Abstract
Background
Malaria remains a major cause of morbidity and mortality especially in sub-Saharan Africa. Whereas herbal medicines have long been used for disease remedy in many African communities, there is limited evidence on the extent of use, their safety, and efficacy. This study, sought to identify herbal medicinal plants used by communities in low and high malaria transmission settings in Uganda for managing of malaria.
Method
An Ethnobotanical survey was conducted across four geographical regions purposively selected to represent moderate-to-high (Apac, Arua and Tororo districts) and low (Kabale district) malaria transmission settings. One-hundred and two (102) traditional medicine practitioners (TMPs) in Ugandan local communities were included in the study. A checklist was used to collect data and covered the following areas; knowledge on malaria transmission, malaria symptoms, diagnosis, medicinal plants used, preparations, preservation methods and doses. Data was analyzed in MS Excel®. Consensus factor, use value metrics and frequencies were calculated.
Results
Ninety-seven plant species distributed across 45 families were mentioned by TMPs in management of malaria in Ugandan communities. Plant family Asteraceae, 15.5% (15/97) had the highest distribution of plants reported by TMPs. Vernonia amygdalina Delile, Aloe vera Burm. F., Artemisia annua L., Vernonia grantii Oliv. and Justicia betonica L. were the most mentioned, with use values of 0.4, 0.3, 0.2, 0.15 and 0.14 respectively. Leaves 64% and root barks 18% were the most harvested plant parts while decoctions (54%) and infusions 26% were the most common methods of preparing herbal products for individuals with malaria. Medicines were stored as dry powders for extended periods although some were prepared as fresh plants. Nearly all medicinal preparations were administered orally with varying dosage (5 ml-500 ml*3times a day) recommendations. Treatment duration varied between 3 and 7 days among practitioners. TMPs mentioned that malaria is transmitted by mosquitoes while others, said poor hygiene, stagnant water and body contact.
Conclusion
A diverse number of plant species, use and preparation methods are documented in this study as a way of preserving traditional knowledge in Uganda. Vernonia amygdalina Del., Aloe vera (L) Burm. f, Vernonia grantii Oliv. and Justicia betonica L. were identified as important plant species that can be further studied to validate their safety, antiplasmodial and active bioactive phytochemicals that can provide novel lead compounds for malaria treatment. These plant species can also be conserved through cultivation for sustainable use.
Keywords: Medicinal plants, Traditional medicine practitioners, Malaria, Ethnobotanical survey, Uganda
Introduction
In Uganda, malaria is estimated to be responsible for 30–50% of out-patient visits, 15–20% of hospital admissions and up to 20% of in-patient deaths [1]. Malaria is thus one of the leading causes of morbidity and mortality in the country especially among children under five years [2]. Uganda is among the six African countries that account for approximately 55% of the global malaria burden [3]. The stagnation in malaria eradication efforts poses a challenge for the attainment of the sustainable development goals which target a reduction of 95% malaria incidence and mortality by 2030 [4].
Artemisinin-based combination therapies are the mainstay in malaria treatment however, recent studies in northern Uganda report prevalence of slow clearing parasites [5, 6]. Furthermore, the studies found increasing prevalence of K13 gene mutations, 469Y and 675 V associated with artemisinin resistance among P. falciparum parasites. With no known effective therapeutic alternatives to artemisinin-based agents, widespread emergence of artemisinin resistance threatens to worsen malaria disease outcomes especially in sub-Saharan Africa [7]. This calls for continuous search for effective alternative agents for malaria treatment especially from medicinal plants.
For millennia, majority of the modern medicines have been developed from natural products or their derivatives. Particularly, the history of antimalarial drug development and treatment is intimately linked with medicinal plants and quinine, the first drug that was used for treatment of malaria was isolated from the Cinchona tree in 1820 [8]. In 1945, Chloroquine, a 4-aminoquinoline was synthesized from quinine and it became the first line treatment of un complicated malaria replacing its congener quinine. However, in the early 1960s, there was a high spread and mortality of P. falciparum chloroquine resistance which inspired researchers to actively develop effective alternative antimalarials against chloroquine-resistant malaria. Accordingly, many antimalarials such mefloquine and halofantrine were synthesized and in 1973, a novel sesquiterpene lactone called qinghaosu and later renamed artemisinin, was isolated from the Chinese herb Artemisia annua L [9]. Artemisinin has a unique mechanism of action which involves the heme-mediated decomposition of the endoperoxide bridge to produce carbon-centered free radicals that are toxic to the parasite [10]. However, because of its poor bioavailability, more potent derivatives such as artesunate, arteether, artemether and dihydroartemisinin were synthesized [9]. In order to increase the sensitivity of P. falciparum to artemisinins and defer drug resistance development WHO recommends Artemisinin based combination therapy (ACTs) which involves combining an artemisinin derivative with another antimalarial drug like lumefantrine, mefloquine, amodiaquine, sulfadoxine/pyrimethamine, piperaquine and chlorproguanil/dapsone [11]. ACTs have thus remained the focal drugs for treatment of P. falciparum malaria. As such the role of plants in the development of novel antimalarial agents cannot be under estimated especially due to the diverse repository of compounds with potential antiplasmodial activity.
Rural communities in Uganda and many parts of the world continue to use plant-sourced remedies against several ailments, including malaria. With the emerging reduction in efficacy of the current antimalarial agents and continued high burden of the disease, the need for efforts towards discovery and development of new effective therapies cannot be overstated. Previous studies [12–15] have documented the use of medicinal plants in treatment of malaria however such studies have mainly focused on the central and western regions of Uganda while high malaria settings like Apac and Arua have not been considered. A comprehensive assessment through ethnobotanical surveys of medicinal plants used in local communities in Uganda is key to preserve traditional knowledge, to conserve the important plant species and to provide baseline information about plant species needed in the drug development pipeline. This study, thus sought to document knowledge on herbal medicinal plants used for managing malaria by Ugandan communities in low, and high malaria transmission settings including Arua, Apac, Tororo and Kabale districts.
Methods
Study design
This was a cross sectional study. We used a descriptive exploratory approach to investigate communities’ knowledge on medicinal plants used for managing malaria. Aspects such as: (i) socio-demographic characteristics, (ii) knowledge on malaria symptoms and transmission, (iii) medicinal plants used for managing malaria, (iv) medicinal plant preparation, (v) source of the plant collection, (vi) drug administration and duration of treatment, and (vii) methods of preservation of the herbal preparations were investigated.
Study area
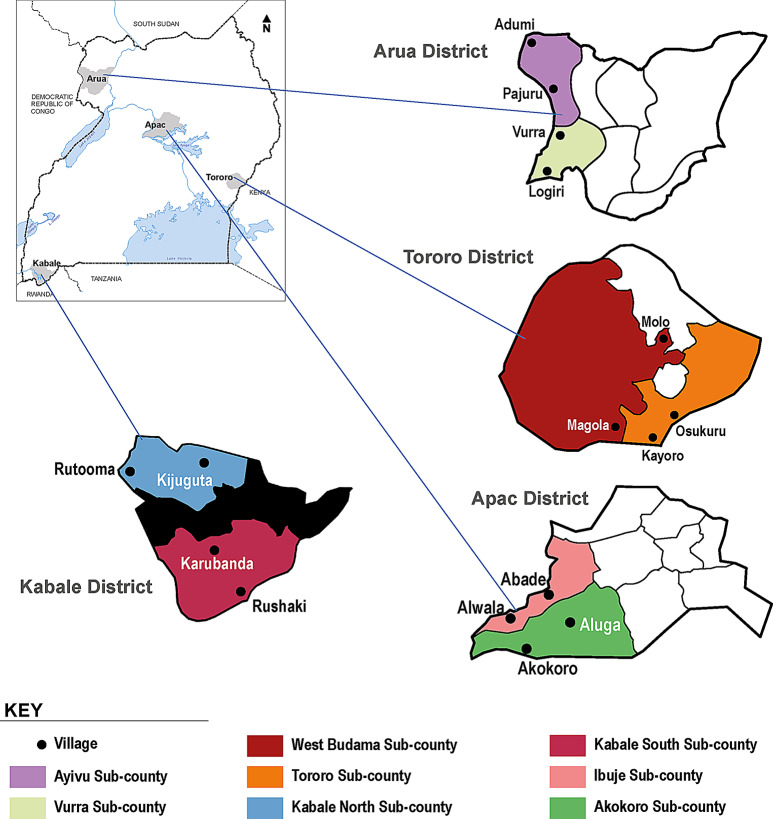
The study was conducted among in four rural districts of northern (Arua and Apac districts), eastern (Tororo district) and western Uganda (Kabale district) (Fig. 1) between October-December 2024. The districts were purposively selected based on malaria transmission intensities. Arua, Apac and Tororo districts are high transmission malaria areas while Kabale district is a low transmission area.
Fig. 1.
