenharter G, Ruëff F, Worm M (2015) Ramipril and metoprolol intake aggravate human and murine anaphylaxis: Evidence for direct mast cell priming. Journal of Allergy and Clinical Immunology 135(2):491–499. 10.1016/j.jaci.2014.09.004 [DOI] [PubMed] [Google Scholar]
- Nguyen, S. M. T., Rupprecht, C. P., Haque, A., Pattanaik, D., Yusin, J., & Krishnaswamy, G. (2021). Mechanisms Governing Anaphylaxis: Inflammatory Cells, Mediators, Endothelial Gap Junctions and Beyond. Int J Mol Sci, 22(15). 10.3390/ijms22157785 [DOI] [PMC free article] [PubMed]
- Nuñez-Borque E, Fernandez-Bravo S, Yuste-Montalvo A, Esteban V (2022) Pathophysiological, cellular, and molecular events of the vascular system in anaphylaxis. Front Immunol 13:836222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nurmatov UB, Rhatigan E, Simons FER, Sheikh A (2014) H2-antihistamines for the treatment of anaphylaxis with and without shock: a systematic review. Ann Allergy Asthma Immunol 112(2):126–131. 10.1016/j.anai.2013.11.010 [DOI] [PubMed] [Google Scholar]
- Panesar SS, Javad S, de Silva D, Nwaru BI, Hickstein L, Muraro A, Anaphylaxis G (2013) The epidemiology of anaphylaxis in Europe: a systematic review. Allergy 68(11):1353–1361. 10.1111/all.12272 [DOI] [PubMed] [Google Scholar]
- Paradis S, Charles A-L, Giannini M, Meyer A, Lejay A, Talha S, Geny B (2024) Targeting Mitochondrial Dynamics during Lower-Limb Ischemia Reperfusion in Young and Old Mice: Effect of Mitochondrial Fission Inhibitor-1 (mDivi-1). Int J Mol Sci 25(7):4025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parish HG, Morton JR, Brown JCJA (2019) Asthma, & Immunology, C. A Systematic Review of Epinephrine Stability and Sterility with Storage in a Syringe 15:1–13 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park J (2020) Anti-Anaphylactic Activity of Isoquercitrin (Quercetin-3-O-β-d-Glucose) in the Cardiovascular System of Animals. Biomedicines 8(6):139. 10.3390/biomedicines8060139 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park, C.-H., Min, S.-Y., Yu, H.-W., Kim, K., Kim, S., Lee, H.-J., . . . Park, Y.-J. (2020). Effects of Apigenin on RBL-2H3, RAW264.7, and HaCaT Cells: Anti-Allergic, Anti-Inflammatory, and Skin-Protective Activities. International Journal of Molecular Sciences, 21(13), 4620. [DOI] [PMC free article] [PubMed]
- Pasha, M. A., Patel, G., Hopp, R., & Yang, Q. (2019). Role of innate lymphoid cells in allergic diseases. Paper presented at the Allergy and asthma proceedings. [DOI] [PMC free article] [PubMed]
- Pastorello EA, Borgonovo L, Preziosi D, Schroeder JW, Pravettoni V, Aversano MG, Losappio LM (2021) Basal tryptase high levels associated with a history of arterial hypertension and hypercholesterolemia represent risk factors for severe anaphylaxis in hymenoptera venom-allergic subjects over 50 years old. Int Arch Allergy Immunol 182(2):146–152 [DOI] [PubMed] [Google Scholar]
- Patel N, Chong KW, Yip AY, Ierodiakonou D, Bartra J, Boyle RJ, Immunology C (2021) Use of Multiple Epinephrine Doses in Anaphylaxis: a Systematic Review and Meta-Analysis. 148(5):1307–1315 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Patel, N., Isaacs, E., Duca, B., Mohammed, H., Nagaratnam, N., Donovan, J., & Turner, P. (2020). What Dose of Epinephrine? Safety and Pharmacokinetics of 0.5mg versus 0.3mg Epinephrine by Autoinjector in Food-allergic Teenagers: a Randomized Cross-over Trial. Journal of Allergy and Clinical Immunology, 145(2, Supplement), AB6. 10.1016/j.jaci.2019.12.848
- Payus A, O., I., A., M., & , N. (2018) “Two Stones on One Bird”: a case report on severe biphasic anaphylaxis masquerading as life-threatening acute asthma. Open Access Macedonian Journal of Medical Sciences 6(11):2136 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pi X, Liu J, Sun Y, Ban Q, Cheng J, Guo M (2023) Protein modification, IgE binding capacity, and functional properties of soybean protein upon conjugation with polyphenols. Food Chem 405:134820. 10.1016/j.foodchem.2022.134820 [DOI] [PubMed] [Google Scholar]
