Abstract
Spontaneous thought is an adaptive cognitive process that can produce novel and insightful thought sequences useful in guiding future behavior. In many psychiatric disorders, spontaneous thinking becomes intrusive and uncontrolled, and can trigger symptoms such as craving, repetitive negative thinking and trauma-related memories. We link studies using clinical imaging and rodent modeling towards understanding the neurocircuitry and neuroplasticity of intrusive thinking. We propose a framework in which drugs or stress change the homeostatic set point of brain reward circuitry, which then impacts subsequent plasticity induced by drug/stress conditioned cues (metaplastic allostasis). We further argue for the importance of examining not only the canonical pre- and postsynapse, but also the adjacent astroglial protrusions and extracellular matrix that together form the tetrapartite synapse and that plasticity throughout the tetrapartite synapse is necessary for cue-induced drug or stress behaviors. This analysis reveals that drug use or trauma cause long-lasting allostatic brain plasticity that sets the stage for subsequent drug/trauma-associated cues to induce transient plasticity that can lead to intrusive thinking.
Keywords: intrusive thinking, PTSD, substance use disorder, resting state MRI, brain imaging, circuitry, tetrapartite synapse, astroglia, extracellular matrix
Introduction to Spontaneous and Intrusive Thinking
--Thoughts meander like a restless wind inside a letter box, they tumble blindly as they make their way across the universe— Lennon/McCartney (1969)
Our stream of consciousness is often punctuated with spontaneous thoughts. Spontaneous thinking occupies 30-50% of our mental activity (Killingsworth and Gilbert, 2010) and is defined as “thought that unfolds effortlessly and unrestrictedly … including concepts such as mind wandering, day-dreaming, creativity and dreaming” (Mildner and Tamir, 2019). Spontaneous thoughts are frequently associated with recent semantic and episodic memories. However, rather than simply replaying recent memories, the spontaneous stream of thoughts often makes seemingly random associations between memories. The relatively unconstrained associations made, along with replaying recent memories, is hypothesized to promote memory consolidation of the most salient and common features of our conscious experiences (Wamsley, 2019). Thus, spontaneous thought is a normal, adaptive cognitive process that facilitates memory consolidation, and can produce novel and insightful thought sequences potentially useful in guiding future behavior.
In addition to being influenced by recent experiences and associated memories (Song and Wang, 2012), spontaneous thoughts are state-dependent, being shaped not only by preceding associations, but by our emotional and physical state, as well as the state of the world around us (Mildner and Tamir, 2019; Poerio et al., 2013). State dependence imposes constraints by linking the spontaneous exploration of memory associations to interoceptive and external stimuli, thereby increasing the probability that spontaneous thinking will be relevant to our changing internal and external world. But what happens to state-dependent limiting of spontaneous thinking if a person’s internal state is impaired by psychopathology?
In many neuropsychiatric disorders, the state-dependent shaping of spontaneous thinking leads to thoughts becoming repetitive and difficult to control. This category of spontaneous thinking is referred to as intrusive thinking and is defined as “any distinct, identifiable cognitive event that is unwanted, unintended, and recurrent. It interrupts the flow of thought, interferes in task performance, is associated with negative affect, and is difficult to control” (Clark and O'Connor, 2005). By this definition, intrusive thinking is a maladaptive, potentially pathological form of spontaneous thought that is a transdiagnostic symptom across many psychiatric disorders (Brewer et al., 2020; Kalivas and Kalivas, 2016; May et al., 2015; Watkins, 2008). For example, the impaired capacity to constrain spontaneous thinking can initiate cardinal psychiatric symptoms, such as craving-induced relapse in substance use disorders (SUDs), rumination-induced anhedonia in major depression (MD), trauma memory-induced anxiety in post-traumatic stress disorder (PTSD), or emotionally destabilizing repetitive behaviors in obsessive-compulsive disorder (OCD) (Brewer et al., 2020; Kalivas and Kalivas, 2016). The fact that spontaneous thoughts can become intrusive and initiate psychopathology in vulnerable individuals suggests that deeper neurobiological knowledge of intrusive thinking as a transdiagnostic symptom will also contribute to understanding the substantial comorbidity between disorders such as SUDs, PTSD and MD (Peskin et al., 2018; Sofuoglu et al., 2014; Suh and Ressler, 2018).
Unfortunately, spontaneous and intrusive thinking in humans is largely declarative (Mildner and Tamir, 2019; Smallwood and Schooler, 2015), and as such cannot be directly modeled in experimental organisms where neurobiological origins can be more readily studied. However, we can back-translate from human imaging studies of intrusive thinking into established animal models for investigating symptoms of psychiatric disorders, such as models of cue-induced drug seeking (SUDs) and anxiety (PTSD) (Gourley et al., 2020). Through this iterative translation, brain circuits and cellular dysregulations are emerging that provide insight into the neurobiology of intrusive thinking and how repetitive intrusions can initiate and maintain psychiatric disorders. Here, we explore the neurocircuitry of spontaneous thinking through human brain imaging studies and hypothesize how this circuitry elicits intrusive thinking. We then examine models that use an animal’s memories to evoke drug seeking or behaviors thought to convey stress or fear and explore the neurocircuitry and cellular neuroplasticity that underpin drug seeking and stress/threat reactivity. Lastly, we explore how therapeutics may be informed by understanding the neurocircuitry and neuroplasticity of intrusive thinking.
Neurocircuitry Underpinning Spontaneous and Deliberate Thinking in Human Imaging
Medial Temporal Lobe and the Default Mode Network:
Spontaneous thinking, like other memory consolidation and recall functions, relies on the medial temporal lobe (MTL) for guiding memory associations; including, entorhinal and parahippocampal cortices and the hippocampus (Figure 1A)(Christoff et al., 2016; McGaugh, 2000). This process is relatively unconstrained in day-dreaming and mind wandering when spontaneous thought transitions occur at a rate of ~4.3 transitions per minute (Tseng and Poppenk, 2020). However, even at its most unconstrained, spontaneous thinking relies in part on the content of the preceding recalled memories. Thus, in the absence of conscious attention during sleep, spontaneous transitions are most frequently to topics related to the immediate previous thought (Bar et al., 2007). From human imaging studies measuring resting state circuit connectivity (rs-fMRI), the MTL is part of the default mode network (DMN) (Figure 1).
Figure 1. Diagram illustrating the differences between adaptive transitions from spontaneous to deliberate thinking versus transitioning from spontaneous to intrusive thinking.
A) Overall organization of key brain areas into the default mode (DMN), salience mode (SN) and executive mode (ECN) networks. Note that the SN has substantial overlap with the DMN and ECN. B) Illustration of how increasing motivation and attention converts unrestrained spontaneous thoughts into constrained deliberate thinking through a hierarchical dominance of ECN connectivity over SN and DMN. C) In some psychiatric disorders, the ECN-dominant hierarchy is disrupted and the transition from spontaneous thinking is dominated by the SN, resulting in intrusive thinking. SN dominance over ECN is indicated by a more bold SN and a smaller, dashed ECN. MTL- medial temporal lobe, PFC- prefrontal cortex, VTA- ventral tegmental area
Dorsolateral Prefrontal Cortex (dlPFC) – Parietal Attention Circuit and the Executive Control Network:
The extent to which spontaneous thoughts transition closer or further away from the content of a preceding thought is state dependent (Mildner and Tamir, 2019; Poerio et al., 2013). State dependence is strongly modulated by the motivational relevance of spontaneous thoughts and environmental stimuli. Sufficient motivational relevance engages the frontal cortex, including the dlPFC, and parietal cortex circuitry underpinning arousal and attention, which are considered key components of the executive control network (ECN) in human fMRI studies (Figure 1A). Increasing ECN connectivity constrains DMN-dependent spontaneous thought transitions (Christoff et al., 2016; Mildner and Tamir, 2019), resulting in top-down control over spontaneous thinking that favors ECN-guided deliberate memory foraging, planning and initiating adaptive behaviors. The spectrum of spontaneous thinking ranging from unconstrained DMN-guided to increasing restraint by ECN attentional processing is illustrated in Figure 1B.
Limbic PFC and Salience Network:
Increased ECN attentional processing often results from encountering spontaneous thoughts or external stimuli that contain motivationally relevant information. Thus, constraining spontaneous thinking often arises from parallel increases in the ECN and limbic cortical regions, containing anterior cingulate, orbital, subgenual anterior cingulate and insular cortices, as well as subcortical regions such as the amygdala, nucleus accumbens and brainstem nuclei whose connectivity may differ depending on whether the motivational salience of the stimulus has positive (rewarding) or negative (stressful) valence (Christoff et al., 2016; Haber and Knutson, 2010; Herman, 2012). Connectivity between these brain areas is referred to as the salience network (SN) in human neuroimaging literature (Borsook et al., 2013; Pariyadath et al., 2016). Regardless of the valence of the thoughts or environmental stimuli initiating SN connectivity, if ECN circuitry is recruited, both SN and DMN connectivity are inhibited to constrain spontaneous thought transitions and produce more deliberate thinking (Figure 1B). In this way, SN activity can initiate but does not guide ECN-dependent (top-down) deliberate thinking involved in planning and executing adaptive behavioral responses to salient environmental stimuli.
In summary, the arrangement between unconstrained spontaneous thinking to uncover motivationally relevant information and transitioning to top-down deliberate thinking increases the probability of developing a plan of action to manage important stimuli towards adaptive outcomes. How might the adaptive emergence of deliberate thinking be subverted into intrusive thinking?