Map of Uganda showing location of study districts, sub counties and villages
Arua district is located in west Nile, covering a total of 3,236.4 km2 between latitude 20º 30’N and 30º 50’N and longitude 30º 30’E and 31º 30’E with a population of 820,500 people. It is bordered by Maracha district in the North West; Yumbe in the North East; Democratic Republic of Congo in the West; Nebbi in the South; Zombo in the South East; and Amuru district in the East. The major economic activity in the district is crop farming.
Apac district is in northern Uganda, located between longitudes 32o E and 34o E and latitudes 2o N and 3o N. It occupies an area of 3,255.9 km2 of which 9% is under swamps and water while 15% is under forest. It has a population of approximately 349,000 people.
Tororo district is a predominantly rural district in the Eastern region of Uganda that is 241,551 square kilometres large and located between latitude: 0° 44’ 59.99” N and longitude: 34° 04’ 60.00” E. It has a population of approx. 517,000 people. Wetland rice growing is the major economic activity in Tororo District favoring mosquito breeding especially in rainy seasons that are characterized with flooding.
Kabale district is in the western region of Uganda between longitudes 29° 34’ E and 35° 0’ E latitude 1° 29’ S and 4° 12’ N, it harbors a population of approximately 248,700 distributed in 19 subcounties and 1,345 villages. The altitude of Kabale district ranges between 1,219 and 2,347 m above sea level and temperatures average 18ºC during day and 10ºC at night. The major economic activity is agriculture.
Study population
The study was done among adult TMPs and were purposively selected based on their reputation of using medicinal plants to manage malaria and willingness to participate in the study by providing written consent.
Sampling criteria and data collection
In each of the four districts (Arua, Apac, Tororo and Kabale), two sub counties were randomly selected i.e. Ayiva and Vurra from Arua District, West Budama and Tororo from Tororo District, Ibuje and Akokoro from Apac, Kabale North and Kabale south from Kabale Districts. From each of the sub counties, two villages were selected (Fig. 1). Within each village, TMPs were purposively selected. The interviews were conducted using a pretested key informant interview guide adopted from a previous study [16] with few modifications. The interview was conducted for an average of 30–45 min and written consent was sought and obtained from all TMPs that participated in the study. For each plant species that was mentioned in the study, a voucher specimen indexed as L 001- L 110 was prepared, identified by a taxonomist (Dr. Ssegawa Paul), and achieved at the Makerere University Museum. All plant species were named using Flora of Tropical East Africa (FTEA). Administrative clearance to conduct the study in the different Districts and villages was sought from the Resident District Commissioner and local councils respectively.
Data management and analysis
Numerical data were entered in MS Excel© program where the relevant metrics were generated. Informant Consensus Factor (FIC) and Use Value (UV) metrics were computed. The FIC estimates homogeneity of information concerning use of medicinal plants for a particular ailment. It is calculated using the following formula [17].
 |
where Nur = the number of use-reports in each category and nt = number of taxa used. On the other hand, the UV metric estimates the commonness of use of each plant species in each area and was determined using the following formula (20):
 |
Where Nur is the number of use reports and Ni is total number of informants surveyed.
Results
Participant social demographics
A total of 102 TMPs from high 74.5% (76/102) and low 25.4% (26/102) malaria transmission settings participated in the study. Of these 65.7% (67/102) were males (Table 1). The mean age of the TMPs was 54 ± 2 years (range of 27–85). Majority, 71.5% (73/102) of the TMPs had completed the primary level of education while 19.6% (20/102) had no formal education. Most participants practiced herbal medicine as a secondary occupation and majority were primarily subsistence farmers.
Table 1.
Socio-demographic characteristics of traditional medicine practitioners interviewed across four districts in Uganda
| Sociodemographic characteristics (n = 102) | ||
|---|---|---|
| Characteristics | Description | Frequency N (%) |
| Sex | Male | 65.8 |
| Female | 34.2 | |
| Education | Primary | 71.8 |
| No formal education | 29.2 | |
| Mean age ± SD | 54 ± 2 | |
| Age categories (years) | 18–29 | 2.9 |
| 30–45 | 13.7 | |
| 46–55 | 33.3 | |
| 56–65 | 19.6 | |
| Marital Status | Married | 85.3 |
| Single | 3 | |
| Separated | 11.7 | |
| Occupation | Farmer | 96 |
| Businessman | 3 | |
| Reverend | 1 | |
| Malaria burden | High | 74.5 |
| Low | 25.5 | |
| Belonged to a professional association | No | 74.5 |
| Yes | 25.5 | |
Knowledge on malaria cause, symptoms, transmission, and diagnosis
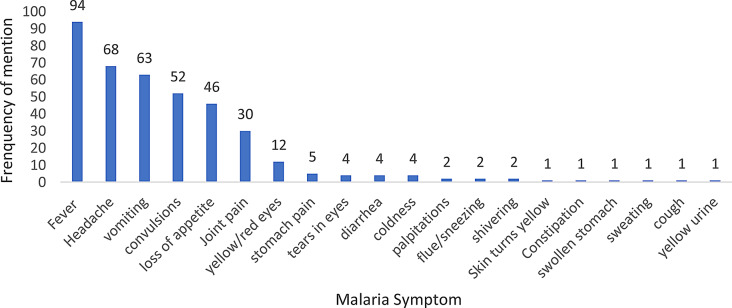
The common symptoms of malaria mentioned by traditional medicine practitioners included fever 94% (96/102), headache 67.6% (69/102), vomiting 62.7% (64/102) and convulsions 51.9% (53/102) (Fig. 2). Majority, 69.6% (71/102) of TMPs mentioned that malaria was spread through mosquito bites and over half, 64.7% (66/102) reported using symptoms in diagnosing malaria among individuals seeking care. A third 35.2% (36/102) of the TMPs mentioned managing individuals who already had malaria diagnosis from hospitals. TMPs neither had knowledge on the malaria causative parasite nor the conventional management of the disease as per the national malaria treatment guidelines. Participants also mentioned body contact, sharing utensils, drinking dirty water, stagnant waters, not sleeping under a mosquito net and a bushy environment as some of the other factors aiding malaria spread.
Fig. 2.
Shows symptoms of Malaria as mentioned by Traditional medicine practitioners and their frequency of mention
Malaria treatment practices and experiences among the study participants
TMPs were on average receiving 8±2 malaria patients per month in high transmission areas and 2–3 patients in low transmission areas. The duration of treatment for each malaria symptomatic patient varied among different TMPs ranging from 3 to 7 days. Traditional medicine practitioners mentioned that in most cases (96/102), their patients recovered, and this was assessed through self-reports. Those that did not recover were referred to health centers for further management. The cost of treatment varied widely amongst different TMPs. For some, treatment was free, and patients simply appreciated or gifted the practitioner after recovery. In high malaria transmission settings, treatment costs ranged between Uganda shillings 2000–10,000 (equivalent range 0.5-3 USD).
Plants used for management of malaria by the traditional medicine practitioners
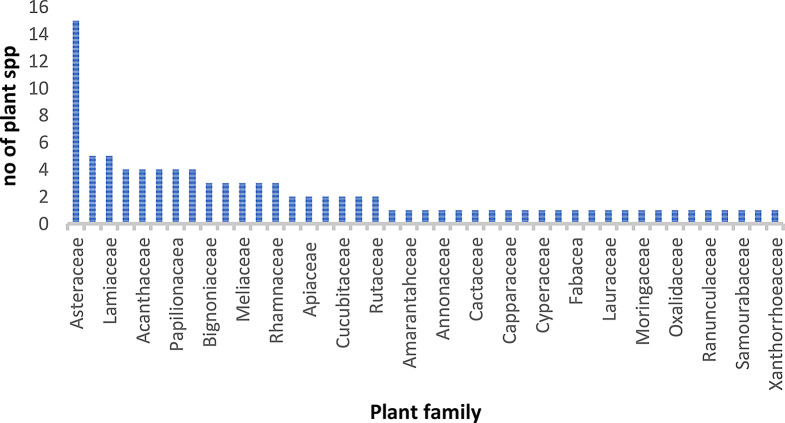
A total of ninety seven (97) plant species were mentioned by study participants across the four districts for treatment of malaria (Table 2). Plant species were distributed across 45 families with Asteraceae 15.5% (15/97) having the highest frequency (Fig. 3). Vernonia amygdalina Del (41) and Aloe vera (L.) Burm. F. (32) were the most mentioned with use values of 0.4 and 0.3 respectively. These were followed by Artemisia annua L. (21), Vernonia grantii Oliv. (15), Justicia betonica L. (14), Senna hirsute (L.) H.S. Irwin and Barneby (10), Psidium guajava L. (8), Schkuhria pinnata (Lam.) O. Ktze (8), Tetradenia riparia (Hochst.) Codd (5) and Sarcocephalus latifolius (smith)Bruce (5) among others. Plant spp like Aloe vera and Justicia betonica L. were mentioned by practitioners in all the four regions surveyed. The Informant Consensus factor in this study was calculated as 0.82.
Table 2.
