Abstract
Introduction:
Transperineal laser ablation (TPLA) of the prostate is a new, minimally invasive technique for benign prostatic hyperplasia (BPH) with promising effectiveness and safety outcomes. This systematic review aims to provide an update of existing literature.
Methods:
A literature review was performed in Pubmed/MEDLINE, Embase, Cochrane Library, and clinicaltrials.gov from January 2000 up to April 2023. Data extraction and risk of bias were performed independently by three authors.
Results:
A total of 11 studies were included, among which 9 were observational, 1 randomized controlled trial, 1 animal study, while 2 of them were comparative (1 with prostatic artery embolization and 1 with transurethral resection of the prostate). Functional outcomes were improved in the majority of studies both for objective (maximum flow rate and post-void residual) and subjective outcomes (improvement of International Prostate Symptom Score and quality of life). Complication rates ranged between 1.9% and 2.3% for hematuria, 3.7% and 36.3% for dysuria, 1.9% and 19% for acute urinary retention, 0.6% and 9.1% for orchitis/urinary tract infections, and 0.6% and 4.8% for prostatic abscess formation. Regarding sexual function, >95% of patients retained their ejaculation while erectile function was maintained or improved.
Conclusion:
TPLA of the prostate is an innovative, minimally invasive technique for managing patients with BPH. Existing studies indicate an effective technique in reducing International Prostate Symptom Score and quality of life scores, post-void residual reduction, and increase in Qmax, albeit the measured improvements in terms of Qmax are not equal to transurethral resection of the prostate. Although sexual function is maintained, the mean catheterization time is 7 days, and no long-term data are available for most patients.
Keywords: BPH, laser, laser ablation, LUTS, prostate, TPLA
Introduction
Benign prostatic hyperplasia (BPH) is a very common clinical condition with a progressive natural history in men aged over 45–50 years. The reported prevalence among men aged over 60 years is up to 40%. 1 Lower urinary tract symptoms (LUTS) and coexisting erectile dysfunction in up to 50–60% are the main symptoms while patients may also encounter serious complications such as recurrent infections, bleeding, or renal failure.1,2 Treatment algorithm entails a stepwise approach with conservative management for mild symptoms, monotherapy, or combined pharmacotherapy for moderate symptoms (α-blockers, 5-α-reductase inhibitors, β3-adrenergic agonists, anticholinergics, Phosphodiesterase-5 (PDE5)-inhibitors) and surgical management for severe symptoms, recurrent disease, or in case of complications. 1
Transurethral resection of the prostate (TURP) has been the gold-standard treatment for many and has withstood the test of time, but nowadays enucleation techniques using several energy types (holmium: YAG-HoLEP, thulium laser – ThuLEP, bipolar energy – bipoLEP) are proved to be at least as effective as resection, while a recent network meta-analysis indicates that they may be even superior to TURP.3,4 TURP is a technique indicated for prostates sized up to 80–100 ml, is accompanied by excellent functional outcomes, but is also associated with side effects such as bleeding, urethral strictures, infections, bladder neck contracture and, importantly, permanent sexual dysfunction, mainly in the form of retrograde ejaculation in more than 60% of the patients. 5 Since quality of life (QoL) is greatly affected by sexual function, especially in younger men, TURP sustainable effectiveness is often hindered by these side effects. Enucleation techniques with laser/bipolar energy are also commonly accompanied by sexual dysfunction. 1
In the last 10 years, a number of minimally invasive surgical therapies (MIST) for BPH have emerged: Aquablation therapy, convective water vapor therapy (Rezum), temporary implantable nitinol device (iTIND), Urolift, and prostatic artery embolization (PAE). 1 Although these techniques offer inferior clinical outcomes compared to TURP and enucleation techniques, it is the preservation of sexual function and ability to perform them in an outpatient setting, which have rendered them a quite popular option among the male population. One of the newest MIST is transperineal laser ablation (TPLA) of the prostate, which is still not incorporated in the European Association of Urology Guidelines, but accumulating evidence indicate that it is an effective and safe method. In this systematic review of the literature, we aim to provide an updated summary of the existing evidence on TPLA.
Methods
Search strategy
This systematic review was conducted according to the Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) statement. 6 A literature search was performed using Pubmed/MEDLINE, Embase, Cochrane Library for Clinical Trials, and clinicaltrails.gov for studies focusing on TPLA and published in the English language between January 2000 and April 2023. Reference lists of all included studies were screened as well. The following key terms were used in combination: benign prostatic enlargement, BPH, bladder outlet obstruction, BPE, BPH, BOO, LUTS, and TPLA. Duplicates were removed using a citation manager tool (Clarivate Analytics, version 20).
Selection of studies, assessment of risk of bias, and data extraction
Two authors (LT and SN) performed an independent screen of the literature by title and abstract, to identify eligible studies. After initial screening, a full-text evaluation followed to decide the studies to be included. All disagreements were dissolved upon consensus with a third author (AP). Eligible studies were observational, either prospective or retrospective, single arm or comparative, and randomized controlled trials (RCTs). Animal studies, case reports, conference abstracts, trial protocols with no reported outcomes, editorial comments, non-English studies, and those with a sample size less than 10 patients were excluded.
Data extraction was performed independently by three authors (LT, SN, and AP) using a predefined Excel spreadsheet, which included baseline study characteristics (author name, year of publication, country, study design, inclusion and exclusion criteria, description of technique, sample size, and follow-up) and patient characteristics [age, prostate volume (PV), International Prostate Symptom Score (IPSS) at baseline and postoperatively, post-void residual (PVR) at baseline and postoperatively, maximum flow rate (Qmax) at baseline and postoperatively, questionnaire responses and scores, complication rates, length of catheterization, and hospital stay]. Any disagreements were resolved upon consensus among the three authors after carefully revising the eligible studies.