Dysregulated Circuit Network Transitions Cause Intrusive Thinking:
In persons with underlying psychiatric vulnerability, perceived threat- or reward-induced constraint of spontaneous thought can lead to repetitive intrusive thinking rather than adaptive deliberate thinking. Examples of environmental stimuli initiating intrusive thinking in different psychiatric disorders include craving induced by drug cues (Kalivas and Kalivas, 2016; Renaud et al., 2021), rumination initiation and maintenance of depression (Watkins and Nolen-Hoeksema, 2014) or rumination and re-experiencing a traumatic event in PTSD (Moulds et al., 2020; Wisco et al., 2023). In a state of intrusive thinking, the person repetitively and unproductively explores the same or closely related memories and potentially improbable or maladaptive future outcomes (Clark and O'Connor, 2005). Figure 1C illustrates impaired transitioning of motivated spontaneous thinking to ECN-guided deliberate thinking, which we propose can arise from two circuitry malfunctions that are not mutually exclusive. 1) Intrusive thinking can occur if the motivational content of a stimulus is too great to be managed by the ECN, such as hearing of the death of a loved one or being in a life-threatening situation (Visser et al., 2020). In healthy individuals, the situational emergence of intrusive thinking is temporary, and terminated by entering a problem-solving ECN-guided thought format (Lu et al., 2023; Lu et al., 2022), or perhaps by engaging in unrelated behaviors that compete with the repetitive thought intrusions, such as exercise or socializing (Carroll, 2021; Venniro et al., 2019). 2) Intrusive thinking can result from a failure of ECN to regulate SN- and DMN-guided spontaneous thinking (Donofry et al., 2020). Human imaging studies cannot readily distinguish between these two cognitive impairments because some brain areas contribute to multiple networks (Figure 1A). For example, fMRI analysis of SUD patients in a drug cue reactivity test show excessive activation of limbic cortex, but also impaired activation of inhibitory ECN circuitry (Feil et al., 2010). Moreover, rumination in major depression is associated with suppressed ECN (dlPFC in particular) activation but activation in DMS core regions (Zhou et al., 2020). Regardless of the relative impairments in SN, DMN or ECN or both, intrusive thinking resulting from uncoupling of the ECN from the SN and DMN occurs during intrusive thinking in many neuropsychiatric disorders (Fedota and Stein, 2020), such as craving in SUDs (Feil et al., 2010; Zilverstand et al., 2016), gambling urges (Moccia et al., 2017), anxiety in PTSD (Malejko et al., 2017; Steward et al., 2020) or rumination in MD (Manning et al., 2019).
Conclusions:
Intrusive thinking is an unproductive state of highly constrained and motivated repetitive thought transitions. We hypothesize that this state arises most often from a combination of excessive activity in the SN and DMN and reduced or dysfunctional hyperactivity in the ECN. Thus, instead of increasing attention and motivation transitioning unconstrained spontaneous thought into ECN guided deliberate thinking (Figure 1B), thought transitions are SN guided, emotionally laden and can be maladaptive in generating correct behavioral responses. We argue that this cognitive impairment in thought transitioning is a symptom of many psychiatric disorders and in some, such as PTSD and SUDs, can be responsible for initiating cardinal symptoms of the psychopathology, such as intense anxiety (terror) or drug relapse, respectively. However, intrusive thinking should be considered only one of multiple factors that can precipitate these key symptoms of psychopathology.
Animal Models of Intrusive Thinking
Using nonhuman animals to model symptoms of psychiatric disorders is useful to explore detailed neurocircuitry and molecular mechanisms. Unfortunately, engagement of the ECN to transition spontaneous thinking into deliberate thinking in humans is a declarative cognitive process that cannot be directly modeled in rodents. However, using salient stimuli to engage learned behaviors reflective of human psychopathology may provide insight into the circuitry and molecular plasticity of intrusive thinking. Supporting this experimental perspective for reverse translating from human to animal studies is a recent study using a combination of the Think/No-Think task and a subsequent stop signal task, which found that preventing a thought from coming to mind recruits fast prefrontal stopping, similar to the electrophysiological signature of action-stopping in the stop-signal task. This finding indicates that the same neural mechanisms underlie both thought suppression and action-stopping (Castiglione et al., 2019). Two more classic examples are animal models of SUDs, in which the pathogenic environmental insult is repeated use of addictive drug(s), and models of PTSD, in which animals are exposed to a single or repeated uncontrollable stressor(s). In addition to examining how the drugs or stressors change brain function, discrete cues and contexts can be paired with drug infusion or stress exposure to initiate drug seeking or stress coping behaviors (Deslauriers et al., 2018; Kuhn et al., 2019). We propose that the changes in brain function produced by such conditioned stimuli in model organisms may reflect thought intrusions in SUDs or PTSD. This phenomenon is especially well studied in operant models of drug use, in which operant seeking of a natural reward, such as sucrose or social interaction, can be used as comparative non-pathological form of brain plasticity (Nall et al., 2021; Venniro et al., 2020).
Shared Neurocircuitry of Cue-induced Drug Seeking and Stress Coping:
Following up on the hypothesis laid out above that intrusive thinking emerges from an imbalance between SN and ECN, we focus on key nuclei in these networks known to regulate motivated drug seeking and stress coping in rodents. Figure 2 illustrates a circuit comprised of brain nuclei shared by both the SN and ECN, where adaptations could contribute to the imbalance between SN and ECN underlying intrusive thinking, including the medial PFC (containing the prelimbic [PL] and infralimbic [IL] cortex), basolateral amygdala (BLA) and nucleus accumbens. Within this circuit, projections are glutamatergic, though the output from the nucleus accumbens critical to initiating motivated behavior (Root et al., 2015) is GABAergic and peptidergic (Koob and Volkow, 2016; Smith et al., 2013). Not shown in Figure 2 is circuitry containing other corticolimbic regions that interface with this circuit, including the orbital and insular cortices, and brainstem limbic structures that monosynaptically innervate these regions, such as dopaminergic neurons of the ventral tegmental area (VTA) and noradrenergic neurons of the locus coeruleus (LC) (Koob and Volkow, 2010). While substantial preclinical and translational research on cue-induced drug seeking or stress reactivity involves brain nuclei not shown in Figure 2 (Gourley and Taylor, 2016; Hanlon et al., 2017; Jasinska et al., 2014; Wichary and Smolen, 2016), understanding synaptic plasticity associated with conditioned cues is relatively advanced at cortical and allocortical (e.g., hippocampus and amygdala) glutamatergic synapses in the nucleus accumbens, on which we will focus.
Figure 2. Illustration of key brain circuitry at the SN – ECN interface.
This drawing is based on the rat atlas by Paxinos and Watson (Paxinos and Watson, 2007), and shows projections between the PFC (green), basolateral amygdala (BLA; red) and core and shell of the nucleus accumbens (NAcore and NAshell, respectively). Arrows are color coded according to PFC or BLA outputs and double headed arrows indicate reciprocal projections between two regions. ac- anterior commissure, cc- corpus callosum, CnA- central nucleus of the amygdala, ic- internal capsule, Pir- pyriform cortex
Many studies have inactivated the PFC or amygdala inputs to the nucleus accumbens to reveal necessity of the circuit in cue-induced drug seeking and cued stress responding (Do Monte et al., 2016; Luscher and Janak, 2021), with a focus on activity and plasticity in accumbens dopamine D1-receptor expressing medium spiny neurons (D1-MSNs) as critical for cued drug seeking (Garcia-Keller et al., 2020a; Lobo and Nestler, 2011; Pardo-Garcia et al., 2019). Importantly, there is a subcircuit within the circuit in Figure 2, in which neuronal activity is necessary for drug seeking, but not for seeking natural rewards, such as sucrose (Nall et al., 2021). Specifically, pharmacological or genetic inhibition of the BLA and shell subcompartment of the accumbens (NAshell) reduces cued seeking for both drug and natural reward (Guercio et al., 2015; Millan et al., 2017), while inhibition of the subcircuit containing projections from the PL to the core of the accumbens (Nacore) reduces cued seeking for addictive drugs but not sucrose (McFarland and Kalivas, 2001; McGlinchey et al., 2016). These data mark the glutamatergic PL – Nacore projection as a site where drug-induced pathological plasticity may exist to create the prepotent cue-reactivity of drug versus biological reward seeking in rodents (Bobadilla et al., 2020) and intrusive thinking (craving) in SUDs (Antons et al., 2020).
Animal models have not brought as much focus to stress-induced adaptations in the PL – Nacore circuit (Gourley and Taylor, 2016; Liberzon and Abelson, 2016; Selemon et al., 2019). However, stress exposure and stress-associated cues potentiate the pharmacological effects and the use of addictive drugs in animal models (Mantsch et al., 2016). Akin to drug cues that recruit PL – Nacore projections in drug seeking, presenting stress cues induces seeking in rodents for cocaine, heroin and alcohol, but not sucrose (Garcia-Keller et al., 2020b; Mantsch et al., 2016). Also, as outlined in detail below, stress exposure elicits enduring changes at glutamatergic synapses in the Nacore that are similar to changes produced by using many addictive drugs (Garcia-Keller et al., 2016), and stress and cocaine both trigger dendritic spine attrition on PL projection neurons (McEwen and Morrison, 2013; Radley et al., 2015). Moreover, both stress and cocaine cause organisms to favor familiar behaviors over novel, more adaptive behaviors (DePoy et al., 2017; Dias-Ferreira et al., 2009; Miles et al., 2003; Schoenbaum and Setlow, 2005) – a phenomenon that has been likened to diminished control over intrusions in neuropsychiatric illness (Visser et al., 2020). The overlap in circuit-, neuron- and synaptic-level plasticity following stress and addictive drugs is of clinical significance, given the high incidence of PTSD and SUDs comorbidity, and the observation that PTSD-associated cues induce drug craving and increase vulnerability to relapse (Back et al., 2020; Suh and Ressler, 2018). For these reasons, we will explore the contribution of PL – Nacore projections in stress or drug cue initiation of synaptic plasticity contributing to drug seeking or stress coping behaviors as a surrogate measure of intrusive thinking in SUDs and PTSD.