Shows plant species used in the treatment of malaria as reported by TMPs, local names, frequency of mention, parts used and use values
| Scientific name (District*) | Local names | Family | Voucher No. | Preparation | Plant part | Frequency | Use value |
|---|---|---|---|---|---|---|---|
| Vernonia amygdalina Del | Muluswa/ Omubirizi | Asteraceae | L080 |
Decoction Infusion |
L | 41 | 0.40 |
| Aloe Vera (L.) Burm.f. | Aloe Vera/ Nzoo/lukaka | Xanthorrhoeaceae | L081 | Infusion Decoction | L | 32 | 0.31 |
| Artemisia annua L. | Artemisia | Asteraceae | L 069 | Decoction Infusion | L | 21 | 0.21 |
| Vernonia grantii Oliv. | okello-kello Ecoro ego/ etwatwa | Asteraceae | L 042 | Maceration | L | 15 | 0.15 |
| Justicia betonica L. | Quinine/ Nalongo | Acanthaceae | L 059 | Decoction | FL, L, WS | 14 | 0.14 |
| Senna hirsute (L.) H.S. Irwin and Barneby | Oyed apar/ Gasia | Caesalpinioideae | L 038 | Decoction | L | 10 | 0.10 |
| Psidium guajava L | Boke mapeera | Myrtaceae | L 052 | Decoction | L, G | 8 | 0.08 |
| Schkuhria pinnata (Lam.) O. Ktze | Quinine, akweyo | Asteraceae | L 034 | Decoction Infusion Maceration | L | 8 | 0.08 |
| Tetradenia riparia (Hochst.) Codd | Omuharavumba | Lamiaceae | L 082 | Decoction | L | 5 | 0.05 |
| Sarcocephalus latifolius (smith)Bruce | Ibeleg, Ebeleng, Akeng | Rubiaceae | L 046 | Decoction | RB/L | 5 | 0.05 |
| Persea americana Mill | Boke ovacado | Lauraceae | L 083 | Decoction Infusion | L | 4 | 0.04 |
| Eucalyptus sp | Kalatunsi | Myrtaceae | L 083 | Maceration Decoction | L | 4 | 0.04 |
| Annona senegalensis Pers. | Omukoonya | Annonaceae | L 084 | Decoction | L | 4 | 0.04 |
| Schkuhria pinnata (Lam) O.Ktze. | Ekadwarit (Ateso) | Asteraceae | L 085 |
Decoction Infusion |
L | 3 | 0.03 |
| Senna occidentalis (L.) Link | Yekeyeke | Fabacea | L 086 | Maceration Infusion | L, RB | 3 | 0.03 |
| Markhamia lutea (Benth.) K.Schum | Emusoliat (Ateso), Musambya | Bignoniaceae | L 065 | Decoction, Infusion | SB/L | 3 | 0.03 |
| Warbugia ugandesis Sprague | Abac | Canellaceae | L 087 | Maceration | SB/L | 3 | 0.03 |
| Mangifera indica L. | Muyembe | Anacardiaceae | L 088 | Maceration | SB | 3 | 0.03 |
| Senna didymobotrya Fresen. | Omugabagaba | Caesalpinioideae | L 072 | Decoction | L | 3 | 0.03 |
| Maesa lanceolata Forssk | Omuhanga | Myrsinaceae | L 089 | Decoction | L | 3 | 0.03 |
| Clerodendrum umbellatum Poir | Lolobi | Lamiaceae | L 027 | Decoction | L | 3 | 0.03 |
| Bidens pilosa L | Sere (JAP) | Asteraceae | L 090 | Infusion | R/L | 2 | 0.02 |
| Cissampelos mucronata A. Rich. | Kavamagombe/ agwaya-gwaya | Mennispermaceae | L 037 | Infusion | SB/L | 2 | 0.02 |
| Tagetes minuta L. | mukazi mulofu | Asteraceae | L 073 | Infusion | L | 2 | 0.02 |
| Gouania longispicata | omufulula | Rhamnaceae | L 091 |
Maceration Infusion |
L | 2 | 0.02 |
| Momordica foetida schumach | kirerabaana / bombo | Cucubitaceae | L 068 | Infusion | L | 2 | 0.02 |
| Tithonia diversifolia Gray | Okangali | Asteraceae | L 092 | Decoction | L | 2 | 0.02 |
| Hyptis suaveolens Poir | Ajingurua | Lamiaceae | L 007 | Decoction | WP | 2 | 0.02 |
| Harrisonia abyssinica Oliv. | Kawakawa | Samourabaceae | L 017 | Powder | RB/SD | 2 | 0.02 |
| Aristolochia elegans mast | Emanimani, koolo | Aristolochiaceae | L 093, L027 | Powder | F, L, SD | 2 | 0.02 |
| Moringa oleifera L. | Moringa | Moringaceae | L 033 | Decoction | L/RB/SB/SD | 1 | 0.01 |
| Dicliptera laxata C.B.CL | Fula | Acanthaceae | L 064 | Decoction | L | 1 | 0.01 |
| Hoslundia opposita (Vahl) | Kamunye | Asteraceae | L 066 | Decoction | RB | 1 | 0.01 |
| Toddalia asiatica (L.) Lam. | Kawule/ Thwolikiluwi | Rutaceae | L 094 | Decoction | RB | 1 | 0.01 |
| Conyza floribunda HB K | Emelliat | Asteraceae | L 061 | Infusion | L | 1 | 0.01 |
| Azadirachta indica (A. Juss) | Neem/ Arubaine | Meliaceae | L 095 | Infusion | L | 1 | 0.01 |
| Mondia whitei (Hook. f.) Skeels | Omulondo (Samia) | Apocynaceae | L 096 | powder | L | 1 | 0.01 |
| Crassocephalum vitellinum Benth | Ensununu | Asteraceae | L 070 | Decoction | 1 | 0.01 | |
| Mukia maderaspatana (L) M.J Roem | Akabindizi | Cucubitaceae | L 069 | Decoction | L | 1 | 0.01 |
| Hibiscus fuscus Garcke | omusinga | Malvaceae | L 074 | Decoction | L | 1 | 0.01 |
| Ageratum conyzoides L. | omubuza | Asteraceae | L 075 | Decoction | L | 1 | 0.01 |
| Ipomoea batatas (L.) Lam. | emikamba | Convolvulaceae | L 097 | Infusion | L | 1 | 0.01 |
| Dicliptera laxata C. B. CL | nyarwehindura | Acanthaceae | L 098 | Decoction | L | 1 | 0.01 |
| Ajuga remota Benth. | kitinwa | Lamiaceae | L 099 | Decoction | L | 1 | 0.01 |
| Indigofera arrecta A. Rich | omushorooza | Papilionacaea | L 071 | Decoction | L | 1 | 0.01 |
| Antidesma venosum E. Mey. ex Tul. | mulingwa/ embatabata | Phyllanthaceae | L 100 | Decoction | L | 1 | 0.01 |
| Dracaena fragrans (L.) Ker Gawl. | ndugaho/ omugolora | Asparagaceae | L 101 | Decoction | RB | 1 | 0.01 |
| Chenopodium opulifolium Schrad. ex W.D.J.Koch & Ziz |
Omwetango, Atiko |
Chenopodiaceae | L102, L055 | Decoction | L | 4 | 0.01 |
| Agrocharis incognita (Norman) Hey &Jury | akatankolora/akatampuhi | Apiaceae | L 103 | Decoction | L | 1 | 0.01 |
| Spathodea campanulata Buch. -Harm. ex DC. | Omwatashare | Bignoniaceae | L 104 | Decoction | SB | 1 | 0.01 |
| Cactus spp | Lubamba | Cactaceae | L 105 | Decoction | L | 1 | 0.01 |
| Sida rhombifolia L. | Omunyiganyenje/ muzimba ndegeya | Malvaceae | L 106 | Decoction | L | 1 | 0.01 |
| Plectranthus cyaneus (Forssk.) Schweinf. Ex. Sprenger | Kibwankula | Lamiaceae | L 107 | Decoction | L | 1 | 0.01 |
| Physalis peruviana L. | Entuntunu | Solanaceae | L 108 | Decoction | L | 1 | 0.01 |
| Markhamia lutea (Benth.) K. Schum. | Musambya | Bignoniaceae | L 109 | Decoction | RB/L | 1 | 0.01 |
| Allium Sativum L. | Garlic | Amaryllidaceae | L 110 | Decoction | B | 1 | 0.01 |
