Summary
Diabetic peripheral neuropathy (DPN) occurs in up to half of individuals with types 1 (T1D) and 2 diabetes (T2D). DPN is a distal-to-proximal loss of peripheral nerve function causing physical disability and possibly pain, lowering quality-of-life. Early diagnosis improves clinical outcomes, but lifetime prognosis remains poor. Hyperglycemia is a DPN risk factor and glycemic control slows DPN in T1D. However, it has modest or no benefit in individuals with T2D, likely due to the high prevalence of comorbidities, including the metabolic syndrome, an additional risk factor. DPN pathophysiology is complex but converges on a unifying theme of bioenergetic failure in peripheral nerves secondary to their unique anatomy. Current management focuses on controlling diabetes, the metabolic syndrome, and pain, but remains suboptimal. Thus, research is on-going to improve early diagnosis, identify specific molecular pathways to develop mechanism-based therapies, and investigate lifestyle interventions to improve clinical outcomes and disease prognosis.
Keywords: Diabetic peripheral neuropathy, diagnosis, epidemiology, management, pain, pathophysiology, risk factors
Introduction
There is an epidemic of diabetes worldwide.1 In 2019, 463 million people, or 9.3% of the world’s population had either type 1 (T1D) or type 2 (T2D) diabetes and 4.2 million people died from diabetes-related complications.1 Diabetes will affect an estimated 578 million people by 2030 and 700 million by 2045.1 This growth is largely attributed to an increase in T2D prevalence in the setting of aging. While diabetes prevalence will increase worldwide, the greatest increases are expected in countries with economies moving from low- to middle-income status, particularly in the Middle East and North Africa.1 As the prevalence of diabetes increases, the burden of diabetes-related complications is also expected to grow.
Among diabetes complications, damage to the peripheral and autonomic nervous system is the most prevalent. Distal symmetric polyneuropathy, which presents as lower limb followed by the upper limb sensory loss, is the most common type of diabetic nerve damage. It will be the focus of this review and will be referred to as diabetic peripheral neuropathy (DPN). The impact of DPN is substantial. It predisposes to lower quality-of-life, falls, and superficial injuries, which can lead to infection and amputation.2 DPN is also associated with higher all-cause and cardiovascular mortality in individuals with diabetes.3
Unfortunately, DPN management remains suboptimal. In T1D, glucose control slows DPN progression.4 However, in T2D, glucose control only marginally affects DPN progression, due to the presence of the metabolic syndrome (MetS). Therefore, managing DPN in T2D currently revolves on weight loss and exercise to mitigate the MetS.5 Earlier intervention may slow DPN progression, making timely diagnosis critical. This review will cover DPN epidemiology, clinical presentation, and diagnosis, focusing on emerging approaches for earlier detection. It will also outline on-going DPN pathophysiology research, which seeks to develop mechanism-based therapies. Current optimal treatment approaches and research into lifestyle interventions to improve patient outcomes will be addressed, as well as the obstacles that must be overcome to secure these outcomes.
Epidemiology and risk factors
Studies of DPN incidence and prevalence among individuals with diabetes most frequently utilize standardized measures, which combine symptoms and physical exam findings. Cross-sectional and cohort studies conducted since 2016 report a DPN incidence of about 8.8/1,000 person-years among individuals with T1D6 and 24–26.9/1,000 person-years among individuals with T2D.6,7 DPN prevalence is generally around 30% (Table 1).8,9 A recent worldwide meta-analysis (29 studies, 50,112 participants) found individuals with T2D had higher DPN prevalence (31.5%, 95%CI 24.4–38.6%) versus those with T1D (17.5%, 95%CI 13.1–36.5%).20 Since diabetes duration is a strong DPN determinant, it is present in fewer adolescents than adults.9,11 However, the US SEARCH study still reported substantial age-adjusted DPN prevalence in adolescents (T2D: 17.7% versus T1D: 8.5%).9 Further, DPN prevalence varies by country and ranges from 1% to 80%.16 This large variation likely arises from multiple factors, including disease severity, diabetes duration, DPN definition, and comorbid conditions predisposing to neuropathy development, especially the MetS.
Table 1.
DPN incidence and prevalence studies since 2017
| Year, First author | Country | Study Type & Population | DPN measure | Prevalence |
|---|---|---|---|---|
| 2017, Abdel-Motal 10 | Algeria, Bahrain, Egypt, Libya, Jordan, Morocco, South Africa, Sudan | Systematic review 2,243 T1D |
Variable | 18% |
| 2017, Dabelea 9 | United States | Cross-sectional study 1,746 T1D, 272 T2D youth |
MNSI-E | T1D: 8·5% T2D: 17·7% |
| 2017, Jaiswal 11 | United States | Cohort study 1,734 T1D youth 258 T2D youth |
MNSI-E | T1D: 7% T2D: 22% |
| 2018, Cardinez 12 | Canada | Longitudinal study 361 T1D >50 years |
MNSI-Q | 42·7% |
| 2019, Ponirakis 13 | Qatar | Cross-sectional study 1,095 T2D |
Douleur Neuropathique en 4 questions | 34·5% |
| 2020, Christensen 14 | Denmark | Longitudinal cohort study 5,249 T2D |
MNSI-Q | 17·9% |
| 2020, Jeyam 15 | Scotland | Cohort study 5,558 T1D |
MNSI-Q | 13% |
| 2020 Lu 16 | Argentina, Bangladesh, China, Germany, India, Italy, Kenya, Mexico, Pakistan, Poland, Russia, Serbia, Uganda, Ukraine | Cross-sectional study 2,733 T2D in clinics |
Sensory symptoms for >3 months | Overall: 26·7% Kenya 0·58% Ukraine: 79·55% |
| 2020, Mizokami-Stout 17 | United States | Cohort study 5,936 T1D |
MNSI-Q | 11% |
| 2020, Sun 18 | Australia, Bangladesh, China, France, India, Iran, Malaysia, Nigeria, Sri Lanka, Sweden Taiwan, Turkey, UK, USA | Meta-analysis 50,112 participants |
Variable | T1D: 17·5% T2D: 31·5% |
| 2021, Amutha 6 | India | Longitudinal cohort study T1D 3,252, T2D 889 |
Vibratory perception threshold | Incidence T1D: 8·8/1000 person-years Incidence T2D: 24·0/1000 person-years |
| 2021, An 7 | United States | Retrospective chart review 135,119 T2D in 1 health system |
Medical diagnosis codes | Incidence: 26·9/1,000 person-years |
| 2021, Aronson 8 | Canada | Cross-sectional study 471 T1D, 3,903 T2D |
Modified Toronto Clinical Neuropathy Score | T1D: 16·7% T2D: 29·3% |
| 2021, TODAY Study Group 19 | United States | Cohort study 674 T2D youth |
MNSI-E, MNSI-QE | 34·9% |
MNSI-E, Michigan Neuropathy Screening Instrument Exam; MNSI-Q, Michigan Neuropathy Screening Instrument Questionnaire; MNSI-QE, Michigan Neuropathy Screening Instrument Questionnaire and Exam·
Diabetes is the strongest determinant of DPN, along with disease characteristics, such as diabetes duration and diabetes severity, measured by hemoglobin A1C (HbA1C).21,22 Additionally, several studies suggest an association between glycemic variability and DPN presence,23 although there is some discordance.24 The UK Prospective Diabetes Study Group found that intensive glucose control lowered the relative risk of DPN onset in T2D patients, assessed by biothesiometer, but only after a 15-year follow-up;25 notably there was significant cohort sample size attrition. The Rio de Janeiro Type 2 Diabetes Cohort Study found glycemic variability did not correlate with incident DPN, determined by clinical symptoms or exam, over a median 9.3-year follow-up, although it did correlate with a composite outcome of both incident and worsening DPN symptoms.26 Moreover, nerve damage may begin long before individuals with hyperglycemia develop overt diabetes, as supported by the growing body of literature supporting an association between prediabetes and early small-fiber symptoms.27
Controlling hyperglycemia impacts DPN progression in patients with T1D.4 However, overall, glucose control may only moderately affect DPN onset and progression in patients with T2D, suggesting the presence of additional risk factors. Clinical research has identified the MetS as the critical risk factor, which includes, in addition to elevated fasting glucose, obesity, dyslipidemia (high triglycerides, low high-density lipoprotein [HDL]), and hypertension. Obesity measured centrally by waist circumference or overall by body mass index (BMI) especially increases DPN risk, in both the context of diabetes and independent of glycemia, in adults14,28–30 and youth (Table 2).11,19
Table 2. MetS-related DPN risk factors identified from clinical studies since 2017.
Search criteria were “diabetic peripheral neuropathy” with “obesity, metabolic syndrome” and “risk factors, metabolic syndrome”, as of 2017. Inclusion criteria were studies with over 100 T1D, T2D, or combined participants.
