Abstract
Since the discovery of mesenchymal stem cells (MSCs) in the 1970s, they have been widely used in the treatment of a variety of diseases because of their wide sources, strong differentiation potential, rapid expansion in vitro, low immunogenicity, and so on. At present, most of the related research is on mesoderm-derived MSCs (M-MSCs) such as bone marrow MSCs and adipose-derived MSCs. As a type of MSC, ectoderm-derived MSCs (E-MSCs) have a stronger potential for self-renewal, multidirectional differentiation, and immunomodulation and have more advantages than M-MSCs in some specific conditions. This paper analyzes the relevant research development of E-MSCs compared with that of M-MSCs; summarizes the extraction, discrimination and culture, biological characteristics, and clinical application of E-MSCs; and discusses the application prospects of E-MSCs. This summary provides a theoretical basis for the better application of MSCs from both ectoderm and mesoderm in the future.
1. Introduction
As an important member of the stem cell family, mesenchymal stem cells (MSCs) are widely distributed and easy to extract and culture and have self-replication ability and strong differentiation potential. MSCs also have the ability to migrate to damaged tissues and regulate the immune response according to the microenvironment, which is why they are being increasingly applied in tissue engineering and clinical research.
MSCs are derived from the mesoderm and ectoderm in early development. Leucht et al. [1] proposed that damaged tissue from different germ layers will recruit MSCs from the corresponding germ layers for repair. Moreover, the proliferation and differentiation abilities of MSCs from different tissues are also different [2]. This paper comprehensively compares the biological characteristics and clinical application of mesenchymal stem cells from the mesoderm and ectoderm, as well as the possible development direction in the future.
2. Basic Introduction
2.1. Mesoderm-Derived MSCs (M-MSCs)
A rich source of M-MSCs is an important basis for their extensive research and application. Bone marrow is an important source of MSCs. In addition to bone marrow, M-MSCs also exist in various tissues and organs. In 2000, human umbilical cord blood stem cells were first reported [3]. Subsequently, in 2001, adipose tissue and synovium were also proven to be rich sources of M-MSCs [4, 5]. The extraction method of M-MSCs, in short, includes separating various tissues, digesting the tissues to obtain cells, culturing the cells for 3 to 5 days, discarding nonadherent cells, and continuously culturing adherent cells to the desired passage [6].
2.2. Ectoderm-Derived MSCs (E-MSCs)
E-MSCs are mainly divided into three types: osteogenic (T.Q. [7]), odontogenic, and olfactory mucosal. Odontogenic stem cells include dental pulp stem cells (DPSCs), dental follicle stem cells (DFSCs), apical dental papilla stem cells (SCAPs), deciduous dental pulp stem cells (SHEDs), and periodontal ligament stem cells (PDLSCs). Sources and markers of odontogenic stem cells are listed in Figure 1. DPSCs were first isolated from adult dental pulp in 2001 [8]. DFSCs were isolated and identified from the dental sac of human third molars by Morsczeck et al. [9] in 2005. Sonoyama et al. [10] first found and identified SCAPs from the apical papilla of extracted third molars in 2006. SCAPs come from developing tissues. Therefore, SCAPs may have better regeneration potential than other mature tissues. Miura et al. (S. Shi) first discovered SHEDs in 2003. SHEDs can differentiate into a variety of cell types, including nerve cells, adipocytes, and odontoblasts. In addition, after injection into the dentate gyrus of the mouse hippocampus, SHEDs can differentiate into neural tissue and express neuronal and glial markers, indicating that SHEDs can be used for dental pulp and nerve regeneration. PDLSCs were isolated and identified from the surface of tooth roots by Seo et al. [11] in 2004. In 2009, Zhang et al. [12] first reported the isolation, characterization, and immunomodulatory properties of gingival mesenchymal stem cells (GMSCs) and found that GMSCs can inhibit the proliferation of peripheral blood monocytes induced by phytohemagglutinin. Olfactory mucosal stem cells (OMSCs) are MSCs isolated from the olfactory mucosal epithelium. Studies have shown that MSCs may play a better role in promoting the formation of the myelin sheath in the central nervous system and in repairing nerve injury than MSCs from other tissues [13].
Figure 1.
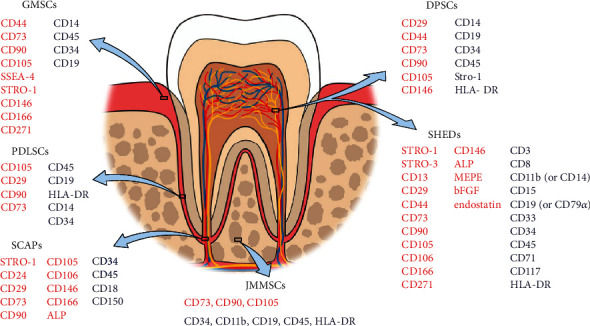
Sources and markers of some representative E-MSCs (DPSC-dental pulp stem cells, SCAP-apical dental papilla stem cells, SHED-deciduous dental pulp stem cells, PDLSC-periodontal ligament stem cells, GMSC-gingival mesenchymal stem cells, and JMMSC-jaw marrow-derived mesenchymal stem cells. Red indicates positively expressed while blue indicates negatively expressed surface markers).
3. Extraction, Discrimination, and Culture
Theoretically, MSCs can be isolated from all tissues. For example, M-MSCs mainly come from the bone marrow, adipose tissue, placenta and human umbilical cord blood, while E-MSCs mainly come from the dental pulp, the jaw, the frontal bone, the periodontal ligament, the gingiva, and the dental papilla. Han et al. [6] summarized the extraction, identification, and culture methods of several common M-MSCs. For comparison, in Table 1, the authors summarized the extraction, identification, and culture of several common E-MSCs. In Figure 2, the extraction process of DPSCs is described in the form of a schematic.
Table 1.
Extraction, discrimination, and culture of E-MSCs.
| MSC type | First found time | Source | Extraction approach | Culture medium | Marker | Reference | |
|---|---|---|---|---|---|---|---|
| DPSCs | 2002 | Pulp of human third molars | DPSC-ED | (1) Tooth surfaces were cleaned and cut around the cementum-enamel junction by using sterilized dental fissure burs to reveal the pulp chamber (2) Rinse with culture medium and cut dental pulp tissue into small pieces of 1-2 mm3 (3) Pulp fragments were digested in a solution of 3 mg/ml collagenase type I and 4 mg/ml dispase II for 1 h at 37°C (4) After digestion, it was centrifuged at a speed of 300 g, and the resuspended tissue fragments were passed through 70 μM cell filter (5) The cells were cultured in 6-well plates in standard DPSC medium. All cell cultures were incubated at 37°C in 5% CO2 |
Alpha modification of Eagle's medium supplemented with 10% FBS+100 μM L-ascorbic acid 2-phosphate+2 mM L-glutamine+100 U/ml penicillin+100 μg/ml streptomycin | Positive: CD29, CD44, CD49f, CD73, CD81, CD90, CD105, and CD146 Negative: CD14, CD19, CD34, CD45, Stro-1, and HLA-DR |
[8, 14–19] |
| DPSC-OG | (1) After disinfecting the tooth surface, mechanically break the tooth and gently separate the dental pulp with tweezers (there is no need to drill, because it may adversely affect the viability of dental pulp stem cells) (2) Rinse with culture medium and cut dental pulp tissue into small pieces of 1-2 mm3 (3) Pulp pieces were cultured in 6-well plates in standard DPSC medium. All cell cultures were incubated at 37°C in 5% CO2 |
||||||
|
| |||||||
| DFSCs | 2005 | Normal human impacted third molars | (1) Normal human impacted third molars were surgically removed and collected. Attached dental follicles were separated from the mineralized tooth (2) The surfaces of the follicle tissues were cleaned and minced by using a sterilized scalpel (dental papilla tissue was discarded) (3) Tissues were digested in a solution of 3 mg/ml collagenase type I and 4 mg/ml dispase II for 1 h at 37°C (4) Minced and digested tissues of dental follicle explants were seeded into 60 mm plates or T25 flasks in media at 37°C in 5% CO2 in a humidified atmosphere |
Alpha modification of Eagle's medium supplemented with 10% FBS+100 μM L-ascorbic acid 2-phosphate+2 mM L-glutamine+100 units/ml penicillin+100 μg/ml streptomycin | Positive: CD105, CD29, CD90, and CD73 Negative: CD45, CD19, HLA-DR, CD14, and CD34 |
[9, 20–23] | |
|
| |||||||
| SCAPs | 2006 | Normal human impacted third molars | (1) Root apical papilla was gently separated from the surface of the root (2) Tissues were minced and digested in a solution of 3 mg/ml collagenase type I and 4 mg/ml dispase for 30 minutes at 37°C (3) Single cell suspensions of SCAP were obtained by passing through a 70 μM strainer (4) Minced and digested tissues of dental follicle explants were seeded into 60 mm plates or T25 flasks in media at 37°C in 5% CO2 in a humidified atmosphere |
Alpha modification of Eagle's medium supplemented with 15% FBS+100 μM L-ascorbic acid 2-phosphate+2 mM L-glutamine+100 U/ml penicillin+100 μg/ml streptomycin | Positive: STRO-1, CD24, CD29, CD73, CD90, CD105, CD106, CD146, CD166, and ALP Negative: CD34, CD45, CD18, and CD150 |
[10] | |