Enduring and transient synaptic plasticity:
When considering the long-lasting brain adaptations produced by stress and drug use, concepts of metaplasticity and allostasis are useful (Figure 3A). Metaplasticity was defined in 1996 by Abraham and Bear as plasticity produced by environmental stimuli that changes the neuroplasticity produced by subsequent stimuli (Abraham, 2008; Abraham and Bear, 1996). In 2001, Koob and Le Moal provided a seminal description of SUDs as a state of allostasis resulting from underlying changes in the homeostatic set point in brain reward and stress circuitry shown in Figure 2, thereby making the user prone to relapse (Koob and Le Moal, 2001, 2008). Linking these concepts in disorders such as SUDs and PTSD, the allostatic state of psychopathology contains a vulnerability scar arising from underlying enduring metaplasticity produced by drug use or trauma (Figure 3A)(Nestler and Luscher, 2019). Correspondingly, drug or stress cue-induced intrusive thinking is reflected by additional transient neuroplasticity that requires the drug/trauma-induced metaplasticity (Mulholland et al., 2016). Thus, allostatic metaplasticity is a change in steady-state brain function that is induced by drug use or trauma but that may not actively manifest behavioral symptoms in the absence of environmental triggers. However, metaplasticity sets the stage for transient neuroplasticity to be triggered by drug- or trauma-associated cues that initiates intrusive thinking and can precipitate other SUDs and PTSD symptoms, such as relapse and anxiety.
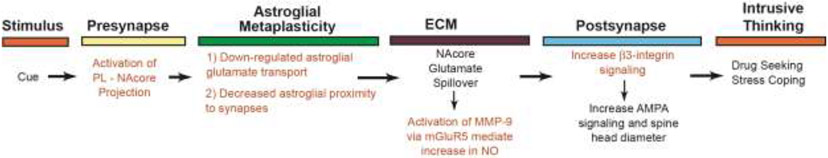
Figure 3. The enduring metaplasticity and transient synaptic plasticity possibly underpinning intrusive thinking.
A) Addictive drug or stress exposure converts homeostatic plasticity into enduring allostatic metaplasticity, characterized after cocaine or stress as long-term potentiation (LTP) and after opioids as long-term depression (LTD). A subsequent drug or stress associated cue produces intrusive thinking (craving/rumination) and additional transient plasticity. The intrusive thinking terminates for example by actual drug use (relapse) and the allostatic condition of metaplasticity-induced vulnerability to intrusive thinking is restored. B) An illustration of a glutamatergic tetrapartite synapse in the NAcore during cue-induced sucrose seeking where astroglial metaplasticity is not induced by sucrose self-administration. Note the intact astroglial morphology and glutamate uptake via GLT-1. C) The state of metaplasticity after drug use or stress is characterized by astroglial down regulation of GLT-1 and withdrawal of the astroglia protrusions from synaptic proximity. D) The transient plasticity induced by a drug or stress cue that is necessary for cues to trigger drug, but not sucrose seeking. Glutamate spillover due to astroglial metaplasticity indirectly activates MMP-9 (see Figure 4), which catalytically liberates β3-integrin ligands from the ECM. Increased signaling through β3-integrin promotes spine head expansion and increased AMPA receptor signaling. In parallel, the astroglial actin binding protein ezrin is phosphorylated, causing a transient return of the astroglial protrusion to the synapse. FAK- focal adhesion kinase, GLT-1- astroglia-selective glutamate transporter, MMP- matrix metalloprotease (blue inactive, red active), RGD- arginine-glycine-aspartate
Importantly, metaplastic allostasis is not induced from homeostatic behaviors such as operant sucrose self-administration, and correspondingly there is no measurable sucrose cue-induced transient plasticity (Figure 3B)(Bobadilla et al., 2017; Kruyer et al., 2020; Scofield et al., 2016). In contrast, glutamatergic synapses in the nucleus accumbens undergo both metaplasticity that endures for weeks or months after discontinuing drug use or stress exposure (Figure 3C)(Centanni et al., 2019; Koob and Volkow, 2016; Nestler and Luscher, 2019), and transient synaptic plasticity following cues predicting drug availability or stress (Figure 3D)(Scofield et al., 2016). We focus almost entirely on metaplasticity and transient synaptic plasticity in the Nacore, which is arguably the most experimentally advanced as a model system in SUDs. However, it is important to note that complementary studies exist for other brain nuclei within the circuitry in Figure 2 (Nestler and Luscher, 2019; Suh and Ressler, 2018).
Tetrapartite synapses in drug- and stress-induced plasticity:
It has become clear over the last decade that to understand metaplasticity in stress and drug-induced neuroadaptations and transient cue-induced plasticity, the canonical two compartment synapse (pre- and postsynapse) is not fully adequate (Fouyssac and Belin, 2019; Kruyer et al., 2020; Wang et al., 2021a). It is necessary to also understand plasticity in the two other domains of the tetrapartite synapse, including perisynaptic astroglial processes (PAPs) and the extracellular proteinaceous matrix (ECM) within and surrounding the synaptic cleft (Kruyer et al., 2020; Kruyer and Kalivas, 2020; Liu et al., 2017; Murphy-Royal et al., 2020; Wang et al., 2021b). Table 1 and Figure 3 illustrate that exposure to addictive drugs or stress impacts all four compartments as part of enduring metaplasticity and cue-induced transient plasticity. Experimental details of tetrapartite plasticity shown in Figure 3 and Table 1 are reviewed elsewhere (Hodebourg et al., 2021; Mulholland et al., 2016). Here we highlight the metaplasticity and cue-induced transient plasticity that are consistent across drug classes and stress as the most relevant candidates for contributing to the cue-induced behavioral responses that we hypothesize reflect intrusive thinking in humans with SUDs or PTSD.
Table 1.
Tetrapartite enduring and transient synaptic adaptations in the nucleus accumbens after drugs, stress and cues.
| Enduring Metaplasticity | Transient Plasticity | ||||||
|---|---|---|---|---|---|---|---|
| Compartment | Measure | Stress | Cocaine | Opioids | Stress | Cocaine | Opioids |
| Neuronal | A:N | + | + (D1) | + (D2) | ? | + (D1) | + |
| dh | + | + (D1) | − | + | + (D1) | + (D1) | |
| Astroglial | PAPs | − | − | + (D1), − (D2) | + | ? | − (D1), + (D2) |
| GLT-1 | − | − | − | ? | ? | + (D1), + (D2) | |
| ECM | MMP-2,9 | + | + | + (D2) | + (D1) | + | + (D1), − (D2) |
indicates increase in function or PAP proximity to synapses; − indicates decreased function or proximity; ? indicates no data. A:N- AMPA:NMDA ratio, dh- dendritic spine head diameter, PAPs- perisynaptic astroglial processes. Experimental details are provided elsewhere (Hodebourg et al., 2021; Kruyer et al., 2022a; Mulholland et al., 2016).
Enduring tetrapartite metaplasticity:
Researchers have brought greatest experimental focus to drug- and stress-induced metaplasticity in the postsynaptic subcompartment of neurons in the Nacore. However, the precise nature of the metaplastic changes often depends on drug class (Figure 3A). For example, the metaplastic adaptations produced by stress and psychostimulants in the postsynapse are similar, with most studies showing long-term potentiation (LTP)-like changes, such as increased AMPA glutamate receptor signaling, dendritic spine density and/or spine head diameter (Conrad et al., 2008; Garcia-Keller et al., 2016; Gipson et al., 2013; Wang et al., 2021c), and potentiated neurotrophic factor expression and signaling (Koo et al., 2019; Li and Wolf, 2015; McGrath and Briand, 2019). However, opioids and cannabis elicit decreases or no change in AMPA receptor signaling and spine density/head diameter (Figure 3A)(Shen et al., 2011; Spencer et al., 2018).
An additional form of postsynaptic metaplasticity induced by addictive drugs is the induction of silent synapses that contain NMDA but not AMPA glutamate receptors on nucleus accumbens medium spiny neurons (MSNs) (Dong et al., 2017; Huang et al., 2015; Ma et al., 2014). These silent synapses can either mature to become functional excitatory synapses or are eliminated during drug withdrawal. Interestingly, while psychostimulants increase pre- and postsynaptic strength in the D1-receptor expressing neuron subtype (D1-MSNs), opioids reduce synaptic strength in D2-MSNs through differential unsilencing or elimination of silent synapses on each MSN subtype (Graziane et al., 2016; Hearing et al., 2018). Thus, both drugs share metaplastic increases in the strength of glutamatergic inputs to D1- relative to D2-MSNs. Importantly, the formation of silent synapses in the nucleus accumbens after addictive drugs is regulated by cocaine-induced astroglial release of synaptogenic molecules, such as thrombospondin (Wang et al., 2021b), pointing to a role for astroglial tetrasynaptic compartment in drug-induced metaplastic allostasis.