| Vernonia spp | Ecolo | Asteraceae | L 010 | maceration | L | 1 | 0.01 |
| Oxalis corniculata | Ewaandre | Oxalidaceae | L 005 | Decoction | WP | 1 | 0.01 |
| Lactuca capensis thumb. | Embereko | Asteraceae | L 026 | Decoction | WP | 1 | 0.01 |
| Citrus lemon Burm. F | lemon | Myrtaceae | L 043 | Maceration | fruit | 1 | 0.01 |
| Senna occidentalis (L.) Link | Boyo boyo | Caesalpinioideae | L 035 | Decoction |
L/ WP |
1 | 0.01 |
| Solanum nigrum Linn | Ocuga | Solanaceae | L 044 | Infusion | L | 1 | 0.01 |
| Clematis hirsuta per.&guill | Adwe | Ranunculaceae | L 046 | Infusion | WR | 1 | 0.01 |
| Dyschoriste nagchana (Nees) Bennet | pilopilo | Acanthaceae | L 30 |
Decoction Infusion |
L/SB/RB | 1 | 0.01 |
| Maerua triphylla A. Rich | Okwenyo/Ayebi | Capparaceae | L 047 | Infusion | RB/L | 1 | 0.01 |
| Euclea racemosa Murray subsp. Schimperi | Jewee | Ebenaceae | L051 | Power | L | 1 | 0.01 |
| Solanum incanum L. | Ocokocok | Solanaceae | L049 | Decoction | L | 1 | 0.01 |
| Crotalaria ochroleuca G. Don | Alayo | Papilionacaea | L 039 |
Maceration Powder |
RB | 1 | 0.01 |
| Leonotis nepetifolia(L.) Ait. F. | Ating ating | Lamiaceae | L 053 | Decoction | L | 1 | 0.01 |
| Periploca Linearifolia Quart. Dill. & A, Rich | Agaba | Apocynaceae | L 052 | Decoction | L | 1 | 0.01 |
| Indigofera spicata Forssk. | Icac | Papilionacaea | L 062 | Decoction | RB | 1 | 0.01 |
| Zanthoxylum chalybeum Engl. | Iucu | Rutaceae | L 050 | Decoction | WR | 1 | 0.01 |
| Rhamnus prinoides L. Herit | Kirikolo | Rhamnaceae | L 011 | Decoction | L | 1 | 0.01 |
| Cyperus rigidifolius Steudel | enkumbo/ omulemampango | Cyperaceae | L079 | Deccoction | L | 1 | 0.01 |
| Guoania longispicata Hemsl. | Omufurura | Rhamnaceae | L 078 | Infusion | L | 1 | 0.01 |
| Euphorbia splendens Hook. | Labi | Euphorbiacea | L 029 | Maceration | L/SB | 1 | 0.01 |
| Lepidotrichilia volkensii (Guerke) Leroy | Okaa | Meliaceae | L 008 | Swallowed fresh | SD | 1 | 0.01 |
| Canavalia afriacana Dunn | Osnosu | Papilionacaea | L 006 | Powder | SD | 1 | 0.01 |
| Chenopodium Procerum Moq. | Omujumbajumba | Chenopodiaceae | L 411 | Infusion | L | 1 | 0.01 |
| Crassocephalum gracile (Hook.f.) Milne-Redh. | Unknown | Asteraceae | L 002 | Maceration | L | 1 | 0.01 |
| Phyllanthus amarus Schumach | Unknown | Euphorbiaceae | L 005 | Decoction | L | 1 | 0.01 |
| Caesalpinia decapetala (Roth) Alston | Unknown | Apiaceae | L 412 | Infusion | L | 1 | 0.01 |
| Ekebergia capensis Sparrm. | Unknown | Meliaceae | L014 | Infusion | L | 1 | 0.01 |
| Ricinodendron heudelotii (Baill) Heckel | Unknown | Euphorbiaceae | L016 | Infusion | L | 1 | 0.01 |
| Senna alata (L.) Roxb. | Unknown | Caesalpinioideae | L 020 | Maceration | L | 1 | 0.01 |
| Allamanda cathartica L. | Unknown | Aponynaceae | L019 | Maceration | L | 1 | 0.01 |
| Lippia woodii Mold. | Unknown | Verbanaceae | L009 | Decoction | L | 1 | 0.01 |
| Burnatia enneandra Micheli | Unknown | Alismataceae | L025 | Decoction | L | 1 | 0.01 |
| Solanum macrocarpon L. | Olianziri | Solanaceae | L015 | Infusion | L | 1 | 0.01 |
| Sclerocarya birrea (A.Rich.) Hochst. subsp. Birrea | Unknown | Anacardiaceae | L 032 | Decoction | L | 1 | 0.01 |
| Chenopodium ambrosioides L. | Anjigun | Chenopodiaceae | L 030 | Infusion | L | 1 | 0.01 |
| Gomphrena celosioides Mast. | Unknown | Amarantahceae | L 041 | Decoction | L | 1 | 0.01 |
| Carissa spinarum L. | Acuga | Apocynaceae | L 036 | Infusion | L | 1 | 0.01 |
| Senna obtusifolia L. | Unknown | Caesalpinioideae | L 026 | Decoction | L | 1 | 0.01 |
Key: L=Leaves, SB=stem bark, WS=whole stem, RB=rootbark, WP=whole plant, SD=seeds, FL=Flower, B=bulb
Fig. 3.
Number of plant spp distributed among different families.
Medicinal plant preparation, packaging and administration in management of malaria
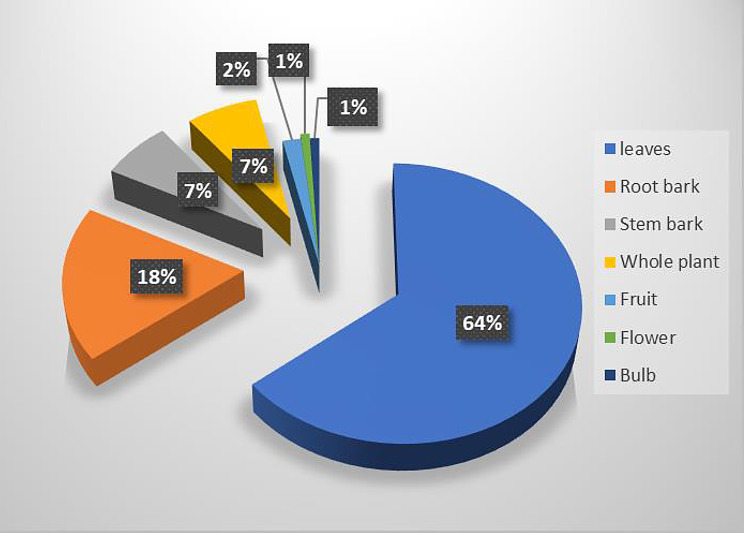
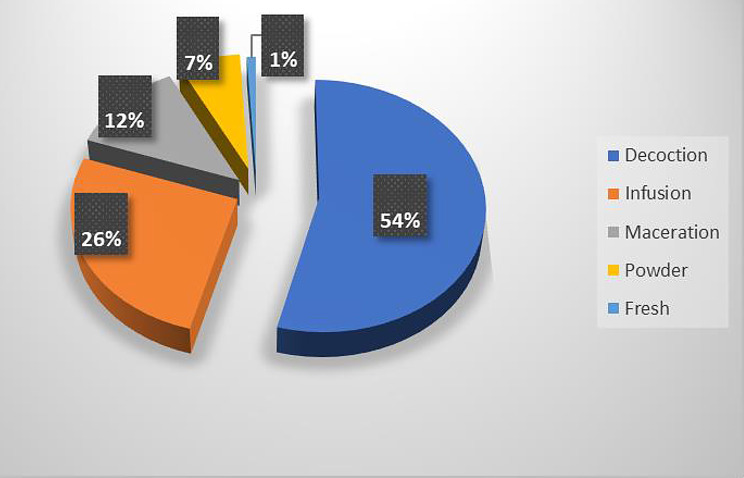
Leaves were most frequently used (69%) in herbal medicine preparations followed by root bark (20%) and stem bark (7%) (Fig. 4). Most medicines were prepared in combination using methods such as by decoctions (54%) followed by infusions (26%), maceration (12%), and powder (7%) (Fig. 5). Medicinal products were stored as powders if the place of collection was far from the TMP however, most medicines were prepared and used fresh as and when needed. The oral route was principally used for treatment administration. The dose recommendations varied widely amongst the different TMPs and ranged from ½ teaspoon (~ 5mls) 2 or 3 times daily to 1 cup (~ 500 ml) taken 2 or 3 times daily. Plastic bottles, polythene bags and papers were used as storage material. In Kabale district, most TMPs cultivated their own medicinal plants as compared to other districts where plants were picked from nearby gardens, forests, or hills where they are abundant.
Fig. 4.
Plant parts used by TMPs for preparation of antimalarial herbal medicine in different parts of Uganda
Fig. 5.
Preparation methods used by traditional medicinal practitioners as reported in different parts of Uganda
Discussion
Uganda ranks third among countries with the highest burden of Malaria in the world [2]. Malaria affects communities in rural settings in endemic areas with favorable climatic conditions for mosquito vector breeding [73]. Such conditions include ample rainfall (average 200 mm), optimal temperatures (26–47ºC), swamps, water bodies and forest cover. The high malaria transmission intensity in the three study districts of Apac, Arua and Tororo range from 390 to 1586 infective bites per person per year. Worse still, in such impoverished communities, the cost of antimalarial treatment coupled with long distances to health facilities fuels the use of alternative medicines such as medicinal plants for malaria treatment.