| Minipigs: canine | (1) The canines of Wuzhishan minipigs were extracted, and the root apical papilla was gently separated from the surface of the root (2) Apical papilla was minced and digested in a solution of 3 mg/ml collagenase type I and 4 mg/ml dispase for 30 minutes at 37°C (3) Then passed through a 70 μm strainer to obtain a single cell suspension and seeded into 25 cm2 culture flasks containing an basic medium |
Positive:STRO-1, CD146, CD24 | [24] | ||||
|
| |||||||
| SHEDs | 2003 | Intact caries free primary teeth | (1) The pulp was separated from a remnant crown under strict aseptic conditions (2) The pulp was minced by using a sterilized scalpel (3) Tissues were digested in a solution of 3 mg/ml collagenase type I and 4 mg/ml dispase for 30 minutes at 37°C (4) Then seeded into 25 cm2 culture flasks containing an basic medium |
Dulbecco's modified Eagle's medium supplemented with 10% FBS+100 U/ml penicillin+100 μg/ml streptomycin | Positive: STRO-1, STRO-3, CD13, CD29, CD44, CD73, CD90, CD105, CD106, CD166, CD271, CD146, ALP, MEPE, bFGF, and endostatin Negative: CD3, CD8, CD11b (or CD14), CD15, CD19 (or CD79α), CD33, CD34, CD45, CD71, CD117, and HLA-DR |
[14, 25–27] | |
|
| |||||||
| PDLSCs | 2004 | Normal impacted third molars | (1) PDL was gently separated from the surface of the root (2) Tissues was minced and digested in a solution of 3 mg/ml collagenase type I and 4 mg/ml dispase for 1 hour at 37°C (3) Single cell suspensions of PDLSCs were obtained by passing through a 70 μM strainer (4) Minced and digested tissues were seeded into 60 mm plates or T25 flasks in media at 37°C in 5% CO2 in a humidified atmosphere |
Alpha modification of Eagle's medium supplemented with 10% FBS+100 μM L-ascorbic acid 2-phosphate+2 mM L-glutamine+100 units/ml penicillin+100 μg/ml streptomycin | Positive: CD105, CD29, CD90, and CD73 Negative: CD45, CD19, HLA-DR, CD14, and CD34 |
[11, 22, 28, 29] | |
|
| |||||||
| GMSCs | 2009 | Attached keratinized gingival tissues | (1) The tissues were deepithelialized and minced into 1–2 mm2 fragments (2) The minced tissues were digested in 2 mg/ml collagenase and 1 mg/ml dispase for 30 min (3) After discarding the first digested cell suspension, the tissues were digested in the same solution for 90 min at 37°C (4) Single cell suspensions of GMSCs were obtained by passing through a 70 μM strainer (5) Minced and digested tissues were seeded into 60 mm plates or T25 flasks in media at 37°C in 5% CO2 in a humidified atmosphere |
Alpha modification of Eagle's medium containing 15% FBS+100 U/ml penicillin+100 μg/ml streptomycin +200 mM L-glutamine +10 mM ascorbic acid 2-hosphate | Positive: CD44, CD73, CD90, CD105, SSEA-4, STRO-1, CD146, CD166, and CD271 Negative: CD14, CD45, CD34, and CD19 |
[30] | |
|
| |||||||
| OMSCs | 2005 | Human upper middle turbinates | (1) All biopsies were collected on ice in Hanks' balanced salt solution containing penicillin (100 U/ml), streptomycin (100 mg/ml), and Fungizone (amphotericin B, 1.25 mg/ml) (2) After being minced with a scalpel blade, the tissue was digested using 1.33% collagenase for 20 min (3) Then, the tissues were incubation with DNAse to reduce cell clumping (0.04 mg/ml bovine pancreas DNAse, 3.0 mg/ml bovine serum albumin-fraction A in L15) (4) Cells were mechanically dissociated by pipetting and then triturating through a 23G needle and centrifuged at 1200 rpm for 5 min, and the pellet resuspended in low-glucose Dulbecco's modified Eagle's medium |
Low-glucose Dulbecco's modified Eagle's medium supplemented with 10% FBS+100 U/ml penicillin+100 μg/ml streptomycin | Positive: CD90, CD54, CD105, CD73, nestin, CD166, and p75NTR Negative: STRO-1 |
[13] | |
|
| |||||||
| JMSCs | 2006 | Human undergoing orthognathic surgery | The resected bone mass was cut into small fragments (<1 mm3) and cultured in T25 flasks with α-MEM containing 10% FBS in a 37°C humidified incubator with a 5% CO2 atmosphere | Alpha modification of Eagle's medium containing 10% FBS+100 U/ml penicillin+100 μg/ml streptomycin | Positive: CD73, CD90, and CD105 Negative: CD34, CD11b, CD19, CD45, and HLA-DR |
[31–34] | |
Figure 2.
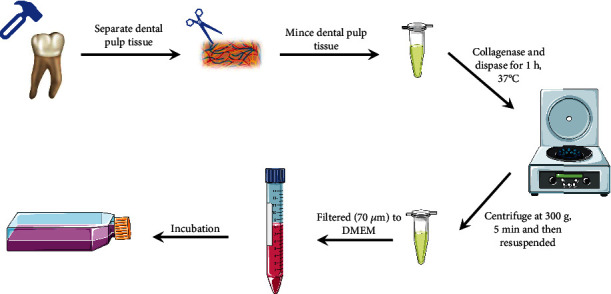
Typical extraction process of DPSCs of human.
4. Biological Properties
Regardless of the source of MSCs, they all show some common characteristics, such as fibroblast-like morphology, cell surface markers, cell proliferation ability, and multidirectional differentiation potential. However, the biological properties of MSCs from different tissues or the same kind of MSCs under different conditions are slightly different.
4.1. Cell Proliferation Ability
According to Miura et al. (S. Shi), SHEDs have a higher proliferation rate and population doubling efficiency than DPSCs and bone marrow mesenchymal stem cells (BMMSCs). In addition, SHEDs are separated from deciduous teeth, so they can be easily obtained without ethical considerations. The transition from deciduous teeth to permanent teeth is a unique dynamic process. The dental pulp of deciduous teeth already exists before birth, indicating that these stem cells are not affected or are less affected by environmental factors than other stem cells [14]. Akintoye et al. [35] compared MSCs from maxillofacial and lower limb bone marrow, and the results showed that the proliferation rate of jaw bone marrow MSCs from maxillofacial-bone marrow was faster than that of iliac bone marrow MSCs from lower limb bone marrow. Moreover, compared with long bone marrow MSCs, jaw bone marrow MSCs showed stronger proliferation and antiapoptotic potential [7, 20]. In embryology, the iliac bone and long bone are derived from the mesoderm, while the jaw is derived from the ectodermal neural crest. Interestingly, studies have shown that cells expressing neural crest markers have stronger stem cell properties and stronger proliferation potential than M-MSCs [2]; thus, they contribute to the survival of MSCs under hypoxia after transplantation [21, 36].
4.2. Multidirectional Differentiation
Regardless of the kind of tissue source, MSCs have multidirectional differentiation potential. Multidirectional differentiation potential is one of the important characteristics of MSCs. The differentiation trend of different tissue sources is also different. The jaw develops from the neural crest cells of ectoderm [37], while the mesenchymal cells of mesoderm develop into the long bones of the limbs [38], and the osteogenic processes experienced by the two bone tissues are also different in the development process [37]. In some diseases, such as osteoporosis and hyperthyroidism, the involvement of the long bone is significantly higher than that of the jaw [39]. Some studies have shown that compared with bone marrow MSCs of long bones of limbs, jawbone marrow MSCs have stronger osteogenic activity. This may be due to the high expression of BMP-4, nestin, and other neural crest-related genes in jawbone marrow MSCs at the transcriptional level [2], which is consistent with the research results of Aghaloo et al. [40].
Central and peripheral nerve injuries are difficult to treat, because the ability of the nervous system to repair damaged cells and tissues is limited. In this regard, E-MSCs have obvious advantages because they can differentiate into neuron-like cells and express neuronal markers, such as STRO-1, nestin, c-FOS, GFAP, and β III-tubulin [41–45]. Although these cells can differentiate into neuron-like cells, they do not further differentiate into functional neurons [41]. On this basis, Kiraly et al. found that simultaneous activation of PKC and cAMP can induce hDPSCs to differentiate into functional neurons [43].
At present, research in the field of nerve regeneration mainly focuses on DPSCs and SHEDs [19, 46, 47]. This may be due to the relationship between their tissue origins. In the process of tooth development, the tooth germ is composed of an enamel organ, dental papilla, and dental sac. DPSCs, SHEDs, and SCAPs are derived from dental papilla; DFSCs and PDLSCs are derived from dental sac; and dental pulp is derived from dental papilla. This shows that DPSCs, SHEDs, and SCAPs are highly homologous with dental pulp at the histological level. At the same time, the extraction difficulty and conditions of SCAPs are more stringent than those of DPSCs and SHEDs. Therefore, DPSCs and SHEDs may be a possible direction for pulp regeneration and even central nerve repair in the future.
Compared with bone marrow MSCs, odontogenic MSCs are more convenient to obtain and easy to expand and preserve, have high activity, and have low immunogenicity and tumorigenicity, and these characteristics make them more conducive to clinical application [7, 48]. The multiple differentiation potential and the derivation of MSCs derived from ectodermal cells are summarized in Table 2. The difference in germ layer origin makes the phenotype of odontogenic MSCs different from that of MSCs, such as BMMSCs.
Table 2.