As far as has been studied, the astroglial compartment of the tetrapartite synapse contains adaptations that are often similar across drug use and acute stress (Figure 3C). Specifically, weeks following cocaine or heroin self-administration or a single exposure to restraint stress, astroglia show marked down-regulation of glutamate transporters (Garcia-Keller et al., 2016; Knackstedt et al., 2010; Shen et al., 2014) that are positioned on PAPs adjacent to the synapse to efficiently eliminate extracellular glutamate (Murphy-Royal et al., 2017; Robinson and Jackson, 2016). Chronic stress also causes a durable loss of striatal transporter content (Rappeneau et al., 2016; Shimamoto et al., 2018). Moreover, many astroglia decrease their morphological proximity to nAcore synapses, thereby exacerbating the impact of down-regulated glutamate transport by pulling remaining transporters further from the synapse (Fischer-Smith et al., 2012; Garcia-Keller et al., 2021). Importantly, measurable metaplasticity is not produced in any of the nAcore tetrapartite synaptic compartments by operant training to self-administer sucrose (Figure 3B)(Bobadilla et al., 2017; Kruyer et al., 2020; Scofield et al., 2016). After drug/stress-induced astroglial metaplasticity, drug/stress cues elicit similar transient plasticity in all tetrapartite synaptic compartments (Table 1). The lack of effect by sucrose and the stability of astroglial metaplasticity across drug classes and stress indicates that metaplasticity in the astroglial compartment may be a critical allostatic adaptation underpinning cue-induced transient plasticity. Restoring astroglial glutamate transport prevents cue- or stress-induced drug seeking (Hodebourg et al., 2021; Mulholland et al., 2016) and inhibits intrusive thinking in SUDs and PTSD (Back et al., 2016; Duailibi et al., 2017). Moreover, many components of cue-induced transient tetrapartite synaptic plasticity are prevented by restoring astroglial glutamate transport (Garcia-Keller et al., 2016; Moussawi et al., 2011).
Despite uniformities across independent investigations, some open questions remain. For instance, self-administration of different classes of abused drug lowers GLT-1 only in some rodent lines, such as in alcohol preferring rats (Rao and Sari, 2012) or not consistently across studies, such as after methamphetamine self-administration (Siemsen et al., 2019). However, elevating astroglial glutamate transport with N-acetylcysteine or ceftriaxone generally inhibits cue-induced reinstatement of seeking these drugs (Smaga et al., 2020). This observation poses the possibility of off-target effects of these drugs and/or different consequences based on the amount or frequency of drug use (Smaga et al., 2020). Along these lines, recent studies employing polydrug use in rodent models reveal that alcohol use prevents cocaine-induced down-regulation of astroglial transporter and the synaptic spillover of glutamate normally induced in the nucleus accumbens by cocaine cues (Stennett and Knackstedt, 2020; Stennett et al., 2020). These surprising findings have important implications in clinical studies in which poly-drug use is common and pharmacological efforts to restore astroglial glutamate transporters have not proven effective at preventing relapse in most studies (see discussion below). These disparate findings also point to the multiple mechanisms whereby drug-induced astroglial metaplasticity dysregulates synaptic glutamate in the extracellular space (Hodebourg et al., 2022). For example, methamphetamine does not reduce astroglial glutamate transport in the accumbens but promotes retraction of astroglia from synaptic proximity (Siemsen et al., 2019), and would therefore be expected to promote synaptic plasticity (Henneberger et al., 2020; Kruyer et al., 2022b).
Transient cue-induced tetrapartite synaptic plasticity:
It has long been known that most drug or stress cues induce transient elevations in extracellular glutamate in the NAcore that are derived from synaptic activity in PFC afferents (LaLumiere and Kalivas, 2008; McFarland et al., 2004; McFarland et al., 2003). The enduring astroglial metaplasticity (decreased GLT-1 and astroglial ensheathing of synapses; Figure 3C) allows cue-induced release of synaptic glutamate to penetrate the adjacent neuropil outside of the synapse and stimulate extrasynaptic glutamate receptors (Shen et al., 2014). This facilitates synaptically released glutamate interaction with interneurons that are a minority of neurons in the NAcore but have individual contacts simultaneously with many MSNs, giving these neurons outsized potential to regulate NAcore output through tonic and phasic inhibition of ensembles of MSNs (Hu et al., 2014). Moreover, the microcircuit within the NAcore defined by interneuronal GABAergic regulation of MSNs and inputs from the PL and BLA (Figure 2) undergoes adaptations after cocaine use that are just now being experimentally explored (Schall et al., 2021) and will no doubt provide strong contributions to our understanding of how drug and stress induced adaptations in the NAcore dysregulate behavior.
One key extrasynaptic glutamate receptor in cue-induced transient synaptic potentiation is mGluR5 located on NAcore interneurons expressing neuronal nitric oxide synthase (nNOS) (Smith et al., 2017). Stimulating mGluR5 increases NOS, leading to activation of the ECM signaling enzyme, matrix metalloprotease-9 (MMP-9), selectively around D1-MSNs (Chioma et al., 2021; Smith et al., 2014). Activating MMP-9 digests ECM proteins, such as fibronectin that contain gelatin motifs, thereby creating ligands for β3-integrin receptors (Garcia-Keller et al., 2020a). β3-integrin binding in the postsynapse promotes both increased AMPA receptor signaling and dendritic spine head enlargement in D1-MSNs that is characteristic of drug and stress cue-induced transient postsynaptic potentiation in NAcore D1-MSNs (Garcia-Keller et al., 2021; Garcia-Keller et al., 2019; Garcia-Keller et al., 2020a). In summary (Figure 4), stress and operant drug use, but not sucrose use, produce enduring metaplasticity in NAcore astroglia that allows for drug or stress cue-induced transient postsynaptic potentiation via increasing MMP-9 signaling in the ECM. Importantly, both reversing astroglial metaplasticity by restoring glutamate transport and inhibiting any aspect of cue-induced tetrapartite transient plasticity, including inhibiting mGluR5, nNOS, MMP-9 or β3-integrin receptors, reduces cue-induced drug seeking (Hodebourg et al., 2021; Mulholland et al., 2016).
Figure 4. Sequence of neurobiological events at NAcore glutamatergic synapses contributing to the cue-induced transient plasticity that underpins cue- or stress-induced drug seeking and possibly intrusive thinking.
The drug use or stress exposure induces metaplasticity in astroglia that makes the animal prone to drug/stress, not sucrose, cue-induced seeking. On this metaplastic background, the cue induces a sequence of signaling events involving presynaptic glutamate release and spillover from the synapse. The extrasynaptic glutamate stimulates mGluR5 receptors on nNOS interneurons to promote nitric oxide (NO) synthesis. Elevated extracellular NO promotes MMP-9 activation which catalyzes the formation of ligands in the ECM for β3-integrin receptors that produces postsynaptic transient potentiation. Red lettering refers to proven targets for disrupting transient tetrapartite synaptic plasticity and preventing cue-induced drug/stress, but not sucrose seeking.
In addition to tetrapartite glutamate homeostasis undergoing marked morphological and signaling adaptions that underpin cue-induced cocaine an opioid reinstatement, the silent synapses that mature on accumbens MSNs during cocaine withdrawal also are profoundly and transiently altered by retrieval of a cocaine memory (such as drug associated contexts). Thus, presenting a cocaine associated context transiently silences the MSN excitatory synapses that had matured and become active during cocaine withdrawal (Wright et al., 2020). Furthermore, preventing silent synapse re-maturation reduces subsequent cue-induced cocaine seeking, indicating that the silencing and subsequent re-maturation of accumbens synapses may be a key component of cocaine memory reconsolidation (Wright et al., 2020).
Integrating the clinical circuitry and the preclinical plasticity associated with intrusive thinking:
We hypothesize above that intrusive thinking arises from excessive activation of the SN by environmental or interoceptive stimuli, in combination with impaired activation of the ECN. Clinical studies examining cue reactivity in SUDs and PTSD reveal marked cue-induced activation of the PFC, nucleus accumbens and amygdala, among other limbic brain nuclei, which indicates excessive activation of the SN (Fedota and Stein, 2020). Moreover, many studies reveal that symptoms of SUDs and PTSD initiated by drug- or trauma-paired cues are associated with poor recruitment of the ECN (Volkow et al., 2016). Paradoxically, a recent study indicated that higher functioning ECN may contribute to inhibiting distractors that could interfere with drug craving (Jakubiec et al., 2022). This general view is consistent with other fMRI findings that drug cue exposure increases ECN along with SN activity (Zilverstand et al., 2018; Zilverstand et al., 2016). In view of these studies, we hypothesize that dysregulated activity by the ECN, either excessive activation or hypoactivation releases the SN from constraint and guidance by the ECN, and that this is a process underlying the vulnerability of drug or stress associated cues to initiate symptoms of intrusive thinking such as craving or anxiety.