| Year, First Author | Country | Population | DPN measure | MetS and diabetes findings related to DPN |
|---|---|---|---|---|
| 2017, Hanewinckel. (105) | Netherlands | Rotterdam Study of 908 participants. | Symptom questionnaire, neurological examination, NCS | BMI independently associated with lower sural SNAP (OR 1·53 95%CI 1·13–2·09) and peroneal CMAP (OR 1·49 95%CI 1·11–1·99) amplitudes. |
| 2017 Jaiswal (11) | United States | SEARCH for Diabetes in Youth study of 1,734 T1D youths and 258 T2D youths. | MNSI-E, MNSI-Q | Of MetS components, higher LDL-c, TG, obesity, diastolic BP and lower HDL-c were DPN risk factors in T1D youths; lower HDL-c was a DPN risk factor in T2D youths. Poor glycemic control correlated with DPN in T1D OR 1·53 (95%CI 1·24–1·88) but not T2D youths OR 1·05 (95%CI 0·7–1·56). |
| 2018, Andersen (22) | Denmark | Danish arm of the Anglo-Danish-Dutch study of Intensive Treatment of Diabetes in Primary Care (ADDITION) of 1,256 T2D participants without DPN at baseline. | MNSI-Q | Baseline weight HR 1·09 (95%CI 1·03–1·16), WC HR 1·14 (95%CI 1·05–1·24), BMI HR 1·14 (95%CI 1·06–1·23), log2(methylglyoxal) HR 1·45 (95%CI 1·12–1·89), HDL-c HR 0·82 (95%CI 0·69–0·99), and LDL-c HR 0·92 (95%CI 0·86–0·98) significantly associated with incident DPN. |
| 2018, Callaghan (13) | China | Pinggu, China study of 4,002 participants, 37·2% normoglycemic, 18·9% diabetic, 44·0% prediabetic. | MNSI-E, MNSI-Q | Diabetes OR 2·60 (95%CI 1·77–3·80), weight OR 1·09 (95%CI 1·02–1·18), and the number of MetS components OR 1·17 (95%CI 1·03–1·32) significantly associated with DPN. |
| 2019, Kurisu (106) | Japan | 625 participants, 68·8% normoglycemic, 12·0% diabetic, 19·2% prediabetic. | Interview, ATR, QVT, NCS | T2D OR 3·65 (95%CI 1·68–7·93) and dyslipidemia OR 0·53 (95%CI 0·30–0·96) significantly associated with DPN in multivariable models, but not prediabetes OR 1·47 (95%CI 0·69–3·12) or WC OR 1·02 (95%CI 0·95–1·10). No parameters associated with DPN when T2D participants were excluded. |
| 2019, Schlesinger (107) | Germany | Cooperative Health Research in the Region of Augsburg (KORA) F4/FF4 cohort of 513 participants. | MNSI | Overweight OR 3·06 (95%CI 1·57–5·97), obese OR 3·47 (95%CI 1·72–7·00), WC OR 1·22 (95%CI 1·07–1·38) significantly associated with DPN in multivariable models. Interaction analyses did not reveal any differences by diabetes status. |
| 2020, Callaghan (25) | United States | University of Michigan bariatric surgery clinic of 138 obese participants. | Primary, Toronto consensus definition of probable DPN; secondary, IENFD in distal leg and 4 NCS in sural, tibial, ulnar nerves. | BMI was comparable in obese participants with and without DPN (p=0·86). WC OR 1·39 (95%CI 1·10–1·75), TGs OR 1·31 (95%CI 1·00–1·70), systolic BP OR 2·89 (95%CI 1·49–5·61) significantly associated with DPN. |
| 2020, Callaghan (45) | United States | University of Michigan bariatric surgery clinic of 138 obese participants. | Primary, Toronto consensus definition of probable DPN; secondary, IENFD in distal leg and one NCS in sural nerve. | WC, HDL-c, systolic BP significantly associated with cognitive decline by NIH Toolbox composite, after adjusting for age, WRAT4, and education level |
| 2020, Christensen. (23) | Denmark | Danish Centre for Strategic Research in Type 2 Diabetes (DD2) Cohort of 5,249 T2D participants. | MNSI-Q, DN4 | In regression analyses, central obesity (WC, WHR, waist-to-height ratio) associated with DPN. Of MetS components, TG ≥1·7 mmol/l aPR 1·36 (95%CI 1·17–1·59), HDL-c <1·0/1·2 mmol/l (male/female) aPR 1·35 (95%CI 1·12–1·62), hs-CRP ≥3·0 mg/l aPR 1·66 (95%CI 1·42–1·94), and HbA1c ≥78 mmol/mol (9·3%) aPR 1·42 (95%CI 1·06–1·88) significantly associated with DPN. |
| 2020, Reynolds (14) | India | Chennai study of 652 participants, 19·9% normoglycemic, 44·8% diabetic, 35·5% prediabetic. | Primary, MNSI combined index; secondary, MSNI-E, MNSI-Q, monofilament, biothesiometer | DPN prevalence increased with poorer glycemic status (p<0·01), but not with number of MetS components. In normoglycemic participants, neuropathy trend was present as the number of MetS components increased (p=0·04). Diabetes (OR 3·41 95%CI 1·28–9·11), but not WC (OR 1·002 95%CI 0·88–1·14), significantly associated with DPN in multivariable models. |
| 2020, van der Velde (108) | Netherlands | Maastricht Study of 2,401 participants, 59·3% normoglycemic, 25·3% diabetic, 15·4% prediabetic. | NCV, VPT, DN4 | FBG or HbA1c significantly associated with 6 DPN measures and higher VPT; WC associated with worse sural nerve function and higher VPT; TGs, HDL-C, LDL-C, and BP were not associated with DPN; inflammation associated with DPN and higher VPT, but only in T2D participants. |
| 2021, Today Study Group (24) | United States | Treatment Options for type 2 Diabetes in Adolescents and Youth (TODAY) study of 674 T2D youths. | MNSI-E, MNSI-Q | BMI (per 5 kg/m2) HR 1·28 (95%CI 1·15–1·43), HbA1c HR 1·26 (95%CI 1·14–1·40), male sex HR 1·81 (95%CI 1·25–2·62), age HR 1·11 (95%CI 1·01–1·21) significantly associated with DPN in multivariable models. |
aPR, adjusted prevalence ratio; ATR, Achilles tendon reflexes; BMI, body mass index; BP, blood pressure; CI, confidence interval; CMAP, compound motor action potential; DN4, Douleur Neuropathique en 4 Questions; FBG, fasting blood glucose; HbA1c, hemoglobin A1c; HDL-c, high-density lipoprotein cholesterol; HR, hazard ratio; hs-CRP, high-sensitivity C-reactive protein; IENFD, intraepidermal nerve fiber density; LDL-c, low-density lipoprotein cholesterol; MetS, metabolic syndrome; MNSI-Q, Michigan Neuropathy Screening Instrument Examination; MNSI-Q, Michigan Neuropathy Screening Instrument Questionnaire; NCS, nerve conduction study; OR, odds ratio; QVT, quantitative vibration threshold; SNAP, sural sensory nerve action potential; T2D, type 2 diabetes; TG, triglycerides; VPT, vibration perception threshold; WC, waist circumference; WHR, waist-to-hip ratio; WRAT4, Wide Range Achievement Test 4.
Other risks span lower HDL11,14,29,30 and higher weight,21,30 triglycerides,14,34 blood pressure,34 and serum biomarkers of inflammation,14 and oxidative stress, i.e., methylglyoxal.30 Furthermore, the presence of more MetS components increases DPN likelihood, particularly small-fiber symptoms, and shortens the time to symptom onset, suggesting a possible dose-response relationship in prediabetic21,29 and diabetic individuals.22 Other independent risk factors include older age and lack of physical activity.14,28 Importantly, although MetS components are more frequent in T2D, MetS components also increase DPN risk in individuals with T1D.11,37 Statin use has previously been reported to enhance DPN risk; however, in a large nationwide registry, statin use was not linked to increased long-term risk of developing DPN.38
A newly emerging area addresses the role of genetic risk factors in DPN. Although predominantly a metabolically acquired neuropathy, DPN pathophysiology is multifactorial and polygenic39 risk factors are increasingly being identified. Single nucleotide polymorphisms (SNPs) that are predisposing or protective for DPN have been identified in metabolism and vasculature genes, However, further studies are needed before clinical implementation. Increasingly, genome-wide association studies (GWAS) are identifying potential SNP risk modifiers of DPN.40,41 For example, SNPs to mitogen-activated protein kinase 14 linked to DPN (rs3761980, rs80028505)40 and foot ulcers (rs80028505)41 in two independent cohorts of European descent. A multi-ancestry meta-analysis of T2D (n=228,499) versus controls (n=1,178,783) developed a polygenic risk score, which moderately, but significantly, associated with DPN.39
Epigenetic determinants of DPN are another emerging research avenue.42,43 The epigenome is modifiable through environmental cues, including metabolism, and may constitute a mechanism of metabolically acquired DPN. Indeed, among individuals with T2D and DPN, there is decreased peripheral nerve genomic DNA methylation and loci-specific differential DNA methylation, supporting a role for epigenetic regulation in neuropathy development.43 The role of DNA methylation on DPN development in T1D is less clear.42 Another putative, but preliminary research area, is the genetics and epigenetics of microRNAs in DPN.44
Overall, metabolic parameters, encompassing both diabetes and the MetS, are strong DPN determinants. Genetic and epigenetic factors remain in the research domain, though studies have highlighted potentially interesting findings.
Clinical presentation and diagnosis
Diabetes can cause several patterns of peripheral nerve injury, including DPN, autonomic neuropathy, radiculoplexus neuropathy, radiculopathy, and mononeuropathy. DPN, the focus of this review, is the most common (Figure 1).45 DPN presents with sensory symptoms, which begin symmetrically in the toes, and slowly advance up to the calves before beginning in the fingers followed by the arms. Symptoms encompass numbness and tingling, and, in some patients, pain (burning, stinging, shooting, deep aching). Neurologic examination can reveal decreased sensation to multiple modalities, including vibration, pinprick, and proprioception, following the same distribution pattern as the symptoms. Some patients have symptoms (pain) and signs (decreased pinprick sensation) attributable to injury of unmyelinated nerves, known as small fibers. Other patients experience symptoms (numbness) and signs (decreased vibration and proprioception) attributable to injury of large, myelinated nerves, known as large fibers. Pressure is perceived via thinly myelinated fibers, which fall under the large fiber umbrella. Most patients exhibit both small and large fiber involvement. Motor symptoms and signs are much less common, although motor involvement is often seen on electrodiagnostic testing.
Figure 1. DPN presentation.
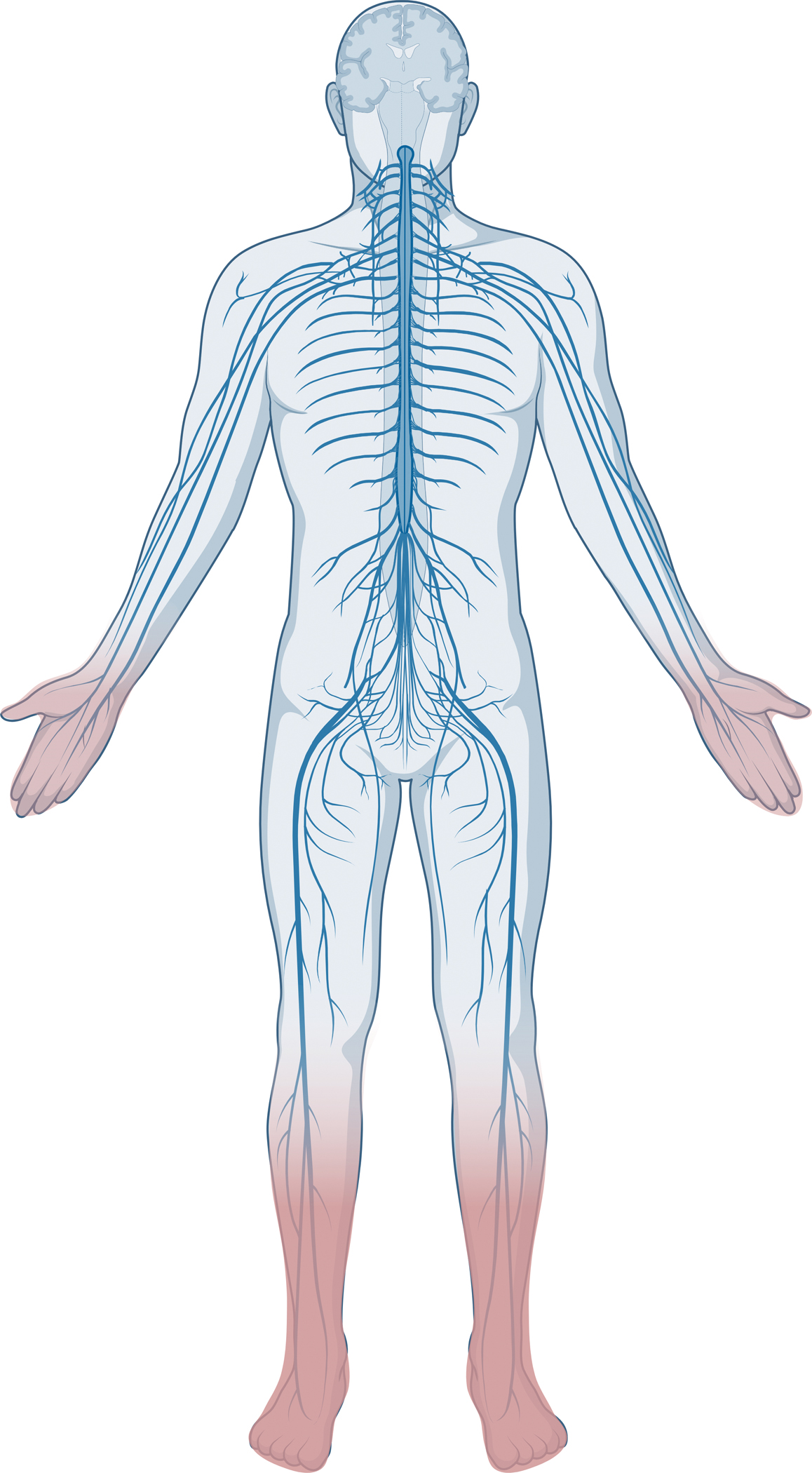
The most common manifestation of DPN is a distal symmetric polyneuropathy, which manifests in the lower limbs first followed by the upper limbs in a ‘stocking-glove’ configuration. DPN symptoms and signs start in the toes (darker pink), which progress proximally towards the calves, at which point, nerve injury initiates in the fingers and moves up to encompass the hands (lighter pink).