Risk of bias (RoB) was assessed independently by three authors (LT, SN, and AP) using the Cochrane tools for non-randomized studies (Risk Of Bias in Non-randomised Studies of Interventions - I (ROBINS)). 7 For RCTs, the updated version of the Cochrane risk-of-bias tool for randomized trials (RoB 2) was used. 8 The quality of this systematic review has been evaluated using the updated AMSTAR 2. 9
Results
Study selection
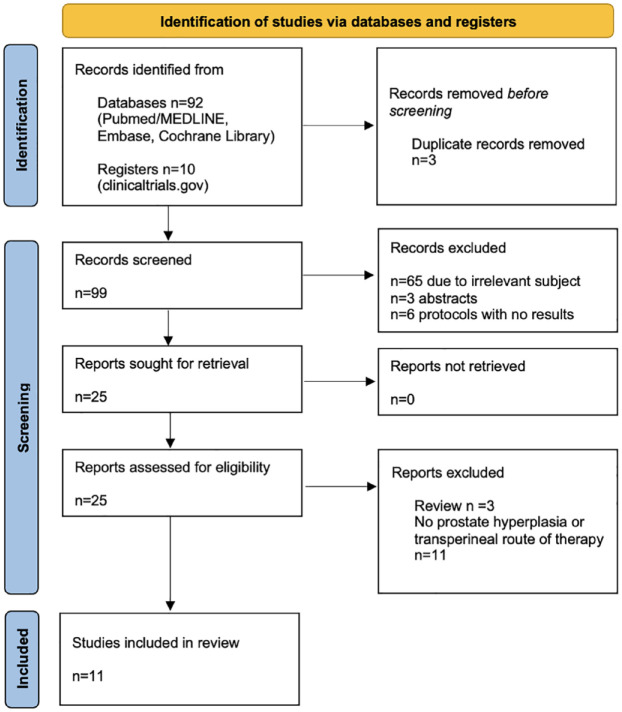
The literature search revealed a total of 102 studies among the four databases. After the removal of 3 duplicates, 99 records were screened by title and abstract. After excluding 74 records (65 due to irrelevant subjects, 3 conference abstracts, and 6 protocols with no published outcomes), 25 full texts were screened for eligibility. Finally, after excluding 3 reviews and 11 studies not on BPH or transperineal route of therapy, 11 studies were deemed eligible for inclusion: 1 RCT, 10 1 animal study, 11 and 9 observational studies,12–20 from which one was comparative. 12 Since only two comparative studies were found, one of which was an RCT and they compared TPLA with either PAE or TURP, no meta-analysis was performed. PRISMA flow diagram is shown in Figure 1. Baseline study characteristics are depicted in Table 1. RoB for selected studies is shown in Supplemental Tables 1–3. A MeaSurement Tool to Assess systematic Reviews 2 scale is shown in Supplemental Files.
Figure 1.
Preferred Reporting Items for Systematic Review and Meta-Analyses (PRISMA) flowchart of the included studies.
Table 1.
Baseline study characteristics.
| Study details author/year | Study design | Inclusion | Follow-up (months) | Technique |
|---|---|---|---|---|
| Patelli/2017 | Prospective single-center pilot trial | LUTS and at least one of the following criteria: (a) male subject >50 years who have symptomatic BPH; (b) IPSS score ⩾ 13; (c) prostate volume ⩾30 ml on transrectal ultrasonographic images; (d) peak urinary flow rate: ⩾5 to ⩽15 ml/s; (e) PVR ⩾50 ml | 3 | - Lithotomy position - Three-way 18-F Foley catheter was inserted and followed by continuous irrigation with saline during and after the maneuver - Using a two-plane TRUS probe (TRT 33, Esaote, Genoa, Italy) up to two 21G introducer needles for each lobe were inserted in the para-urethral sites and placed on planes as parallel as possible to the longitudinal plane of the prostate - For prostate volume <40 ml, two fibers were used, while four fibers were used for prostates larger than 40 ml - 300-lm bare flat-tip optical laser fiber was introduced and advanced up to needle tip. The introducer needle was designed to expose the fiber tip of 5 mm. Care was taken to maintain a safety distance of 8 mm from the needle to the outer wall of the urethra and of at least 15 mm from the needle tip to the bladder floor - The applicators were inserted one at a time, spaced at least 1.5–2.0 cm one to the other - The optical fibers were then connected with a continuous wave diode laser source operating at 1064 nm (Echolaser XVG system; Elesta s.r.l. 50041, Calenzano, Italy) - A fix power protocol (3 W) changing the illumination time case by case according to prostate size. Each ablation time ranged from a minimum of 400 s to a maximum of 600 s to maintain the total energy applied between 1200 and 1800 J per fiber - Depending on the size of the prostate, from one to two consecutive illuminations were performed with a ‘pull-back’ technique - Treatment was concluded when the gas formed during the ablation covered the entire desired area or 1800 J per illumination was reached |
| Pacella/2019 | Multicentric retrospective study | - >50 years with IPSS ⩾ 12, maximum urinary flow rate <15 ml, estimated prostate volume >30 ml on transrectal ultrasonographic, PVR urine volume <400 ml | 12 | - Lithotomy position - Three-way Foley 18-F catheter was inserted with continuous irrigation of a saline solution during and after the maneuver - Positioning up to four applicators (one to two per lobe, depending on prostate volume and shape) - 21G Chiba needle (Sterylab, Rho, Milano, Italy) as an introducer in whose lumen is inserted a bare optic fiber of quartz of 300 μm until it protrudes by 10 mm from the tip of the thin introducer which was done under ultrasound guidance - The needle insertion and positioning are performed using a dedicated guidance device of the biplanar probe - The optic fibers were connected to a multisource laser system operating at 1.064 nm (EchoLaser X4, Elesta s.r.l., Calenzano, Italy) - Volumes less than or equal to 40 ml, a single applicator per lobe can be inserted; while in the case of a prostate with a volume greater than 40 ml two applicators per lobe are used - The tip of the fiber should be placed 8–10 mm away from the outer wall of the urethra, 15 mm from the bottom of the bladder, and 10 mm from the outer edge of the prostate capsule - In the case of two applicators, these must be positioned one after the other at a mutual distance of 8–10 mm - Applicators must be positioned along a path that is as parallel as possible to the longitudinal plane of the prostate. - Fixed power of 3 W changing the illumination time case by case according to prostate size. One to three consecutive illuminations are performed with a ‘pullback’ technique (retraction of the fibers of ~10–12 mm) during the same treatment session - The treatment ends when 1800 J is reached for a single illumination for each single fiber (3600 J for two illuminations, 5400 J for three illuminations). So the time needed to release the maximum energy dose ranged 600–1800 s (total energy 3600–21,600 J) |
| De Rienzo/2020 | Prospective single arm | 40–90 years old Moderate LUTS (IPSS ⩾ 12) Prostate volume <100 ml Failed medical treatment |
6 (at 12 phone calls) | - TRUS probe and a diode laser generator, with a 1064 nm wavelength, having four independent channels for simultaneous firing were used. The laser light is conveyed from the source to the tissue through 300 mm caliber flat-tipped optical fibers introduced percutaneously through 21G Chiba needles. The energy delivered into the tissue produces a lesion of ellipsoid shape, one-third of which is located behind the tip of the fiber and two-thirds in front of it. The longitudinal diameter of the ellipsoid is 22.5 mm, and the transversal diameter is 16 mm - 1800 J of energy is applied in every side of firing. The tailoring of the procedure consisted of modifying the number and the site of applications per lobe, depending on the volume of the transition zone of the gland. After the check of security distances, the lasing began delivering a starting power of 4.5 W, reduced to 3.5 W after 1–2 min, when bubbles of vaporized tissue became visible at ultrasound At least one fiber per lateral lobe was placed, inserting the second fiber if it did not impair the respect of security distances (according to experience, in prostate larger than 55–60 ml), with a minimum distance of 10–15 mm from each other. If a median lobe was present, another needle was placed for its ablation. When the prostate mainly developed in a longitudinal direction, the pull-back of the fibers allowed the ablation of the distal half of the gland |