- Piotin A, Oulehri W, Charles A-L, Tacquard C, Collange O, Mertes P-M, Geny B (2024) Oxidative Stress and Mitochondria Are Involved in Anaphylaxis and Mast Cell Degranulation: A Systematic Review. Antioxidants 13(8):920 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pouessel, G., Dribin, T. E., Tacquard, C., Tanno, L. K., Cardona, V., Worm, M., . . . Allergy, E. (2024). Management of Refractory Anaphylaxis: An Overview of Current Guidelines. [DOI] [PMC free article] [PubMed]
- Pu P, Zheng X, Jiao L, Chen L, Yang H, Zhang Y, Liang G (2021) Six flavonoids inhibit the antigenicity of β-lactoglobulin by noncovalent interactions: A spectroscopic and molecular docking study. Food Chem 339:128106. 10.1016/j.foodchem.2020.128106 [DOI] [PubMed] [Google Scholar]
- Pushparajah V, Fatima A, Chong CH, Gambule TZ, Chan CJ, Ng ST, Lim RLH (2016) Characterisation of a New Fungal Immunomodulatory Protein from Tiger Milk mushroom. Lignosus Rhinocerotis. Scientific Reports 6(1):30010. 10.1038/srep30010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Putney JW (1986) A model for receptor-regulated calcium entry. Cell Calcium 7(1):1–12. 10.1016/0143-4160(86)90026-6 [DOI] [PubMed] [Google Scholar]
- Putney JW (2009) Capacitative calcium entry: from concept to molecules. Immunol Rev 231(1):10–22. 10.1111/j.1600-065X.2009.00810.x [DOI] [PubMed] [Google Scholar]
- Qian, B. C., Gong, W. G., Chen, J., Zhang, Y. Q., Xu, H. J., & Zhang, L. X. (1980). [Pharmacological studies on anti-asthmatic and anti-anaphylactic activities of the essential oil of Litsea cubeba (Lour.) Pers. (author's transl)]. Yao Xue Xue Bao, 15(10), 584–589. [PubMed]
- Quah SY, Chin JH, Akowuah GA, Khalivulla SI, Yeong SW, Sabu MC (2017) Cytotoxicity and cytochrome P450 inhibitory activities of Clinacanthus nutans. Drug Metabolism Personalized Therapy 32(1):59–65. 10.1515/dmpt-2016-0034 [DOI] [PubMed] [Google Scholar]
- Quoc QL, Bich TCT, Jang JH, Park HS (2021) Recent update on the management of anaphylaxis. Clin Exp Emerg Med 8(3):160–172. 10.15441/ceem.21.121 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rachid, O. M. (2013). Evaluation of the effects of non-medicinal ingredients on the in vitro characteristics and in vivo bioavailability of a sublingual tablet formulation of epinephrine: University of Manitoba (Canada).
- Rakha A, Umar N, Rabail R, Butt MS, Kieliszek M, Hassoun A, Aadil RM (2022) Anti-inflammatory and anti-allergic potential of dietary flavonoids: A review. Biomed Pharmacother 156:113945. 10.1016/j.biopha.2022.113945 [DOI] [PubMed] [Google Scholar]
- Rana A, Samtiya M, Dhewa T, Mishra V, Aluko RE (2022) Health benefits of polyphenols: A concise review. J Food Biochem 46(10):e14264. 10.1111/jfbc.14264 [DOI] [PubMed] [Google Scholar]
- Rawas-Qalaji, M. M., Rachid, O., Simons, F. E. R., Simons, K. J. J. A. o. A., Asthma, & Immunology. (2013). Long-term stability of epinephrine sublingual tablets for the potential first-aid treatment of anaphylaxis. 111(6), 568–570. [DOI] [PubMed]
- Rebello, C. J., Beyl, R. A., Lertora, J. J. L., Greenway, F. L., Ravussin, E., Ribnicky, D. M., . . . Redman, L. M. (2020). Safety and pharmacokinetics of naringenin: A randomized, controlled, single-ascending-dose clinical trial. Diabetes, Obesity and Metabolism, 22(1), 91–98. 10.1111/dom.13868 [DOI] [PMC free article] [PubMed]
- Regateiro FS, Marques ML, Gomes ER (2020) Drug-induced anaphylaxis: an update on epidemiology and risk factors. Int Arch Allergy Immunol 181(7):481–487 [DOI] [PubMed] [Google Scholar]
- Roberts, G., Pfaar, O., Akdis, C., Ansotegui, I., Durham, S., Gerth van Wijk, R., . . . Pitsios, C. J. A. (2018). EAACI guidelines on allergen immunotherapy: allergic rhinoconjunctivitis. 73(4), 765–798. [DOI] [PubMed]
- Rossi CM, Lenti MV, Di Sabatino A (2022) Adult anaphylaxis: A state-of-the-art review. Eur J Intern Med 100:5–12. 10.1016/j.ejim.2022.03.003 [DOI] [PubMed] [Google Scholar]