We propose that by paralleling behaviors between animal models and cardinal symptoms of SUDs and PTSD that are strongly associated with intrusive thinking, such as cue-induced drug seeking (craving) or stress-induced rumination, it is possible to identify enduring metaplasticity and transient cue-induced plasticity that may contribute vulnerability to intrusive thinking. This is especially possible for drug use, where a biological reward can be substituted for the drug to identify the consequences of adaptive reward learning and cue-initiated behaviors that may overlap with drug-associated plasticity. Through this approach and with a focus on a relevant circuit that intersects the SN and ECN, the cortical and allocortical glutamatergic projections to the accumbens, it is possible to identify enduring synaptic adaptations produced by stress and drug use (i.e. allostatic metaplasticity), as well as transient synaptic plasticity elicited by exposure to drug or stress cues, none of which occurs in rodents trained to take and seek sucrose. To discover these adaptations, it has been necessary to expand our understanding of synaptic plasticity to include not just the pre- and postsynapse but also the adjacent astroglia and ECM. Indeed, it appears that the most consequential metaplasticity shared by drug use and stress exposure is in astroglia. Moreover, the transient plasticity and seeking induced by cues requires the astroglial metaplasticity and involves all four tetrapartite synaptic compartments, at least on D1-MSNs in NAcore. Importantly, cue-induced drug seeking is reduced by inhibiting key cue-induced signaling events in any of the four synaptic compartments (Kruyer et al., 2023; Scofield et al., 2016; Wang et al., 2021b). Moreover, it seems likely that tetrapartite enduring metaplasticity and transient cue-induced plasticity occurs widely at synapses in other brain nuclei (Whyte et al., 2021).
Uncovering the circuitry and molecular plasticity associated with vulnerability to enter intrusive thinking instead of ECN-guided deliberate thinking provides potential targets for treating this transdiagnostic symptom. In as much as the animal models described herein reflect aspects of intrusive thinking in SUDs and PTSD, it appears that molecular targets have been identified that can disrupt the behaviors based on the neurobiology of enduring metaplasticity and cue-induced transient plasticity shown in Figures 3 and 4. However, the only compound that has entered clinical trials for reversing astroglial metaplasticity is N-acetylcysteine, which normalizes down-regulated astroglial glutamate transport. Unfortunately, while N-acetylcysteine consistently reduces drug cravings (Duailibi et al., 2017), it is only effective in relapse prevention in subcategories of SUDs patients, such as adolescent cannabis users and already abstinent cocaine users (Gray et al., 2012; Larowe et al., 2013). Similarly, results for N-acetylcysteine ameliorating PTSD symptoms are also mixed and may be effective only in certain subpopulations (Back et al., 2016). To evaluate the therapeutic relevance of targeting brain plasticity and intrusive thinking, future clinical trials are needed that explicitly include reducing intrusive thinking as an outcome measure and incorporate new compounds targeting the brain plasticity identified in rodent models of SUDs and PTSD.
Highlights.
Intrusive thinking is a transdiagnostic feature of many psychiatric disorders
Intrusive thinking involves dysregulated top-down control over motivational circuitry
Drugs and stress induce synaptic changes in prefrontal regulation of behavior
Drug or stress cues stress induce transient tetrapartite synaptic plasticity
Acknowledgments:
We thank Julia R. Lupp and her team at the Ernest Strungmann Institute for sponsoring the Forum titled: Intrusive Thinking: From molecules to free will, and our many scientific colleagues at the Forum for the open-ended discussions that spawned many of the ideas in this review. We thank other members of the program advisory committee, including Aikaterini Fotopoulou, Rita Z. Goldstein, Trevor W. Robbins and Peter Tse, whose insights and leadership in their respective fields resulted in the amazing roster of scientists attending the Forum.
Funding and Disclosures:
PWK is supported in part by NIH DA012513, DA0453000, DA016511, DA046373 and a VA Merit Award BX004727. SLG is supported in part by NIH DA044297. The Emory National Primate Research Center is supported by NIH OD011132. The authors declare no conflicts of interest.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- Abraham WC (2008). Metaplasticity: tuning synapses and networks for plasticity. Nat Rev Neurosci 9, 387–399. [DOI] [PubMed] [Google Scholar]
- Abraham WC, and Bear MF (1996). Metaplasticity: the plasticity of synaptic plasticity. Trends Neurosci 19, 126–130. [DOI] [PubMed] [Google Scholar]
- Antons S, Brand M, and Potenza MN (2020). Neurobiology of cue-reactivity, craving, and inhibitory control in non-substance addictive behaviors. J Neurol Sci 415, 116952. [DOI] [PubMed] [Google Scholar]
- Back SE, Gray K, Santa Ana E, Jones JL, Jarnecke AM, Joseph JE, Prisciandaro J, Killeen T, Brown DG, Taimina L, et al. (2020). N-acetylcysteine for the treatment of comorbid alcohol use disorder and posttraumatic stress disorder: Design and methodology of a randomized clinical trial. Contemp Clin Trials 91, 105961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Back SE, McCauley JL, Korte KJ, Gros DF, Leavitt V, Gray KM, Hamner MB, DeSantis SM, Malcolm R, Brady KT, et al. (2016). A Double-Blind, Randomized, Controlled Pilot Trial of N-Acetylcysteine in Veterans With Posttraumatic Stress Disorder and Substance Use Disorders. J Clin Psychiatry 77, e1439–e1446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bar M, Aminoff E, Mason M, and Fenske M (2007). The units of thought. Hippocampus 17, 420–428. [DOI] [PubMed] [Google Scholar]
- Bobadilla AC, Dereschewitz E, Vaccaro L, Heinsbroek JA, Scofield MD, and Kalivas PW (2020). Cocaine and sucrose rewards recruit different seeking ensembles in the nucleus accumbens core. Mol Psychiatry 25, 3150–3163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bobadilla AC, Garcia-Keller C, Heinsbroek JA, Scofield MD, Chareunsouk V, Monforton C, and Kalivas PW (2017). Accumbens Mechanisms for Cued Sucrose Seeking. Neuropsychopharmacology 42, 2377–2386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borsook D, Edwards R, Elman I, Becerra L, and Levine J (2013). Pain and analgesia: the value of salience circuits. Prog Neurobiol 104, 93–105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brewer J, de Wit H, Cortese A, Denys D, Hanlon CA, Paulus MP, Schwarzbach J, and Tse P (2020). Interventions and implications. In Intrusive Thinking: from molecules to free will, Kalivas PW, and Paulus MP, eds. (Cambridge, MA: The MIT Press; ), pp. 347–377. [Google Scholar]
- Carroll ME (2021). Voluntary exercise as a treatment for incubated and expanded drug craving leading to relapse to addiction: Animal models. Pharmacol Biochem Behav 208, 173210. [DOI] [PubMed] [Google Scholar]
- Castiglione A, Wagner J, Anderson M, and Aron AR (2019). Preventing a Thought from Coming to Mind Elicits Increased Right Frontal Beta Just as Stopping Action Does. Cereb Cortex 29, 2160–2172. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Centanni SW, Bedse G, Patel S, and Winder DG (2019). Driving the Downward Spiral: Alcohol-Induced Dysregulation of Extended Amygdala Circuits and Negative Affect. Alcohol Clin Exp Res. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chioma VC, Kruyer A, Bobadilla AC, Angelis A, Ellison Z, Hodebourg R, Scofield MD, and Kalivas PW (2021). Heroin Seeking and Extinction From Seeking Activate Matrix Metalloproteinases at Synapses on Distinct Subpopulations of Accumbens Cells. Biol Psychiatry 89, 947–958. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Christoff K, Irving ZC, Fox KC, Spreng RN, and Andrews-Hanna JR (2016). Mind-wandering as spontaneous thought: a dynamic framework. Nat Rev Neurosci 17, 718–731. [DOI] [PubMed] [Google Scholar]
- Clark DA, and O'Connor K (2005). Thinking Is Believing: Ego-Dystonic Intrusive Thoughts in Obsessive-Compulsive Disorder. In Intrusive thoughts in clinical disorders: Theory, research, and treatment (New York, NY, US: Guilford Press; ), pp. 145–174. [Google Scholar]
- Conrad KL, Tseng KY, Uejima JL, Reimers JM, Heng LJ, Shaham Y, Marinelli M, and Wolf ME (2008). Formation of accumbens GluR2-lacking AMPA receptors mediates incubation of cocaine craving. Nature 454, 118–121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- DePoy LM, Zimmermann KS, Marvar PJ, and Gourley SL (2017). Induction and Blockade of Adolescent Cocaine-Induced Habits. Biol Psychiatry 81, 595–605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deslauriers J, Toth M, Der-Avakian A, and Risbrough VB (2018). Current Status of Animal Models of Posttraumatic Stress Disorder: Behavioral and Biological Phenotypes, and Future Challenges in Improving Translation. Biol Psychiatry 83, 895–907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dias-Ferreira E, Sousa JC, Melo I, Morgado P, Mesquita AR, Cerqueira JJ, Costa RM, and Sousa N (2009). Chronic stress causes frontostriatal reorganization and affects decision-making. Science 325, 621–625. [DOI] [PubMed] [Google Scholar]