| Frego/2021 | Retrospective single arm | Men ⩾45 years with IPSS ⩾ 8; PSA <4 ng/ml or men with PSA >4 ng/ml with a previously negative prostate biopsy and a negative digital rectal examination; peak urinary flow (Qmax) ⩽ 15 ml/s and PVR ⩽150 ml; prostate volume between 30 and 100 cc | 12 | - Procedure is done under conscious sedation by IV of midazolam (3 mg) and with local anesthesia of the perineum with lidocaine 2%, 10 ml, followed by a US-guided periprostatic block with lidocaine 2%, 10 ml. All patients were treated with antibiotic prophylaxis with levofloxacin 500 mg the day before treatment and for the subsequent 5 days - The SoracteLite™ TPLA treatment was performed using an EchoLaser system (EchoLaser X4, EIEn Spa and EchoLaser Smart Interface, Elesta Spa, Calenzano (FI), Italy) with four independent laser sources, operating at 1064 nm wavelength - The laser light is guided through a flexible quartz optical fiber of 272 μm (Oberon GmbH, Wildau, Germany). The patient was placed in a lithotomy position. A three-way 18-F catheter was inserted and followed by continuous irrigation with saline solution during the procedure evaluation of the prostatic volume and morphology through a biplanar transrectal US probe (BK 5000, BK Medical System, Herlev, Denmark), up to two 21G introducer needles for each lobe were inserted parallel to the urethra, with a minimal distance of 10 mm from the urethral margin. The introducer needles were placed under US guidance using an articulated arm, a stepper, and a brachytherapy template Grid - For prostate volume <60 ml, a single optical fiber per lobe was used, while for prostate larger than 60 ml, two optical fibers per lobe were requested. When a median lobe was present, an additional needle was placed, the optical laser fibers were introduced, and advanced up to the needle tip, exposing the fiber tip for about 10 mm, taking care to maintain the safety distance of at least 15 mm from fiber tip to the bladder neck and 10 mm from the prostatic capsule. When two fibers were allocated within the same lobe, a 10–15 mm distance between them was ensured - Ablation was performed with a predetermined power of 3 W, delivering a maximum total energy of 1800 J per fiber in 600 s. For prostate volume >80 ml, a second ablation cycle called pull-back was performed, retracting the fiber 10 mm along its trajectory to deliver another 1200–1800 J in 400–600 s - Treatment was concluded when the gas formed during the ablation covered the entire desired area or 1800 J per fiber was reached |
| Manenti/2021 | Prospective, single arm | 1. Age ⩾50 years 2. IPSS ⩾ 12 3. Prostate volume ⩾30 ml 4. Intolerance or/and lack of results of combined medical treatment (α-adrenergic receptor blockers and 5-α-reductase inhibitors) |
12 | - Lithotomy position - The bladder and urethra were continuously irrigated by saline through a three-way catheter to avoid any thermal damage. Perineal and periprostatic local anesthesia was administered (20 ml lidocaine solution 2%); none of the patients underwent moderate conscious sedation - The US machine (MyLab9 eXP Platform, Esaote S.p.A, Genoa, Italy) was connected to a support planning system (Echolaser Smart Interface, Elesta S.p.A., Calenzano, Florence, Italy) - Chiba needles 21G (Elesta S.p.A, Calenzano, Italy) were inserted into the perineal space inside the prostatic gland and then they were used as introducers - Up to two needles for each lobe were positioned depending on the shape and volume of the prostate (one fiber per lobe if the prostate volume was <45 ml). If multiple in one lobe, these were consequently positioned at a distance of 8–10 mm from each other inside the central gland adenoma - Needles were positioned as parallel as possible to the prostatic longitudinal axis. Needles tip placement on the axial plane was tailored according to the relation of the urethral position and its longitudinal width to generate a symmetric cavity of ablation, to reduce urethral stromal compression and to shift the urethra lumen as close as possible to the midline - In the lumen of the needles bare optic quartz fibers were then introduced with a diameter of 272 μm (Oberon GmbH, Wildau, Germany), that protruded from the applicator tip by 10 mm and were connected to a multisource laser machine operating at 1064 nm (EchoLaser X4, ElEn S.p.A, Calenzano, Italy). Distances over 10 mm from the urethral wall, 15 mm from the bladder, and 10 mm from the prostatic capsule outer edge were maintained, and a fixed power of 3 W was applied during the procedure after an initial 2-min 5-W pulse ablation was adapted to prostatic volume (not more than two ablation sessions per each treatment). One pullback maneuver (retraction of the fibers by 1 cm in the longitudinal axis) was performed to widen the ablative area. Procedural ablation time ranged from 400 to 600 s for each fiber. Each fiber operated at 1800 J, with a maximum total energy delivery of 7200 J |
| Cai/2021 | Retrospective single arm | ⩾50 years, PV ⩾30 ml (TRUS/MRI), PVR ⩾ 50 ml ⩽ 400 ml IPSS ⩾ 12 Qmax ⩽ 15 ml/s |
6 | - Under the guidance of a transrectal biplanar linear array probe (4–13 Hz), 2% lidocaine was used from the skin to prostate lobules - The needle was inserted into the hyperplastic area surrounding the urethra of one side of the prostate, by ensuring that the distance from the tip of the needle to the bladder was >15 mm and the distance from the needle to the urethra was 8–10 mm from the side view - A second needle was inserted with the first needle used as a reference. After the needle sheath was withdrawn approximately 1 cm, ablation was initiated - The power of the ablation instrument was set to 3 W, and the energy value of the single fiber was set to 1800 J - If necessary, the proximal end of the prostate was ablated with a ‘pull-back technique’ to ensure complete vaporization of the targeted tissue. The contralateral prostate was ablated in the same manner - Finally, the ablation range was confirmed in contrast-enhanced ultrasound If the hyperplastic area surrounding the urethra was not completely ablated, the needle could be used again for ablation - Technical success was defined as complete ablation of the hyperplastic area surrounding the urethra |
| Cai/2022 | Retrospective comparative study (comparator –prostatic arterial embolization) | Men aged >50 years diagnosed with BPH with LUTS, with no fertility requirements; IPSS ⩾ 13; PV ⩾ 30 ml; maximum urinary flow rate (Qmax) ⩽15 ml/s; and PVR ⩽400 ml | 6 | - Under the guidance of a transrectal biplanar linear array probe (4–13 Hz), 2% lidocaine was used from the skin to prostate lobules - The needle was inserted into the hyperplastic area surrounding the urethra of one side of the prostate, by ensuring that the distance from the tip of the needle to the bladder was >15 mm and the distance from the needle to the urethra was 8–10 mm from the side view - A second needle was inserted with the first needle used as a reference. After the needle sheath was withdrawn approximately 1 cm, ablation was initiated - The power of the ablation instrument was set to 3 W, and the energy value of the single fiber was set to 1800 J - If necessary, the proximal end of the prostate was ablated with a ‘pull-back technique’ to ensure complete vaporization of the targeted tissue. The contralateral prostate was ablated in the same manner - Finally, the ablation range was confirmed in contrast-enhanced ultrasound If the hyperplastic area surrounding the urethra was not completely ablated, the needle could be used again for ablation - Technical success was defined as complete ablation of the hyperplastic area surrounding the urethra |