- Ruiz-Garcia M, Bartra J, Alvarez O, Lakhani A, Patel S, Tang A, Boyle RJ (2021) Cardiovascular changes during peanut-induced allergic reactions in human subjects. Journal of Allergy and Clinical Immunology 147(2):633–642. 10.1016/j.jaci.2020.06.033 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulze-Osthoff K, Ferrari D, Riehemann K, Wesselborg S (1997) Regulation of NF-κB activation by MAP kinase cascades. Immunobiology 198(1–3):35–49 [DOI] [PubMed] [Google Scholar]
- Shahgordi S, Sankian M, Yazdani Y, Mashayekhi K, Hasan Ayati S, Sadeghi M, Hashemi M (2020) Immune responses modulation by curcumin and allergen encapsulated into PLGA nanoparticles in mice model of rhinitis allergic through sublingual immunotherapy. Int Immunopharmacol 84:106525. 10.1016/j.intimp.2020.106525 [DOI] [PubMed] [Google Scholar]
- Shahid H, Shahzad M, Shabbir A, Saghir G (2019) Immunomodulatory and Anti-Inflammatory Potential of Curcumin for the Treatment of Allergic Asthma: Effects on Expression Levels of Pro-inflammatory Cytokines and Aquaporins. Inflammation 42(6):2037–2047. 10.1007/s10753-019-01066-2 [DOI] [PubMed] [Google Scholar]
- Shaik, Y., Caraffa, A., Ronconi, G., Lessiani, G., & Conti, P. (2018). Impact of polyphenols on mast cells with special emphasis on the effect of quercetin and luteolin. Central European Journal of Immunology, 43(4), 476–481. 10.5114/ceji.2018.81347 [DOI] [PMC free article] [PubMed]
- Shaker, M. S., Wallace, D. V., Golden, D. B., Oppenheimer, J., Bernstein, J. A., Campbell, R. L., . . . Immunology, C. (2020). Anaphylaxis—a 2020 practice parameter update, systematic review, and Grading of Recommendations, Assessment, Development and Evaluation (GRADE) analysis. 145(4), 1082–1123. [DOI] [PubMed]
- Shaker W, G., & , O. B. (2020) Anaphylaxis—a 2020 practice parameter update, systematic review, and Grading of Recommendations, Assessment, Development and Evaluation (GRADE) analysis. Journal of Allergy and Clinical Immunology 145(4):1082–1123. 10.1016/j.jaci.2020.01.017 [DOI] [PubMed] [Google Scholar]
- Shakeri F, Eftekhar N, Roshan NM, Rezaee R, Moghimi A, Boskabady MH (2019) Rosmarinic acid affects immunological and inflammatory mediator levels and restores lung pathological features in asthmatic rats. Allergol Immunopathol 47(1):16–23. 10.1016/j.aller.2018.04.004 [DOI] [PubMed] [Google Scholar]
- Shakoor H, Feehan J, Apostolopoulos V, Platat C, Al Dhaheri AS, Ali HI, Stojanovska L (2021) Immunomodulatory Effects of Dietary Polyphenols. Nutrients 13(3):728 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shalaby AS, Eid HH, El-Shiekh RA, Mohamed OG, Tripathi A, Al-Karmalawy AA, Tadros SH (2023) Taming Food-Drug Interaction Risk: Potential Inhibitory Effects of Citrus Juices on Cytochrome Liver Enzymes Can Safeguard the Liver from Overdose Paracetamol-Induced Hepatotoxicity. ACS Omega 8(29):26444–26457 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shankar A, McAlees JW, Lewkowich IP (2022) Modulation of IL-4/IL-13 cytokine signaling in the context of allergic disease. Journal of Allergy and Clinical Immunology 150(2):266–276. 10.1016/j.jaci.2022.06.012 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma S, Sethi GS, Naura AS (2020) Curcumin Ameliorates Ovalbumin-Induced Atopic Dermatitis and Blocks the Progression of Atopic March in Mice. Inflammation 43(1):358–369. 10.1007/s10753-019-01126-7 [DOI] [PubMed] [Google Scholar]
- Shi Y-N, Su J-Z, Wang J, Geng J-Q (2023) Myricetin alleviates ovalbumin-induced allergic rhinitis in mice by regulating Th1/Th2 balance. Asian Pac J Trop Biomed 13(7):306–314 [Google Scholar]
- Sidhu, N., Jones, S., Perry, T., Thompson, T., Storm, E., Castro, M. S. M., & Nick, T. G. J. P. e. c. (2016). Evaluation of anaphylaxis management in a pediatric emergency department. 32(8), 508–513. [DOI] [PubMed]
- De Silva, D., Halken, S., Singh, C., Muraro, A., Angier, E., Arasi, S., Immunol, c. (2020). European Academy of Allergy. 31(7), 813–826. [DOI] [PubMed]