The multiple differentiation potential and the derivation of ectodermal MSCs.
| Cell type | PD | Multipotentiality | Source | Reference |
|---|---|---|---|---|
| DPSCs | 60-70 | Osteogenic | Human impacted third molar (age 18–22 years old) | [49] |
| Rat | [50] | |||
| Angiogenic | Human impacted third molar (age 18-25 years) | [51] | ||
| Adipogenic | Supernumerary tooth (female, 8 years, and male, 12 years) | [52] | ||
| Neurogenic | Human impacted third molars (age 20-30 years) | [53] | ||
| Human impacted third molars | [54] | |||
| Human impacted third molars (age 18–22 years) | [55] | |||
| Human impacted third molars | [56] | |||
| Dentin/pulp-like | Human impacted third molars (age 15-25 years) | [57] | ||
| Human impacted third molar (11 years old) | [58] | |||
|
| ||||
| DFSCs | — | Chondrogenesis | Rat | [59] |
| Healthy children (age 6-12 years) | [60] | |||
| Osteogenic | Rat | [61] | ||
| Healthy children (age 6-12 years) | [60] | |||
| Adipogenic | Healthy children (age 6-12 years) | [60] | ||
| Human impacted third molar (female, 22 years) | [52] | |||
| Neurogenic | Impacted third molar (age 18–22 years) | [55] | ||
|
| ||||
| SCAPs | 70 | Angiogenesis | Human impacted third molars (age 12-15 years) | [62] |
| Neurogenic | Human impacted third molars (age 18-22 years) | [55] | ||
| Osteo/dentinogenic | Human impacted third molars (age 12-15 years) | [63] | ||
|
| ||||
| SHEDs | <140 | Neurogenic | Healthy children (age 7-8 years) | [64] |
| Adipogenic | Healthy children (age 7-8 years) | [64] | ||
| Osteo/dentinogenic | Healthy children (age 7-8 years) | [64] | ||
| Chondrogenesis | Healthy children (age 7-8 years) | [64] | ||
| Angiogenesis | Healthy children (age 7-8 years) | [65] | ||
|
| ||||
| PDLSCs | — | Adipogenic | Supernumerary tooth (male, 12 years) | [52] |
| Osteogenesis | Human periodontal ligament | [66] | ||
| [67] | ||||
| [68] | ||||
| Angiogenesis | Human impacted third molars | [69] | ||
| Adipogenic | Human periodontal ligament | [70] | ||
| Chondrogenesis | Human periodontal ligament | [70] | ||
|
| ||||
| GMSCs | — | Adipogenic | Human gingiva | [12] |
| Chondrogenesis | Human gingiva | [12] | ||
| Osteogenesis | Human gingiva | [12] | ||
| Human gingiva | [71] | |||
| Human gingiva (16 to 22 years old) | [72] | |||
| Angiogenesis | Human gingiva | [73] | ||
| Neurogenic | Human gingiva (20 to 40 years old) | [74] | ||
|
| ||||
| OMSCs | — | Neurogenic | Rat | [75] |
| Human olfactory mucosa | [76] | |||
| Osteogenesis | Human olfactory mucosa | [76] | ||
| [77] | ||||
|
| ||||
| JMSCs | 50-60 | Osteogenesis | Mouse jaw bone | [78] |
| [35] | ||||
| Adipogenic | Mouse jaw bone | [35] | ||
| Chondrogenesis | Mouse jaw bone | [35] | ||
Abbreviation: PD: population doubling.
4.3. Age-Related Changes
As the application potential of E-MSCs has been gradually explored, the preparation of sufficient E-MSCs has become a research hotspot, but it is also one of the obstacles that hinders the clinical application of E-MSCs. The age of the MSC donor has a great impact on cell proliferation activity, differentiation potential, and paracrine effect, but the specific effect is still not clear; particularly, whether elderly patients can undergo autologous stem cell therapy is currently controversial [79].
At present, research on the age-related changes in E-MSCs mainly focuses on DPSCs, and there are relatively few studies on other stem cells. In the dental pulp of aged individuals, the proportion of cells decreases, and the proportion of fiber and collagen components gradually increases with age [80]. Mitsiadis et al. [81] showed that pulp volume gradually decreases with age due to continuous production of dentin matrix by odontoblasts, which may explain, at least in part, why DPSC extraction from permanent teeth from old donors is less efficient.
Current studies on age-related changes in the biological activity of DPSCs have shown conflicting results, with some studies showing that the proliferation and differentiation potential of MSCs are independent of age [82]. However, other studies have shown that the proliferation ability, differentiation potential, and cell surface marker expression of DPSCs are affected by age [83, 84]. Therefore, young DPSCs should be collected and preserved as soon as possible, as this may be a potential treatment for elderly patients with dental diseases in the future.
4.4. Immunomodulation
4.4.1. Immune Regulation Mechanism of MSCs
MSCs can interact with a variety of immune cells, including T cells, dendritic cells (DCs), B cells, macrophages, neutrophils, and natural killer (NK) cells [85]. Studies have shown that the immunosuppressive effect of MSCs is mainly the result of the joint action of intercellular contact and soluble immune factors [86, 87]. Soluble immune factors, including a variety of immune regulatory factors, cytokines, and growth factors, such as prostaglandin E2 (PGE-2), indoleamine 2,3-dioxygenase (IDO), and nitric oxide (NO), can respond to immune cells and activate the immune regulation of MSCs [88, 89]. In addition, indirect or direct cell contact can also cause the immunosuppressive effects of MSCs, which are mainly mediated by programmed cell death ligand 1, programmed cell death ligand 2, and membrane-bound human leukocyte antigen [90].
MSCs can also induce chemotaxis to inflammatory sites to exert immune regulation and repair damaged cells and tissues [91, 92]. Interestingly, the immunomodulatory effect of MSCs can not only inhibit the immune response but also enhance it, and which effect they have mainly depends on the function of immunosuppressants, the types of inflammatory factors, and the state of the immune system [93]. MSCs not only respond to inflammatory cytokines but also secrete immunoregulatory molecules and participate in the regulation of the inflammatory process. For example, IDO, NO, and chemokines secreted by MSCs play key roles in MSC-mediated immune regulation [94].
4.4.2. Immunomodulatory Effect of E-MSCs
E-MSCs are similar to MSCs from other tissues and can regulate the activities of different immune cells [26, 95]. The immunomodulatory activity of E-MSCs is usually activated by inflammatory cytokines produced by immune cells, which indicates that there is an interaction between E-MSCs and activated immune cells. The interactions between E-MSCs and immune cells are listed in Table 3. MSCs can have a significant impact on immune cells.
Table 3.
The function of E-MSCs in mediating immune cells.
| Immune cell type | E-MSC functions |
|---|---|
| PBMCs | Inhibiting PBMC proliferation |
| DCs | Inhibiting DC differentiation and maturation |
| Macrophage | Activating M2 macrophage polarization in general; activating M1 macrophage polarization in specific microenvironment |
| Mast cells | Inhibiting mast cell exocytosis |
| T cell | Inhibiting T cell proliferation, differentiation, and apoptosis |
| B cell | Inhibiting B cell exocytosis |
Abbreviation: PBMCs: peripheral blood mononuclear cells; DCs: dendritic cells.
(1) Peripheral Blood Mononuclear Cells (PBMCs). Peripheral blood mononuclear cells (PBMCs) are mononuclear cells in peripheral blood, including lymphocytes and monocytes. E-MSCs can inhibit the proliferation of peripheral blood monocytes through paracrine signaling [12, 17, 28, 95–97], and γ-interferon treatment can enhance this ability [95].
(2) Myeloid Dendritic Cells (DCs). Myeloid dendritic cells (DCs) maintain and regulate the immune response by accelerating the process of antigen-specific T cells and the activation of cells in the innate immune response after DC maturation [98, 99]. Studies have shown that E-MSCs have an immunosuppressive function on DCs, which can inhibit DC maturation and differentiation through a prostaglandin E2-dependent mechanism [100, 101].
(3) Mast Cells. Mast cells are widely distributed around microvessels under the skin and visceral mucosa and can secrete a variety of cytokines. It has been reported that E-MSCs can inhibit the release of inflammatory cytokines by mast cell 1 (HMC-1) through a prostaglandin E2-dependent mechanism but have no effect on the proliferation of HMC-1 cells [101].
(4) Macrophages. Macrophages are cells with significant plasticity in the immune system [102], and they can polarize into M1 or M2 macrophages [103]. Generally, M1 macrophages have significant antibacterial properties enacted by the release of a variety of chemokines and inflammatory cytokines, while M2 macrophages can reduce inflammation and accelerate tissue repair by secreting IL-10 and nutritional factors [104]. In addition, macrophages can be cocultured with MSCs to induce M2 macrophages [27, 105]. Transplantation of DPSCs into unilateral hind limb skeletal muscle can inhibit the occurrence of sciatic nerve inflammation [106]. In specific cases, for example, lipopolysaccharide-treated PDLSCs can promote the polarization of macrophages to the inflammatory M1 phenotype [107].
(5) T Cells. T cells are widely distributed in animal and human tissues. Once activated, they can differentiate into helper T cell (Th) 1 and the regulatory T cell (Treg) subsets Th2, Th9, and Th17 according to the stimulation intensity and microenvironment [108, 109]. It has been proven that MSCs have a close relationship with T cells [87, 110].
MSCs secrete a large number of immunosuppressive factors, chemokines, and adhesion molecules that can effectively inhibit the proliferation, apoptosis, and differentiation of T cells [92, 111]. It has been reported that E-MSCs can inhibit T cell proliferation [24, 29, 112, 113], induce T cell apoptosis, and stimulate regulatory T cell differentiation [114]. E-MSCs induce the immunomodulatory effect of T cell apoptosis, which has an anti-inflammatory effect in vivo [115]. Interestingly, although there are few relevant studies comparing the immunomodulatory ability of MSCs from different germ layers, there is evidence that human gingival MSCs have a stronger inhibitory effect on the proliferation and Th1/Th2/Th17 differentiation of mouse CD4+ T cells than BMSCs [26]. Comparison of the immunomodulatory ability of MSCs from different germ layers may be a future research focus of E-MSCs.