The American Diabetes Association (ADA) recommends evaluating DPN in patients with T2D at diagnosis and patients with T1D five years after diagnosis and then annually thereafter.5 Screening should comprise a detailed clinical history and examination, including assessment of temperature or pinprick sensation (small fiber), vibratory sensation (large fiber), and ability to perceive pressure using a 10 gram monofilament, (large fiber) (Panel 1). Electrodiagnostic testing and/or referral to a neurologist is rarely needed, except for patients presenting with atypical features, e.g., rapid onset, asymmetrical signs, non-length dependent neuropathy, or pronounced motor greater than sensory symptoms and signs.
Panel 1. DPN and pain diagnosis.
Diagnostic methods for DPN in routine clinical use
Clinical history:
Known diabetes diagnosis; diabetes is the most common cause of peripheral neuropathy. Longer diabetes duration, uncontrolled diabetes (higher HbA1C), obesity, dyslipidemia, hypertension, and older age are DPN risks.
Symptom presentation:
Positive, e.g., tingling, burning and lancinating pain, and negative sensations, e.g., numbness, usually occurring in a symmetric distal-to-proximal formation, initiating in the feet up to the calves, then progressing to the fingers.
Physical examination:
Temperature or pinprick sensation assess small fiber neuropathy. Vibration assesses large fiber neuropathy. Monofilament assesses ulcer risk. Small fiber neuropathy usually precedes large fiber neuropathy, but not always. Isolated small and large fiber neuropathy can also occur.
Methods for clinical diagnosis of atypical DPN presentation or clinical research studies
Nerve conduction studies (NCS):
Electrodiagnostic, measures nerve conduction velocities (NCVs) in sensory (sural, peroneal, tibial) and motor (peroneal, tibial) nerves using surface or needle electrodes.45 NCVs and amplitudes drop with progressive large fiber neuropathy. Abnormality (velocity or amplitude) in the sural nerve with at least one other NCS abnormality signifies large fiber neuropathy. Alternatively, a composite Z-score summation from multiple nerves is compared to normative values.
Intraepidermal nerve fiber density (IENFD):
Gold standard for small fiber neuropathy.45 Immunohistochemical test on skin punch biopsy, generally on the distal leg. Stained small fibers are counted and compared to normative values. IENFD drops with progressive small fiber neuropathy. Morphological, e.g., fiber length, branching, axonal swellings, and molecular, e.g., substance P, calcitonin gene-related peptide, growth associated protein 43, changes can be assessed.
Criteria:
Hierarchical classification schemes, rate the degree of certainty in the diagnosis, ranging from possible, to probable, to definite, using a combination of symptoms and signs, e.g., Toronto Consensus Panel.45
Diagnostic methods for DPN in the research setting
Corneal confocal microscopy (CCM):
Noninvasive imaging of corneal fibers, which are counted and compared to normative values.45 Studies suggest decline in CCM nerve fiber density may correlate with DPN progression. CCM can also be represented by fiber length and branching. Sensitivity (60–91%) and specificity (40–87%).53
DPNCheck:
A handheld point-of-care device, which measures sural NCV and response amplitude in a few minutes.52 Sensitivity (84.3–90.5%) and specificity (68.3–86.1%).
Neuropad:
A sticker affixed to the plantar surface of the foot, which measures moisture (sweat) by turning from blue to pink.52 Sensitivity (65.1–100.0%) and specificity (32.0–78·5%).
Sudoscan:
Measures conductance of chloride ions from sweat released from hands and soles of the feet after electrical stimulation of sweat glands. Measures sudomotor function in a few minutes.52 Sensitivity (87.5%) and specificity (76.2%).
Diagnostic methods for painful DPN in the research setting
Quantitative sensory testing (QST):
Standardized protocols measuring the response to well-defined sensory stimuli.45 Parameters include thermal and mechanical detection, pain, vibration, and pressure pain thresholds, dynamic mechanical allodynia, and wind-up ratio, which assess function in all fiber types (Aβ large myelinated, Aδ thinly myelinated, C unmyelinated).
Microneurography:
Needle electrode measures spontaneous activity and stimulus-evoked of unmyelinated C fibers in peripheral nerve, usually peroneal.75 Irregular “saw-tooth” baselines in abnormal nerves have been observed. This is a labor-intensive technique, which requires further cross laboratory validation.
Hoffman-Reflex rate-dependent depression:
Differentiates pain of spinal disinhibition origin from pain of peripheral origin by measuring the deep tendon reflex response neural pathways.75
Functional brain imaging:
fMRI uses different protocols, including the BOLD response, arterial spin labelling, and connectivity analysis, and relates these changes to both spontaneous and evoked pain states, illustrating the distributed cortical network involved in the discriminative and affective pain components, as well as the descending pain modulatory system.
Clinical history and examination are the mainstays of clinical diagnosis. However, confirmatory testing is available for large fiber (electrodiagnostic) and small fiber (intraepidermal nerve fiber density [IENFD]) nerve injury. Electrodiagnostic tests have good test characteristics, with areas under the curve (AUCs) ranging from 0.76 to 0.90,46 but face issues defining the parameters that constitute an abnormal test. Moreover, they do not assess small fiber involvement. IENFD also has good test characteristics (AUCs 0.75 to 0.82)46 but is invasive. Further, many countries lack facilities to perform IENFD assessments, and, where available, it is uncertain whether commercial laboratories perform comparably to academic centers. Overall, confirmatory testing is not usually needed for clinical care, but is used in clinical research and in patients with atypical presentations.
In addition to confirmatory tests, provider administered clinical scales are also available to identify patients with neuropathy (Panel 2). At least 18 such scales exist, mostly assessing similar domains, e.g., sensory (large and small fiber), motor, autonomic, reflexes, but attributing variable weight to these domains.47,48 Studies show that many of these scales have similar diagnostic characteristics. Like the confirmatory tests above, the diagnostic characteristics of clinician rated scales are good, but not great.49 A limitation of the scales is the need for trained personnel for administering tests. However, these clinical scales perform comparably to confirmatory testing, but without the associated costs, time, and discomfort.46
Panel 2. DPN clinical scales and symptom questionnaires in clinical use.
Clinical scales, in alphabetical order
Used to screen for and monitor DPN progression using a combination of symptoms and signs.48
Diabetic Neuropathy Examination Score (DNE):
Eight-item examination tool that consists of muscle strength; muscle jerk reflex at the triceps; pinprick in the index finger and great toe; and vibration, joint position, and touch in the great toe.
Michigan Neuropathy Screening Instrument (MNSI):
Includes 15 questions related to history and symptoms and a physical exam consisting of great toe vibration with a 128-Hz tuning fork, muscle jerk reflex at the ankle joint, monofilament testing, and foot exam (appearance, ulcerations). The MNSI Index expands the MNSI with a weighted scoring algorithm.
Modified Toronto Clinical Neuropathy Score (mTCNS):
Includes six questions regarding impact of symptoms on daily living and examination of the loss of sensation to five modalities (touch, pinprick, temperature, vibration, and proprioception) in lower extremities. The mTCNS is a modification of the Toronto Clinical Neuropathy Score which included ankle and knee jerk reflexes and did not include gradation of symptom impact or severity of sensory loss.
Neuropathy Impairment Score (NIS):
Examination-based measure that includes muscle strength in 24 muscle groups; jerk reflexes at the biceps, triceps, brachioradalis, quadriceps, and ankle; and sensation to touch, vibration, joint position in the index finger and great toe. The NIS-LL is a condensed measure focused on the lower extremities, The NIS-LL+7 was developed specifically to measure impairment in DPN and includes nerve conduction studies of sural, tibial, and peroneal nerves, vibratory threshold at the great toe using quantitative sensory testing (QST), and heart rate response to deep breathing.
Total Neuropathy Score (TNS):
Multimodality measure that includes history of sensory, motor, and autonomic symptoms; strength at major muscle groups; muscle jerk reflex at the ankle joint; vibratory threshold at the great toes bilaterally and right index finger using QST; and nerve conduction studies of the bilateral sural nerves, right common peroneal, and bilateral posterior tibial nerves.
Utah Early Neuropathy Score (UENS):
Examination-based measure that includes muscle strength at the great toes; extent of sensory loss to pinprick and vibration in the lower extremities; proprioception at the great toes; muscle jerk reflexes at the ankle joints; and presence of allodynia in the toes or foot.
Screening and assessment symptom scales, in alphabetical order
Douleur Neuropathique en 4 Questions (DN4):
Includes questions related to history of seven types of pain, examination of loss of sensation to touch and pinprick, and hyperesthesia.108
Leeds Assessment of Neuropathic Symptoms and Signs Pain Scale (LANSS):
Includes five questions related to type of pain and associated signs and examination for decreased pinprick and hyperesthesia.108
Michigan Neuropathy Screening Instrument Questionnaire (MNSI-Q):
Includes 15 questions related to history and neuropathy symptoms. The MNSI-Q Index expands the MNSI-Q with a weighted scoring algorithm.108
At least five neuropathic pain questionnaires are also available to identify DPN patients experiencing neuropathic pain (Panel 2).50 Pain is often underreported and undertreated; therefore, identifying painful DPN is critical for clinical management.45,51 Importantly, not all pain is neuropathic in nature; therefore, distinguishing neuropathic pain from other pain generators, such as joint pain, may inform changes to clinical management.
DPN, and peripheral neuropathy more broadly, often go undiagnosed. The reasons are manifold, but the lack of systematic and widespread screening contribute to this problem. Moreover, current diagnostic methods are of suboptimal sensitivity and earlier diagnosis may improve prognosis. Therefore, new diagnostic tests for DPN are needed, which are rapid, sensitive, specific, and inexpensive. Emerging tests that have been most extensively studied include corneal confocal microscopy, DPNCheck, Neuropad, and Sudoscan (Panel 1).52 Corneal confocal microscopy is less invasive than IENFD and has acceptable diagnostic accuracy in most studies.53 However, the test requires specialized personnel and equipment and, as a result, is primarily used in research. DPNCheck is quick, reliable, and inexpensive, and may have diagnostic accuracy nearly equal to nerve conduction studies.52 Neuropad has not been studied as extensively as DPNCheck, but early results indicate its diagnostic test characteristics are modest at best.52 Similarly, studies to date do not support Sudoscan as a reliable test to diagnose neuropathy.52 In summary, while new diagnostic tests are being developed and tested, none are currently ready for clinical use.