- Simons FER, Ardusso LR, Bilò MB, El-Gamal YM, Ledford DK, Ring J, Thong BYJWAOJ (2011) World Allergy Organization Guidelines for the Assessment and Management of Anaphylaxis. 4(2):13–37 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simons FE, Ardusso LR, Bilo MB, Cardona V, Ebisawa M, El-Gamal YM, Worm M (2014) International consensus on (ICON) anaphylaxis. World Allergy Organ J 7(1):9. 10.1186/1939-4551-7-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simons FER, Ebisawa M, Sanchez-Borges M, Thong BY, Worm M, Tanno LK, Sheikh A (2015) 2015 update of the evidence base: World Allergy Organization anaphylaxis guidelines. World Allergy Organ J 8:32. 10.1186/s40413-015-0080-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simons, F. E. R., & Sampson, H. (2015). Anaphylaxis: unique aspects of clinical diagnosis and management in infants (birth to age 2 years). 135(5), 1125–1131. [DOI] [PubMed]
- Simons, F. E. R., Gu, X., Johnston, L. M., & Simons, K. J. J. P. (2000). Can epinephrine inhalations be substituted for epinephrine injection in children at risk for systemic anaphylaxis? , 106(5), 1040–1044. [DOI] [PubMed]
- Simons, F. E. R., Gu, X., Simons, K. J. J. J. o. a., & immunology, c. (2001). Epinephrine absorption in adults: intramuscular versus subcutaneous injection. 108(5), 871–873. [DOI] [PubMed]
- Simons, F. E. R., Gu, X., Silver, N. A., Simons, K. J. J. J. o. a., & immunology, c. (2002). EpiPen Jr versus EpiPen in young children weighing 15 to 30 kg at risk for anaphylaxis. 109(1), 171–175. [DOI] [PubMed]
- Simons, F. E. (2009). Anaphylaxis: recent advances in assessment and treatment. 124(4), 625–636. [DOI] [PubMed]
- Singh A, Holvoet S, Mercenier A (2011) Dietary polyphenols in the prevention and treatment of allergic diseases. Clin Exp Allergy 41(10):1346–1359. 10.1111/j.1365-2222.2011.03773.x [DOI] [PubMed] [Google Scholar]
- Soares-Silva, M., Diniz, F. F., Gomes, G. N., & Bahia, D. (2016). The Mitogen-Activated Protein Kinase (MAPK) Pathway: Role in Immune Evasion by Trypanosomatids. Frontiers in Microbiology, 7. 10.3389/fmicb.2016.00183 [DOI] [PMC free article] [PubMed]
- Sonoda, J., Mizoguchi, I., Inoue, S., Watanabe, A., Sekine, A., Yamagishi, M., . . . Katahira, Y. J. I. J. o. M. S. (2023). A Promising Needle-Free Pyro-Drive Jet Injector for Augmentation of Immunity by Intradermal Injection as a Physical Adjuvant. 24(10), 9094. [DOI] [PMC free article] [PubMed]
- Soucy N, Michaud A (2023) Anaphylaxis: What You Need to Know. Physician Assistant Clinics 8(4):729–738. 10.1016/j.cpha.2023.05.008 [Google Scholar]
- Srisawat C, Pipitpreecha R, Chandranipapongse W, Chatsiricharoenkul S, Pongnarin P, Kongpatanakul S et al. (2022) Systemic absorption of epinephrine compared between the intranasal and intramuscular routes of administration in healthy adults. 10.12932/AP-120821-1209 [DOI] [PubMed]
- Stevens WW, Kraft M, Eisenbarth SC (2023) Recent insights into the mechanisms of anaphylaxis. Curr Opin Immunol 81:102288. 10.1016/j.coi.2023.102288 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sturm GJ, Varga EM, Roberts G, Mosbech H, Bilo MB, Akdis CA, Jakob TJA (2018) EAACI Guidelines on Allergen Immunotherapy: Hymenoptera Venom Allergy. 73(4):744–764 [DOI] [PubMed] [Google Scholar]
- Su Z, Liao J, Liu Y, Liang Y, Chen H, Chen X, Li Y (2015) Protective effects of patchouli alcohol isolated from Pogostemon cablin on lipopolysaccharide-induced acute lung injury in mice. Exp Ther Med 11(2):674–682. 10.3892/etm.2015.2918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Su Z, Liao J, Liu Y, Liang Y, Chen H, Chen X, Li Y (2016) Protective effects of patchouli alcohol isolated from Pogostemon cablin on lipopolysaccharide-induced acute lung injury in mice. Exp Ther Med 11(2):674–682. 10.3892/etm.2015.2918 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Subramanian H, Gupta K, Ali H (2016) Roles of Mas-related G protein–coupled receptor X2 on mast cell–mediated host defense, pseudoallergic drug reactions, and chronic inflammatory diseases. Journal of Allergy and Clinical Immunology 138(3):700–710. 10.1016/j.jaci.2016.04.051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sugiura, Y., Katsuzaki, H., Imai, K., & Amano, H. (2021). The Anti-Allergic and Anti-Inflammatory Effects of Phlorotannins from the Edible Brown Algae, Ecklonia sp. and Eisenia sp. Natural Product Communications, 16(12), 1934578X211060924. 10.1177/1934578X211060924