(6) B Cells. B cells mainly resist and hunt down foreign pathogens by producing specific antibodies [116, 117]. At present, there are relatively few studies on the effect of E-MSCs on B cells. Kwack et al. [17] found that DPSCs can inhibit the production of immunoglobulin by B cells. However, it has also been reported that MSCs inhibit the production of antibodies by B cells depending on the intensity of inflammatory stimulation and the ratio of BMSCs to B cells [118, 119].
The immunomodulatory properties of MSCs depend on the surrounding microenvironment. Activation of E-MSCs by inflammatory factors, such as γ-interferon, tumor necrosis factor α, and interleukin-1 β, can significantly enhance their immunomodulatory ability [120]. Activated immune cells can upregulate the expression of MSC-related proteins [121]. Activated immune cells play a key role in inducing the immunomodulatory potential of MSCs, and there is a close relationship between these cells.
5. Clinical Application
Studies have proven that E-MSCs are ideal seed cells for tissue engineering. DPSCs have been used to treat severe limb ischemia, tissue defects, and bone necrosis, to regenerate skin damage caused by burns, and to generate liver, nerve, skeletal muscle, blood vessels, and skin [122, 123] and have been shown to have good application prospects [124]. Odontogenic MSCs from third molars, orthodontic teeth, and deciduous teeth have been applied to dentin, periodontal tissue, dental pulp tissue, jaw defect repair, and other in vivo and in vitro studies and have been shown to have good regeneration ability [125].
Due to the same source of dental pulp tissue, E-MSCs have incomparable advantages in the field of dental pulp regeneration compared with M-MSCs, which is also a research hotspot of clinical application of E-MSCs. The majority of tooth loss is due to dental caries and root fractures. At present, root canal therapy is still the main treatment for pulpitis. However, the risk of root fracture is greatly increased due to the lack of nutrition from the pulp of the tooth after endodontic treatment. Therefore, the regenerative restoration of dental pulp has become the goal of functional tooth restoration. Gronthos et al. [8] first demonstrated the ability of DPSCs to differentiate into odontoblasts in 2000. SHEDs can be injected into the dental pulp cavity using injectable scaffold materials, which can not only maintain the nerve activity of dental pulp but also reconstruct the vascularized dental pulp tissue and have the ability to differentiate into odontoblasts [126].
Pulp regeneration of pulpless teeth has always been a dream of dentists and researchers. However, there are still many problems, including the longtime pulp regeneration and the use of scaffold materials that increase the risk of inflammation and infection. The dental pulp regeneration therapy technology established by scaffold-free 3D DPSC constructs avoids the potential problems caused by scaffold materials in transplanted pulp-like tissues [126]. Histological analysis showed that the transplanted DPSC constructs were differentiated into odontoblast-like cells at the site of contact with dentin and were able to form a vascular pulp-like tissue without the need for scaffolds or growth factors. The establishment and development of this technique suggest that the transplantation of DPSCs holds promise for the regeneration of pulp tissue in pulpless teeth.
Odontogenic MSCs also have a strong immunomodulatory effect. They can induce immune tolerance and reduce tissue damage caused by inflammatory reactions, which is conducive to the recovery and prognosis of damaged tissues. They have been applied to the immunomodulatory treatment of a variety of immune system diseases, such as systemic lupus erythematosus, colitis, and multiple sclerosis. After receiving stem cells or their secretions, symptoms related to these diseases can be alleviated. DPSCs are also expected to be able to treat type 2 diabetes and rheumatoid arthritis [127].
By May 2023, more than 12,000 clinical trials of MSCs had been retrieved from the ClinicalTrials.gov website. Internationally, approved MSC drugs have been listed in the United States, South Korea, Japan, and the European Union, and stem cell therapy has become a reality. Although E-MSCs have good biological properties and immunomodulatory ability, it is worth considering that their clinical application is far from that of M-MSCs. According to incomplete statistics, there are 11 MSC drugs approved for marketing worldwide (Table 4), including the United States (1), the European Union (2), Japan (3), South Korea (4), India (1), Australia (1), and Canada (1). From a review of the clinical trials obtained from the ClinicalTrials.gov website, we discovered that all the seed cells of stem cell drugs approved for clinical application are from the mesoderm, and no stem cell drugs from the ectoderm have been listed anywhere in the world.
Table 4.
11 stem cell therapeutic drugs approved for marketing worldwide.
| Country | Trade name | Cell type | Indication | Approved time |
|---|---|---|---|---|
| The United States | Prochymal | Bone marrow mesenchymal stem cells | Graft versus host disease (GVHD), Crohn's disease | 2010.05 |
|
| ||||
| The European Union | Stempeucel | Bone marrow mesenchymal stem cells | Thromboangiitis obliterans | 2015.06 |
| Alofisel | Adipose-derived mesenchymal stem cells | Crohn's disease with complex perianal fistula | 2018.03 | |
|
| ||||
| South Korea | Cell gram | Bone marrow mesenchymal stem cells | Acute myocardial infarction | 2011.07 |
| Cartistem | Umbilical cord blood mesenchymal stem cells | Degenerative arthritis and knee cartilage injury | 2012.01 | |
| Cuepistem | Adipose-derived mesenchymal stem cells | Complex Crohn's disease complicated with anal fistula | 2012.01 | |
| NeuroNATA-R | Bone marrow mesenchymal stem cells | Amyotrophic lateral sclerosis, motor neuron disease | 2014.07 | |
|
| ||||
| Canada | Prochymal | Bone marrow mesenchymal stem cells | Graft versus host disease (GVHD) in children | 2012.05 |
|
| ||||
| Australia | MPC | Autologous mesenchymal precursor cells | Repair of damaged bone tissue | 2010.07 |
|
| ||||
| Japan | Temcell | Bone marrow mesenchymal stem cells | Graft versus host disease (GVHD) | 2016.02 |
| RNL-Astrostem | Adipose-derived mesenchymal stem cells | Alzheimer's disease | 2018.04 | |
| Stemirac | Bone marrow mesenchymal stem cells | Spinal cord injury | 2018.12 | |
|
| ||||
| India | Stempeucel | Bone marrow mesenchymal stem cells | Severe lower limb ischemia caused by Burger's disease | 2017 |
What is the reason for this? We hypothesized that although the biological properties of E-MSCs are more suitable for tissue engineering, researchers have spent a relatively short amount of time researching them. BMSCs were first discovered 50 years ago, but the earliest E-MSCs were discovered approximately 20 years ago. Compared with MSCs from other sources, such as BMSCs and ADSCs, the foundation of E-MSC research is not strong enough, and relevant supporting research is not sufficient. It will take more time to study the biological properties and immune regulation characteristics of E-MSCs. Second, due to the short research time and imperfect supporting conditions, different researchers have different methods to isolate and culture E-MSCs. Different culture conditions, such as serum, cell inoculation density, and oxygen partial pressure, may affect cell proliferation and differentiation potential [128, 129]. Therefore, it is necessary to formulate an international unified standard process for the isolation, extraction, identification, and culture of E-MSCs. The age of donors also affects the proliferation and differentiation potential of MSCs. Studies have shown that MSCs from young donors show less damage and better proliferation [130].
In the field of tissue engineering, scaffold material is an indispensable factor. It can provide an environment for MSCs to perform their functions and is conducive to the further development of the therapeutic role of MSCs. The scaffold material for E-MSCs can improve the therapeutic effect of stem cells, and there is relatively little research in this field, which may also be one of the factors that hinders the further application of E-MSCs in regenerative medicine.
6. Summary
Both E-MSCs and M-MSCs have good self-renewal and multidirectional differentiation potential; are convenient and safe to extract, expand, and preserve; and have fewer ethical concerns. They are potential seed cells for tissue regeneration, repair, and clinical treatment in the future. However, the clinical application of E-MSCs is still limited. Research on the biological role, mechanism, and regulation after entering the host and differentiation into other tissues is still in the initial stage.
E-MSCs are valuable resources for regenerative medicine. The excellent differentiation potential of E-MSCs provides a new opportunity for the development of different research fields such as metabolic diseases, tumors, and injury repair. DPSC culture technology based on tissue engineering 3D scaffolds has great potential in dental pulp tissue regeneration. It is worth mentioning that the advantages of E-MSCs in neuronal differentiation are helpful for the research of many neurodegenerative diseases, such as Alzheimer's disease, Parkinson's disease, TBI, and peripheral nerve injury. E-MSC transplantation may become an effective treatment for restoring neurological function.
Further development of materials, science, molecular biology, and tissue engineering technology combined with increased understanding of the biological properties of MSCs from different germ layers will promote the clinical application of E-MSCs. And E-MSCs are expected to become a mature clinical technology and have a bright application prospect in the field of regenerative medicine, creating new alternative treatment options for a variety of diseases.
Acknowledgments
This study was supported by grants from the National Natural Science Foundation of China (No. 82170986 to LLX).
Contributor Information
Xiaoxia Jiang, Email: smilovjiang@163.com.
Lulu Xu, Email: xululu@301hospital.com.cn.