The results of this study show that communities in Arua, Apac, Tororo and Kabale districts use a diverse number of plant species for management of malaria. Plant species like Vernonia amygdalina Del, Aloe Vera (L.) Burm.f., Artemisia annua L., Vernonia grantii Oliv. and Justicia betonica L. were the most used across the four surveyed local communities and they contribute to the high informant consensus factor. The use of Vernonia amygdalina, Artemisinin annua, Aloe vera and Justicia betonica L. for treatment of malaria is well documented across Uganda in previous studies [66, 74, 75]. The wide use of the plant species by different communities across Uganda poses a challenge of sustainability and plant conservation strategies should be encouraged.
Artemisinin annua, from family Asteraceae has strong antimalarial activity and is the source of current antimalarial drugs including dihydroartemisinin, artemether and artesunate. However, recently, there is increasing artemisinin drug resistance reported especially in northern Uganda, calling for more efforts to search for novel compounds [76]. Vernonia amygdalina, a renowned antimalarial plant in communities, yielded two alkaloids namelyvernodalin and vernolide with median inhibitory concentrations of 0.52 µg/ml and 1.87 µg/ml against a chloroquine sensitive strain of P. falciparum (D10) [19] (Table 3). Similarly, fagamine a secondary carboxamide, isolated from Zanthoxyllum Chalybeum had an MIC value of 2.85 µg/ml [77]. The three phytochemicals can be used as leads for development of new antimalarial drugs.
Table 3.
Literature on antiplasmodial activity and isolated compounds from some of the mentioned plant species
| No | Plant species | Plant part | Extract (IC50) |
Invitro activity(µg/ml) / Plasmodium strain | Isolated compound (IC50 µg/ml) | Citation |
|---|---|---|---|---|---|---|
| 1. | Vernonia amygdalina | Leaves | methanol/ ethyl acetate | 2.7 mg/ml |
Vernodalin (0.52) Vernolide (1.87) |
[18, 19] |
| 2. | Aloe Vera | Leaves |
Aloin (67 µg/ml) Aloe-emodin (22 µg/ml) |
[20] | ||
| 3. | Artemisia annua L. | Leaves |
Dichloromethane Methanol |
3.79 3.00 |
Arteether (3.88nM) Artemether (3.71nM) Artelinate (3.46nM) |
[21, 22] |
| 4. | Justicia betonica L | Leaves | Methanol | 13.36 | Justiciosides A–D, Jusbetonin | [23–25] |
| 5. | Psidium guajava | Leaves | Dichloromethane | 3.3–4.6/ Dd2, D10 |
Ellagic acid Gallic acid Guavin B Gualjaverine Quacetrine |
[26, 27] |
| 6. | Tetradenia riparia | Root | 13.2/NF54 | Abietane and Royleanone | [28] | |
| 7. | Sarcocephalus latifolius | Leaves | Methanol | 14.7/Field isolates | Quinovic acid Strictosamide | [29, 30] |
| 8. | Annona senegalensis Pers. | Leaves | Ethanol | 17–19/ 3D7 & Dd2 | [31] | |
| 9. | Senna occidentalis (L.) Link | Roots | Methanol | 1.7/3D7 | [32] | |
| 10. | Warbugia ugandesis Sprague | Stem bark | Ethanol | 15.6–8.92/ 3D7&Dd2 |
Polygodial 1a Warburganal 2a Muzigadial 2b |
[33, 34] |
| 11. | Senna didymobotrya Fresen. | Leaf | Dichloromethane | 23–24/ D6& W2 | Bianthraquinones | [35, 36] |
| 12. | Maesa lanceolata Forssk | Twig, Leaf | Dichloromethane: Methanol | P. falciparum, D10 | [37] | |
| 13. | Bidens pilosa L | Leaf | Water | 5–11/D10 | [38] | |
| 14. | Cissampelos mucronata A. Rich. | RB | Ethanol | 8.0-9.2/ W2, D6 | Stigmasterol Hentriacontane Simiarenol Nonacosene | [39, 40] |
| 15. | Momordica foetida schumach | Leaves | Ethyl acetate | 29.3/K1 |
Phenolic glycosides Sitosterylglucoside 5,25-stigmastadien-3β-ylglucoside |
[41, 42] |
| 16. | Tithonia diversifolia Gray | Leaves | Water | 18–19/D7, W2 |
Sesquiterpene lactones Tephrostachin Quercetin |
[35, 43] |
| 17. | Harrisonia abyssinica Oliv. | Stem bark | Methanol | 4.7–10/ Dd2, 3D7 | 2-hydroxymethylalloptaeroxylin. | [44] |
| 18. | Moringa oleifera L | Leaves | Methanol, Hexane | 3.3–3.4/ D7 |
Kaempferol Myricetin Quercetin Gallic acid Luteolin |
[45, 46] |
| 19. | Aristolochia elegans mast | Seeds | Methanol | > 50/ 3D7 | β-caryophyllene | [47, 48] |
| 20. | Hoslundia opposita (Vahl) | Leaf | Water | 4–12/ P. falciparum |
1,4- Citric acid Tetracontane-1,40-diol |
[49, 50] |
| 21. | Toddalia asiatica (L.) Lam. | Leaf | Methanol | 6.8–13.9/ D6, W2 |
Toddanolic acid Toddalic acid |
[51, 52] |
| 22. | Azadirachta indica (A. Juss) | Leaf | Methanol | 1.7–5.8/D7, DD5 |
Dodecanoic acid Oleic acid 13-octadecyl 15-tetracosanoic acid methyl ester |
[44, 53] |
| 23. | Ageratum conyzoides L. | Whole plant | Dichloromethane | 2.15–3.44/ W2, D6 |
Precocene II β-caryophyllene Eugenol |
[54, 55] |
| 24. | Chenopodium opulifolium Schrad. | leaves | Dichloromethane | 0.1/ 3D7 |
Allantoin Decan-2-one |
[56] |
| 25. | Spathodea campanulata Buch. | Fruit | Ethyl acetate | 28.1/3D7 |
5,7-Dihydroxy-4-Metilcoumarin Quercetin Kaempferol |
[57, 58] |
| 26. | Sida rhombifolia L. | Whole plant | Ethyl acetate Fraction | 3.56/ Dd2 | Rhombifoliamide | [59] |
| 27. | Physalis peruviana L. (KB) | Leaf | Dichloromethane | 8-14.7/ D6, W2 |
Kaempferol Quercetin Luteolin Apigenin |
[60, 61] |
| 28. | Citrus lemon Burm. F | Leaf | 10% Methanol: water | 12/ P. falciparum |
Hesperidin eriocitrin, diosmin Citric acid |
[62, 63] |
| 29. | Leonotis nepetifolia(L.) Ait. F. | Whole plant | Dichloromethane: Methanol | 15/ d10 |
Cirsiliol Catechin P-coumaric acid Rosmarinic acid |
[64, 65] |
| 30. | Zanthoxylum chalybeum Engl. Var. Molle | Husk | Ethyl acetate | 3.7–9.3/ INDO, 3D7 | Fagaramide (2.85) | [66] |
| 31. | Phyllanthus amarus Schumach | leaves | Water | 23.1/ P. falciparum |
Niranthin Phyllanthin Nirtetralin |
[67, 68] |
| 32. | Ekebergia capensis Sparrm. | Husks, Stem bark | Methanol | > 5/ K1 | [69] | |
| 33. | Ricinodendron heudeloti (Baill) Heckel | Leaf | Methanol | 28/D6 | [70] | |
| 34. | Carissa spinarum L. | Husks, Root bark | Methanol | < 3-14.5/ 3D7, NIDO, D6 |
Scopoletin Evomonoside |
[71, 72] |
35% (34/96) of the plant species in this study have been evaluated for their in-vitro antiplasmodial activity (Table 3). This verifies and confirms their local use in the Ugandan communities for treatment of malaria. From previous studies, plant species such as; Psidium guajava, Senna occidentalis, Harrisonia abyssinica, Moringa oleifera, Hoslunda opposita, Azadiracha indica, Ageratum conyzoides, Chemopodium opulifolium, sida rhombifolia and Carissa spinarum were very active with minimum inhibitory concentrations < 5 µg/ml [26, 32, 44, 45, 49, 54, 56, 71]. With the high antiplasmodial activity, these plants can be potential sources for new antimalarial drugs. On the other hand, medicinal plants such as Vernonia granti Oliv. with a high use value is yet to be investigated for its anti-plasmodial activity.
Most of the active and mentioned plant species belong to family Asteraceae which is one of the largest families in the plant kingdom with over 2500 species distributed widely all over the world. The family is knowned for its diverse pharmacological activities owed to presence of numerous phytochemicals including phenolic compounds like quercetin (a) that has shown significant antimalarial activity invitro [78–80].