- Sun S, Jiang T, Gu Y, Yao L, Du H, Luo J, Che H (2023) Contribution of five major apple polyphenols in reducing peanut protein sensitization and alleviating allergencitiy of peanut by changing allergen structure. Food Res Int 164:112297. 10.1016/j.foodres.2022.112297 [DOI] [PubMed] [Google Scholar]
- Suwan, P., Praphaiphin, P., Chatchatee, P. J. A. P. J. o. A., & Immunology. (2018). Randomized comparison of caregivers' ability to use epinephrine autoinjectors and prefilled syringes for anaphylaxis. 36(4), 248–256. [DOI] [PubMed]
- Suzuki Y, Yoshimaru T, Inoue T, Ra C (2005) Mitochondrial Ca2+ flux is a critical determinant of the Ca2+ dependence of mast cell degranulation. J Leukoc Biol 79(3):508–518. 10.1189/jlb.0705412 [DOI] [PubMed] [Google Scholar]
- Švajger U, Obermajer N, Jeras M (2010) Dendritic cells treated with resveratrol during differentiation from monocytes gain substantial tolerogenic properties upon activation. Immunology 129(4):525–535. 10.1111/j.1365-2567.2009.03205.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svetlov SI, Siafaka-Kapadai A, Hanahan DJ, Olson MS (1996) Signaling Responses to Alkyllysophosphatidic Acid: The Activation of Phospholipases A2and C and Protein Tyrosine Phosphorylation in Human Platelets. Arch Biochem Biophys 336(1):59–68. 10.1006/abbi.1996.0532 [DOI] [PubMed] [Google Scholar]
- Tagen M, Elorza A, Kempuraj D, Boucher W, Kepley CL, Shirihai OS, Theoharides TC (2009) Mitochondrial Uncoupling Protein 2 Inhibits Mast Cell Activation and Reduces Histamine Content1. J Immunol 183(10):6313–6319. 10.4049/jimmunol.0803422 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takata, M., Sabe, H., Hata, A., Inazu, T., Homma, Y., Nukada, T., . . . Kurosaki, T. (1994). Tyrosine kinases Lyn and Syk regulate B cell receptor‐coupled Ca2+ mobilization through distinct pathways. The EMBO Journal, 13(6), 1341–1349–1349. 10.1002/j.1460-2075.1994.tb06387.x [DOI] [PMC free article] [PubMed]
- Takazawa, T., Yamaura, K., Hara, T., Yorozu, T., Mitsuhata, H., Morimatsu, H., & Working Group for the Preparation of Practical Guidelines for the Response to Anaphylaxis, S. C. o. t. J. S. o. A. J. J. o. a. (2021). Practical guidelines for the response to perioperative anaphylaxis. 1–16. [DOI] [PubMed]
- Takekawa M, Furuno T, Hirashima N, Nakanishi M (2012) Mitochondria Take Up Ca<sup>2+</sup> in Two Steps Dependently on Store-Operated Ca<sup>2+</sup> Entry in Mast Cells. Biol Pharm Bull 35(8):1354–1360. 10.1248/bpb.b110576 [DOI] [PubMed] [Google Scholar]
- Tan, E., O'Sullivan, M., Crozier, T., Kolawole, H., Mantas, S., Said, M., . . . Barnes, S. J. A. J. o. G. P. (2022). Acute management of anaphylaxis in pregnancy. 51(6), 405–408. [DOI] [PubMed]
- Taylor CW, Rahman T, Tovey SC, Dedos SG, Taylor EJA, Velamakanni S (2009) IP3 receptors: some lessons from DT40 cells. Immunol Rev 231(1):23–44. 10.1111/j.1600-065X.2009.00807.x [DOI] [PubMed] [Google Scholar]
- Tomasiak-Łozowska MM, Klimek M, Lis A, Moniuszko M, Bodzenta-Łukaszyk A (2018) Markers of anaphylaxis – a systematic review. Adv Med Sci 63(2):265–277. 10.1016/j.advms.2017.12.003 [DOI] [PubMed] [Google Scholar]
- Tortola L, Jacobs A, Pohlmeier L, Obermair F-J, Ampenberger F, Bodenmiller B, Kopf M (2020) High-Dimensional T Helper Cell Profiling Reveals a Broad Diversity of Stably Committed Effector States and Uncovers Interlineage Relationships. Immunity 53(3):597-613.e596. 10.1016/j.immuni.2020.07.001 [DOI] [PubMed] [Google Scholar]
- Turner, P. J., Ruiz-Garcia, M., Durham, S. R., Boyle, R. J. J. T. J. o. A., & Practice, C. I. I. (2021). Limited effect of intramuscular epinephrine on cardiovascular parameters during peanut-induced anaphylaxis: an observational cohort study. 9(1), 527–530. e521. [DOI] [PMC free article] [PubMed]
- Tuttle, R., Popescu, L., Hill, S., Slanczka, A., Jankowski, J., Barre, K., . . . Dretchen, K. L. (2020). Intranasal epinephrine effects on epinephrine pharmacokinetics and heart rate in a nasal congestion canine model. Respir Res, 21(1), 78. 10.1186/s12931-020-01343-x [DOI] [PMC free article] [PubMed]