DPN and cognitive decline
Several clinical studies demonstrate that diabetes54 and obesity55 predispose individuals to cognitive impairment and frank dementia, e.g., Alzheimer’s disease, later in life. Even early in life, people with diabetes54 or obesity35 exhibit subtle, but detectable decline in cognition. These findings suggest shared pathological processes may occur in the PNS and CNS. Like DPN, Alzheimer’s disease is characterized by mitochondrial dysfunction and bioenergetic failure.56 Moreover, the risks between DPN and cognitive dysfunction are shared, and encompass both glucose and lipid centric mechanisms.54,55 In clinical studies of patients with T1D and T2D, DPN is a risk for poorer cognitive function57,58 and brain structural changes.59 The possibility of shared or similar neurological damage in the periphery and centrally in diabetes is intriguing and the increasing burden of diabetes has implications for the future burden of dementia. Given their shared risks, early diabetes and DPN management could have important implications for future occurrence of dementia.
DPN management
DPN prevention and/or treatment is not straightforward and presents a challenge for both the treating physician and the affected patient. While there is a salutary effect of glycemic control for preventing DPN in patients with T1D, multiple clinical trials have failed to show a similar effect of this intervention in patients with T2D.4 The ADA guidelines for treating DPN recommend adding diet and exercise to glycemic control as critical therapeutic interventions for patients with T2D and DPN. Thus, while glycemic control is likely important in T2D patients, other interventions are clearly needed and suggested as standard of care.5
With respect to dietary modifications as a first line treatment, medical weight loss improves multiple MetS risk factors, making it an attractive intervention for DPN in T2D patients. Recent studies demonstrate the importance and limitations of this intervention. The Look Ahead study randomized 5,145 diabetes patients to an intervention primarily focused on dietary weight loss for 9 to 11 years.60 Dietary weight loss ameliorated Michigan Neuropathy Screening Instrument (MNSI) questionnaire scores, but did not impact MNSI examination scores. Similarly, an observational study by our group demonstrated that severely obese participants (n=131, mean baseline BMI 40.8 kg/m2) placed on a dietary weight loss intervention had the same findings, with improved MNSI questionnaire findings but a stable MSNI exam.61,62 Therefore, medical weight loss is a promising disease-modifying intervention for DPN, but more dramatic improvements may require earlier intervention and/or different interventions. Exercise studies have been more limited, with small samples and/or lacking randomization. Despite these limitations, T2D patients with DPN show improvement in IENFD with exercise.63 Interestingly, exercise interventions lead to only minimal weight loss indicating that, if exercise is effective, the mechanisms are likely independent of weight loss.
Lastly, there are no effective pharmacological interventions for DPN although sodium-glucose cotransporter (SGLT)-2 inhibitors provide a novel direction. SGLT-2 inhibitors block glucose resorption in the kidneys, increasing glucose excretion, which lowers blood levels. SGLT-2 inhibitors improve cardiovascular outcomes in diabetes patients,64 and recent animal studies indicate SGLT-2 inhibitors may be promising for improving neuropathy outcomes, although possibly to a greater extent in the context of T1D;65 however, human studies are lacking.
Given the lack of established disease-modifying therapies, the clinical management of DPN patients is focused on educating patients about DPN, the importance of good foot care, proper shoe ware and an annual foot exam.2 Another essential component of DPN management is the control of pain. Four drug classes of medications are effective for painful DPN, including serotonin norepinephrine reuptake inhibitors, tricyclic antidepressants, gabapentinoids, and sodium channel blockers (Figure 2).66 All these medications have similar effect sizes and differences within a class are likely minimal to nonexistent. Therefore, choosing a neuropathic pain medication should focus on factors beyond efficacy, including tolerability, other contraindications, and cost. Topical medications are also available with capsaicin the best studied.66 Effect sizes for capsaicin are comparable to oral therapies. Overall, however the effect sizes of all these medications are small, and only around 1 in 7 patients with painful DPN experience pain relief,45 emphasizing the clear need for more effective interventions for pain.
Figure 2. Pain medication selection protocol for patients with painful DPN. Adapted from Callaghan et al. JAMA, 2020.70.
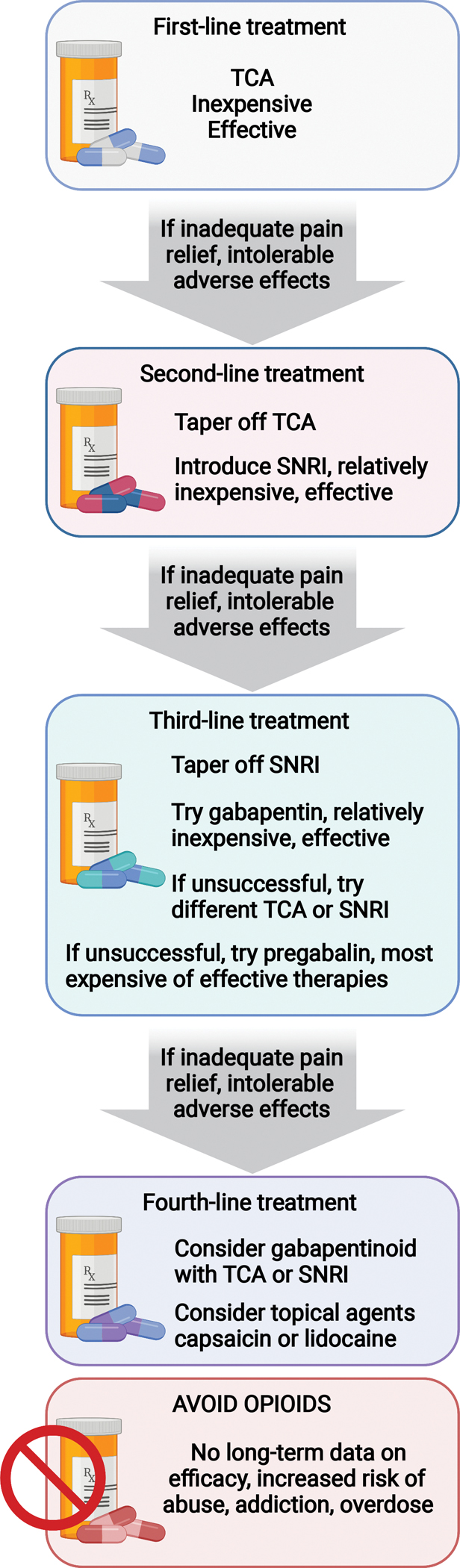
Tiered approach to pain management in patients with painful DPN considers. All medications, tricyclic antidepressants (TCAs), serotonin norepinephrine reuptake inhibitors (SNRIs), gabapentinoids (e.g., gabapentin, pregabalin), and sodium channel blockers (e.g., lidocaine) have similar effect sizes. Treatment selection, therefore, should focus on alternative parameters, e.g., tolerability, cost etc. Created with BioRender.com.
Behavioral interventions also exist for treating painful DPN. Exercise, cognitive behavioral therapy, and mindfulness have all been studied with early promising results.67 While definitive trials are needed, behavioral interventions may provide another possible avenue for treating pain, especially given the emerging evidence in other chronic pain conditions, such as fibromyalgia.68 Combination therapy, when behavioral therapy, pharmacological, or topical medications are employed together, may be warranted to address painful DPN, which is not responsive to one modality alone.
Surgical interventions are also available for treating painful DPN. Unfortunately, the role of spinal cord stimulation remains unclear despite a recent randomized clinical trial of 216 participants.69 Although 79% of participants responded in the intervention arm versus only 5% in controls, the results must be interpreted with great caution because this was an open label study without sham surgery in the control arm. Future surgical intervention studies for painful DPN will need to include a sham control and blinding to understand what role, if any, they have for treating painful DPN.
Although opioids, including tramadol and tapentadol, effectively reduce pain in DPN patients short-term, there is no information regarding long-term efficacy. Moreover, painful DPN is chronic and long-term opioids cause significant harm, including dependence, overdose, and death,66,70 and should be avoided in this population. Furthermore, multiple studies show that opioid treatment is common in patients with painful DPN, despite a lack in efficacy.71,72 Many physicians prescribe tramadol or tapentadol as alternatives to other opioids, but recent evidence suggests they share the same harmful long term effects.73 This evidence has led to guidelines recommending extreme caution when prescribing opioids for chronic non-cancer pain.66,74
An emerging area for treating pain is improved pain phenotyping, which potentially may better predict treatment response to pain medications (Panel 1). Phenotyping measures include sensory profiles, including pain/sensory quality and quantitative sensory testing, microneurography, and Hoffman-Reflex rate-dependent depression.75 Quantitative sensory testing measures the perceptions evoked by distinct sensory stimuli; this produces individual sensory profiles that can be used to stratify patients in a manner which relates to pathophysiological mechanisms.76 Microneurography involves inserting an electrode into a peripheral nerve and measuring spontaneous and stimulus evoked activity of unmyelinated small fibers (autonomic and sensory). Hoffman-Reflex rate-dependent depression is a means of assessing spinal disinhibition from pain of by measuring the same neural pathways that constitute the deep tendon reflex response. Studies using sensory profiling to predict treatment response are still in their infancy,45,77 and more work remains to integrate such measures into clinical trials. Similarly, Hoffman-Reflex rate-dependent depression has been investigated in animals,75 but future studies are needed to determine whether this test can predict treatment response in humans. If supported by future studies, pain phenotyping may further our ability to address DPN pain by targeting patient-specific characteristics. Current guidelines recommend assessing patients for other factors that may affect pain perception, such as mood and sleep disorders. This can help select medications to manage these symptoms, which may produce additional benefits for managing pain.66
Pathophysiology
Peripheral nerve anatomy poses significant challenges to well-orchestrated energy distribution and nerve function. Cell bodies of peripheral neurons are only microns in width, but their corresponding axons can be over a meter in length (Figure 3A); thus, for normal nerve function, neurons need to traffic energy-producing mitochondria from the cell body along the axon across large distances to areas of high energy demand. When mitochondria fail to traffic to the distal portion of a long axon or are dysfunctional upon arrival, there is subsequent energy failure and axonal injury. This distal-to-proximal loss of normal energy supply to axons occurs in both T1D and T2D78 and underlies the stocking-glove pattern of clinical signs and symptoms, which define DPN. Hyperglycemia is present in both T1D and T2D; however, comorbid obesity and dyslipidemia in T2D additionally underscore the delicate balance between systemic and peripheral nervous system (PNS) metabolic requirements for normal nerve function. Thus, bioenergetic failure and mitochondrial dysfunction secondary to excessive circulating glucose and lipids provide a unifying mechanism to explain both the pathology and clinical presentation of DPN.
Figure 3. Neuron anatomy and DPN pathophysiology.