| Lagana/2022 | Prospective single arm | - BPH with several comorbidities - Patients with a desire to spare anterograde ejaculation - Patients intolerant of or poorly compliant to medical therapy, with no indication for surgery |
12 | - Patients were treated using the EchoLaser system (Elesta SpA, Calenzano, Italy), a 1064-nm continuous wave multisource diode laser, in conjunction with a MyLab Eight Ultrasound device (Esaote, Genoa, Italy) - Dedicated planning software in the ultrasound device was used to optimize applicator positioning within the prostate volume, taking into account safety distances to maintain from the urethra, prostatic capsule, and bladder neck - After the planning phase, either one or two 21G introducer needles were inserted per lobe, according to prostate dimensions and shape. The needles were inserted parallel to the prostatic capsule and urethra under US guidance using a transperineal approach - Then, a 300-μm optical laser fiber was introduced into each needle, with 10 mm protruding from the needle tip. Fiber tips were placed at a security distance of at least 15 mm from the bladder neck and 8–10 mm from the prostatic capsule and urethra - Ablation was performed at a fixed power of 3 W, with an energy delivery of 1800 J per fiber and illumination - Where necessary, a pull-back maneuver and reillumination were performed. When two fibers were placed in the same lobe, a distance of 10 mm was maintained between them. Patients were discharged the same day as the SoracteLite™ TPLA procedure, with the urinary catheter in place |
| Sessa/2022 | Prospective single arm | - Age >45 years with moderate/severe LUTS due to benign prostatic obstruction and IPSS ⩾ 12, prostate volume ⩾ 30 ml and up to 100 ml, ineffectiveness of medical therapies - No severe detrusor hypo-contractility - No large median lobe |
6 | - SoracteLite™ was used with EchoLaser diode laser with four independent laser sources operating at 1064 nm wavelength. A dedicated planning tool with simulation software was also used to plan treatment and place applicators in the prostate safely - The laser light is conveyed by the source to the tissues through 300 μm quartz optical fibers with a flat tip, inserted percutaneously within 21G Chiba needles. The laser light produces an ellipsoidal shape area of coagulative necrosis around the tip of the fiber - Intravenous prophylaxis with 2 g cephazolin was given within an hour before the procedure and a three-way 18Fr Foley catheter was placed with continuous irrigation to ensure cooling of the urethral wall during lasing time - A superficial anesthesia of perineum skin and subcutaneous tissue with lidocaine-prilocaine 5% cream and 10 ml of lidocaine 2%. Benzodiazepine solution was used for conscious sedation. A periprostatic anesthesia was also given under guidance with the TRUS probe and 10 ml of 2% lidocaine at each lobe of the prostate - Procedure was done in an outpatient setting - A 21G needle was inserted transperineally and located in the middle of each lobe with the orientation parallel to the longitudinal axis of the gland - Security distances from the urethra (8 mm) and from the bladder neck (15 mm) were checked. The procedure continued with the guidance of planning software for real-time user assistance to establish the proper position of applicators in an ellipsoidal shape area, ensuring safe distances - Stopper devices were applied to fix the needle in the desired position, preventing misplacement, and the 300 μm optical fibers were introduced. The starting power energy was 5 W, reduced in 2 min to 3.5 W when a cavity started to grow with vapor formation resulting in bubbles hyperechoic images at US. A fixed protocol to deliver energy was used, consisting of 1400 J per fiber. In the case of large prostates, a pill back, retraction of the applicator of 10 mm along its trajectory was applied to ablate another part of the prostatic tissue, delivering additional energy up to 1800 J per fiber - At the end of the treatment, 20 mg of methylprednisolone was administered intravenously for anti-edema and anti-inflammatory purposes. After an observation of a few hours in the outpatient clinic, patients were discharged with an indwelling catheter for 7 days |
| Bertolo/2023 | Open-label, single-center RCT (TPLA versus TURP) | 18–75 years, normal Ej-MSHQ preoperatively, normal antegrade ejaculation, IPSS ⩾ 10, Qmax <15 ml/s, prostate volume <100 ml, normal preop urine test | 6 | - EchoLaser EVO which consists of a continuous wave multisource (four sources) laser system with a wavelength of 1064 nm and EchoLaser Smart Interface, a simulation and planning device for safer insertion of 21G introducer needles (Introducer; Elesta SpA, Calenzano, Italy) and optical fibers (Fiber Optic for PLA; Elesta SpA) into the prostatic tissue under ultrasound guidance - Laser light is conveyed from the source to the tissue through 300-lm caliber flat-tipped optical fibers introduced percutaneously through the introducer needles. The result of the combined action of local heating and exposure time is irreversible necrosis of the cells. The energy delivered into the tissue produces a lesion of ellipsoid shape, one-third of which is located behind the tip of the fiber and two-thirds in front of it. The longitudinal diameter of the ellipsoid is 22.5 mm, and the transversal diameter is 16 mm -Under TRUS guidance one or two 21G introducer needles for each lobe are inserted into the adenoma and placed on planes as parallel as possible to the longitudinal plane of the prostate. To ease needle insertion, dedicated software displaying a grid overlaying the TRUS image is used. One 300-lm bare flat-tip optical laser fiber per needle is subsequently introduced and advanced up to the tip of the needle (the reader should note that 5 mm of the fiber tip will protrude from the tip of the needle). The integrated EchoLaser Smart Interface aids in achieving an exact and safe needle position so that a security distance from the urethral wall, the prostatic capsule, and the bladder neck is maintained (8, 8, and 15 mm, respectively) -The optical fibers are then connected with the laser source. TPLA is performed according to a standard protocol, which consists of delivering 1800 J of energy to every side of firing at a starting power of 4.5 W, reduced to 3.5 W after 1–2 min when bubbles of vaporized tissue became visible on TRUS. The procedure can be tailored by modifying the number and the site of applications per lobe, depending on the volume of the transition zone of the prostate. At least one fiber per lobe was placed, inserting the second fiber while respecting the security distances, with a minimum distance of 10–15 mm from each other - In the case of prostate glands with a mainly longitudinal growth pattern, the pull-back of the fibers allows the ablation of the distal half of the gland. Specifically for the purpose of the study, standard spinal anesthesia was performed to limit potential confounders (the reader should note that the procedure can be performed under conscious sedation plus local anesthesia of the perineal region and periprostatic anesthesia using lidocaine 10 mg/ml) |
BPH, benign prostatic hyperplasia; IPSS, International Prostate Symptom Score; LUTS, lower urinary tract symptoms; PSA, Prostate Specific Antigen; PV, prostate volume; PVR, post-void residual; Qmax, maximum urinary flow rate; RCT, randomized controlled trial; TPLA, Transperineal Laser Ablation; TRUS, transrectal ultrasound; TURP, transurethral resection of the prostate; US, ultrasound.