- Ueda Y, Oku H, Iinuma M, Ishiguro K (2005) Antianaphylactic and antipruritic effects of the flowers of Impatiens textori MIQ. Biol Pharm Bull 28(9):1786–1790. 10.1248/bpb.28.1786 [DOI] [PubMed] [Google Scholar]
- Umasunthar T, Leonardi-Bee J, Hodes M, Turner PJ, Gore C, Habibi P, Boyle RJ (2013) Incidence of fatal food anaphylaxis in people with food allergy: a systematic review and meta-analysis. Clin Exp Allergy 43(12):1333–1341. 10.1111/cea.12211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valenta R, Karaulov A, Niederberger V, Gattinger P, van Hage M, Flicker S, Pickl WF (2018) Molecular Aspects of Allergens and Allergy. Adv Immunol 138:195–256. 10.1016/bs.ai.2018.03.002 [DOI] [PubMed] [Google Scholar]
- Valentová K, Vrba J, Bancířová M, Ulrichová J, Křen V (2014) Isoquercitrin: Pharmacology, toxicology, and metabolism. Food Chem Toxicol 68:267–282. 10.1016/j.fct.2014.03.018 [DOI] [PubMed] [Google Scholar]
- Vo TS, Le TT, Kim S-Y, Ngo D-H (2020) The role of myricetin from Rhodomyrtus tomentosa (Aiton) Hassk fruits on downregulation of FcɛRI-mediated mast cell activation. J Food Biochem 44(3):e13143. 10.1111/jfbc.13143 [DOI] [PubMed] [Google Scholar]
- Wang S, Li C, Sun P, Shi J, Wu X, Liu C, Wang Z (2021a) PCV2 Triggers PK-15 Cell Apoptosis Through the PLC–IP3R–Ca2+ Signaling Pathway. Front Microbiol. 10.3389/fmicb.2021.674907 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Z, Zeng M, Wang Z, Qin F, Chen J, He Z (2021b) Dietary luteolin: a narrative review focusing on its pharmacokinetic properties and effects on glycolipid metabolism. J Agric Food Chem 69(5):1441–1454. 10.1021/acs.jafc.0c08085 [DOI] [PubMed] [Google Scholar]
- Wang L, Xian Y-F, Loo SKF, Ip SP, Yang W, Chan WY, Wu JCY (2022) Baicalin ameliorates 2,4-dinitrochlorobenzene-induced atopic dermatitis-like skin lesions in mice through modulating skin barrier function, gut microbiota and JAK/STAT pathway. Bioorg Chem 119:105538. 10.1016/j.bioorg.2021.105538 [DOI] [PubMed] [Google Scholar]
- Wang Y, Zhang K, Mao Y-Y, Shao Y-H, Liu J, Tu Z-C (2023) A comparative study on the allergenic potential of β-lactoglobulin conjugated to glucose, caffeic acid and caffeoyl glucopyranose. Food Funct 14(9):4354–4367. 10.1039/D3FO00195D [DOI] [PubMed] [Google Scholar]
- Wang, Q.-Q., Gao, H., Yuan, R., Han, S., Li, X.-X., Tang, M., Feng, J. (2020). Procyanidin A2, a polyphenolic compound, exerts anti-inflammatory and anti-oxidative activity in lipopolysaccharide-stimulated RAW264. 7 cells. PLoS One, 15(8), e0237017. [DOI] [PMC free article] [PubMed]
- Worm M, Eckermann O, Dölle S, Aberer W, Beyer K, Hawranek T, Nemat K (2014) Triggers and treatment of anaphylaxis: an analysis of 4000 cases from Germany Austria and Switzerland. Dtsch Arztebl Int 111(21):367 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Worm M, Francuzik W, Renaudin JM, Bilo MB, Cardona V, Scherer Hofmeier K, Cichocka-Jarosz E (2018) Factors increasing the risk for a severe reaction in anaphylaxis: An analysis of data from The European Anaphylaxis Registry. Allergy 73(6):1322–1330 [DOI] [PubMed] [Google Scholar]
- Wu X, Lu Y, Xu H, Lin D, He Z, Wu H, Wang Z (2018) Reducing the allergenic capacity of β-lactoglobulin by covalent conjugation with dietary polyphenols. Food Chem 256:427–434. 10.1016/j.foodchem.2018.02.158 [DOI] [PubMed] [Google Scholar]
- Wu YF, Chen YQ, Li Q, Ye XY, Zuo X, Shi YL, Yang Y (2020) Supplementation with Tetrahydrocurcumin Enhances the Therapeutic Effects of Dexamethasone in a Murine Model of Allergic Asthma. Int Arch Allergy Immunol 181(11):822–830. 10.1159/000509367 [DOI] [PubMed] [Google Scholar]
- Wu T, Li Z, Wu Y, Yang X, Li L, Chen S, Zhao Y (2023) Exploring plant polyphenols as anti-allergic functional products to manage the growing incidence of food allergy. Front Nutr. 10.3389/fnut.2023.1102225 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J, Hao M, Sun Q, Tang L (2019) Comparative studies of interaction of β-lactoglobulin with three polyphenols. Int J Biol Macromol 136:804–812. 10.1016/j.ijbiomac.2019.06.053 [DOI] [PubMed] [Google Scholar]