(A) Peripheral neuron cell bodies are only microns in width, but axons are up to several feet in length. For normal nerve function, neurons traffic mitochondria from the cell body along the axons to areas of high energy demand. Failure to traffic mitochondria results in energy failure at axon termini followed by distal (pink) -to-proximal (blue) nerve injury, which underlies the stocking-glove pattern of clinical signs and symptoms. (B) Homeostatic conditions (blue), healthy nerve: Axons take up glucose (GLU) in an insulin-independent manner through glucose transporter 3 (GLUT3; green transporter), which is metabolized to pyruvate (PYR) in the cytoplasm and further to ATP in mitochondria. Schwann cells take up glucose in an insulin-dependent manner through GLUT1 (blue transporter); part of the glucose is metabolized for energy use by Schwann cells, part is metabolized to pyruvate then lactate (LAC) for transport to axons through monocarboxylate transporter 1 (MCT1; grey, on Schwann cells) then MCT2 (purple, on axons). Schwann cells take up fatty acids (FA) using fatty acid transporter/ translocase/ binding protein (brown), primarily for myelin production. Mitochondria (blue) are trafficked in the anterograde and retrograde directions to areas of high energy demand. Mitochondria generate a mitochondrial membrane potential for ATP production, powering Na+/K+ ATPase channel activity (pink; purple spheres are cations). Mitochondrial biogenesis occurs to replenish dysfunctional mitochondria (brown), which are eliminated by mitophagy. Pathologic conditions (pink), DPN nerve: Hyperglycemia increases glucose flux (wider arrow) into axons through GLUT3; excess glucose that is not metabolized by glycolysis enters or activates other pathways, e.g., polyol, advanced glycation end products (AGEs), and protein kinase C (PKC) pathways. Downstream, this triggers inflammation and disrupts Na+/K+ ATPase channel activity (no cations pumped, stop sign). Insulin resistance (IR) impairs insulin-dependent GLUT1-facilitated glucose uptake by Schwann cells, disrupting MCT-mediated fuel transport to axons (stop sign) and possibly the balance with lipid metabolism and myelin production. Hyperglycemia and hyperlipidemia also enhance Schwann cell oxidative stress and apoptosis. Hyperlipidemia disrupts axonal mitochondrial biogenesis and trafficking, lowering the fraction and velocity of motile mitochondria. Palmitate also depolarizes axonal mitochondria and reduce their ability to dissipate mitochondrial membrane potential to meet physiological energy demand. Mitophagy is also impaired. Created with BioRender.com.
The idea that axons are also dependent on glia for energy substrates and normal function is well recognized in the central nervous system (CNS);79 however, the importance of this concept to our understanding of DPN pathophysiology has only recently emerged.80 Glucose enters both axons and supporting Schwann cells via specific glucose transporters. Through a process known as metabolic axoglial coupling,80 Schwann cells can then supply energy requiring long axons with fuel by transferring lactate through certain monocarboxylate transporters.81 The MetS results in poor glucose uptake in the PNS, which develops insulin resistance (IR) similar to peripheral muscle and fat tissue.82 This in turn disrupts axoglial coupling and depletes axonal energy stores, especially in the distal aspects of axons, leading to nerve injury and DPN over time. This novel idea underlying DPN pathogenesis bears out in preclinical studies. High-fat fed mice develop the MetS, placing long axons in an energy crisis with resultant clinical neuropathy, along with impaired axonal mitochondrial trafficking and biogenesis (Figure 3B).78
High-fat feeding also depolarizes energy starved axonal mitochondria, further reducing their ability to provide ATP to meet physiological energy demands during nerve firing. As mitochondrial damage accumulates with disease progression, it triggers persistent global tissue damage due to energy failure,83 oxidative84 and endoplasmic reticulum stress,85,86 and loss of normal ion flux.83,87 Glucose accumulation in the nerve leads to an increase in advanced glycation end products, and enhances the polyol, hexosamine, and protein kinase C pathways. These changes in nerve metabolism induce inflammation and disrupt osmotic balance, membrane resting potential, axon electrophysiology, and vascular function.83
Importantly, the impact of fatty acids on neuronal mitochondrial function is chain length- and saturation-dependent; longer-chain, saturated fatty acids are more detrimental to neuronal mitochondria versus their shorter-chain, unsaturated counterparts.88 This finding suggests potential dietary interventions could slow DPN progression. In obese prediabetic rodents, supplementing the diet with unsaturated fatty acids ameliorates neuropathy, without reducing body fat mass or improving systemic IR,88 possibly indicative of local energy effects directly within the nerve. In human T2D89 and obesity90 studies, DPN correlates with distinct plasma signatures of specific lipid classes and/or species. Although causality in the DPN disease process has not been established for these lipids in humans, ceramides, sphingomyelins, and lipid signaling inhibitors are being investigated preclinically in the context of T2D and obesity.91
While recent research has focused on the investigation of direct metabolic dysfunction in DPN pathogenesis, additional emerging directions involve metabolism regulated through extracellular vesicles92 and the gut microbiome.93 We anticipate these new areas will continue to converge on the basic tenet that neural anatomy results in unique energy demands, which, if not met, leads to injury and ultimately neuropathy.
DPN pain mechanisms
Neuropathic pain, i.e., arising from lesions or disease affecting the somatosensory nervous system, usually manifests in DPN as spontaneous burning pain sometimes associated with hyperalgesia and/or allodynia and often accompanied by a background sensory loss.45 Determinants of painful DPN remain incompletely known; however, it likely occurs from complex interactions of lesion characteristics (e.g., severity), diabetes severity, MetS, genetics, and environment.83
Hyperexcitability of sensory neurons in the form of spontaneous activity and an enhanced response to sensory stimuli is thought to be a key driver of neuropathic pain in DPN. In the healthy nerve, ion channels sense and transduce the initial stimulus to sensory receptors (Figure 4A–B). Neural ion channels then generate the action potential, which propagates along afferent axons towards the CNS. Finally, within the spinal cord dorsal horn, ion channels trigger neurotransmitter release. Mutations or changes in expression and/or posttranslational modifications to ion channels alter their functional properties, triggering hyperexcitability. Several ion channels are implicated in painful DPN, including voltage-gated sodium channels (VGSCs; Nav1.7, 1.8, 1.9),45,83,94 voltage-gated potassium channels,95 ligand-gated transduction channels such as transient receptor potential cation channel, subfamily A, member 1 (TRPA1),96 calcium channels (Cav3.2),83 and hyperpolarization-activated cyclic nucleotide-gated channels (HCN2).97 Some of these channels have been validated in human studies of painful DPN, e.g., VGSCs,94 TRPA1,96 whereas others remain within the preclinical domain. Nevertheless, clinical studies of painful DPN targeting these channels have been launched.98
Figure 4. DPN pain mechanisms.
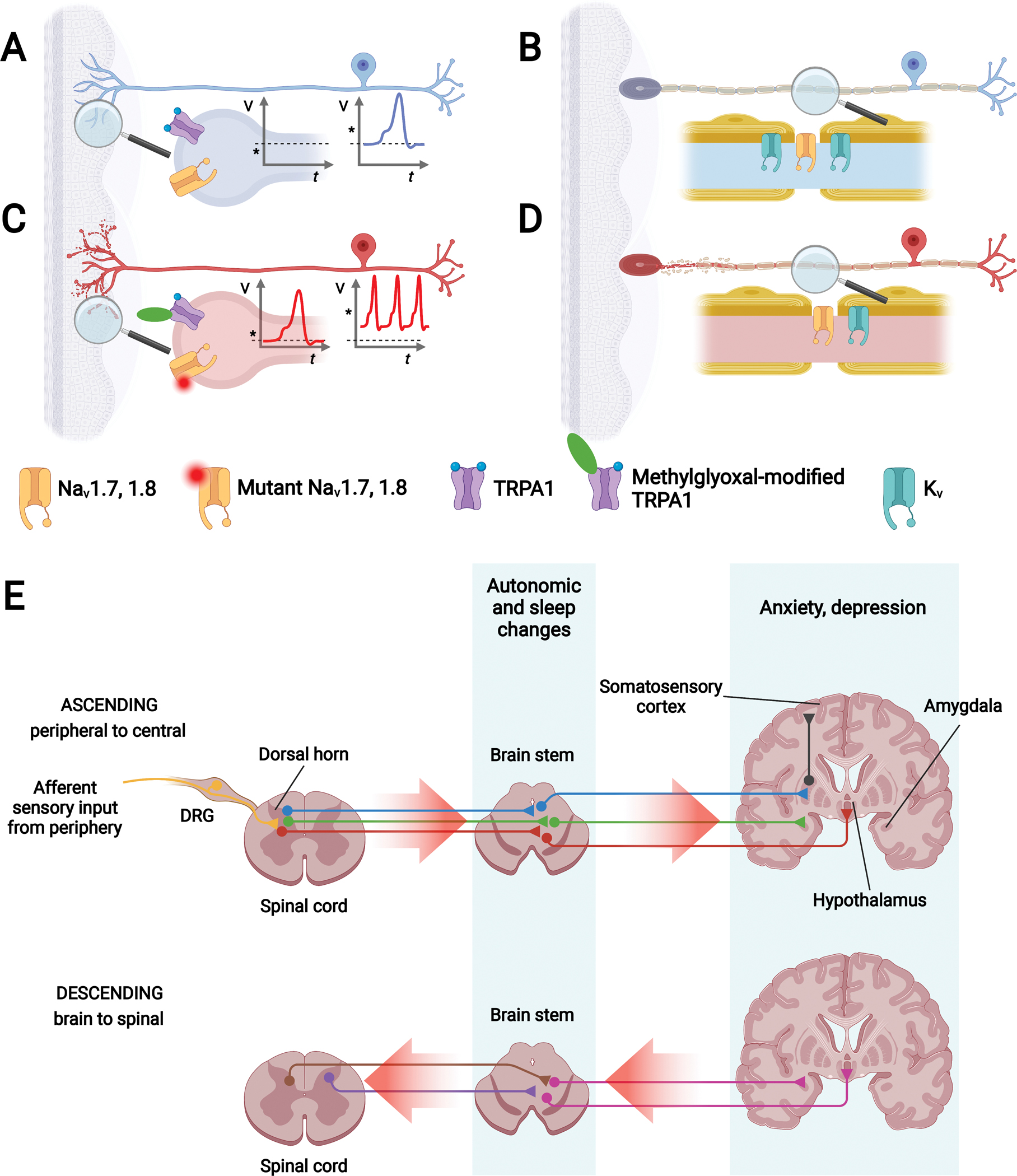
(A) Healthy unmyelinated C fiber: Healthy unmyelinated C fiber (blue) with free nerve endings embedded in skin. Inset: Voltage-gated sodium channels (e.g., Nav1.7, 1.8; yellow channel) and ligand-gated channels (e.g., TRPA1; purple channel) transmit an afferent signal if the stimulus (asterisk) is above the channel threshold. (B) Neuropathic unmyelinated C fiber: Unmyelinated C fiber with distal-to-proximal length-dependent neurodegeneration (red) starting in the free nerve endings. Inset: Channel modification by reactive metabolites, e.g., methylglyoxal, reactive species (green oval), or channel mutations (red shaded circle) can lower the threshold potential. Less intense stimulus (asterisk) now falls below the channel threshold, causing neuronal hyperexcitability. (C) Healthy myelinated fiber: Healthy myelinated fiber (blue) with encapsulated nerve ending embedded in skin. Inset: Voltage-gated potassium (Kv; green channel, located to the juxtaparanode) and sodium (Nav; yellow, located to the nodes) channels propagate signals along nodes of Ranvier by saltatory conduction. (D) Neuropathic myelinated fiber: Myelinated fiber with distal-to-proximal length-dependent neurodegeneration (red) starting in the encapsulated nerve ending. Inset: Kv channels expression level drops, leading to hyperexcitability. (E) Simplified schematic of CNS pain mechanisms. Top panel: Ascending pathways of CNS dysfunction arising from peripheral to central pain signal amplification (red arrows). Sensory dorsal root ganglion (DRG; yellow) neurons carrying afferent signals connect with CNS neurons within the spinal cord dorsal horn, which relay within the brainstem to the brain. Different neuron colors (red, green, blue) represent various ascending pain pathways. Bottom panel: Descending pathways of CNS dysfunction arising from brain to spinal cord pain signal amplification (red arrows). Different neuron colors (pink, purple, brown) represent various descending pain pathways. CNS pain pathways give rise to autonomic dysfunction and anxiety, depression, and disturbed sleep. Created with BioRender.com.