Most herbal medicines were prepared as decoctions or infusions. An infusion is prepared by soaking fresh plant parts for a short period of time with cold or boiling water while a decoction is prepared by boiling two or more plant spp at minimum heat for a given period of time [81]. Decoction is a common method of medicine preparation among local communities; for instance, one of the TMPs in this study mentioned that Zanthoxylum chalybeum and Sarcocephalus latifolius are dried, pounded and boiled together in water and then 150 ml are taken three times a day. Another TMP mentioned that Vernonia grantii, Crotalaria ochroleuca, aloe vera and Justicia betonica are boiled together and 200mls are taken once per day. The combination of different medicinal herbs ensures use of less plant materials thus potentially minimizing toxic effects while maximizing efficacy through additive or synergistic effects. Also, some plant species in the decoction play different roles for example, Azadirachta indica (A. Juss) in a preparation can act as an antipyretic to control high temperatures [82]. On the other hand, Euclea racemosa contains phytosterols that possess anti-inflammatory and anti-pain activity; it was noted that in this study, TMPs rub the plant powder on joints to control joint pain [83]. Eucalyptus is also added in herbal medicines as a preservative to prolong the shelf life of the herbal products [84].
This study showed that TMPs in the surveyed communities had knowledge about malaria disease and its mode of transmission. This was evident by the symptoms most frequently mentioned which were consistent with the well-known clinical symptoms of malaria. On the contrary, practitioners demonstrated limited knowledge about the malaria causative agent and lacked sufficient knowledge on conventional treatment of malaria. Furthermore, the variable nature of dosing among different practitioners, even within the same village was concerning as it shows a lack of dose standardization. Dose standardization presents a challenge that has long been experienced among TMPs mainly due to the numerous and diverse bioactive phytochemicals contained in plant based medicines, numerous range of indigenous healing practices, lack of training for the TMPs, lack of a basic knowledge of the possible side effects and efficacy of the plant remedies. Since TMPs play an important role in rural health care, this calls for training and education on such pertinent issues especially in poverty-stricken regions where the malaria transmission and disease burden remain high.
Conclusion
A diverse number of plant species, plant use and preparation methods are documented in this study as a way of preserving traditional knowledge in Uganda. Vernonia amygdalina Del., Aloe vera (L) Burm. f, Vernonia grantii Oliv. and Justicia betonica L. were identified as important plant species that can be further studied to validate their safety, antiplasmodial and active bioactive phytochemicals that can provide novel lead compounds for malaria treatment. These plant species can also be conserved through cultivation for sustainable use.
Acknowledgements
We would like to acknowledge the Traditional medicine practitioners in Arua, Kabale, Apac and Tororo districts who participated in this study. We thank the Government of Uganda Office of the President through the Science technology and innovation (STI) for funding this research study.
Author contributions
LB conceived the research idea, collected field data, analyzed and interpreted the data as well as prepared the first draft of the manuscript. MO, FWO, SN, COO, GO and AL conceived the research idea and collected field data. All authors revised and efficiently contributed to improvement of drafts and approved the final manuscript.
Funding
The research study was funded by the government of Uganda under the office of the president Science and Technology innovation (OP-STI) research grant. The funding agent was not involved in the study design and implementation.
Data availability
The study tools used in acquiring the data presented in this manuscript are available as raw data in questionnaire documents. This can be acquired from the corresponding author when needed.
Declarations
Ethics approval and consent to participate
The study protocol was reviewed and received ethics approval from Makerere University School of Biomedical Sciences’ Research and Ethics Committee (#SBS-2022-213). Clearance to conduct the study in Uganda was granted by the Uganda National Council of Science and Technology (#NS431ES). A written informed consent was obtained from each traditional medicine practitioner prior to the interview. The study adhered to ethical principles in the Declaration of Helsinki (2013).
Consent of publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ministry of Health. Uganda National Malaria Annual Report 2017–2018. In: Epidemiological Bulletin. vol. 4; 2019.
- 2.WHO. World health organization world malaria report 2023. In. World Health Organization; 2023.
- 3.Anjorin S, Okolie E, Yaya S. Malaria profile and socioeconomic predictors among under-five children: an analysis of 11 sub-Saharan African countries. Malar J. 2023;22(1):55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lawal L, Buhari AO, Jaji TA, Alatare AS, Adeyemo AO, Olumoh AO, Yusuff YA, Osborn G, Mogaji AB, Adoto BH, et al. Lingering challenges in malaria elimination efforts in sub-Saharan africa: insights and potential solutions. Health Sci Rep. 2024;7(6):e2122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Balikagala B, Fukuda N, Ikeda M, Katuro OT, Tachibana S-I, Yamauchi M, Opio W, Emoto S, Anywar DA, Kimura E. Evidence of artemisinin-resistant malaria in Africa. N Engl J Med. 2021;385(13):1163–71. [DOI] [PubMed] [Google Scholar]
- 6.Asua V, Conrad MD, Aydemir O, Duvalsaint M, Legac J, Duarte E, Tumwebaze P, Chin DM, Cooper RA, Yeka A. Changing prevalence of potential mediators of aminoquinoline, antifolate, and Artemisinin resistance across Uganda. J Infect Dis. 2021;223(6):985–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Malaria RB. World malaria report 2005. World Health Organization and UNICEF 2005.
- 8.Zhou B, Yue J-M. Natural products are the treasure pool for antimalarial agents. Natl Sci Rev. 2022;9(11):nwac112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen C. Development of antimalarial drugs and their application in china: a historical review. Infect Dis Poverty. 2014;3(1):9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Meshnick SR. Artemisinin: mechanisms of action, resistance and toxicity. Int J Parasitol. 2002;32(13):1655–60. [DOI] [PubMed] [Google Scholar]
- 11.WHO. Guidelines for the treatment of malaria. World Health Organization; 2015. [PubMed]
- 12.Stangeland T, Alele PE, Katuura E, Lye KA. Plants used to treat malaria in Nyakayojo sub-county, Western Uganda. J Ethnopharmacol. 2011;137(1):154–66. [DOI] [PubMed] [Google Scholar]
- 13.Philip K, Elizabeth MM, Cheplogoi PK, Samuel KT. Ethnobotanical survey of antimalarial medicinal plants used in Butebo County, Eastern Uganda. 2017.
- 14.Okello D, Kang Y. Exploring antimalarial herbal plants across communities in Uganda based on electronic data. Evidence-Based Complement Altern Med. 2019;2019(1):3057180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Tabuti JRS, Obakiro SB, Nabatanzi A, Anywar G, Nambejja C, Mutyaba MR, Omara T, Waako P. Medicinal plants used for treatment of malaria by Indigenous communities of Tororo district, Eastern Uganda. Trop Med Health. 2023;51(1):34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bunalema L, Obakiro S, Tabuti JR, Waako P. Knowledge on plants used traditionally in the treatment of tuberculosis in Uganda. J Ethnopharmacol. 2014;151(2):999–1004. [DOI] [PubMed] [Google Scholar]
- 17.Trotter RT, Logan MH. Informant consensus: a new approach for identifying potentially effective medicinal plants. Plants and indigenous medicine and diet. edn.: Routledge; 2019. pp. 91–112.
- 18.Egharevba C, Erharuyi O, Imieje V, Ahomafor J, Akunyuli C, Udu-Cosi A, Theophilus O, Osakue J, Ali I, Falodun A. Significance of bitter leaf (Vernonia Amagdalina) in tropical diseases and beyond: A review. Malaria Chemotherapy, Control & Elimination; 2014.
- 19.Chukwujekwu J, Lategan CA, Smith P, Van Heerden F, Staden J. Antiplasmodial and cytotoxic activity of isolated sesquiterpene lactones from the acetone leaf extract of Vernonia colorata. South Afr J Bot. 2009;75:176–9. [Google Scholar]
- 20.Kumar S, Yadav M, Yadav A, Rohilla P, Yadav JP. Antiplasmodial potential and quantification of Aloin and aloe-emodin in Aloe vera collected from different Climatic regions of India. BMC Complement Altern Med. 2017;17:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Basco LK, Le Bras J. In vitro activity of Artemisinin derivatives against African isolates and clones of plasmodium falciparum. Am J Trop Med Hyg. 1993;49(3):301–7. [DOI] [PubMed] [Google Scholar]
- 22.Gruessner BM, Weathers PJ. In vitro analyses of Artemisia extracts on plasmodium falciparum suggest a complex antimalarial effect. PLoS ONE. 2021;16(3):e0240874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bbosa G, Kyegombe D, Lubega A, Musisi N, Ogwal-Okeng J, Odyek O. Anti-Plasmodium falciparum activity of Aloe dawei and Justicia betonica. Afr J Pharm Pharmacol. 2013;7:2258–63. [Google Scholar]
- 24.Kanchanapoom T, Noiarsa P, Ruchirawat S, Kasai R, Otsuka H. Triterpenoidal glycosides from Justicia betonica. Phytochemistry. 2004;65(18):2613–8. [DOI] [PubMed] [Google Scholar]
- 25.Subbaraju GV, Kavitha J, Rajasekhar D, Jimenez JI. Jusbetonin, the first indolo[3,2-b]quinoline alkaloid glycoside, from Justicia betonica. J Nat Prod. 2004;67(3):461–2. [DOI] [PubMed] [Google Scholar]
- 26.Gutiérrez RMP, Mitchell S, Solis RV. Psidium guajava: A review of its traditional uses, phytochemistry and Pharmacology. J Ethnopharmacol. 2008;117(1):1–27. [DOI] [PubMed] [Google Scholar]
- 27.Adebajo AC, Odediran SA, Aliyu FA, Nwafor PA, Nwoko NT, Umana US. In vivo antiplasmodial potentials of the combinations of four Nigerian antimalarial plants. Molecules Vol. 2014;19:13136–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kumari A, Baskaran P, Chukwujekwu JC, de Kock CA, Smith PJ, Van Staden J. The changes in morphogenesis and bioactivity of tetradenia riparia, Mondia whitei and cyanoptis speciosa by an aeroponic system. Ind Crops Prod. 2016;84:199–204. [Google Scholar]
- 29.Ac E, Ezeji-Chigbu N, Aa E, Ene C, Gn O, Ujowundu C, Chinedu E. Anti-plasmodial and antioxidant evaluation of the methanol and aqueous extracts of sarcocephaluslatifolius. 2018, 1:1–9.