Data Availability
All the data indicated in this study are available upon request by contacting the corresponding authors.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
References
- 1.Leucht P., Kim J. B., Amasha R., James A. W., Girod S., Helms J. A. Embryonic origin and Hox status determine progenitor cell fate during adult bone regeneration. Development . 2008;135(17):2845–2854. doi: 10.1242/dev.023788. [DOI] [PubMed] [Google Scholar]
- 2.Lloyd B., Tee B. C., Headley C., Emam H., Mallery S., Sun Z. Similarities and differences between porcine mandibular and limb bone marrow mesenchymal stem cells. Archives of Oral Biology . 2017;77:1–11. doi: 10.1016/j.archoralbio.2017.01.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Erices A., Conget P., Minguell J. J. Mesenchymal progenitor cells in human umbilical cord blood. British Journal of Haematology . 2015;109(1):235–242. doi: 10.1046/j.1365-2141.2000.01986.x. [DOI] [PubMed] [Google Scholar]
- 4.Bari C. D., Dell'Accio F., Tylzanowski P., Luyten F. P. Multipotent mesenchymal stem cells from adult human synovial membrane. Arthritis & Rheumatology . 2001;44(8):1928–1942. doi: 10.1002/1529-0131(200108)44:8<1928::AID-ART331>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- 5.Zuk P. A., Zhu M., Mizuno H., et al. Multilineage cells from human adipose tissue: implications for cell-based therapies. Tissue Engineering Part A . 2001;7(2):211–228. doi: 10.1089/107632701300062859. [DOI] [PubMed] [Google Scholar]
- 6.Han Y., Li X., Zhang Y., Han Y., Chang F., Ding J. Mesenchymal stem cells for regenerative medicine. Cell . 2019;8(8) doi: 10.3390/cells8080886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Li T. Q., Meng X. B., Shi Q., Zhang T. Research progress in biological characteristics and influencing factors of jaw bone marrow mesenchymal stem cell. Zhonghua Kou Qiang Yi Xue Za Zhi . 2022;57(1):107–112. doi: 10.3760/cma.j.cn112144-20211009-00457. [DOI] [PubMed] [Google Scholar]
- 8.Gronthos S., Mankani M., Brahim J., Robey P. G., Shi S. Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proceedings of the National Academy of Sciences of the United States of America . 2000;97(25):13625–13630. doi: 10.1073/pnas.240309797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Morsczeck C., Gotz W., Schierholz J., et al. Isolation of precursor cells (PCs) from human dental follicle of wisdom teeth. Matrix Biology . 2005;24(2):155–165. doi: 10.1016/j.matbio.2004.12.004. [DOI] [PubMed] [Google Scholar]
- 10.Sonoyama W., Liu Y., Fang D., et al. Mesenchymal stem cell-mediated functional tooth regeneration in swine. PLoS One . 2006;1(1, article e79) doi: 10.1371/journal.pone.0000079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Seo B.-M., Miura M., Gronthos S., et al. Investigation of multipotent postnatal stem cells from human periodontal ligament. The Lancet . 2004;364(9429):149–155. doi: 10.1016/s0140-6736(04)16627-0. [DOI] [PubMed] [Google Scholar]
- 12.Zhang Q., Shi S., Liu Y., et al. Mesenchymal stem cells derived from human gingiva are capable of immunomodulatory functions and ameliorate inflammation-related tissue destruction in experimental colitis. Journal of Immunology . 2009;183(12):7787–7798. doi: 10.4049/jimmunol.0902318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Johnstone S. A., Liley M., Dalby M. J., Barnett S. C. Comparison of human olfactory and skeletal MSCs using osteogenic nanotopography to demonstrate bone-specific bioactivity of the surfaces. Acta Biomaterialia . 2015;13:266–276. doi: 10.1016/j.actbio.2014.11.027. [DOI] [PubMed] [Google Scholar]
- 14.Shi X., Mao J., Liu Y. Pulp stem cells derived from human permanent and deciduous teeth: biological characteristics and therapeutic applications. Stem Cells Translational Medicine . 2020;9(4):445–464. doi: 10.1002/sctm.19-0398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bakopoulou A., Apatzidou D., Aggelidou E., et al. Isolation and prolonged expansion of oral mesenchymal stem cells under clinical-grade, GMP-compliant conditions differentially affects "stemness" properties. Stem Cell Research & Therapy . 2017;8(1):p. 247. doi: 10.1186/s13287-017-0705-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Laino G., d'Aquino R., Graziano A., et al. A new population of human adult dental pulp stem cells: a useful source of living autologous fibrous bone tissue (LAB) Journal of Bone and Mineral Research . 2005;20(8):1394–1402. doi: 10.1359/jbmr.050325. [DOI] [PubMed] [Google Scholar]
- 17.Kwack K. H., Lee J. M., Park S. H., Lee H. W. Human dental pulp stem cells suppress alloantigen-induced immunity by stimulating T cells to release transforming growth factor Beta. Journal of Endodontics . 2017;43(1):100–108. doi: 10.1016/j.joen.2016.09.005. [DOI] [PubMed] [Google Scholar]
- 18.Hossein-Khannazer N., Hashemi S. M., Namaki S., Ghanbarian H., Khojasteh A. Study of the immunomodulatory effects of osteogenic differentiated human dental pulp stem cells. Life Sciences . 2018;216:111–118. doi: 10.1016/j.lfs.2018.11.040. [DOI] [PubMed] [Google Scholar]
- 19.Li L., Ge J. Exosome-derived lncRNA-Ankrd26 promotes dental pulp restoration by regulating miR-150-TLR4 signaling. Molecular Medicine Reports . 2022;25(5) doi: 10.3892/mmr.2022.12668. [DOI] [PubMed] [Google Scholar]
- 20.Dong W., Ge J., Zhang P., et al. Phenotypic characterization of craniofacial bone marrow stromal cells: unique properties of enhanced osteogenesis, cell recruitment, autophagy, and apoptosis resistance. Cell and Tissue Research . 2014;358(1):165–175. doi: 10.1007/s00441-014-1927-4. [DOI] [PubMed] [Google Scholar]
- 21.Millman J. R., Tan J. H., Colton C. K. The effects of low oxygen on self-renewal and differentiation of embryonic stem cells. Current Opinion in Organ Transplantation . 2009;14(6):694–700. doi: 10.1097/MOT.0b013e3283329d53. [DOI] [PubMed] [Google Scholar]
- 22.Açil Y., Yang F., Gulses A., Ayna M., Wiltfang J., Gierloff M. Isolation, characterization and investigation of differentiation potential of human periodontal ligament cells and dental follicle progenitor cells and their response to BMP-7 in vitro. Odontology . 2016;104(2):123–135. doi: 10.1007/s10266-015-0198-1. [DOI] [PubMed] [Google Scholar]
- 23.Tomic S., Djokic J., Vasilijic S., et al. Immunomodulatory properties of mesenchymal stem cells derived from dental pulp and dental follicle are susceptible to activation by toll-like receptor agonists. Stem Cells and Development . 2011;20(4):695–708. doi: 10.1089/scd.2010.0145. [DOI] [PubMed] [Google Scholar]
- 24.Ding G., Liu Y., An Y., et al. Suppression of T cell proliferation by root apical papilla stem cells in vitro. Cells, Tissues, Organs . 2010;191(5):357–364. doi: 10.1159/000276589. [DOI] [PubMed] [Google Scholar]
- 25.Dahake P. T., Panpaliya N. P., Kale Y. J., Dadpe M. V., Kendre S. B., Bogar C. Response of stem cells from human exfoliated deciduous teeth (SHED) to three bioinductive materials - an in vitro experimental study. The Saudi Dental Journal . 2020;32(1):43–51. doi: 10.1016/j.sdentj.2019.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Yamaza T., Kentaro A., Chen C., et al. Immunomodulatory properties of stem cells from human exfoliated deciduous teeth. Stem Cell Research & Therapy . 2010;1(1) doi: 10.1186/scrt5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Gao X., Shen Z., Guan M., et al. Immunomodulatory Role of Stem Cells from Human Exfoliated Deciduous Teeth on Periodontal Regeneration. Tissue engineering, Part A . 2018;24(17-18):1341–1353. doi: 10.1089/ten.tea.2018.0016. [DOI] [PubMed] [Google Scholar]
- 28.Li C., Wang X., Tan J., Wang T., Wang Q. The immunomodulatory properties of periodontal ligament stem cells isolated from inflamed periodontal granulation. Cells, Tissues, Organs . 2014;199(4):256–265. doi: 10.1159/000367986. [DOI] [PubMed] [Google Scholar]
- 29.Shin C., Kim M., Han J. A., et al. Human periodontal ligament stem cells suppress T-cell proliferation via down- regulation of non-classical major histocompatibility complex-like glycoprotein CD1b on dendritic cells. Journal of Periodontal Research . 2017;52(1):135–146. doi: 10.1111/jre.12378. [DOI] [PubMed] [Google Scholar]
- 30.Jin S. H., Lee J. E., Yun J. H., Kim I., Ko Y., Park J. B. Isolation and characterization of human mesenchymal stem cells from gingival connective tissue. Journal of Periodontal Research . 2015;50(4):461–467. doi: 10.1111/jre.12228. [DOI] [PubMed] [Google Scholar]
- 31.Cicconetti A., Sacchetti B., Bartoli A., et al. Human maxillary tuberosity and jaw periosteum as sources of osteoprogenitor cells for tissue engineering. Oral Surgery, Oral Medicine, Oral Pathology, Oral Radiology, and Endodontics . 2007;104(5):618.e1–618.e12. doi: 10.1016/j.tripleo.2007.02.022. [DOI] [PubMed] [Google Scholar]