Several mechanisms of ion channel-mediated hyperexcitability and pain can occur (Figure 4C–D).83 Increased ion channel expression increases conduction in unmyelinated C fiber nociceptors, intensifying impulse transmission to the CNS.83 Ion channel mutations can lower action potential thresholds or impair channel inactivation, causing increased resurgence currents, enhancing spontaneous activity and firing frequency.94 Post-translational modifications can also alter channel characteristics; for instance, modification of TRPA1 by methylglyoxal, an oxidized reactive metabolite that is elevated in diabetes, results in activation of this channel.83,96 TRPA1 senses noxious stimuli, such as exogenous reactive oxygen species and noxious cold. Therefore, oxidized (4-hydroxynonenal) or reactive oxygen species (H2O2) that are increased in diabetes can enhance such TRPA1 activation.
GWAS studies of painful DPN shed light on additional potential mechanisms; one study identified SNPs around the locus for phosphate carrier protein, mitochondrial (SLC25A3), involved in oxidative phosphorylation.99 DOLORisk is a large multicenter observational study of patients with neuropathic pain, including painful DPN with robust phenotyping and gene sets available for interrogation; these data are actively being analyzed to identify additional pathogenic SNPs.100 Additionally, differences in circulating pro-inflammatory cytokines in clinical studies101 and the C-X-C chemokine receptor type 4 (CXCR4) signaling axis are implicated in preclinical models.102
In addition to peripheral mechanisms, CNS dysfunction also contributes to pain (Figure 4E). Changes within the dorsal horn of the spinal cord further amplify nociceptive signals contributing to painful DPN. This includes altered dorsal horn neuron morphology and spinal disinhibition.103 The descending pain modulatory system, mediated by projections from the brainstem, can either inhibit or facilitate transmission of nociceptive information within the dorsal horn. Conditioned pain modulation provides a means to test the integrity of the descending pain modulatory system and is predictive of treatment response.2 Functional MRI studies suggest that facilitation via the ventrolateral periaqueductal grey is increased in patients with versus without painful DPN.104 These changes are not restricted to neurons, but preclinical models also suggest a contribution of pro-inflammatory microglia.105
A full review is outside the scope of this paper; however, multimodal MRIs show structural and functional differences in the somatosensory cortex in patients with painful versus nonpainful DPN.106 These changes could partly reflect deafferentation. Painful DPN is also associated with important psychological changes and comorbidities, including greater anxiety and depression and impaired sleep, which can contribute to maladaptive pain states. Moreover, it is possible to leverage brain MRI phenotypes to identify painful DPN patients likely to respond to specific pain treatments.107 Pain management will be discussed further in the “DPN management” section.
Conclusions and future directions
Unfortunately, diabetes patients continue to grapple with DPN, a prevalent complication, which, despite being long-recognized, still lacks disease-modifying therapies. DPN can develop despite well-controlled diabetes,4 suggesting determinants of disease in addition to glucose centric parameters. A better understanding of additional risk, particularly the contribution of modifiable factors, such as MetS components,11,14,21,28–30,34 can help identify diabetes patients most susceptible to DPN development. Awareness of modifiable risks also advocates potential interventions to slow DPN. Early indications are that medical weight loss and lifestyle interventions, such as diet and exercise, may slow DPN progression,60,61,63 leading to stable disease. Indeed, the ADA now recommends diet and exercise as first-line prevention for DPN.5 Since these interventions slow DPN, it suggests patients would benefit from earlier intervention. Thus, earlier, more sensitive, and specific diagnosis may help prognosis; however, clinical diagnosis remains, by and large, essentially unchanged over the past few decades and innovation is needed. Lack of effective therapies for painful DPN is a significant roadblock to pain management and mindfulness of long-term opioid side effects advocates a tiered approach, which avoids opioid prescription. We anticipate that a deeper understanding of the pathogenesis of DPN and the diverse mechanisms underlying distinct types of pain will lead to more effective treatment of DPN and, when present, pain. This prevalent disorder needs a disease-modifying therapy, a goal that underlies all current and newly evolving DPN research.
Search strategy and selection criteria:
References for this Review were identified by searches of PubMed between January 2017 and December 2021 with the terms “diabetic peripheral neuropathy”, “brain”, “cognitive decline”, “dementia”, “differential diagnosis”, “diagnosis”, “epidemiology”, “EWAS”, “genetic risk”, “GWAS”, “hyperexcitability”, “insulin resistance”, “ion channel”, “metabolic syndrome”, “neuropathic”, “pain”, “painful”, “pathophysiology”, “prevalence”, “scales”, “screening”. In addition, authors supplemented the search with references from the identified articles. There were no language restrictions. The final reference list was generated based on relevance to the topics covered in this Review.
Acknowledgments
MAE acknowledges grants from NIH NINDS (5R25NS089450), grants from NIH NCATS (UL1TR002240), grants from NIH NIDDK (P30-DK-02926 & P30-DK089503). HA acknowledges grants from Novo Nordisk Foundation (NFOC140011633). DLB acknowledges grants from the International Diabetic Neuropathy Consortium and is funded by UKRI/Versus (MR/W002388/1 PAINSTORM), the MRC (MR/T020113/1) and Diabetes UK (19/0005984).BCC acknowledges grants from NIH NIDDK R01DK115687, grants from JDRF 5COE-2019-861-S-B. ELF acknowledges grants from NIH NIDDK R01DK129320, grants from NIH NIDDK R24DK082841, grants from NIH NINDS R21NS102924, grants from IDNC - Intl Diabetic Neuropathy Consortium (NNF14OC0011633), grants from JDRF 5COE-2019-861-S-B, grants from Nathan and Rose Milstein Research Fund, grants from Sinai Medical Staff Foundation, grants from A. Alfred Taubman Medical Research Institute, grants from NeuroNetwork for Emerging Therapies.
Footnotes
Conflict of interest
MAE has nothing to disclose. HA has nothing to disclose. MGS has nothing to disclose. VV has nothing to disclose. DLB reports grants from AstraZeneca, grants from Lilly, grants from Diabetes UK, during the conduct of the study; DLB has acted as a consultant on behalf of Oxford Innovation for Amgen, Bristows, LatigoBio, GSK, Ionis, Lilly, Olipass, Orion, Regeneron and Theranexus over the last 2 years, outside the submitted work; in addition, DLB has a patent application ‘a method for the treatment or prevention of pain, or excessive neuronal activity, or epilepsy’ Application No. 16/337,428 pending. BCC reports personal fees from Medical legal work, grants from Veterans Affairs, personal fees from DynaMed, grants and personal fees from American Academy of Neurology, personal fees from Vaccine Injury Compensation Program, outside the submitted work. ELF has nothing to disclose.
REFERENCES
- 1.Saeedi P, Petersohn I, Salpea P, et al. Global and regional diabetes prevalence estimates for 2019 and projections for 2030 and 2045: Results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res Clin Pract 2019; 157: 107843. [DOI] [PubMed] [Google Scholar]
- 2.Feldman EL, Callaghan BC, Pop-Busui R, et al. Diabetic neuropathy. Nat Rev Dis Primers 2019; 5(1): 41. [DOI] [PubMed] [Google Scholar]
- 3.Hicks CW, Wang D, Matsushita K, Windham BG, Selvin E. Peripheral Neuropathy and All-Cause and Cardiovascular Mortality in U.S. Adults : A Prospective Cohort Study. Ann Intern Med 2021; 174(2): 167–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Callaghan BC, Little AA, Feldman EL, Hughes RA. Enhanced glucose control for preventing and treating diabetic neuropathy. Cochrane Database Syst Rev 2012; 6: CD007543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Pop-Busui R, Boulton AJ, Feldman EL, et al. Diabetic Neuropathy: A Position Statement by the American Diabetes Association. Diabetes Care 2017; 40(1): 136–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Amutha A, Ranjit U, Anjana RM, et al. Clinical profile and incidence of microvascular complications of childhood and adolescent onset type 1 and type 2 diabetes seen at a tertiary diabetes center in India. Pediatr Diabetes 2021; 22(1): 67–74. [DOI] [PubMed] [Google Scholar]
- 7.An J, Nichols GA, Qian L, et al. Prevalence and incidence of microvascular and macrovascular complications over 15 years among patients with incident type 2 diabetes. BMJ Open Diabetes Res Care 2021; 9(1). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Aronson R, Chu L, Joseph N, Brown R. Prevalence and Risk Evaluation of Diabetic Complications of the Foot Among Adults With Type 1 and Type 2 Diabetes in a Large Canadian Population (PEDAL Study). Can J Diabetes 2021; 45(7): 588–93. [DOI] [PubMed] [Google Scholar]
- 9.Dabelea D, Stafford JM, Mayer-Davis EJ, et al. Association of Type 1 Diabetes vs Type 2 Diabetes Diagnosed During Childhood and Adolescence With Complications During Teenage Years and Young Adulthood. JAMA 2017; 317(8): 825–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Abdel-Motal UM, Abdelalim EM, Abou-Saleh H, Zayed H. Neuropathy of type 1 diabetes in the Arab world: A systematic review and meta-analysis. Diabetes Res Clin Pract 2017; 127: 172–80. [DOI] [PubMed] [Google Scholar]
- 11.Jaiswal M, Divers J, Dabelea D, et al. Prevalence of and Risk Factors for Diabetic Peripheral Neuropathy in Youth With Type 1 and Type 2 Diabetes: SEARCH for Diabetes in Youth Study. Diabetes Care 2017; 40(9): 1226–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cardinez N, Lovblom LE, Bai JW, et al. Sex differences in neuropathic pain in longstanding diabetes: Results from the Canadian Study of Longevity in Type 1 Diabetes. J Diabetes Complications 2018; 32(7): 660–4. [DOI] [PubMed] [Google Scholar]
- 13.Ponirakis G, Elhadd T, Chinnaiyan S, et al. Prevalence and risk factors for painful diabetic neuropathy in secondary healthcare in Qatar. J Diabetes Investig 2019; 10(6): 1558–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Christensen DH, Knudsen ST, Gylfadottir SS, et al. Metabolic Factors, Lifestyle Habits, and Possible Polyneuropathy in Early Type 2 Diabetes: A Nationwide Study of 5,249 Patients in the Danish Centre for Strategic Research in Type 2 Diabetes (DD2) Cohort. Diabetes Care 2020; 43(6): 1266–75. [DOI] [PubMed] [Google Scholar]