- 30.Ajiboye AT, Asekun OT, Familoni OB, Ali Z, Wang Y-H, Ghanadian M, Zulfiqar F, Larbie C, Samuel OA, Khan IA. Phytochemicals isolated from the root bark of sarcocephalus latifolius (Sm.) e.a.bruce. Biochem Syst Ecol. 2019;86:103909. [Google Scholar]
- 31.Wele M, Kirkman L, Diarra N, Goita Y, Doumbia M, Traore K, Diallo D. Antiplasmodial potential and phytochemical screening of ten plants used as antimalarial in Mali. Eur J Med Plants. 2017;19(4):1–9. [Google Scholar]
- 32.Mogaka S, Molu H, Kagasi E, Ogila K, Waihenya R, Onditi F, Ozwara H. Senna occidentalis (L.) link root extract inhibits plasmodium growth in vitro and in mice. BMC Complement Med Ther. 2023;23(1):71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Okello D, Gathirwa J, Wanyoko A, Komakech R, Chung Y, Gang R, Omujal F, Kang Y. Comparative antiplasmodial activity, cytotoxicity, and phytochemical contents of Warburgia ugandensis stem bark against aspilia Africana wild and in vitro regenerated tissues. J Plant Biotechnol. 2023;50(1):97–107. [Google Scholar]
- 34.Kitte R, Tretbar M, Dluczek S, Beckmann L, Marquardt P, Duenkel A, Schubert A, Fricke S, Tretbar US. Chemical and cytotoxic activity of three main sesquiterpenoids from Warburgia ugandensis. Results Chem. 2021;3:100242. [Google Scholar]
- 35.Waiganjo B, Moriasi G, Onyancha J, Elias N, Muregi F. Antiplasmodial and cytotoxic activities of extracts of selected medicinal plants used to treat malaria in Embu county, Kenya. J Parasitol Res. 2020;2020:1–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Alemayehu G, Hailu A, Abegaz BM. Bianthraquinones from Senna didymobotrya. Phytochemistry. 1996;42(5):1423–5. [Google Scholar]
- 37.Muchiri J, Amanya M, Mfitundinda W, Nyabayo J, Odda J, Extension KP. Antimalarial activity and levels of inorganic ions of the aqueous leaf extract of Maesa lanceolata Forssk. J Sci Res. 2023;10:89–101. [Google Scholar]
- 38.Owuor B, Ochanda J, Kokwaro J, Cheruiyot A, Yeda R, Okudo C, Akala H. In vitro antiplasmodial activity of selected Luo and Kuria medicinal plants. J Ethnopharmacol. 2012;144(3):779–81. [DOI] [PubMed] [Google Scholar]
- 39.Mucrorata C. In-vivo antiplasmodial activity and screening of ethanolic extract of the leaves of Cissampelos mucronata. Nigerian J Pharm Sci 2007, 6(2).
- 40.Akande RT, Fouche G, Famuyide IM, Makhubu FN, Nkadimeng SM, Aro AO, Kayoka-Kabongo PN, McGaw LJ. Anthelmintic and antimycobacterial activity of fractions and compounds isolated from Cissampelos mucronata. J Ethnopharmacol. 2022;292:115130. [DOI] [PubMed] [Google Scholar]
- 41.Waako PJ, Gumede B, Smith P, Folb PI. The in vitro and in vivo antimalarial activity of Cardiospermum halicacabum L. and Momordica foetida schumch. Et Thonn. J Ethnopharmacol. 2005;99(1):137–43. [DOI] [PubMed] [Google Scholar]
- 42.Muronga M, Quispe C, Tshikhudo PP, Msagati TAM, Mudau FN, Martorell M, Salehi B, Abdull Razis AF, Sunusi U, Kamal RM, et al. Three selected edible crops of the genus Momordica as potential sources of phytochemicals: biochemical, nutritional, and medicinal values. Front Pharmacol. 2021;12:625546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kerebba N, Oyedeji AO, Byamukama R, Kuria SK, Oyedeji OO. Pesticidal activity of Tithonia diversifolia (Hemsl.) A. Gray and Tephrosia vogelii (Hook f.); phytochemical isolation and characterization: A review. South Afr J Bot. 2019;121:366–76. [Google Scholar]
- 44.El Tahir A, Satti GM, Khalid SA. Antiplasmodial activity of selected Sudanese medicinal plants with emphasis on Maytenus senegalensis (Lam.) Exell. J Ethnopharmacol. 1999;64(3):227–33. [DOI] [PubMed] [Google Scholar]
- 45.Bezerra JJL, Pinheiro AAV, Dourado D. Antimalarial potential of Moringa oleifera Lam.(Moringaceae): A review of the ethnomedicinal, pharmacological, toxicological, and phytochemical evidence. J Venom Anim Toxins Including Trop Dis. 2023;29:e20220079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Hassan MA, Xu T, Tian Y, Zhong Y, Ali FAZ, Yang X, Lu B. Health benefits and phenolic compounds of Moringa oleifera leaves: A comprehensive review. Phytomedicine. 2021;93:153771. [DOI] [PubMed] [Google Scholar]
- 47.Muganga R, Angenot L, Tits M, Frédérich M. Antiplasmodial and cytotoxic activities of Rwandan medicinal plants used in the treatment of malaria. J Ethnopharmacol. 2010;128(1):52–7. [DOI] [PubMed] [Google Scholar]
- 48.Alois KM, Sangiwa GC, Marciale CM, Sahini MG. Phytochemical constituents and larvicidal efficacy of leaf extracts of Aristolochia elegans (Aristolochiaceae). South Afr J Bot. 2022;146:383–94. [Google Scholar]
- 49.Moustapha K, Karim T, Offianan T, Sylvain B, David A, Albert G, Stéphane Y, Philippe B. Assessment of antiplasmodial and anti-anaemic activities of Hoslundia opposita, an Ivorian medicinal plant. J Adv Microbiol. 2018;11(1):1–11. [Google Scholar]
- 50.Namuga C, Muwonge H, Nasifu K, Sekandi P, Sekulima T, Kirabira JB. Hoslundia opposita vahl; a potential source of bioactive compounds with antioxidant and antibiofilm activity for wound healing. BMC Complement Med Ther. 2024;24(1):236. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Reinhardt JK, Zimmermann-Klemd AM, Danton O, Smieško M, Gründemann C, Hamburger M. Compounds from Toddalia asiatica: immunosuppressant activity and absolute configurations. J Nat Prod. 2020;83(10):3012–20. [DOI] [PubMed] [Google Scholar]
- 52.Orwa J, Ngeny L, Mwikwabe N, Ondicho J, Jondiko I. Antimalarial and safety evaluation of extracts from Toddalia Asiatica (L) Lam.(Rutaceae). J Ethnopharmacol. 2013;145(2):587–90. [DOI] [PubMed] [Google Scholar]
- 53.Igwenyi IO, Onodugo CA, Aja PM, Elom SO, Awoke JN, Ibhadode OS, Uraku AJ, Edwin N, Obasi AN, David EE, et al. Azadirachta indica fruit juice clears malaria parasites and replenishes blood levels in plasmodium berghei-infected mice. Phytomedicine Plus. 2024;4(4):100615. [Google Scholar]
- 54.Malebo H, Tanja W, Cal M, Swaleh S, Omolo M, Hassanali A, Séquin U, Hamburger M, Brun R, Ndiege I. Antiplasmodial, anti-trypanosomal, anti-leishmanial and cytotoxicity activity of selected Tanzanian medicinal plants. Tanzan J Health Res 2009, 11(4). [DOI] [PubMed]
- 55.Chahal R, Nanda A, Akkol EK, Sobarzo-Sánchez E, Arya A, Kaushik D, Dutt R, Bhardwaj R, Rahman MH, Mittal V. Ageratum conyzoides L. and its secondary metabolites in the management of different fungal pathogens. Molecules 2021, 26(10). [DOI] [PMC free article] [PubMed]
- 56.Juma S. Anti-plasmodial activity and phytochemical analysis of leaf extract of Chenopodium Opulifolium schrad. EX WDJ Koch and Ziz used in Uganda. Makerere University; 2022.