- 32.Pu Y., Wang M., Hong Y., Wu Y., Tang Z. Adiponectin promotes human jaw bone marrow mesenchymal stem cell chemotaxis via CXCL1 and CXCL8. Journal of Cellular and Molecular Medicine . 2017;21(7):1411–1419. doi: 10.1111/jcmm.13070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hu H., Pu Y., Lu S., et al. The osteogenesis effect and underlying mechanisms of local delivery of gAPN in extraction sockets of beagle dogs. International Journal of Molecular Sciences . 2015;16(10):24946–24964. doi: 10.3390/ijms161024946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pu Y., Wu H., Lu S., et al. Adiponectin promotes human jaw bone marrow stem cell osteogenesis. Journal of Dental Research . 2016;95(7):769–775. doi: 10.1177/0022034516636853. [DOI] [PubMed] [Google Scholar]
- 35.Akintoye S. O., Lam T., Shi S., Brahim J., Collins M. T., Robey P. G. Skeletal site-specific characterization of orofacial and iliac crest human bone marrow stromal cells in same individuals. Bone . 2006;38(6):758–768. doi: 10.1016/j.bone.2005.10.027. [DOI] [PubMed] [Google Scholar]
- 36.Pan H., Cai N., Li M., Liu G. H., Izpisua Belmonte J. C. Autophagic control of cell 'stemness'. EMBO Molecular Medicine . 2013;5(3):327–331. doi: 10.1002/emmm.201201999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Chai Y., Jiang X., Ito Y., et al. Fate of the mammalian cranial neural crest during tooth and mandibular morphogenesis. Development . 2000;127(8):1671–1679. doi: 10.1242/dev.127.8.1671. [DOI] [PubMed] [Google Scholar]
- 38.Mackie E. J., Ahmed Y. A., Tatarczuch L., Chen K. S., Mirams M. Endochondral ossification: how cartilage is converted into bone in the developing skeleton. The International Journal of Biochemistry & Cell Biology . 2008;40(1):46–62. doi: 10.1016/j.biocel.2007.06.009. [DOI] [PubMed] [Google Scholar]
- 39.Mavropoulos A., Rizzoli R., Ammann P. Different responsiveness of alveolar and tibial bone to bone loss stimuli. Journal of Bone and Mineral Research . 2007;22(3):403–410. doi: 10.1359/jbmr.061208. [DOI] [PubMed] [Google Scholar]
- 40.Aghaloo T. L., Chaichanasakul T., Bezouglaia O., et al. Osteogenic potential of mandibular vs. long-bone marrow stromal cells. Journal of Dental Research . 2010;89(11):1293–1298. doi: 10.1177/0022034510378427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Aanismaa R., Hautala J., Vuorinen A., Miettinen S., Narkilahti S. Human dental pulp stem cells differentiate into neural precursors but not into mature functional neurons. Stem Cell Discovery . 2012;2(3):85–91. doi: 10.4236/scd.2012.23013. [DOI] [Google Scholar]
- 42.Karaöz E., Demircan P. C., Sağlam O., Aksoy A., Kaymaz F., Duruksu G. Human dental pulp stem cells demonstrate better neural and epithelial stem cell properties than bone marrow-derived mesenchymal stem cells. Histochemistry and Cell Biology . 2011;136(4):455–473. doi: 10.1007/s00418-011-0858-3. [DOI] [PubMed] [Google Scholar]
- 43.Kiraly M., Porcsalmy B., Pataki A., et al. Simultaneous PKC and cAMP activation induces differentiation of human dental pulp stem cells into functionally active neurons. Neurochemistry International . 2009;55(5):323–332. doi: 10.1016/j.neuint.2009.03.017. [DOI] [PubMed] [Google Scholar]
- 44.Nosrat I. V., Smith C. A., Mullally P., Olson L., Nosrat C. A. Dental pulp cells provide neurotrophic support for dopaminergic neurons and differentiate into neurons in vitro; implications for tissue engineering and repair in the nervous system. The European Journal of Neuroscience . 2004;19(9):2388–2398. doi: 10.1111/j.0953-816X.2004.03314.x. [DOI] [PubMed] [Google Scholar]
- 45.Song M., Jue S. S., Cho Y. A., Kim E. C. Comparison of the effects of human dental pulp stem cells and human bone marrow-derived mesenchymal stem cells on ischemic human astrocytes in vitro. Journal of Neuroscience Research . 2015;93(6):973–983. doi: 10.1002/jnr.23569. [DOI] [PubMed] [Google Scholar]
- 46.Saharkhiz M., Ayadilord M., Emadian Razavi F., Naseri M. Effects of phytosomal curcumin treatment on modulation of immunomodulatory and pulp regeneration genes in dental pulp mesenchymal stem cells. Odontology . 2022;110(2):287–295. doi: 10.1007/s10266-021-00659-4. [DOI] [PubMed] [Google Scholar]
- 47.Wang H., Sun M., Sun J., Gong P., Liu N., Wang M. Dental pulp stem cell therapy in ischemic stroke: a meta-analysis of preclinical studies. Journal of Stroke and Cerebrovascular Diseases . 2022;31(6, article 106453) doi: 10.1016/j.jstrokecerebrovasdis.2022.106453. [DOI] [PubMed] [Google Scholar]
- 48.Huang A. H., Chen Y. K., Lin L. M., Shieh T. Y., Chan A. W. Isolation and characterization of dental pulp stem cells from a supernumerary tooth. Journal of Oral Pathology & Medicine . 2008;37(9):571–574. doi: 10.1111/j.1600-0714.2008.00654.x. [DOI] [PubMed] [Google Scholar]
- 49.Fujii Y., Kawase-Koga Y., Hojo H., et al. Bone regeneration by human dental pulp stem cells using a helioxanthin derivative and cell-sheet technology. Stem Cell Research & Therapy . 2018;9(1):p. 24. doi: 10.1186/s13287-018-0783-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Tian J., Chen W., Xiong Y., et al. Small extracellular vesicles derived from hypoxic preconditioned dental pulp stem cells ameliorate inflammatory osteolysis by modulating macrophage polarization and osteoclastogenesis. Bioactive Materials . 2023;22:326–342. doi: 10.1016/j.bioactmat.2022.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Angelopoulos I., Brizuela C., Khoury M. Gingival mesenchymal stem cells outperform haploidentical dental pulp-derived mesenchymal stem cells in proliferation rate, migration ability, and angiogenic potential. Cell Transplantation . 2018;27(6):967–978. doi: 10.1177/0963689718759649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Mercado-Rubio M. D., Pérez-Argueta E., Zepeda-Pedreguera A., et al. Similar features, different behaviors: a comparative in vitro study of the adipogenic potential of stem cells from human follicle, dental pulp, and periodontal ligament. Journal of Personalized Medicine . 2021;11(8):p. 738. doi: 10.3390/jpm11080738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lin L., Zheng Y., Wang C., Li P., Xu D., Zhao W. Concentration-dependent cellular uptake of graphene oxide quantum dots promotes the odontoblastic differentiation of dental pulp cells via the AMPK/mTOR pathway. ACS Omega . 2023;8(6):5393–5405. doi: 10.1021/acsomega.2c06508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sramkó B., Földes A., Kádár K., Varga G., Zsembery Á., Pircs K. The wisdom in teeth: neuronal differentiation of dental pulp cells. Cellular Reprogramming . 2023;25(1):32–44. doi: 10.1089/cell.2022.0102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Yang C., Sun L., Li X., et al. The potential of dental stem cells differentiating into neurogenic cell lineage after cultivation in different modes in vitro. Cellular Reprogramming . 2014;16(5):379–391. doi: 10.1089/cell.2014.0026. [DOI] [PubMed] [Google Scholar]
- 56.Lott K., Collier P., Ringor M., Howard K. M., Kingsley K. Administration of epidermal growth factor (EGF) and basic fibroblast growth factor (bFGF) to induce neural differentiation of dental pulp stem cells (DPSC) isolates. Biomedicine . 2023;11(2) doi: 10.3390/biomedicines11020255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Bai Y., Cheng X., Liu X., et al. Transforming growth factor-β1 promotes early odontoblastic differentiation of dental pulp stem cells via activating AKT, Erk1/2 and p38 MAPK pathways. Journal of Dental Sciences . 2023;18(1):87–94. doi: 10.1016/j.jds.2022.06.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Matsui M., Kobayashi T., Tsutsui T. W. CD146 positive human dental pulp stem cells promote regeneration of dentin/pulp-like structures. Human Cell . 2018;31(2):127–138. doi: 10.1007/s13577-017-0198-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Genç D., Sezer Kürkçü M., Yiğittürk G., et al. Synovial fluid niche promoted differentiation of dental follicle mesenchymal stem cells toward chondrogenesis in rheumatoid arthritis. Archives of Rheumatology . 2022;37(1):94–109. doi: 10.46497/ArchRheumatol.2022.8891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Shoi K., Aoki K., Ohya K., Takagi Y., Shimokawa H. Characterization of pulp and follicle stem cells from impacted supernumerary maxillary incisors. Pediatric Dentistry . 2014;36(3):79–84. [PubMed] [Google Scholar]
- 61.Rezai Rad M., Liu D., He H., et al. The role of dentin matrix protein 1 (DMP1) in regulation of osteogenic differentiation of rat dental follicle stem cells (DFSCs) Archives of Oral Biology . 2015;60(4):546–556. doi: 10.1016/j.archoralbio.2014.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Liang H., Li Q., Wang N., et al. KDM4D enhances osteo/dentinogenic differentiation and migration of SCAPs via binding to RPS5. Oral Diseases . 2022 doi: 10.1111/odi.14479. [DOI] [PubMed] [Google Scholar]