- 15.Jeyam A, McGurnaghan SJ, Blackbourn LAK, et al. Diabetic Neuropathy Is a Substantial Burden in People With Type 1 Diabetes and Is Strongly Associated With Socioeconomic Disadvantage: A Population-Representative Study From Scotland. Diabetes Care 2020; 43(4): 734–42. [DOI] [PubMed] [Google Scholar]
- 16.Lu Y, Xing P, Cai X, et al. Prevalence and Risk Factors for Diabetic Peripheral Neuropathy in Type 2 Diabetic Patients From 14 Countries: Estimates of the INTERPRET-DD Study. Front Public Health 2020; 8: 534372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mizokami-Stout KR, Li Z, Foster NC, et al. The Contemporary Prevalence of Diabetic Neuropathy in Type 1 Diabetes: Findings From the T1D Exchange. Diabetes Care 2020; 43(4): 806–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sun J, Wang Y, Zhang X, Zhu S, He H. Prevalence of peripheral neuropathy in patients with diabetes: A systematic review and meta-analysis. Prim Care Diabetes 2020; 14(5): 435–44. [DOI] [PubMed] [Google Scholar]
- 19.Risk Factors for Diabetic Peripheral Neuropathy in Adolescents and Young Adults With Type 2 Diabetes: Results From the TODAY Study. Diabetes Care 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sun J, Wang Y, Zhang X, Zhu S, He H. Prevalence of peripheral neuropathy in patients with diabetes: A systematic review and meta-analysis. Prim Care Diabetes 2020. [DOI] [PubMed] [Google Scholar]
- 21.Callaghan BC, Gao L, Li Y, et al. Diabetes and obesity are the main metabolic drivers of peripheral neuropathy. Ann Clin Transl Neurol 2018; 5(4): 397–405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Reynolds EL, Callaghan BC, Banerjee M, Feldman EL, Viswanathan V. The metabolic drivers of neuropathy in India. J Diabetes Complications 2020: 107653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhang X, Yang X, Sun B, Zhu C. Perspectives of glycemic variability in diabetic neuropathy: a comprehensive review. Commun Biol 2021; 4(1): 1366. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lachin JM, Bebu I, Bergenstal RM, et al. Association of Glycemic Variability in Type 1 Diabetes With Progression of Microvascular Outcomes in the Diabetes Control and Complications Trial. Diabetes Care 2017; 40(6): 777–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Intensive blood-glucose control with sulphonylureas or insulin compared with conventional treatment and risk of complications in patients with type 2 diabetes (UKPDS 33). UK Prospective Diabetes Study (UKPDS) Group. Lancet 1998; 352(9131): 837–53. [PubMed] [Google Scholar]
- 26.Cardoso CRL, Leite NC, Moram CBM, Salles GF. Long-term visit-to-visit glycemic variability as predictor of micro- and macrovascular complications in patients with type 2 diabetes: The Rio de Janeiro Type 2 Diabetes Cohort Study. Cardiovasc Diabetol 2018; 17(1): 33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kirthi V, Perumbalath A, Brown E, et al. Prevalence of peripheral neuropathy in pre-diabetes: a systematic review. BMJ Open Diabetes Res Care 2021; 9(1). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Callaghan BC, Xia R, Reynolds E, et al. Association Between Metabolic Syndrome Components and Polyneuropathy in an Obese Population. JAMA Neurol 2016; 73(12): 1468–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Callaghan BC, Xia R, Banerjee M, et al. Metabolic Syndrome Components Are Associated With Symptomatic Polyneuropathy Independent of Glycemic Status. Diabetes Care 2016; 39(5): 801–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Andersen ST, Witte DR, Dalsgaard EM, et al. Risk Factors for Incident Diabetic Polyneuropathy in a Cohort With Screen-Detected Type 2 Diabetes Followed for 13 Years: ADDITION-Denmark. Diabetes Care 2018; 41(5): 1068–75. [DOI] [PubMed] [Google Scholar]
- 31.Hanewinckel R, Ikram MA, Franco OH, Hofman A, Drenthen J, van Doorn PA. High body mass and kidney dysfunction relate to worse nerve function, even in adults without neuropathy. J Peripher Nerv Syst 2017; 22(2): 112–20. [DOI] [PubMed] [Google Scholar]
- 32.Kurisu S, Sasaki H, Kishimoto S, et al. Clinical polyneuropathy does not increase with prediabetes or metabolic syndrome in the Japanese general population. J Diabetes Investig 2019; 10(6): 1565–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Schlesinger S, Herder C, Kannenberg JM, et al. General and Abdominal Obesity and Incident Distal Sensorimotor Polyneuropathy: Insights Into Inflammatory Biomarkers as Potential Mediators in the KORA F4/FF4 Cohort. Diabetes Care 2019; 42(2): 240–7. [DOI] [PubMed] [Google Scholar]
- 34.Callaghan BC, Reynolds E, Banerjee M, Chant E, Villegas-Umana E, Feldman EL. Central Obesity is Associated With Neuropathy in the Severely Obese. Mayo Clin Proc 2020; 95(7): 1342–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Callaghan BC, Reynolds EL, Banerjee M, et al. The Prevalence and Determinants of Cognitive Deficits and Traditional Diabetic Complications in the Severely Obese. Diabetes Care 2020; 43(3): 683–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.van der Velde J, Koster A, Strotmeyer ES, et al. Cardiometabolic risk factors as determinants of peripheral nerve function: the Maastricht Study. Diabetologia 2020; 63(8): 1648–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Braffett BH, Gubitosi-Klug RA, Albers JW, et al. Risk Factors for Diabetic Peripheral Neuropathy and Cardiovascular Autonomic Neuropathy in the Diabetes Control and Complications Trial/Epidemiology of Diabetes Interventions and Complications (DCCT/EDIC) Study. Diabetes 2020; 69(5): 1000–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kristensen FP, Christensen DH, Callaghan BC, et al. Statin Therapy and Risk of Polyneuropathy in Type 2 Diabetes: A Danish Cohort Study. Diabetes Care 2020. [DOI] [PubMed] [Google Scholar]
- 39.Vujkovic M, Keaton JM, Lynch JA, et al. Discovery of 318 new risk loci for type 2 diabetes and related vascular outcomes among 1.4 million participants in a multi-ancestry meta-analysis. Nat Genet 2020; 52(7): 680–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ustinova M, Peculis R, Rescenko R, et al. Novel susceptibility loci identified in a genome-wide association study of type 2 diabetes complications in population of Latvia. BMC Med Genomics 2021; 14(1): 18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Meng W, Veluchamy A, Hébert HL, Campbell A, Colhoun HM, Palmer CNA. A genome-wide association study suggests that MAPK14 is associated with diabetic foot ulcers. Br J Dermatol 2017; 177(6): 1664–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Roshandel D, Chen Z, Canty AJ, et al. DNA methylation age calculators reveal association with diabetic neuropathy in type 1 diabetes. Clin Epigenetics 2020; 12(1): 52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Guo K, Elzinga S, Eid S, et al. Genome-wide DNA methylation profiling of human diabetic peripheral neuropathy in subjects with type 2 diabetes mellitus. Epigenetics 2019; 14(8): 766–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Ciccacci C, Latini A, Colantuono A, et al. Expression study of candidate miRNAs and evaluation of their potential use as biomarkers of diabetic neuropathy. Epigenomics 2020; 12(7): 575–85. [DOI] [PubMed] [Google Scholar]
- 45.Jensen TS, Karlsson P, Gylfadottir SS, et al. Painful and non-painful diabetic neuropathy, diagnostic challenges and implications for future management. Brain 2021; 144(6): 1632–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Callaghan BC, Xia R, Reynolds E, et al. Better diagnostic accuracy of neuropathy in obesity: A new challenge for neurologists. Clinical Neurophysiology: Official Journal of the International Federation of Clinical Neurophysiology 2018; 129(3): 654–62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gewandter JS, Gibbons CH, Campagnolo M, et al. Clinician-rated measures for distal symmetrical axonal polyneuropathy: ACTTION systematic review. Neurology 2019; 93(8): 346–60. [DOI] [PubMed] [Google Scholar]
- 48.Fernández-Torres R, Ruiz-Muñoz M, Pérez-Panero AJ, García-Romero JC, Gónzalez-Sánchez M. Clinician Assessment Tools for Patients with Diabetic Foot Disease: A Systematic Review. J Clin Med 2020; 9(5). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gylfadottir SS, Itani M, Kroigard T, et al. Diagnosis and prevalence of diabetic polyneuropathy: a cross-sectional study of Danish patients with type 2 diabetes. Eur J Neurol 2020; 27(12): 2575–85. [DOI] [PubMed] [Google Scholar]
- 50.Attal N, Bouhassira D, Baron R. Diagnosis and assessment of neuropathic pain through questionnaires. The Lancet Neurology 2018; 17(5): 456–66. [DOI] [PubMed] [Google Scholar]
- 51.Daousi C, MacFarlane IA, Woodward A, Nurmikko TJ, Bundred PE, Benbow SJ. Chronic painful peripheral neuropathy in an urban community: a controlled comparison of people with and without diabetes. Diabet Med 2004; 21(9): 976–82. [DOI] [PubMed] [Google Scholar]
- 52.Selvarajah D, Kar D, Khunti K, et al. Diabetic peripheral neuropathy: advances in diagnosis and strategies for screening and early intervention. Lancet Diabetes Endocrinol 2019; 7(12): 938–48. [DOI] [PubMed] [Google Scholar]
- 53.Burgess J, Frank B, Marshall A, et al. Early Detection of Diabetic Peripheral Neuropathy: A Focus on Small Nerve Fibres. Diagnostics 2021; 11(2): 165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Biessels GJ, Despa F. Cognitive decline and dementia in diabetes mellitus: mechanisms and clinical implications. Nat Rev Endocrinol 2018; 14(10): 591–604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.O’Brien PD, Hinder LM, Callaghan BC, Feldman EL. Neurological consequences of obesity. Lancet Neurol 2017; 16(6): 465–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Neth BJ, Craft S. Insulin Resistance and Alzheimer’s Disease: Bioenergetic Linkages. Front Aging Neurosci 2017; 9: 345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Ding X, Fang C, Li X, et al. Type 1 diabetes-associated cognitive impairment and diabetic peripheral neuropathy in Chinese adults: results from a prospective cross-sectional study. BMC Endocr Disord 2019; 19(1): 34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zhao L, Mao L, Liu Q, Chen X, Tang X, An D. Cognitive impairment in type 2 diabetes patients with and without diabetic peripheral neuropathy: a mismatch negativity study. Neuroreport 2021; 32(14): 1223–8. [DOI] [PubMed] [Google Scholar]