Technical aspects
The available studies demonstrated heterogeneity in the type of equipment that was used. While differences existed among the applied techniques. The main principles were similar and are summarized below.
Position and preparation: The procedure was performed in a lithotomy position with the testes and penis lifted and fixed to expose the perineum. An 18Fr three-way catheter was placed, while continuous normal saline irrigation for urethral cooling to prevent urethral wall injury was applied.13,16 The procedure was performed under transrectal ultrasound (TRUS) guidance with a biplanar 4–13 Hz probe. 13 Furthermore, the use of a multi-channel needle applicator with a dedicated software display grid overlapping the ultrasound images could also aid the procedure. 16
Anesthesia: The perineum was disinfected with wipes and the procedure was performed under local anesthesia only,13,17 or conscious sedation and local anesthesia.15,16,18,19 Lidocaine 2% was administered to the perineum and the periprostatic region for local anesthesia.13,15
Equipment and settings: The SoracteLite EchoLaser, Elesta laser ablation system was used. A dedicated planning tool (Echolaser Smart Interface, Elesta S.p.A, Calenzano, Italy) with simulation software allowed the user to plan the treatment and to place applicators in the prostate in a safe manner.13,15–18 A 21G trocar needle was used to accommodate the 300-μm flat-tip optical fiber and a continuous mode with a wavelength of 1064 nm was employed. At this wavelength, the penetration of light in the infrared spectrum was optimal. 13 The energy setting was 1800 J per fiber and illumination at a fixed power of 3 W,13,15,18,19 or 4.5–5 W reduced to 3–3.5 W after 1–2 min.16,17,20
Details of the procedure: The initial planning is conducted on the biplanar TRUS, to ensure adequate margins of safety. The ellipsoid area of coagulative necrosis extends to 22.5 mm longitudinally (of which two-thirds are localized beyond the tip of the fiber and one-third behind the tip) and 16 mm transversely. 16 Hence, the needle tip must be at least 15 mm from the base of the bladder and 8–10 mm from the urethra and prostate capsule to ensure a safe procedure. A 21G Chiba needle was inserted percutaneously in the perineum, under ultrasound guidance, and the energy delivered through the laser fiber was transferred through the needle. The needle was inserted as parallel to the longitudinal axis of the prostate as possible. The laser fiber protrudes 5–10 mm outside of the tip of the needle.13,14,16 Additional fibers may be used, depending on the size of the prostate. Generally, one fiber was needed for each lobe of the prostate. However, for gland sizes more than 45–60 ml,15,17 additional fibers may be required. Also, additional fibers are needed in case a median lobe exists. Whenever additional fibers are used, the distance between them must be 10–15 mm to comply with the created ellipsoid area of coagulative necrosis, as discussed above. Also, if the prostate gland has an increased volume profile longitudinally, a ‘pull-back’ maneuver to ensure adequate ablation may be used. 16 The treatment ablation lasted between 400 and 600 s 17 or was concluded when the maximum energy of 1800 J per fiber was reached.15,18,19 The ablation range evaluation was performed by contrast-enhanced ultrasound in three studies,13,18,19 and MRI in one study. 17
A graphical illustration of the technique is shown in Figure 2, while Figures 3 and 4 show the intraoperative setting and use of the equipment.
Figure 2.
Graphical illustration of TPLA.
TPLA, transperineal laser ablation.
Figure 3.
(a) Intraoperative setting, (b) laser setting screen, (c) transrectal application of ultrasound transducer, and (d) appearance of the prostate using the transrectal ultrasound probe.
Figure 4.
(a) Insertion of needles using the transrectal probe, (b) measurement of distance between needles and urethra, (c) selection of needle, and (d) application of energy.
Inclusion and exclusion criteria for the technique
Among the inclusion criteria of the eligible studies, patients over 18 years old and PV between 30 and 100 ml, based on TRUS or MRI measurements, were eligible for TPLA treatment. In addition to this, LUTS with IPSS ⩾ 12, Qmax ⩽15 ml/s, or PVR 50–400 ml were common inclusion criteria. Prior urethral procedures, PSA >4 ng/ml or suspected prostate cancer, urethral stricture history, neurological disease (spinal cord surgery, Parkinson’s disease, multiple sclerosis), allergy to ultrasound contrast, underactive detrusor, bladder cancer, prior prostatic abscess, acute or chronic prostatitis, active urinary tract infection, gland volume over 100 ml, bladder stones, and active hematuria were common exclusion criteria. A detailed list of them is found in Table 2.
Table 2.