- Do Monte FH, Quirk GJ, Li B, and Penzo MA (2016). Retrieving fear memories, as time goes by. Mol Psychiatry 21, 1027–1036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong Y, Taylor JR, Wolf ME, and Shaham Y (2017). Circuit and Synaptic Plasticity Mechanisms of Drug Relapse. J Neurosci 37, 10867–10876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donofry SD, Stillman CM, and Erickson KI (2020). A review of the relationship between eating behavior, obesity and functional brain network organization. Soc Cogn Affect Neurosci 15, 1157–1181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Duailibi MS, Cordeiro Q, Brietzke E, Ribeiro M, LaRowe S, Berk M, and Trevizol AP (2017). N-acetylcysteine in the treatment of craving in substance use disorders: Systematic review and meta-analysis. Am J Addict 26, 660–666. [DOI] [PubMed] [Google Scholar]
- Fedota JR, and Stein EA (2020). Networks relevant to psychopathology and intrusive thought. In Intrusive Thinking: from molecules to free will, Kalivas PW, and Paulus MP, eds. (Cambridge, MA: The MIT Press; ), pp. 187–202. [Google Scholar]
- Feil J, Sheppard D, Fitzgerald PB, Yucel M, Lubman DI, and Bradshaw JL (2010). Addiction, compulsive drug seeking, and the role of frontostriatal mechanisms in regulating inhibitory control. Neurosci Biobehav Rev 35, 248–275. [DOI] [PubMed] [Google Scholar]
- Fischer-Smith KD, Houston AC, and Rebec GV (2012). Differential effects of cocaine access and withdrawal on glutamate type 1 transporter expression in rat nucleus accumbens core and shell. Neuroscience 210, 333–339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fouyssac M, and Belin D (2019). Beyond drug-induced alteration of glutamate homeostasis, astrocytes may contribute to dopamine-dependent intrastriatal functional shifts that underlie the development of drug addiction: A working hypothesis. Eur J Neurosci. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Keller C, Carter JS, Kruyer A, Kearns AM, Hopkins JL, Hodebourg R, Kalivas PW, and Reichel CM (2021). Behavioral and accumbens synaptic plasticity induced by cues associated with restraint stress. Neuropsychopharmacology 46, 1848–1856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Keller C, Kupchik YM, Gipson CD, Brown RM, Spencer S, Bollati F, Esparza MA, Roberts-Wolfe DJ, Heinsbroek JA, Bobadilla AC, et al. (2016). Glutamatergic mechanisms of comorbidity between acute stress and cocaine self-administration. Mol Psychiatry 21, 1063–1069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Keller C, Neuhofer D, Bobadilla AC, Spencer S, Chioma VC, Monforton C, and Kalivas PW (2019). Extracellular Matrix Signaling Through beta3 Integrin Mediates Cocaine Cue-Induced Transient Synaptic Plasticity and Relapse. Biol Psychiatry 86, 377–387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Keller C, Scofield MD, Neuhofer D, Varanasi S, Reeves MT, Hughes B, Anderson E, Richie CT, Mejias-Aponte C, Pickel J, et al. (2020a). Relapse-Associated Transient Synaptic Potentiation Requires Integrin-Mediated Activation of Focal Adhesion Kinase and Cofilin in D1-Expressing Neurons. J Neurosci 40, 8463–8477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Keller C, Smiley C, Monforton C, Melton S, Kalivas PW, and Gass J (2020b). N-Acetylcysteine treatment during acute stress prevents stress-induced augmentation of addictive drug use and relapse. Addict Biol 25, e12798. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gipson CD, Kupchik YM, Shen H, Reissner KJ, Thomas CA, and Kalivas PW (2013). Relapse induced by cues predicting cocaine depends on rapid, transient synaptic potentiation. Neuron 77, 867–872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gourley SL, Bonci A, Bruchas MR, Flagel SB, Haber SN, Kalivas PW, Milton AL, Phillips PE, Picciotto MR, and Seamans JK (2020). Covergent experimental systems for dissecting the neurobiology of intrusive thought: A road map. In Intrusive Thinking: from molecules to free will, Kalivas PW, and Paulus MP, eds. (Cambridge, MA: The MIT Press; ), pp. 53–78. [Google Scholar]
- Gourley SL, and Taylor JR (2016). Going and stopping: Dichotomies in behavioral control by the prefrontal cortex. Nat Neurosci 19, 656–664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gray KM, Carpenter MJ, Baker NL, DeSantis SM, Kryway E, Hartwell KJ, McRae-Clark AL, and Brady KT (2012). A double-blind randomized controlled trial of N-acetylcysteine in cannabis-dependent adolescents. The American journal of psychiatry 169, 805–812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graziane NM, Sun S, Wright WJ, Jang D, Liu Z, Huang YH, Nestler EJ, Wang YT, Schluter OM, and Dong Y (2016). Opposing mechanisms mediate morphine- and cocaine-induced generation of silent synapses. Nat Neurosci 19, 915–925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guercio LA, Schmidt HD, and Pierce RC (2015). Deep brain stimulation of the nucleus accumbens shell attenuates cue-induced reinstatement of both cocaine and sucrose seeking in rats. Behav Brain Res 281, 125–130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haber SN, and Knutson B (2010). The reward circuit: linking primate anatomy and human imaging. Neuropsychopharmacology 35, 4–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hanlon CA, Dowdle LT, Correia B, Mithoefer O, Kearney-Ramos T, Lench D, Griffin M, Anton RF, and George MS (2017). Left frontal pole theta burst stimulation decreases orbitofrontal and insula activity in cocaine users and alcohol users. Drug Alcohol Depend 178, 310–317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hearing M, Graziane N, Dong Y, and Thomas MJ (2018). Opioid and Psychostimulant Plasticity: Targeting Overlap in Nucleus Accumbens Glutamate Signaling. Trends Pharmacol Sci 39, 276–294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henneberger C, Bard L, Panatier A, Reynolds JP, Kopach O, Medvedev NI, Minge D, Herde MK, Anders S, Kraev I, et al. (2020). LTP Induction Boosts Glutamate Spillover by Driving Withdrawal of Perisynaptic Astroglia. Neuron. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herman JP (2012). Neural pathways of stress integration: relevance to alcohol abuse. Alcohol research : current reviews 34, 441–447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodebourg R, Kalivas PW, and Kruyer A (2021). Extrasynaptic therapeutic targets in substance use and stress disorders. Trends in Pharmacological Science In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodebourg R, Kalivas PW, and Kruyer A (2022). Extrasynaptic therapeutic targets in substance use and stress disorders. Trends Pharmacol Sci 43, 56–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu H, Gan J, and Jonas P (2014). Interneurons. Fast-spiking, parvalbumin(+) GABAergic interneurons: from cellular design to microcircuit function. Science 345, 1255263. [DOI] [PubMed] [Google Scholar]
- Huang YH, Schluter OM, and Dong Y (2015). Silent Synapses Speak Up: Updates of the Neural Rejuvenation Hypothesis of Drug Addiction. Neuroscientist 21, 451–459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jakubiec L, Chirokoff V, Abdallah M, Sanz-Arigita E, Dupuy M, Swendsen J, Berthoz S, Gierski F, Guionnet S, Misdrahi D, et al. (2022). The Executive Functioning Paradox in Substance Use Disorders. Biomedicines 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jasinska AJ, Stein EA, Kaiser J, Naumer MJ, and Yalachkov Y (2014). Factors modulating neural reactivity to drug cues in addiction: a survey of human neuroimaging studies. Neurosci Biobehav Rev 38, 1–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalivas BC, and Kalivas PW (2016). Corticostriatal circuitry in regulating diseases characterized by intrusive thinking. Dialogues Clin Neurosci 18, 65–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Killingsworth MA, and Gilbert DT (2010). A wandering mind is an unhappy mind. Science 330, 932. [DOI] [PubMed] [Google Scholar]
- Knackstedt LA, Melendez RI, and Kalivas PW (2010). Ceftriaxone restores glutamate homeostasis and prevents relapse to cocaine seeking. Biol Psychiatry 67, 81–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koo JW, Chaudhury D, Han MH, and Nestler EJ (2019). Role of Mesolimbic Brain-Derived Neurotrophic Factor in Depression. Biol Psychiatry 86, 738–748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koob GF, and Le Moal M (2001). Drug addiction, dysregulation of reward, and allostasis. Neuropsychopharmacology 24, 97–129. [DOI] [PubMed] [Google Scholar]
- Koob GF, and Le Moal M (2008). Addiction and the brain antireward system. Annu Rev Psychol 59, 29–53. [DOI] [PubMed] [Google Scholar]