- Xue Y-T, Han Y-N, Wang Y, Zhang Y-H, Yin Y-Q, Liu B-H, Zhao X-H (2023) Effect of ferulic acid covalent conjugation on the functional properties and antigenicity of β-lactoglobulin. Food Chem 406:135095. 10.1016/j.foodchem.2022.135095 [DOI] [PubMed] [Google Scholar]
- Yan X, Zeng Z, McClements DJ, Gong X, Yu P, Xia J, Gong D (2023) A review of the structure, function, and application of plant-based protein–phenolic conjugates and complexes. Comprehensive Reviews in Food Science and Food Safety 22(2):1312–1336. 10.1111/1541-4337.13112 [DOI] [PubMed] [Google Scholar]
- Yan Z, Feng X, Li X, Gao Z, Wang Z, Ren G, Long F (2024) Sea buckthorn flavonoid extracted by high hydrostatic pressure inhibited IgE-stimulated mast cell activation through the mitogen-activated protein kinase signaling pathway. Foods 13(4):560 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang R, Shao H, Yan Y, Wu Y, Meng X, Yang A, Chen H (2023a) Changes in structural and functional properties of whey protein cross-linked by polyphenol oxidase. Food Res Int 164:112377. 10.1016/j.foodres.2022.112377 [DOI] [PubMed] [Google Scholar]
- Yang Y, Wang L, Wang S, Wang Y, Du Y, Fan Y (2023b) Luteolin restored Treg/Th17 balance to ameliorate allergic rhinitis in a mouse model. Immunopharmacol Immunotoxicol 45(4):461–468 [DOI] [PubMed] [Google Scholar]
- Yasui Y, Sasao E, Sakata M, Matsui N, Fukuishi N, Akagi R, Akagi M (2007) Upregulation of heme oxygenase-1 by degranulation in rat basophilic leukemia cells. Biol Pharm Bull 30(3):443–446. 10.1248/bpb.30.443 [DOI] [PubMed] [Google Scholar]
- Yoneyama S, Kawai K, Tsuno NH, Okaji Y, Asakage M, Tsuchiya T, Nagawa H (2008) Epigallocatechin gallate affects human dendritic cell differentiation and maturation. Journal of Allergy and Clinical Immunology 121(1):209–214. 10.1016/j.jaci.2007.08.026 [DOI] [PubMed] [Google Scholar]
- Yong PYA, Yip AJW, Islam F, Hong HJ, Teh YE, Tham CL, Tan JW (2023) The anti-allergic potential of stingless bee honey from different botanical sources via modulation of mast cell degranulation. BMC Complement Med Ther 23(1):307. 10.1186/s12906-023-04129-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoo G, Lee K, Lee D-C (2017) Inhibitory effects of 2-oxo-2H-chromen-4-yl 4-methylbenzenesulfonate on allergic inflammatory responses in rat basophilic leukemia cells. Int Immunopharmacol 48:196–202. 10.1016/j.intimp.2017.05.003 [DOI] [PubMed] [Google Scholar]
- Yoon M-S, Lee JS, Choi B-M, Jeong Y-I, Lee C-M, Park J-H, Park Y-M (2006) Apigenin Inhibits Immunostimulatory Function of Dendritic Cells: Implication of Immunotherapeutic Adjuvant. Mol Pharmacol 70(3):1033–1044. 10.1124/mol.106.024547 [DOI] [PubMed] [Google Scholar]
- Yoshioka Y, Inoue M, Yoshioka H, Kitakaze T, Furuyashiki T, Abe N, Ashida H (2020) Enzymatically synthesized glycogen inhibited degranulation and inflammatory responses through stimulation of intestine. J Clin Biochem Nutrition 67(1):67–73. 10.3164/jcbn.20-33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yousef M, Crozier RWE, Hicks NJ, Watson CJF, Boyd T, Tsiani E, MacNeil AJ (2020) Attenuation of allergen-mediated mast cell activation by rosemary extract (Rosmarinus officinalis L.). J Leukocyte Biol 107(5):843–857. 10.1002/jlb.3a0320-434r [DOI] [PubMed] [Google Scholar]
- Yuan J, Dong X, Yap J, Hu J (2020) The MAPK and AMPK signalings: interplay and implication in targeted cancer therapy. J Hematol Oncol 13(1):113. 10.1186/s13045-020-00949-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zarubin T, Han J (2005) Activation and signaling of the p38 MAP kinase pathway. Cell Res 15(1):11–18 [DOI] [PubMed] [Google Scholar]
- Zeke A, Misheva M, Reményi A, Bogoyevitch Marie A (2016) JNK Signaling: Regulation and Functions Based on Complex Protein-Protein Partnerships. Microbiol Mol Biol Rev 80(3):793–835. 10.1128/mmbr.00043-14 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeng B, Jiang T, Xiong W, Che H, Sun S (2023a) Protective properties of polyphenols in food allergy: A review. Allergy 78(6):1654–1656. 10.1111/all.15459 [DOI] [PubMed] [Google Scholar]