- 63.Zhuang X., Ji L., Jiang H., et al. Exosomes derived from stem cells from the apical papilla promote dentine-pulp complex regeneration by inducing specific dentinogenesis. Stem Cells International . 2020;2020:10. doi: 10.1155/2020/5816723.5816723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Miura M., Gronthos S., Zhao M., et al. SHED: stem cells from human exfoliated deciduous teeth. Proceedings of the National Academy of Sciences of the United States of America . 2003;100(10):5807–5812. doi: 10.1073/pnas.0937635100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Wu J., Chen L., Wang R., et al. Exosomes secreted by stem cells from human exfoliated deciduous teeth promote alveolar bone defect repair through the regulation of angiogenesis and osteogenesis. ACS Biomaterials Science & Engineering . 2019;5(7):3561–3571. doi: 10.1021/acsbiomaterials.9b00607. [DOI] [PubMed] [Google Scholar]
- 66.Wang A., Liu J., Zhuang X., et al. Identification and comparison of piRNA expression profiles of exosomes derived from human stem cells from the apical papilla and bone marrow mesenchymal stem cells. Stem Cells and Development . 2020;29(8):511–520. doi: 10.1089/scd.2019.0277. [DOI] [PubMed] [Google Scholar]
- 67.Xu X. Y., Tian B. M., Xia Y., et al. Exosomes derived from P2X7 receptor gene-modified cells rescue inflammation-compromised periodontal ligament stem cells from dysfunction. Stem Cells Translational Medicine . 2020;9(11):1414–1430. doi: 10.1002/sctm.19-0418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Liu T., Hu W., Zou X., et al. Human periodontal ligament stem cell-derived exosomes promote bone regeneration by altering MicroRNA profiles. Stem Cells International . 2020;2020:13. doi: 10.1155/2020/8852307.8852307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhang Z., Shuai Y., Zhou F., et al. PDLSCs regulate angiogenesis of periodontal ligaments via VEGF transferred by exosomes in periodontitis. International Journal of Medical Sciences . 2020;17(5):558–567. doi: 10.7150/ijms.40918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Gay I. C., Chen S., MacDougall M. Isolation and characterization of multipotent human periodontal ligament stem cells. Orthodontics & Craniofacial Research . 2007;10(3):149–160. doi: 10.1111/j.1601-6343.2007.00399.x. [DOI] [PubMed] [Google Scholar]
- 71.Wu W., Xiao Z., Chen Y., et al. CD39 produced from human GMSCs regulates the balance of osteoclasts and osteoblasts through the Wnt/β-catenin pathway in osteoporosis. Molecular Therapy . 2020;28(6):1518–1532. doi: 10.1016/j.ymthe.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Hasani-Sadrabadi M. M., Sarrion P., Pouraghaei S., et al. An engineered cell-laden adhesive hydrogel promotes craniofacial bone tissue regeneration in rats. Science Translational Medicine . 2020;12(534) doi: 10.1126/scitranslmed.aay6853. [DOI] [PubMed] [Google Scholar]
- 73.Jin S., Yang C., Huang J., et al. Conditioned medium derived from FGF-2-modified GMSCs enhances migration and angiogenesis of human umbilical vein endothelial cells. Stem Cell Research & Therapy . 2020;11(1):p. 68. doi: 10.1186/s13287-020-1584-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Zhang Q., Burrell J. C., Zeng J., et al. Implantation of a nerve protector embedded with human GMSC-derived Schwann-like cells accelerates regeneration of crush-injured rat sciatic nerves. Stem Cell Research & Therapy . 2022;13(1):p. 263. doi: 10.1186/s13287-022-02947-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Girard S. D., Devéze A., Nivet E., Gepner B., Roman F. S., Féron F. Isolating nasal olfactory stem cells from rodents or humans. Journal of Visualized Experiments . 2011;54(54) doi: 10.3791/2762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Delorme B., Nivet E., Gaillard J., et al. The human nose harbors a niche of olfactory ectomesenchymal stem cells displaying neurogenic and osteogenic properties. Stem Cells and Development . 2010;19(6):853–866. doi: 10.1089/scd.2009.0267. [DOI] [PubMed] [Google Scholar]
- 77.Murrell W., Féron F., Wetzig A., et al. Multipotent stem cells from adult olfactory mucosa. Developmental Dynamics . 2005;233(2):496–515. doi: 10.1002/dvdy.20360. [DOI] [PubMed] [Google Scholar]
- 78.Son C., Choi M. S., Park J. C. Different responsiveness of alveolar bone and long bone to epithelial-mesenchymal interaction-related factor. JBMR Plus . 2020;4(8, article e10382) doi: 10.1002/jbm4.10382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Park J. S., Park G., Hong H. S. Age affects the paracrine activity and differentiation potential of human adipose-derived stem cells. Molecular Medicine Reports . 2021;23(2) doi: 10.3892/mmr.2020.11799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Morse D. R. Age-related changes of the dental pulp complex and their relationship to systemic aging. Oral Surgery, Oral Medicine, and Oral Pathology . 1991;72(6):721–745. doi: 10.1016/0030-4220(91)90019-9. [DOI] [PubMed] [Google Scholar]
- 81.Mitsiadis T. A., De Bari C., About I. Apoptosis in developmental and repair-related human tooth remodeling: a view from the inside. Experimental Cell Research . 2008;314(4):869–877. doi: 10.1016/j.yexcr.2007.11.001. [DOI] [PubMed] [Google Scholar]
- 82.Sato M., Kawase-Koga Y., Yamakawa D., Fujii Y., Chikazu D. Bone regeneration potential of human dental pulp stem cells derived from elderly patients and osteo-induced by a helioxanthin derivative. International Journal of Molecular Sciences . 2020;21(20):p. 7731. doi: 10.3390/ijms21207731. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Kretlow J. D., Jin Y. Q., Liu W., et al. Donor age and cell passage affects differentiation potential of murine bone marrow-derived stem cells. BMC Cell Biology . 2008;9(1):p. 60. doi: 10.1186/1471-2121-9-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wu W., Zhou J., Xu C. T., Zhang J., Jin Y. J., Sun G. L. Derivation and growth characteristics of dental pulp stem cells from patients of different ages. Molecular Medicine Reports . 2015;12(4):5127–5134. doi: 10.3892/mmr.2015.4106. [DOI] [PubMed] [Google Scholar]
- 85.Wang Y., Chen X., Cao W., Shi Y. Plasticity of mesenchymal stem cells in immunomodulation: pathological and therapeutic implications. Nature Immunology . 2014;15(11):1009–1016. doi: 10.1038/ni.3002. [DOI] [PubMed] [Google Scholar]
- 86.Kean T. J., Lin P., Caplan A. I., Dennis J. E. MSCs: delivery routes and engraftment, cell-targeting strategies, and immune modulation. Stem Cells International . 2013;2013:13. doi: 10.1155/2013/732742.732742 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Matsumura-Kawashima M., Ogata K., Moriyama M., Murakami Y., Kawado T., Nakamura S. Secreted factors from dental pulp stem cells improve Sjögren's syndrome via regulatory T cell-mediated immunosuppression. Stem Cell Research & Therapy . 2021;12(1):p. 182. doi: 10.1186/s13287-021-02236-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Doaa A., Han V. Different effects of insulin-like growth factor-1 and insulin-like growth factor-2 on myogenic differentiation of human mesenchymal stem cells. Stem Cells International . 2017;2017:15. doi: 10.1155/2017/8286248.8286248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Gebler A., Zabel O., Seliger B. The immunomodulatory capacity of mesenchymal stem cells. Trends in Molecular Medicine . 2012;18(2):128–134. doi: 10.1016/j.molmed.2011.10.004. [DOI] [PubMed] [Google Scholar]
- 90.Tipnis S., Viswanathan C., Majumdar A. S. Immunosuppressive properties of human umbilical cord-derived mesenchymal stem cells: role of B7-H1 and IDO. Immunology and Cell Biology . 2010;88(8):795–806. doi: 10.1038/icb.2010.47. [DOI] [PubMed] [Google Scholar]
- 91.Cui S. J., Zhang T., Fu Y., et al. DPSCs attenuate experimental progressive TMJ arthritis by inhibiting the STAT1 pathway. Journal of Dental Research . 2020;99(4):446–455. doi: 10.1177/0022034520901710. [DOI] [PubMed] [Google Scholar]
- 92.Ren G., Zhang L., Xin Z., et al. Mesenchymal stem cell-mediated immunosuppression occurs via concerted action of chemokines and nitric oxide. Cell Stem Cell . 2008;2(2):141–150. doi: 10.1016/j.stem.2007.11.014. [DOI] [PubMed] [Google Scholar]
- 93.Anzalone R., Iacono M. L., Corrao S., et al. New emerging potentials for human Wharton's jelly mesenchymal stem cells: immunological features and hepatocyte-like differentiative capacity. Stem Cells and Development . 2010;19(4):423–438. doi: 10.1089/scd.2009.0299. [DOI] [PubMed] [Google Scholar]
- 94.Wada N., Menicanin D., Shi S., Bartold P. M., Gronthos S. Immunomodulatory properties of human periodontal ligament stem cells. Journal of Cellular Physiology . 2009;219(3):667–676. doi: 10.1002/jcp.21710. [DOI] [PubMed] [Google Scholar]
- 95.Struys T., Moreels M., Martens W., Donders R., Wolfs E., Lambrichts I. Ultrastructural and immunocytochemical analysis of multilineage differentiated human dental pulp- and umbilical cord-derived mesenchymal stem cells. Cells, Tissues, Organs . 2011;193(6):366–378. doi: 10.1159/000321400. [DOI] [PubMed] [Google Scholar]
- 96.Xia Y., Tang H. N., Wu R. X., Yu Y., Gao L. N., Chen F. M. Cell responses to conditioned media produced by patient-matched stem cells derived from healthy and inflamed periodontal ligament tissues. Journal of Periodontology . 2015;87(5):e53–e63. doi: 10.1902/jop.2015.150462. [DOI] [PubMed] [Google Scholar]