- 59.Teh K, Wilkinson ID, Heiberg-Gibbons F, et al. Somatosensory network functional connectivity differentiates clinical pain phenotypes in diabetic neuropathy. Diabetologia 2021; 64(6): 1412–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Look ARG. Effects of a long-term lifestyle modification programme on peripheral neuropathy in overweight or obese adults with type 2 diabetes: the Look AHEAD study. Diabetologia 2017; 60(6): 980–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Callaghan BC, Reynolds EL, Banerjee M, et al. Dietary weight loss in people with severe obesity stabilizes neuropathy and improves symptomatology. Obesity (Silver Spring) 2021; 29(12): 2108–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Callaghan BC RE, Banerjee M, Akinci G, Chant E, Villegas-Umana E, Rothberg AE, Burant CF, Feldman EL Dietary weight loss in the severely obese stabilizes neuropathy and improves symptomatology. Obesity 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Zilliox LA, Russell JW. Physical activity and dietary interventions in diabetic neuropathy: a systematic review. Clin Auton Res 2019; 29(4): 443–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Brown E, Heerspink HJL, Cuthbertson DJ, Wilding JPH. SGLT2 inhibitors and GLP-1 receptor agonists: established and emerging indications. Lancet 2021; 398(10296): 262–76. [DOI] [PubMed] [Google Scholar]
- 65.Eid SA, O’Brien PD, Hinder LM, et al. Differential Effects of Empagliflozin on Microvascular Complications in Murine Models of Type 1 and Type 2 Diabetes. Biology (Basel) 2020; 9(11). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Price R, Smith D, Franklin G, et al. Oral and Topical Treatment of Painful Diabetic Polyneuropathy: Practice Guideline Update Summary: Report of the AAN Guideline Subcommittee. Neurology 2022; 98(1): 31–43. [DOI] [PubMed] [Google Scholar]
- 67.van Laake-Geelen CCM, Smeets R, Quadflieg S, Kleijnen J, Verbunt JA. The effect of exercise therapy combined with psychological therapy on physical activity and quality of life in patients with painful diabetic neuropathy: a systematic review. Scand J Pain 2019; 19(3): 433–9. [DOI] [PubMed] [Google Scholar]
- 68.Mascarenhas RO, Souza MB, Oliveira MX, et al. Association of Therapies With Reduced Pain and Improved Quality of Life in Patients With Fibromyalgia: A Systematic Review and Meta-analysis. JAMA Intern Med 2021; 181(1): 104–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Petersen EA, Stauss TG, Scowcroft JA, et al. Effect of High-frequency (10-kHz) Spinal Cord Stimulation in Patients With Painful Diabetic Neuropathy: A Randomized Clinical Trial. JAMA neurology 2021; 78(6): 687–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Callaghan BC, Price RS, Feldman EL. Distal Symmetric Polyneuropathy in 2020. Jama 2020; 324(1): 90–1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Callaghan BC, Reynolds E, Banerjee M, Kerber KA, Skolarus LE, Burke JF. Longitudinal pattern of pain medication utilization in peripheral neuropathy patients. Pain 2019; 160(3): 592–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hoffman EM, Watson JC, St Sauver J, Staff NP, Klein CJ. Association of Long-term Opioid Therapy With Functional Status, Adverse Outcomes, and Mortality Among Patients With Polyneuropathy. JAMA neurology 2017; 74(7): 773–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Xie J, Strauss VY, Martinez-Laguna D, et al. Association of Tramadol vs Codeine Prescription Dispensation With Mortality and Other Adverse Clinical Outcomes. Jama 2021; 326(15): 1504–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Dowell D, Haegerich TM, Chou R. CDC Guideline for Prescribing Opioids for Chronic Pain--United States, 2016. Jama 2016; 315(15): 1624–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Marshall A, Alam U, Themistocleous A, Calcutt N, Marshall A. Novel and Emerging Electrophysiological Biomarkers of Diabetic Neuropathy and Painful Diabetic Neuropathy. Clin Ther 2021; 43(9): 1441–56. [DOI] [PubMed] [Google Scholar]
- 76.Baron R, Maier C, Attal N, et al. Peripheral neuropathic pain: a mechanism-related organizing principle based on sensory profiles. Pain 2017; 158(2): 261–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Todorovic MS, Frey K, Swarm RA, et al. Prediction of Individual Analgesic Response to Intravenous Lidocaine in Painful Diabetic Peripheral Neuropathy: A Randomized, Placebo-controlled, Cross-over Trial. Clin J Pain 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Sajic M, Rumora AE, Kanhai AA, et al. High Dietary Fat Consumption Impairs Axonal Mitochondrial Function In Vivo. J Neurosci 2021; 41(19): 4321–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Philips T, Rothstein JD. Oligodendroglia: metabolic supporters of neurons. J Clin Invest 2017; 127(9): 3271–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Babetto E, Wong KM, Beirowski B. A glycolytic shift in Schwann cells supports injured axons. Nat Neurosci 2020; 23(10): 1215–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Jha MK, Morrison BM. Lactate Transporters Mediate Glia-Neuron Metabolic Crosstalk in Homeostasis and Disease. Front Cell Neurosci 2020; 14: 589582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Yorek M Treatment for diabetic peripheral neuropathy: What have we learned from animal models? Curr Diabetes Rev 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Feldman EL, Nave KA, Jensen TS, Bennett DLH. New Horizons in Diabetic Neuropathy: Mechanisms, Bioenergetics, and Pain. Neuron 2017; 93(6): 1296–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Eftekharpour E, Fernyhough P. Oxidative stress and mitochondrial dysfunction associated with peripheral neuropathy in type 1 diabetes. Antioxid Redox Signal 2021. [DOI] [PubMed] [Google Scholar]
- 85.Liu YP, Shao SJ, Guo HD. Schwann cells apoptosis is induced by high glucose in diabetic peripheral neuropathy. Life Sci 2020; 248: 117459. [DOI] [PubMed] [Google Scholar]
- 86.Sifuentes-Franco S, Pacheco-Moises FP, Rodriguez-Carrizalez AD, Miranda-Diaz AG. The Role of Oxidative Stress, Mitochondrial Function, and Autophagy in Diabetic Polyneuropathy. J Diabetes Res 2017; 2017: 1673081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Singh JN, Jain G, Sharma SS. In vitro hyperglycemia enhances sodium currents in dorsal root ganglion neurons: an effect attenuated by carbamazepine. Neuroscience 2013; 232: 64–73. [DOI] [PubMed] [Google Scholar]
- 88.Rumora AE, LoGrasso G, Hayes JM, et al. The Divergent Roles of Dietary Saturated and Monounsaturated Fatty Acids on Nerve Function in Murine Models of Obesity. J Neurosci 2019; 39(19): 3770–81. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Rumora AE, Guo K, Alakwaa FM, et al. Plasma lipid metabolites associate with diabetic polyneuropathy in a cohort with type 2 diabetes. Annals of Clinical and Translational Neurology 2021; n/a(n/a). [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Guo K, Savelieff MG, Rumora AE, et al. Plasma Metabolomics and Lipidomics Differentiate Obese Individuals by Peripheral Neuropathy Status. The Journal of Clinical Endocrinology & Metabolism 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Stith JL, Velazquez FN, Obeid LM. Advances in determining signaling mechanisms of ceramide and role in disease. J Lipid Res 2019; 60(5): 913–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wang L, Chopp M, Szalad A, et al. Exosomes Derived From Schwann Cells Ameliorate Peripheral Neuropathy in Type 2 Diabetic Mice. Diabetes 2020; 69(4): 749–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Grasset E, Burcelin R. The gut microbiota to the brain axis in the metabolic control. Rev Endocr Metab Disord 2019; 20(4): 427–38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Bennett DL, Clark AJ, Huang J, Waxman SG, Dib-Hajj SD. The Role of Voltage-Gated Sodium Channels in Pain Signaling. Physiol Rev 2019; 99(2): 1079–151. [DOI] [PubMed] [Google Scholar]
- 95.Djouhri L, Zeidan A, Abd El-Aleem SA, Smith T. Cutaneous Aβ-Non-nociceptive, but Not C-Nociceptive, Dorsal Root Ganglion Neurons Exhibit Spontaneous Activity in the Streptozotocin Rat Model of Painful Diabetic Neuropathy in vivo. Front Neurosci 2020; 14: 530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Düll MM, Riegel K, Tappenbeck J, et al. Methylglyoxal causes pain and hyperalgesia in human through C-fiber activation. Pain 2019; 160(11): 2497–507. [DOI] [PubMed] [Google Scholar]
- 97.Tsantoulas C, Lainez S, Wong S, Mehta I, Vilar B, McNaughton PA. Hyperpolarization-activated cyclic nucleotide-gated 2 (HCN2) ion channels drive pain in mouse models of diabetic neuropathy. Sci Transl Med 2017; 9(409): eaam6072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Bernard Healey SA, Scholtes I, Abrahams M, McNaughton PA, Menon DK, Lee MC. Role of hyperpolarization-activated cyclic nucleotide-gated ion channels in neuropathic pain: a proof-of-concept study of ivabradine in patients with chronic peripheral neuropathic pain. Pain Rep 2021; 6(4): e967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Veluchamy A, Hébert HL, van Zuydam NR, et al. Association of Genetic Variant at Chromosome 12q23.1 With Neuropathic Pain Susceptibility. JAMA Netw Open 2021; 4(12): e2136560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Pascal MMV, Themistocleous AC, Baron R, et al. DOLORisk: study protocol for a multi-centre observational study to understand the risk factors and determinants of neuropathic pain. Wellcome Open Res 2018; 3: 63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Bäckryd E, Themistocleous A, Larsson A, et al. Hepatocyte growth factor, colony-stimulating factor 1, CD40, and 11 other inflammation-related proteins are associated with pain in diabetic neuropathy: exploration and replication serum data from the Pain in Neuropathy Study. Pain 2021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Jayaraj ND, Bhattacharyya BJ, Belmadani AA, et al. Reducing CXCR4-mediated nociceptor hyperexcitability reverses painful diabetic neuropathy. J Clin Invest 2018; 128(6): 2205–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Marshall AG, Lee-Kubli C, Azmi S, et al. Spinal Disinhibition in Experimental and Clinical Painful Diabetic Neuropathy. Diabetes 2017; 66(5): 1380–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Segerdahl AR, Themistocleous AC, Fido D, Bennett DL, Tracey I. A brain-based pain facilitation mechanism contributes to painful diabetic polyneuropathy. Brain 2018; 141(2): 357–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Zhang X, Xia L, Xie A, Liao O, Ju F, Zhou Y. Low concentration of Bupivacaine ameliorates painful diabetic neuropathy by mediating miR-23a/PDE4B axis in microglia. Eur J Pharmacol 2021; 891: 173719. [DOI] [PubMed] [Google Scholar]
- 106.Selvarajah D, Wilkinson ID, Fang F, et al. Structural and Functional Abnormalities of the Primary Somatosensory Cortex in Diabetic Peripheral Neuropathy: A Multimodal MRI Study. Diabetes 2019; 68(4): 796–806. [DOI] [PubMed] [Google Scholar]
- 107.Wilkinson ID, Teh K, Heiberg-Gibbons F, et al. Determinants of Treatment Response in Painful Diabetic Peripheral Neuropathy: A Combined Deep Sensory Phenotyping and Multimodal Brain MRI Study. Diabetes 2020; 69(8): 1804–14. [DOI] [PubMed] [Google Scholar]
- 108.Colloca L, Ludman T, Bouhassira D, et al. Neuropathic pain. Nat Rev Dis Primers 2017; 3: 17002. [DOI] [PMC free article] [PubMed] [Google Scholar]