- Koob GF, and Volkow ND (2010). Neurocircuitry of addiction. Neuropsychopharmacology 35, 217–238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koob GF, and Volkow ND (2016). Neurobiology of addiction: a neurocircuitry analysis. Lancet Psychiatry 3, 760–773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruyer A, Angelis A, Garcia-Keller C, Li H, and Kalivas PW (2022a). Plasticity in astrocyte subpopulations regulates heroin relapse. Science Advances in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruyer A, Angelis A, Garcia-Keller C, Li H, and Kalivas PW (2022b). Plasticity in astrocyte subpopulations regulates heroin relapse. Sci Adv 8, eabo7044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruyer A, Chioma VC, and Kalivas PW (2020). The Opioid-Addicted Tetrapartite Synapse. Biol Psychiatry 87, 34–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruyer A, and Kalivas PW (2020). Astrocytes as cellular mediators of cue reactivity in addiction. Curr Opin Pharmacol 56, 1–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kruyer A, Kalivas PW, and Scofield MD (2023). Astrocyte regulation of synaptic signaling in psychiatric disorders. Neuropsychopharmacology 48, 21–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhn BN, Kalivas PW, and Bobadilla AC (2019). Understanding Addiction Using Animal Models. Front Behav Neurosci 13, 262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LaLumiere RT, and Kalivas PW (2008). Glutamate release in the nucleus accumbens core is necessary for heroin seeking. J Neurosci 28, 3170–3177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larowe SD, Kalivas PW, Nicholas JS, Randall PK, Mardikian PN, and Malcolm RJ (2013). A double-blind placebo-controlled trial of N-acetylcysteine in the treatment of cocaine dependence. The American journal on addictions / American Academy of Psychiatrists in Alcoholism and Addictions 22, 443–452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X, and Wolf ME (2015). Multiple faces of BDNF in cocaine addiction. Behav Brain Res 279, 240–254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liberzon I, and Abelson JL (2016). Context Processing and the Neurobiology of Post-Traumatic Stress Disorder. Neuron 92, 14–30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu B, Teschemacher AG, and Kasparov S (2017). Astroglia as a cellular target for neuroprotection and treatment of neuro-psychiatric disorders. Glia 65, 1205–1226. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lobo MK, and Nestler EJ (2011). The striatal balancing act in drug addiction: distinct roles of direct and indirect pathway medium spiny neurons. Frontiers in neuroanatomy 5, 41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu F, Yang W, and Qiu J (2023). Neural bases of motivated forgetting of autobiographical memories. Cogn Neurosci 14, 15–24. [DOI] [PubMed] [Google Scholar]
- Lu F, Yang W, Wei D, Sun J, Zhang Q, and Qiu J (2022). Superior frontal gyrus and middle temporal gyrus connectivity mediates the relationship between neuroticism and thought suppression. Brain Imaging Behav 16, 1400–1409. [DOI] [PubMed] [Google Scholar]
- Luscher C, and Janak PH (2021). Consolidating the Circuit Model for Addiction. Annu Rev Neurosci. [DOI] [PubMed] [Google Scholar]
- Ma YY, Lee BR, Wang X, Guo C, Liu L, Cui R, Lan Y, Balcita-Pedicino JJ, Wolf ME, Sesack SR, et al. (2014). Bidirectional modulation of incubation of cocaine craving by silent synapse-based remodeling of prefrontal cortex to accumbens projections. Neuron 83, 1453–1467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Malejko K, Abler B, Plener PL, and Straub J (2017). Neural Correlates of Psychotherapeutic Treatment of Post-traumatic Stress Disorder: A Systematic Literature Review. Front Psychiatry 8, 85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Manning K, Wang L, and Steffens D (2019). Recent advances in the use of imaging in psychiatry: functional magnetic resonance imaging of large-scale brain networks in late-life depression. F1000Res 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mantsch JR, Baker DA, Funk D, Le AD, and Shaham Y (2016). Stress-Induced Reinstatement of Drug Seeking: 20 Years of Progress. Neuropsychopharmacology 41, 335–356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- May J, Kavanagh DJ, and Andrade J (2015). The Elaborated Intrusion Theory of desire: a 10-year retrospective and implications for addiction treatments. Addict Behav 44, 29–34. [DOI] [PubMed] [Google Scholar]
- McEwen BS, and Morrison JH (2013). The brain on stress: vulnerability and plasticity of the prefrontal cortex over the life course. Neuron 79, 16–29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFarland K, Davidge SB, Lapish CC, and Kalivas PW (2004). Limbic and motor circuitry underlying footshock-induced reinstatement of cocaine-seeking behavior. J Neurosci 24, 1551–1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFarland K, and Kalivas PW (2001). The circuitry mediating cocaine-induced reinstatement of drug-seeking behavior. J Neurosci 21, 8655–8663. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McFarland K, Lapish CC, and Kalivas PW (2003). Prefrontal glutamate release into the core of the nucleus accumbens mediates cocaine-induced reinstatement of drug-seeking behavior. J Neurosci 23, 3531–3537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGaugh JL (2000). Memory--a century of consolidation. Science 287, 248–251. [DOI] [PubMed] [Google Scholar]
- McGlinchey EM, James MH, Mahler SV, Pantazis C, and Aston-Jones G (2016). Prelimbic to Accumbens Core Pathway Is Recruited in a Dopamine-Dependent Manner to Drive Cued Reinstatement of Cocaine Seeking. J Neurosci 36, 8700–8711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGrath AG, and Briand LA (2019). A potential role for microglia in stress- and drug-induced plasticity in the nucleus accumbens: A mechanism for stress-induced vulnerability to substance use disorder. Neurosci Biobehav Rev 107, 360–369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mildner JN, and Tamir DI (2019). Spontaneous Thought as an Unconstrained Memory Process. Trends Neurosci 42, 763–777. [DOI] [PubMed] [Google Scholar]
- Miles FJ, Everitt BJ, and Dickinson A (2003). Oral cocaine seeking by rats: action or habit? Behav Neurosci 117, 927–938. [DOI] [PubMed] [Google Scholar]
- Millan EZ, Kim HA, and Janak PH (2017). Optogenetic activation of amygdala projections to nucleus accumbens can arrest conditioned and unconditioned alcohol consummatory behavior. Neuroscience 360, 106–117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moccia L, Pettorruso M, De Crescenzo F, De Risio L, di Nuzzo L, Martinotti G, Bifone A, Janiri L, and Di Nicola M (2017). Neural correlates of cognitive control in gambling disorder: a systematic review of fMRI studies. Neurosci Biobehav Rev 78, 104–116. [DOI] [PubMed] [Google Scholar]
- Moulds ML, Bisby MA, Wild J, and Bryant RA (2020). Rumination in posttraumatic stress disorder: A systematic review. Clin Psychol Rev 82, 101910. [DOI] [PubMed] [Google Scholar]
- Moussawi K, Zhou W, Shen H, Reichel CM, See RE, Carr DB, and Kalivas PW (2011). Reversing cocaine-induced synaptic potentiation provides enduring protection from relapse. Proceedings of the National Academy of Sciences of the United States of America 108, 385–390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mulholland PJ, Chandler LJ, and Kalivas PW (2016). Signals from the Fourth Dimension Regulate Drug Relapse. Trends Neurosci 39, 472–485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy-Royal C, Dupuis J, Groc L, and Oliet SHR (2017). Astroglial glutamate transporters in the brain: Regulating neurotransmitter homeostasis and synaptic transmission. J Neurosci Res 95, 2140–2151. [DOI] [PubMed] [Google Scholar]
- Murphy-Royal C, Johnston AD, Boyce AKJ, Diaz-Castro B, Institoris A, Peringod G, Zhang O, Stout RF, Spray DC, Thompson RJ, et al. (2020). Stress gates an astrocytic energy reservoir to impair synaptic plasticity. Nature communications 11, 2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nall RW, Heinsbroek JA, Nentwig TB, Kalivas PW, and Bobadilla AC (2021). Circuit Selectivity in Drug Versus Natural Reward Seeking Behaviors. J Neurochem. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nestler EJ, and Luscher C (2019). The Molecular Basis of Drug Addiction: Linking Epigenetic to Synaptic and Circuit Mechanisms. Neuron 102, 48–59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardo-Garcia TR, Garcia-Keller C, Penaloza T, Richie CT, Pickel J, Hope BT, Harvey BK, Kalivas PW, and Heinsbroek JA (2019). Ventral Pallidum Is the Primary Target for Accumbens D1 Projections Driving Cocaine Seeking. J Neurosci 39, 2041–2051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pariyadath V, Gowin JL, and Stein EA (2016). Resting state functional connectivity analysis for addiction medicine: From individual loci to complex networks. Prog Brain Res 224, 155–173. [DOI] [PubMed] [Google Scholar]
- Paxinos G, and Watson C (2007). The Rat Brain in Stereotaxic Coordinates, 6th Edition edn (Burlington: Elsevier Academic Press; ). [Google Scholar]
- Peskin M, Wyka K, Cukor J, Olden M, Altemus M, Lee FS, and Difede J (2018). The relationship between posttraumatic and depressive symptoms during virtual reality exposure therapy with a cognitive enhancer. J Anxiety Disord. [DOI] [PubMed] [Google Scholar]
- Poerio GL, Totterdell P, and Miles E (2013). Mind-wandering and negative mood: does one thing really lead to another? Conscious Cogn 22, 1412–1421. [DOI] [PubMed] [Google Scholar]
- Radley JJ, Anderson RM, Cosme CV, Glanz RM, Miller MC, Romig-Martin SA, and LaLumiere RT (2015). The Contingency of Cocaine Administration Accounts for Structural and Functional Medial Prefrontal Deficits and Increased Adrenocortical Activation. J Neurosci 35, 11897–11910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rao PS, and Sari Y (2012). Glutamate transporter 1: target for the treatment of alcohol dependence. Current medicinal chemistry 19, 5148–5156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rappeneau V, Blaker A, Petro JR, Yamamoto BK, and Shimamoto A (2016). Disruption of the Glutamate-Glutamine Cycle Involving Astrocytes in an Animal Model of Depression for Males and Females. Front Behav Neurosci 10, 231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Renaud F, Jakubiec L, Swendsen J, and Fatseas M (2021). The Impact of Co-occurring Post-traumatic Stress Disorder and Substance Use Disorders on Craving: A Systematic Review of the Literature. Front Psychiatry 12, 786664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson MB, and Jackson JG (2016). Astroglial glutamate transporters coordinate excitatory signaling and brain energetics. Neurochem Int 98, 56–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Root DH, Melendez RI, Zaborszky L, and Napier TC (2015). The ventral pallidum: Subregion-specific functional anatomy and roles in motivated behaviors. Prog Neurobiol 130, 29–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schall TA, Wright WJ, and Dong Y (2021). Nucleus accumbens fast-spiking interneurons in motivational and addictive behaviors. Mol Psychiatry 26, 234–246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schoenbaum G, and Setlow B (2005). Cocaine makes actions insensitive to outcomes but not extinction: implications for altered orbitofrontal-amygdalar function. Cereb Cortex 15, 1162–1169. [DOI] [PubMed] [Google Scholar]