General inclusion and exclusion criteria.
| Inclusion criteria | Exclusion criteria |
|---|---|
| - Age >18 years - Volume ⩾30 ml (TRUS/MRI) < 100 ml - PVR ⩾ 50ml ⩽ 400 ml - IPSS ⩾ 12 - Qmax ⩽ 15 ml/s - No evidence of prostate cancer |
- Prior to urethral procedure - PSA >4 ng/ml or diagnosis of prostate cancer - Urethral stricture - Spinal cord surgery, Parkinson’s disease, multiple sclerosis - Allergy to ultrasound contrast - Underactive detrusor - Bladder cancer - Prior prostatic abscess - Acute or chronic prostatitis - Gland volume 100 ml - Bladder stones - Active infection - Hematuria |
IPSS, International Prostate Symptom Score; PVR, post-void residual; Qmax, maximum urinary flow; TRUS, transrectal ultrasound.
Operative and perioperative data
Procedural time ranged between 28 and 61 min, with the actual ablation time being between 13 and 42.6 min. In most studies, a short hospitalization time was required (1–2), while Sessa et al., 20 who performed the procedure in an outpatient setting, reported a median monitoring of 6.4 h and only one patient required re-hospitalization for pelvic discomfort. Catheterization time ranged between 4 and 17.3 days, with the majority of patients requiring a catheter for 7–8 days. The measured total energy deployed was 6616.2–10,522 J, while two studies reported the coagulation zone, which was 10.3–11.7 ml.12,19 Operative and perioperative data are summarized in Table 3.
Table 3.
Operative and perioperative data.
| Author/year | Procedure time (min) | Ablation time (min) | Hospitalization days | Energy deployed (J) | Coagulation zone (ml) | Catheter time (days) | No. of fibers used | Time without pullback (min) |
|---|---|---|---|---|---|---|---|---|
| Patelli/2017 | 43.3 ± 8.7 | 15.9 ± 3.9 | 1.5 ± 0.4 | 10,522 ± 3290.5 | 10.3 ± 3.6 | 17.3 ± 10.0 | – | – |
| Pacella/2019 | 44.1 ± 12.9 | 23.4 ± 10.2 | 1.8 ± 0.4 | 6616.2 ± 3880.4 | – | 11.3 ± 11.5 | 1 fiber per lobe 4 patients (22.2%) 2 fibers per lobe 14 patients (77.8%) |
– |
| De Rienzo/2020 | 36.0 ± 9.5 | – | 20.8 ± 3.6 hours | – | – | 8.7 ± 2.5 | 2.2 ± 0.5 | – |
| Frego/2021 | – | 17.22 (10–18.88) | 1–2 | Per fiber 1800 (IQR: 1800–3200) | 7 | 1 fiber per lobe 12 patients (54.5%) 2 fibers per lobe 10 patients (45.5%) |
– | |
| Manenti/2021 | 28.2 ± 10.6 (range 21–75) | – | 0 | – | – | 39 patients 7 days 5 patients 14 days |
3 patients (6.8%) 2 fibers 4 patients (9%) 3 fibers 37 patients (84.2%) 4 fibers |
– |
| Cai/2021 | 60.9 ± 10.8 | 42.6 ± 9.9 | 1.5 ± 0.5 | 7179.2 ± 2815.7 | 11.7 ± 5.8 | 16.5 ± 4.2 | – | – |
| Cai/2022 | – | – | – | – | – | – | – | – |
| Lagana/2022 | 48.8 ± 14.3 | 13.0 ± 1.95 | 0 | 8261.6 ± 3280.1 | – | 14.9 ± 7.5 | 1.82 ± 0.4 per lobe | 10 ± 2.1 |
| Sessa/2022 | 31.5 (IQR: 28–37) | – | 6.4 h (IQR 5.9–7.2) | – | – | 7 | 2 fibers in all cases | – |
| Bertolo/2023 | TPLA 35 (range: 30–55) TURP 68 (range: 60–95) |
– | 2 (2–3) 3 (2–4) |
– | – | 4 (2–7) 3 (3–4) |
– | – |
IQR, interquartile range; TPLA, transperineal laser ablation; TRUS, transrectal ultrasound.
Functional outcomes
The age of patients in the included studies ranged between 61.9 and 73.9 years old, while PV at baseline was measured between 40 and 102 ml, although in the majority of studies except one, 17 median PV before ablation was <100 ml. IPSS at baseline indicated severe disease with a median of 18.3–22.7 among studies and PVR ranged between 60 and 199.9 ml. Qmax at baseline was also low, with a median of 7.6–9.2 ml/s. QoL was severely affected in all studies, with a median score ranging between 5 and 5.8, based on the IPSS QoL-related question. In all studies, IPSS was reduced during follow-up, with median values ranging between 8–13.1 at 3 months and 5–7 at 6–12 months, indicating a moderate-term effectiveness for symptom relief. Similarly, the QoL component of IPSS was also improved, with a reported median of 1–2.1 at 3 months and 1–2.3 at 6–12 months. Qmax improved compared to baseline and ranged between 11–13.3 ml/s at 3 months and 11.5–20.5 ml/s at 6–12 months follow-up. PVR postoperatively ranged between 45.6–54.8 ml at 3 months and 41.5–60.3 ml at 6–12 months of follow-up. All functional outcomes are shown in Tables 4 and 5.
Table 4.
Patient characteristics and outcomes.