- Zeng J, Hao J, Yang Z, Ma C, Gao L, Chen Y, Li J (2023b) Anti-Allergic Effect of Dietary Polyphenols Curcumin and Epigallocatechin Gallate via Anti-Degranulation in IgE/Antigen-Stimulated Mast Cell Model: A Lipidomics Perspective. Metabolites 13(5):628 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J, Berenstein E, Siraganian RP (2002) Phosphorylation of Tyr342 in the linker region of Syk is critical for FcεRI signaling in mast cells. Mol Cell Biol 22:8144 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang R, Yan P, Li Y, Xiong L, Gong X, Peng C (2016) A pharmacokinetic study of patchouli alcohol after a single oral administration of patchouli alcohol or patchouli oil in rats. Eur J Drug Metab Pharmacokinet 41(4):441–448. 10.1007/s13318-015-0272-7 [DOI] [PubMed] [Google Scholar]
- Zhang Y-F, Liu Q-M, Liu B, Shu Z-D, Han J, Liu H, Liu G-M (2019) Dihydromyricetin inhibited ovalbumin-induced mice allergic responses by suppressing the activation of mast cells. Food Funct 10(11):7131–7141. 10.1039/C9FO01557D [DOI] [PubMed] [Google Scholar]
- Zhang T, Hu Z, Cheng Y, Xu H, Velickovic TC, He K, Wu X (2020) Changes in Allergenicity of Ovalbumin in Vitro and in Vivo on Conjugation with Quercetin. J Agric Food Chem 68(13):4027–4035. 10.1021/acs.jafc.0c00461 [DOI] [PubMed] [Google Scholar]
- Zhang Q, Cheng Z, Wang Y, Fu L (2021) Dietary protein-phenolic interactions: characterization, biochemical-physiological consequences, and potential food applications. Crit Rev Food Sci Nutr 61(21):3589–3615. 10.1080/10408398.2020.1803199 [DOI] [PubMed] [Google Scholar]
- Zhang K, Wen Q, Li T, Zhang Y, Huang J, Huang Q, Gao L (2022a) Effect of covalent conjugation with chlorogenic acid and luteolin on allergenicity and functional properties of wheat gliadin. J Cereal Sci 106:103484. 10.1016/j.jcs.2022.103484 [Google Scholar]
- Zhang Z, Zhao Y, Han Y, Yang B, Lin H, Li Z (2022b) The natural substances with anti-allergic properties in food allergy. Trends Food Sci Technol 128:53–67. 10.1016/j.tifs.2022.07.004 [Google Scholar]
- Zhao Y, Zhang Z, Li Z, Li X-M, Wang H, Lin H (2023) Insight into the conformational and allergenicity alterations of shrimp tropomyosin induced by Sargassum fusiforme polyphenol. Food Res Int 165:112521. 10.1016/j.foodres.2023.112521 [DOI] [PubMed] [Google Scholar]
- Zheng H, Zhang Y, Pan J, Liu N, Qin Y, Qiu L, Wang T (2021) The Role of Type 2 innate lymphoid cells in allergic diseases. Front Immunol. 10.3389/fimmu.2021.586078 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou S-D, Lin Y-F, Xu X, Meng L, Dong M-S (2020) Effect of non-covalent and covalent complexation of (−)-epigallocatechin gallate with soybean protein isolate on protein structure and in vitro digestion characteristics. Food Chem 309:125718. 10.1016/j.foodchem.2019.125718 [DOI] [PubMed] [Google Scholar]
- Zhou E, Xue X, Xu H, Zhao L, Wu L, Li Q (2023) Effects of covalent conjugation with quercetin and its glycosides on the structure and allergenicity of Bra c p from bee pollen. Food Chem 406:135075. 10.1016/j.foodchem.2022.135075 [DOI] [PubMed] [Google Scholar]
- Zhou E, Li Q, Zhu D, Chen G, Wu L (2024) Characterization of physicochemical and immunogenic properties of allergenic proteins altered by food processing: a review. Food Sci Human Wellness 13(3):1135–1151. 10.26599/FSHW.2022.9250095 [Google Scholar]
- Zhu Y, Ouyang H, Lin W, Li W, Cao X, Chang Y, He J (2024a) GC-MS method for simultaneous determination and pharmacokinetic investigation of five volatile components in rat plasma after oral administration of the essential oil extract of Pogostemon cablin. Heliyon. 10.1016/j.heliyon.2024.e32444 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu Y, Ouyang H, Lv Z, Yao G, Ge M, Cao X, He J (2024b) Simultaneous determination of multiple components in rat plasma by UPLC-MS/MS for pharmacokinetic studies after oral administration of Pogostemon cablin extract. Front Pharmacol. 10.3389/fphar.2024.1293464 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analyzed during the current study.