- 97.Bassi Ê. J., Aita C. A. M., Câmara N. O. S. Immune regulatory properties of multipotent mesenchymal stromal cells: where do we stand? World Journal of Stem Cells . 2011;3(1):1–8. doi: 10.4252/wjsc.v3.i1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Tuettenberg A., Becker C., Correll A., Steinbrink K., Jonuleit H. Immune regulation by dendritic cells and T cells-basic science, diagnostic, and clinical application. Clinical Laboratory . 2011;57(1-2):1–12. [PubMed] [Google Scholar]
- 99.Silva F. D. S., Ramos R. N., Almeida D. C. D., et al. Mesenchymal stem cells derived from human exfoliated deciduous teeth (SHEDs) induce immune modulatory profile in monocyte-derived dendritic cells. PLoS One . 2014;9(5, article e98050) doi: 10.1371/journal.pone.0098050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Su W. R., Zhang Q. Z., Shi S. H., Nguyen A. L., Le A. D. Human gingiva-derived mesenchymal stromal cells attenuate contact hypersensitivity via prostaglandin E2-dependent mechanisms. Stem Cells . 2011;29(11):1849–1860. doi: 10.1002/stem.738. [DOI] [PubMed] [Google Scholar]
- 101.Wynn T. A., Vannella K. M. Macrophages in tissue repair, regeneration, and fibrosis. Immunity . 2016;44(3):450–462. doi: 10.1016/j.immuni.2016.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Glass C. K., Natoli G. Molecular control of activation and priming in macrophages. Nature Immunology . 2015;17(1):26–33. doi: 10.1038/ni.3306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Mosser D. M., Edwards J. P. Exploring the full spectrum of macrophage activation. Nature Reviews Immunology . 2008;8(12):958–969. doi: 10.1038/nri2448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Zhang Q. Z., Su W. R., Shi S. H., et al. Human gingiva-derived mesenchymal stem cells elicit polarization of M2 macrophages and enhance cutaneous wound healing. Stem Cells . 2010;28(10):1856–1868. doi: 10.1002/stem.503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Omi M., Hata M., Nakamura N., et al. Transplantation of dental pulp stem cells suppressed inflammation in sciatic nerves by promoting macrophage polarization towards anti-inflammation phenotypes and ameliorated diabetic polyneuropathy. Journal of Diabetes Investigation . 2016;7(4):485–496. doi: 10.1111/jdi.12452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Zhao L., Kang I., Fang X., et al. Gamma-tocotrienol attenuates high-fat diet-induced obesity and insulin resistance by inhibiting adipose inflammation and M1 macrophage recruitment. International Journal of Obesity . 2015;39(3):438–446. doi: 10.1038/ijo.2014.124. [DOI] [PubMed] [Google Scholar]
- 107.Kaech S. M., Cui W. Transcriptional control of effector and memory CD8+ T cell differentiation. Nature Reviews Immunology . 2015;261(1):157–168. doi: 10.1038/nri3307. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Soroosh P., Doherty T. A. Th9 and allergic disease. Immunology . 2009;127(4):450–458. doi: 10.1111/j.1365-2567.2009.03114.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.O'Garra A. Cytokines induce the development of functionally heterogeneous T helper cell subsets. Immunity . 1998;8(3):275–283. doi: 10.1016/S1074-7613(00)80533-6. [DOI] [PubMed] [Google Scholar]
- 110.Ren G., Xin Z., Zhang L., et al. Inflammatory cytokine-induced intercellular adhesion molecule-1 and vascular cell adhesion molecule-1 in mesenchymal stem cells are critical for immunosuppression. Journal of Immunology . 2010;184(5):2321–2328. doi: 10.4049/jimmunol.0902023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Özdemir A. T., Özgül Özdemir R. B., Kırmaz C., et al. The paracrine immunomodulatory interactions between the human dental pulp derived mesenchymal stem cells and CD4 T cell subsets. Cellular Immunology . 2016;310:108–115. doi: 10.1016/j.cellimm.2016.08.008. [DOI] [PubMed] [Google Scholar]
- 112.Liu D., Xu J., Liu O., et al. Mesenchymal stem cells derived from inflamed periodontal ligaments exhibit impaired immunomodulation. Journal of Clinical Periodontology . 2012;39(12):1174–1182. doi: 10.1111/jcpe.12009. [DOI] [PubMed] [Google Scholar]
- 113.Demircan P. C., Sariboyaci A. E., Unal Z. S., Gacar G., Subasi C., Karaoz E. Immunoregulatory effects of human dental pulp-derived stem cells on T cells: comparison of transwell co-culture and mixed lymphocyte reaction systems. Cytotherapy . 2011;13(10):1205–1220. doi: 10.3109/14653249.2011.605351. [DOI] [PubMed] [Google Scholar]
- 114.Zhao Y., Wang L., Jin Y., Shi S. Fas ligand regulates the immunomodulatory properties of dental pulp stem cells. Journal of Dental Research . 2012;91(10):948–954. doi: 10.1177/0022034512458690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Depoil D., Weber M., Treanor B., et al. Early events of B cell activation by AntigenA presentation from the 12th Joint Meeting of the Signal Transduction Society, Weimar, Germany, 29 to 31 October 2008. Science Signaling . 2009;2(63) doi: 10.1126/scisignal.263pt1. [DOI] [PubMed] [Google Scholar]
- 116.Silva N. S., Klein U. Dynamics of B cells in germinal centres. Nature Reviews Immunology . 2015;15(3):137–148. doi: 10.1038/nri3804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Bernardo M. E., Locatelli F., Fibbe W. E. Mesenchymal stromal cells. Annals of the New York Academy of Sciences . 2009;1176(1):101–117. doi: 10.1111/j.1749-6632.2009.04607.x. [DOI] [PubMed] [Google Scholar]
- 118.Krampera M., Cosmi L., Angeli R., et al. Role for interferon-γ in the immunomodulatory activity of human bone marrow mesenchymal stem cells. Stem Cells . 2006;24(2):386–398. doi: 10.1634/stemcells.2005-0008. [DOI] [PubMed] [Google Scholar]
- 119.Krampera M. Mesenchymal stromal cell 'licensing': a multistep process. Leukemia . 2011;25(9):1408–1414. doi: 10.1038/leu.2011.108. [DOI] [PubMed] [Google Scholar]
- 120.Andrukhov O., Hong J. S. A., Andrukhova O., Blufstein A., Moritz A., Rausch-Fan X. Response of human periodontal ligament stem cells to IFN-γ and TLR-agonists. Scientific Reports . 2017;7(1):p. 12856. doi: 10.1038/s41598-017-12480-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Kadar K., Kiraly M., Porcsalmy B., et al. Differentiation potential of stem cells from human dental origin - promise for tissue engineering. Journal of Physiology and Pharmacology . 2009;60(Supplement 7):167–175. [PubMed] [Google Scholar]
- 122.Uzunoglu-Ozyurek E., Önal G., Dökmeci S. Odonto/osteogenic differentiation of dental pulp stem cells of type 1 diabetic patients with mineral trioxide aggregate/1α,25-dihydroxyvitamin D3 combination. Journal of Endodontia . 2022;48(4):516–526. doi: 10.1016/j.joen.2022.01.010. [DOI] [PubMed] [Google Scholar]
- 123.Staniowski T., Zawadzka-Knefel A., Skośkiewicz-Malinowska K. Therapeutic potential of dental pulp stem cells according to different transplant types. Molecules . 2021;26(24):p. 7423. doi: 10.3390/molecules26247423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Ledesma-Martínez E., Mendoza-Núñez V. M., Santiago-Osorio E. Mesenchymal stem cells derived from dental pulp: a review. Stem Cells International . 2016;2016:12. doi: 10.1155/2016/4709572.4709572 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Itoh Y., Sasaki J. I., Hashimoto M., Katata C., Hayashi M., Imazato S. Pulp regeneration by 3-dimensional dental pulp stem cell constructs. Journal of Dental Research . 2018;97(10):1137–1143. doi: 10.1177/0022034518772260. [DOI] [PubMed] [Google Scholar]
- 126.Ishikawa J., Takahashi N., Matsumoto T., et al. Factors secreted from dental pulp stem cells show multifaceted benefits for treating experimental rheumatoid arthritis. Bone . 2016;83:210–219. doi: 10.1016/j.bone.2015.11.012. [DOI] [PubMed] [Google Scholar]
- 127.Berniakovich I., Giorgio M. Low oxygen tension maintains multipotency, whereas normoxia increases differentiation of mouse bone marrow stromal cells. International Journal of Molecular Sciences . 2013;14(1):2119–2134. doi: 10.3390/ijms14012119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Tsutsumi S., Shimazu A., Miyazaki K., et al. Retention of multilineage differentiation potential of mesenchymal cells during proliferation in response to FGF. Biochemical and Biophysical Research Communications . 2001;288(2):413–419. doi: 10.1006/bbrc.2001.5777. [DOI] [PubMed] [Google Scholar]
- 129.Stolzing A., Jones E., McGonagle D., Scutt A. Age-related changes in human bone marrow-derived mesenchymal stem cells: consequences for cell therapies. Mechanisms of Ageing and Development . 2008;129(3):163–173. doi: 10.1016/j.mad.2007.12.002. [DOI] [PubMed] [Google Scholar]
- 130.Su J., Chen X., Huang Y., et al. Phylogenetic distinction of iNOS and IDO function in mesenchymal stem cell- mediated immunosuppression in mammalian species. Cell Death & Differentiation . 2014;21(3):388–396. doi: 10.1038/cdd.2013.149. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All the data indicated in this study are available upon request by contacting the corresponding authors.