| Author/year, sample size | Age (years) | IPSS | QoL | Qmax (ml/s) | PVR (ml) | Prostate volume (ml) | Complications | Sexual function | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Baseline | f/u 3 m | f/u 6/12 m | Baseline | f/u 3 m | f/u 6/12 m | Baseline | f/u 3 m | f/u 6/12 m | Baseline | f/u 3 m | f/u 6/12 m | Baseline | f/u 3 m | f/u 6/12 m | ||||
| Patelli/2017 TPLA 18 |
71.7 ± 9.4 (range 51–89 years) | 21.9 ± 6.2 | 10.7 ± 4.7 | 4.7 ± 0.6 | 2.1 ± 1.2 | 7.6 ± 2.7 | 13.3 ± 76.2 | 199.9 ± 147.3 | 81.5 ± 97.8 | 69.8 ± 39.9 | 54.8 ± 29.8 | 0% | – | |||||
| Pacella/2019 TPLA 160 |
69.8 ± 9.6 | 22.5 ± 5.1 | 6 m: 7.7 ± 3.3 83/160 follow-up 12 m: 7.0 ± 2.9 (baseline 22.2 ± 4.5) |
4.5 ± 1.1 | 6 m: 1.8 ± 1.0 83/160 follow-up 12 m : 1.6 ± 0.9 (baseline 4.2 ± 0.6) |
8.0 ± 3.8 | 6 m: 14.3 ± 3.9 83/160 follow-up 12 m: 15.0 ± 4.0 (baseline 8.6 ± 5.2) |
89.5 ± 84.6 | 6 m: 27.2 ± 44.5 83/160 follow-up 12 m: 17.8 ± 51.0 (baseline 71.7 ± 93.9) |
75.0 ± 32.4 | 6 m: 60.3 ± 24.5 83/160 follow-up 12 m: 58.8 ± 22.9 (baseline 87.9 ± 31.6) |
- 7/160 (4.3%) Clavien-Dindo grade I (3 transient hematuria, 3 acute urinary retention, 1 orchitis) - 1/160 (0.6%) grade III (prostatic abscess) - 6/160 (3.7%) patients experienced transient dysuria |
−2/160 (1.2%) reported loss of ejaculatory function | |||||
| De Rienzo/2020 21 TPLA |
62 (IQR 54–69) | 18.3 ± 3.9 | At 1 m 12 ± 5.6 At 3 m −8.3 ± 3.8 |
6.1 ± 2.6 | 4.1 ± 1 | 1 m: 2.4 ± 1.6 3 m: 1.4 ± 0.9 |
1.7 ± 0.8 | 9.2 ± 3.4 | 1 m: 12.1 ± 6.4 3 m: 13.3 ± 6.7 |
13.9 ± 6.2 | 81.8 ± 62.7 | 1 m: 37.4 ± 25.7 3 m: 18.7 ± 21.2 |
14 ± 16.7 | 40 (IQR: 40–50) | – | – | - 1/21 (4.8%) Clavien-Dindo grade III (prostatic abscess) | No change in IIEF Ejaculation returned to normal at 3 and 6 m |
| Frego/2021 22 TPLA |
61.9 (IQR: 55–65.5) | 22 (IQR: 19.5–25.25) | 8 (IQR: 4.5–11) | 6 m: 5 (IQR: 3–8.5) 12 m(n = 10): 6 (IQR: 4.25–7) |
4 (IQR: 4–5) | 1 (IQR: 0.5–2) | 6 m: 1 (IQR: 0–2) 12 m (n = 10): 1 (IQR: 1–2) |
9 (IQR: 5–12.5) | 12 (IQR: 9–16.5) | 6 m: 15 (IQR: 11.5–20.5) 12 m (n = 10): 20.5 (IQR: 14.25–23.75) |
60 (IQR: 25–107.5) | 39 (IQR 10–87.5) | 6 m: 40 (IQR: 16–63) 12 m (n = 10): 30 (IQR: 5–50) |
65 (IQR: 46.5–81) | 46(IQR 28.4–69) | 6 m: 42.3 (IQR 39.5–59) 12 m (n = 10): 41.5 (IQR 36.25–55) |
- 8/22 (36.3%) Clavien-Dindo grade I (dysuria) - 3/22 (13.6%) Clavien-Dindo grade II (retention) - 2/22 (9.1%) Clavien-Dindo grade II (urinary tract infection) |
Ejaculatory function was preserved in all but one of the sexually active patients at 6 m. No change in IIEF-5 10/22 patients (45.5%) completed the 12 m follow-up |
| Manenti/2021 44 TPLA |
18.5 ± 5.5 | 12 m: 6.2 ± 3.8 | 5.8 ± 1.4 | 12 m: 2.1 ± 1.1 | 7.6 ± 4.2 | 12 m: 16.2 ± 4.9 | 138.4 ± 40.8 | 12 m: 18.8 ± 8.5 | 102.4 ± 36.3 | 12 m: 48.12 ± 19.2 | - 1/44 (2.3%) Clavien-Dindo grade I (prolonged hematuria) - 1/44 (2.3%) Clavien-Dindo grade II (orchitis) - 1/44 (2.3%) Clavien-Dindo grade III (bilateral prostatic abscess) |
- All patients retained erectile function - Ejaculatory function, evaluated using the MSHQ-EjD, at month 12 (MSHQ-EjD 7.7 ± 3.2) showed improvement in the mean score (p = 0.030) |
||||||
| Cai/2021 20 TPLA |
73.9 ± 9.2 | 22.7 ± 5.3 | – | 9.1 ± 3.2 | 4.9 ± 1.7 | – | 2.3 ± 1.3 | 8.5 ± 3.0 | – | 15.2 ± 4.8 | 78.7 ± 58.8 | – | 30.3 ± 34.25 | 70.8 ± 23.8 | – | 54.7 ± 20.9 | 0% | – |
| Cai/2022 20 TPLA 20 PAE |
73.9 70.6 |
22.7 24.4 |
13.1 11.9 |
9.1 7.8 |
5 5.1 |
3.5 3.2 |
2.3 2.0 |
8.3 8.2 |
11.8 12.2 |
15.2 16.3 |
82.8 83.9 |
44.3 40.5 |
30.3 25.3 |
– | – | – | - 3 (15%) grade I (hematuria, dysuria) in the US-TPLA group - 2 (10%) grade I (pelvic pain) in PAE group |
– |
| Lagana/2022 63 TPLA |
72 ± 10 | 20.8 ± 7.4 | 11.0 ± 6.6 | 8.4 ± 5.9 | 4.7 ± 1.4 | 1.5 ± 1.2 | 1.2 ± 0.8 | 8.6 ± 3.5 | 13.2 ± 5.7 | 16.2 ± 4.3 | 124 ± 115.4 | 43.6 ± 53.6 | 40.6 ± 53.6 | 63.6 ± 29.7 | 45.6 ± 21.8 | 42.8 ± 14.2 | - 1/63 (1.6%) Clavien-Dindo grade II (orchitis) - 2/63 (3.2%) Clavien-Dindo grade III (prostatic abscess) |
– |
| Sessa/2022 38 TPLA |
71.5 (63.5–79) | 20 (16–25) | 11 (9–16) | 11 (10–13) | 4 (3–5) | 1 (1–3) | 2 (1–3) | 9.1 (8–11.5) | 11 (9.4–13.6) | 12 (9.5–15) | 100 (70–150) | 50 (35–95) | 50 (23–70) | 46 (IQR 38–71) | – | – | 0% | - Ejaculation was preserved in all patients or improved (+15–29%) - Erections based on IIEF-5 answers stable or improved (+2–4%) |
| Bertolo/2023 26 TPLA 25 TURP |
TPLA 63 (57–70.5) TURP 68.5 (59–74) |
24 (16–29) 20 (18.5–24) |
– | 11 (8–15) 8 (3–9) |
5 (3–5) 4 (3–5) |
– | 2 (2–4) 2 (1–2) |
10.2 (8.7–12) 10 (6.5–11.6) |
– | 15.2 (13.5–18.3) 26 (22–48) |
70 (20–100) 30 (20–70) |
– | 0 (0–5) 0 (0–20) |
49 (37–65) 55 (25–88) |
– | – | - 5/26 (19%) in TPLA had Clavien-Dindo grade II (urinary retention) - No complication in TURP group |
- 25/26 (96%) preserved ejaculation in TPLA - 7/25 (28%) in TURP preserved ejaculation - No difference in IIEF-5 between the two groups at 1 m |
PAE, prostatic artery embolization, BPH, benign prostatic hyperplasia; f/u, follow-up; IIES, International Index for Erectile Function; IPSS, International Prostate Symptom Score; IQR, interquartile range; LUTS, lower urinary tract symptoms; m, months; MSHQ-EjD, Male Sexual Questionnaire - Ejaculatory Dysfunction; PV, prostate volume; PVR, post-void residual; Qmax, maximum urinary flow rate; QoL, quality of life; RCT, randomized controlled trial; TRUS, transrectal ultrasound; TPLA, transperineal laser ablation; TURP, transurethral resection of the prostate; US, ultrasound.