- 57.Teinkela JEM, Oumarou H, Noundou XS, Meyer F, Megalizzi V, Hoppe HC, Krause RWM, Wintjens R. Evaluation of in vitro antiplasmodial, antiproliferative activities, and in vivo oral acute toxicity of Spathodea campanulata flowers. Sci Afr. 2023;21:e01871. [Google Scholar]
- 58.Ngnameko CR, Marchetti L, Zambelli B, Quotadamo A, Roncarati D, Bertelli D, Njayou FN, Smith SI, Moundipa PF, Costi MP et al. New insights into bioactive compounds from the medicinal plant Spathodea campanulata P. Beauv. And their activity against Helicobacter pylori. In: Antibiotics vol. 9; 2020. [DOI] [PMC free article] [PubMed]
- 59.Kamdoum BC, Simo I, Wouamba SCN, Tchatat Tali BM, Ngameni B, Fotso GW, Ambassa P, Fabrice FB, Lenta BN, Sewald N. Chemical constituents of two Cameroonian medicinal plants: Sida rhombifolia L. and Sida acuta burm. f.(Malvaceae) and their antiplasmodial activity. Nat Prod Res. 2022;36(20):5311–8. [DOI] [PubMed] [Google Scholar]
- 60.Kamau PK, Ng’ang’a Z, Njeruh FM, Thuita J. In vitro antiplasmodial, cytotoxicity assay and partial chemical characterization of Kenyan Physalis peruviana L.(Solanaceae family) extracts. 2020.
- 61.Muñoz P, Parra F, Simirgiotis MJ, Sepúlveda Chavera GF, Parra C. Chemical characterization, nutritional and bioactive properties of Physalis peruviana fruit from high areas of the Atacama desert. Foods 2021, 10(11). [DOI] [PMC free article] [PubMed]
- 62.Melariri P, Campbell W, Etusim P, Smith P. In vitro antiplasmodial activities of extracts from five plants used singly and in combination against plasmodium falciparum parasites. J Med Plants Res. 2012;6(47):5770–9. [Google Scholar]
- 63.Wang H, Xiao H, Wu Y, Zhou F, Hua C, Ba L, Shamim S, Zhang W. Characterization of volatile compounds and microstructure in different tissues of ‘eureka’lemon (Citrus limon). Int J Food Prop. 2022;25(1):404–21. [Google Scholar]
- 64.Lagnika HO, Medjigbodo AA, Djihinto OY, Saïzonou HM, Mousse WA, Akoton R, Djossou L, Vodounkpe DN, Lagnika L, Djogbénou LS. Beninese plant extracts with antiplasmodial activity select new allele variants Msp1 and Msp2 in plasmodium falciparum. J Parasitol Res. 2024;2024(1):9980715. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Sitarek P, Kowalczyk T, Śliwiński T, Hatziantoniou S, Soulintzi N, Pawliczak R, Wieczfinska J. Leonotis nepetifolia transformed root extract reduces Pro-Inflammatory cytokines and promotes tissue repair in vitro. Int J Environ Res Public Health 2023, 20(6). [DOI] [PMC free article] [PubMed]
- 66.Adia MM, Anywar G, Byamukama R, Kamatenesi-Mugisha M, Sekagya Y, Kakudidi EK, Kiremire BT. Medicinal plants used in malaria treatment by Prometra herbalists in Uganda. J Ethnopharmacol. 2014;155(1):580–8. [DOI] [PubMed] [Google Scholar]
- 67.Aliyu K, Mohammed Y, Abdullahi IN, Umar AA, Bashir F, Sani MN, Kabuga AI, Adamu AY, Akande AO. In vitro antiplasmodial activity of Phyllanthus amarus against plasmodium falciparum and evaluation of its acute toxicity effect in mouse model. Trop Parasitol. 2021;11(1):31–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Nisar MF, He J, Ahmed A, Yang Y, Li M, Wan C. Chemical components and biological activities of the genus phyllanthus: A review of the recent literature. Molecules 2018, 23(10). [DOI] [PMC free article] [PubMed]
- 69.Obbo C, Kariuki S, Gathirwa J, Olaho-Mukani W, Cheplogoi P, Mwangi E. In vitro antiplasmodial, antitrypanosomal and antileishmanial activities of selected medicinal plants from Ugandan flora: refocusing into multi-component potentials. J Ethnopharmacol. 2019;229:127–36. [DOI] [PubMed] [Google Scholar]
- 70.Ogbole OO, Segun PA, Fasinu PS. Antimicrobial and antiprotozoal activities of twenty-four Nigerian medicinal plant extracts. South Afr J Bot. 2018;117:240–6. [Google Scholar]
- 71.Fatima A, Singh PP, Agarwal P, Irchhaiya R, Alok S, Verma A. Treatment of various diseases by Carissa spinarum L.: a promising shrub. Int J Pharm Sci Res. 2013;4(7):2489–95. [Google Scholar]
- 72.Wangteeraprasert R, Lipipun V, Gunaratnam M, Neidle S, Gibbons S, Likhitwitayawuid K. Bioactive compounds from Carissa spinarum. Phytother Res. 2012;26(10):1496–9. [DOI] [PubMed] [Google Scholar]
- 73.Okiring J, Routledge I, Epstein A, Namuganga JF, Kamya EV, Obeng-Amoako GO, Sebuguzi CM, Rutazaana D, Kalyango JN, Kamya MR, et al. Associations between environmental covariates and Temporal changes in malaria incidence in high transmission settings of uganda: a distributed lag nonlinear analysis. BMC Public Health. 2021;21(1):1962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Namukobe J, Kasenene JM, Kiremire BT, Byamukama R, Kamatenesi-Mugisha M, Krief S, Dumontet V, Kabasa JD. Traditional plants used for medicinal purposes by local communities around the Northern sector of Kibale National park, Uganda. J Ethnopharmacol. 2011;136(1):236–45. [DOI] [PubMed] [Google Scholar]
- 75.Ssegawa P, Kasenene JM. Plants for malaria treatment in Southern uganda: traditional use, preference and ecological viability. J Ethnobiol. 2007;27(1):110–31. [Google Scholar]
- 76.Conrad MD, Asua V, Garg S, Giesbrecht D, Niaré K, Smith S, Namuganga JF, Katairo T, Legac J, Crudale RM. Evolution of partial resistance to artemisinins in malaria parasites in Uganda. N Engl J Med. 2023;389(8):722–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Adia MM, Emami SN, Byamukama R, Faye I, Borg-Karlson AK. Antiplasmodial activity and phytochemical analysis of extracts from selected Ugandan medicinal plants. J Ethnopharmacol. 2016;186:14–9. [DOI] [PubMed] [Google Scholar]
- 78.Ali AH, Sudi S, Shi-Jing N, Hassan WRM, Basir R, Agustar HK, Embi N, Sidek HM, Latip J. Dual anti-malarial and GSK3β-mediated cytokine-modulating activities of Quercetin are requisite of its potential as a plant-derived therapeutic in malaria. Pharmaceuticals. 2021;14(3):248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Rolnik A, Olas B. The plants of the Asteraceae family as agents in the protection of human health. Int J Mol Sci 2021, 22(6). [DOI] [PMC free article] [PubMed]
- 80.Chea A, Hout S, Long C, Marcourt L, Faure R, Azas N, Elias R. Antimalarial activity of sesquiterpene lactones from Vernonia cinerea. Chem Pharm Bull. 2006;54(10):1437–9. [DOI] [PubMed] [Google Scholar]
- 81.Suna S, Tamer CE, Özcan-Sinir G. 13 - Trends and Possibilities of the Usage of Medicinal Herbal Extracts in Beverage Production. In: Grumezescu AM, Holban AM, editors. Natural Beverages. Academic Press, 2019. p. 361–398.
- 82.Islas JF, Acosta E, G-Buentello Z, Delgado-Gallegos JL, Moreno-Treviño MG, Escalante B, Moreno-Cuevas JE. An overview of Neem (Azadirachta indica) and its potential impact on health. J Funct Foods. 2020;74:104171. [Google Scholar]
- 83.Taye AD, Bizuneh GK, Kasahun AE. Ethnobotanical uses, phytochemistry and biological activity of the genus euclea: A review. Front Pharmacol. 2023;14:1170145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Tyagi AK, Bukvicki D, Gottardi D, Tabanelli G, Montanari C, Malik A, Guerzoni ME. Eucalyptus essential oil as a natural food preservative: in vivo and in vitro antiyeast potential. Biomed Res Int. 2014;2014:969143. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The study tools used in acquiring the data presented in this manuscript are available as raw data in questionnaire documents. This can be acquired from the corresponding author when needed.