- Scofield MD, Heinsbroek JA, Gipson CD, Kupchik YM, Spencer S, Smith AC, Roberts-Wolfe D, and Kalivas PW (2016). The Nucleus Accumbens: Mechanisms of Addiction across Drug Classes Reflect the Importance of Glutamate Homeostasis. Pharmacol Rev 68, 816–871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selemon LD, Young KA, Cruz DA, and Williamson DE (2019). Frontal Lobe Circuitry in Posttraumatic Stress Disorder. Chronic Stress (Thousand Oaks: ) 3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen H, Moussawi K, Zhou W, Toda S, and Kalivas PW (2011). Heroin relapse requires long-term potentiation-like plasticity mediated by NMDA2b-containing receptors. Proceedings of the National Academy of Sciences of the United States of America 108, 19407–19412. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen HW, Scofield MD, Boger H, Hensley M, and Kalivas PW (2014). Synaptic glutamate spillover due to impaired glutamate uptake mediates heroin relapse. J Neurosci 34, 5649–5657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimamoto A, Rappeneau V, Munjal H, Farris T, Davis C, Wilson A, Edwards M, Moore C, Reynolds C, and Meshul CK (2018). Glutamate-Glutamine Transfer and Chronic Stress-Induced Sex Differences in Cocaine Responses. Neuroscience 391, 104–119. [DOI] [PubMed] [Google Scholar]
- Siemsen BM, Reichel CM, Leong KC, Garcia-Keller C, Gipson CD, Spencer S, McFaddin JA, Hooker KN, Kalivas PW, and Scofield MD (2019). Effects of Methamphetamine Self-Administration and Extinction on Astrocyte Structure and Function in the Nucleus Accumbens Core. Neuroscience 406, 528–541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smaga I, Fierro D, Mesa J, Filip M, and Knackstedt LA (2020). Molecular changes evoked by the beta-lactam antibiotic ceftriaxone across rodent models of substance use disorder and neurological disease. Neurosci Biobehav Rev 115, 116–130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smallwood J, and Schooler JW (2015). The science of mind wandering: empirically navigating the stream of consciousness. Annu Rev Psychol 66, 487–518. [DOI] [PubMed] [Google Scholar]
- Smith AC, Kupchik YM, Scofield MD, Gipson CD, Wiggins A, Thomas CA, and Kalivas PW (2014). Synaptic plasticity mediating cocaine relapse requires matrix metalloproteinases. Nat Neurosci 17, 1655–1657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith AC, Scofield MD, Heinsbroek JA, Gipson CD, Neuhofer D, Roberts-Wolfe DJ, Spencer S, Garcia-Keller C, Stankeviciute NM, Smith RJ, et al. (2017). Accumbens nNOS Interneurons Regulate Cocaine Relapse. J Neurosci 37, 742–756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith RJ, Lobo MK, Spencer S, and Kalivas PW (2013). Cocaine-induced adaptations in D1 and D2 accumbens projection neurons (a dichotomy not necessarily synonymous with direct and indirect pathways). Curr Opin Neurobiol 23, 546–552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sofuoglu M, Rosenheck R, and Petrakis I (2014). Pharmacological treatment of comorbid PTSD and substance use disorder: recent progress. Addict Behav 39, 428–433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Song X, and Wang X (2012). Mind wandering in Chinese daily lives--an experience sampling study. PLoS One 7, e44423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer S, Neuhofer D, Chioma VC, Garcia-Keller C, Schwartz DJ, Allen N, Scofield MD, Ortiz-Ithier T, and Kalivas PW (2018). A Model of Delta(9)-Tetrahydrocannabinol Self-administration and Reinstatement That Alters Synaptic Plasticity in Nucleus Accumbens. Biol Psychiatry 84, 601–610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stennett BA, and Knackstedt LA (2020). A Rat Model of Cocaine-Alcohol Polysubstance Use Reveals Altered Cocaine Seeking and Glutamate Levels in the Nucleus Accumbens. Front Neurosci 14, 877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stennett BA, Padovan-Hernandez Y, and Knackstedt LA (2020). Sequential cocaine-alcohol self-administration produces adaptations in rat nucleus accumbens core glutamate homeostasis that are distinct from those produced by cocaine self-administration alone. Neuropsychopharmacology 45, 441–450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Steward T, Das P, Malhi GS, Bryant RA, and Felmingham KL (2020). Dysfunctional coupling of the parahippocampal cortex and inferior frontal gyrus during memory suppression in posttraumatic stress disorder. Eur Neuropsychopharmacol 41, 146–151. [DOI] [PubMed] [Google Scholar]
- Suh J, and Ressler KJ (2018). Common Biological Mechanisms of Alcohol Use Disorder and Post-Traumatic Stress Disorder. Alcohol research : current reviews 39, 131–145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tseng J, and Poppenk J (2020). Brain meta-state transitions demarcate thoughts across task contexts exposing the mental noise of trait neuroticism. Nature communications 11, 3480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venniro M, Banks ML, Heilig M, Epstein DH, and Shaham Y (2020). Improving translation of animal models of addiction and relapse by reverse translation. Nat Rev Neurosci 21, 625–643. [DOI] [PubMed] [Google Scholar]
- Venniro M, Russell TI, Zhang M, and Shaham Y (2019). Operant Social Reward Decreases Incubation of Heroin Craving in Male and Female Rats. Biol Psychiatry. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Visser RM, Anderson MC, Aron A, Banich MT, Brady KT, Huys QJM, Monfils M-H, Schiller D, Schlagenjauf F, Schooler JW, et al. (2020). Neuropsychological mechanisms of intrusive thinking. In Intrusive Thinking: From molecules to free will, Kalivas PW, and Paulus MP, eds. (Cambridge, MA: The MIT Press; ), pp. 125–184. [Google Scholar]
- Volkow ND, Koob GF, and McLellan AT (2016). Neurobiologic Advances from the Brain Disease Model of Addiction. N Engl J Med 374, 363–371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wamsley EJ (2019). Memory Consolidation during Waking Rest. Trends Cogn Sci 23, 171–173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Holt LM, Huang HH, Sesack SR, Nestler EJ, and Dong Y (2021a). Astrocytes in cocaine addiction and beyond. Mol Psychiatry. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J, Li KL, Shukla A, Beroun A, Ishikawa M, Huang X, Wang Y, Wang YQ, Yang Y, Bastola ND, et al. (2021b). Cocaine Triggers Astrocyte-Mediated Synaptogenesis. Biol Psychiatry 89, 386–397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang YQ, Huang YH, Balakrishnan S, Liu L, Wang YT, Nestler EJ, Schluter OM, and Dong Y (2021c). AMPA and NMDA Receptor Trafficking at Cocaine-Generated Synapses. J Neurosci 41, 1996–2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watkins ER (2008). Constructive and unconstructive repetitive thought. Psychol Bull 134, 163–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watkins ER, and Nolen-Hoeksema S (2014). A habit-goal framework of depressive rumination. J Abnorm Psychol 123, 24–34. [DOI] [PubMed] [Google Scholar]
- Whyte AJ, Trinoskey-Rice G, Davies RA, Woon EP, Foster SL, Shapiro LP, Li DC, Srikanth KD, Gil-Henn H, and Gourley SL (2021). Cell adhesion factors in the orbitofrontal cortex control cue-induced reinstatement of cocaine seeking and amygdala-dependent goal seeking. J Neurosci. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wichary S, and Smolen T (2016). Neural Underpinnings of Decision Strategy Selection: A Review and a Theoretical Model. Front Neurosci 10, 500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wisco BE, Vrshek-Schallhorn S, May CL, Campbell AA, Nomamiukor FO, and Pugach CP (2023). Effects of trauma-focused rumination among trauma-exposed individuals with and without posttraumatic stress disorder: An experiment. J Trauma Stress. [DOI] [PubMed] [Google Scholar]
- Wright WJ, Graziane NM, Neumann PA, Hamilton PJ, Cates HM, Fuerst L, Spenceley A, MacKinnon-Booth N, Iyer K, Huang YH, et al. (2020). Silent synapses dictate cocaine memory destabilization and reconsolidation. Nat Neurosci 23, 32–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou HX, Chen X, Shen YQ, Li L, Chen NX, Zhu ZC, Castellanos FX, and Yan CG (2020). Rumination and the default mode network: Meta-analysis of brain imaging studies and implications for depression. Neuroimage 206, 116287. [DOI] [PubMed] [Google Scholar]
- Zilverstand A, Huang AS, Alia-Klein N, and Goldstein RZ (2018). Neuroimaging Impaired Response Inhibition and Salience Attribution in Human Drug Addiction: A Systematic Review. Neuron 98, 886–903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zilverstand A, Parvaz MA, Moeller SJ, and Goldstein RZ (2016). Cognitive interventions for addiction medicine: Understanding the underlying neurobiological mechanisms. Prog Brain Res 224, 285–304. [DOI] [PMC free article] [PubMed] [Google Scholar]