Table 5.
General advantages and disadvantages of TPLA.
| Advantages | Disadvantages |
|---|---|
| (1) Procedure under LA (can be performed in patients with comorbidities not tolerating GA)(2) Decreased urethral trauma of instrumentation (3) Short hospital stay (4) If performed accurately, very minimal erectile dysfunction or ejaculatory problems (5) The use of anticoagulants was not an exclusion criterion (6) TPLA can be performed using the smallest available applicators on the market (7) The extreme precision of a laser, together with the very simple system of guidance, allows for a very controlled and safe energy delivery and predictable area of ablation, thus minimizing the risk of damage to surrounding structures (8) The constant monitoring in real time of all the phases of the treatment allows avoidance of severe damage to the adjacent structures. Real-time control of the thermo-ablated area allows to preserve the key anatomical landmarks such as the bladder neck and seminal colliculus, maintaining anterograde ejaculation and urinary continence (9) Short learning curve with fine needles used |
(1) New procedure, learning curve (2) Long duration of the catheter and increased risk of retention if the catheter is removed early (3) Poor quality of evidence so far (4) Studies with longer follow-ups are necessary to better understand if results are sustained in the midterm and long-term period |
LA, Local anaesthesia; GA, General anaesthesia; TPLA, transperineal laser ablation.
Complications
In three studies, no complications were recorded.13,19,20 Clavien-Dindo grade I complications were most commonly transient hematuria and dysuria, while acute urinary retention requiring re-catheterization and urinary tract infections/orchitis were the most commonly reported grade II complications. A prostatic abscess requiring drainage and prolonged course of antibiotics was the only grade III complication reported. Complication rates ranged between 1.9% and 2.3% for hematuria, 3.7% and 36.3% for dysuria, 1.9% and 19% for acute urinary retention, 0.6% and 9.1% for orchitis/urinary tract infections, and 0.6% and 4.8% for prostatic abscess formation. All complications are listed in Table 4.
Sexual function
Six studies evaluated erectile function and ejaculation postoperatively.10,15–18,20 Pacella et al. 18 reported a loss of ejaculation in 1.2% of patients, while de Rienzo et al. 16 reported no change in the IIEF questionnaire, while ejaculation returned to baseline after 3–6 months. Frego et al. 15 showed that ejaculation was preserved in 95.6% of patients, while no IIEF change was noticed at 12 months of follow-up, although only 45.5% completed the questionnaire at this timepoint. 15 Similarly, all patients retained erectile function and ejaculation in the study by Manenti et al., 17 with a mean score of ejaculatory function, as assessed by the Male Sexual Questionnaire – Erectile Dysfunction (MSHQ-EiD) questionnaire, showing improvement. Sessa et al. 20 reported preserved ejaculation in all patients and improvement in 15–29%, while erections were stable or even showed some improvement (+2–4%). Finally, Bertolo et al. 10 in their RCT showed that 96% of patients who underwent TPLA preserved their ejaculation.
Comparative studies
Two comparative studies reported the effectiveness and safety of TPLA compared to PAE 12 and TURP. 10 Cai et al. 12 found that both treatments led to improvement compared to baseline for functional outcomes, but no significant differences existed between them. Bertolo et al. 10 reported that both TURP and TPLA were effective in reducing symptoms and improving objective outcomes postoperatively at 6 months of follow-up, but TURP was superior in terms of Qmax improvement (+14.9 versus +5.4 ml/s, p < 0.001), while statistically nonsignificant differences were found regarding PVR reduction, IPSS, and QoL improvement. 10 At 1-month follow-up, 96.2% of TPLA patients retained their ejaculation, in contrast to 28% of TURP patients (p < 0.001) while the IIEF-5 score was similar between groups. 10 More patients in the TURP group were satisfied with their treatment compared to the TPLA group (80% versus 50%, p = 0.02) at 1 month postoperatively. 10
Discussion
The armamentarium for surgical management of BPH has rapidly evolved over the last 10 years. Besides MIST such as TURP and Holmium Laser Enucleation of the Prostate (HoLEP), further development of instruments and miniaturization of tools permitted the design of ultra-minimally invasive surgical techniques (uMIST), including steam injection (Rezu¯m), PAE, intraprostatic injections, insertion of prostatic urethral lift, and iTIND. 21 A recent systematic review and meta-analysis revealed that uMIST can reduce IPSS by 9.8 points, increase Qmax by 3.7 ml/s and decrease PVR without affecting erectile function and QoL, which are commonly affected when using MIST.21,22 One of the most recent additions is the TPLA, with accumulating evidence showing that this is an effective and safe technique.23,24 Two recent systematic reviews of the literature demonstrated the improvement in functional outcomes with simultaneous low complication rates and preservation of sexual function in the majority of patients;23,24 yet, the literature search was confined to a period ending in August 2022, since when a number of new clinical studies have been released. In our updated systematic, we included new evidence, including the first reported RCT on TPLA comparing it with TURP.
Findings from this systematic literature review indicate that most studies do not report in detail the perioperative management of patients; antibiotic coverage is usually prescribed within an hour from the procedure and up to 7 days, using cephalosporins and quinolones, while a common practice is also the administration of steroids to reduce the edema by their anti-inflammatory effect. 20 The decision of when is the appropriate timepoint for catheter removal was also variable, with most patients having it successfully removed after at least 7 days of catheterization. Based on the histopathological findings from an animal study, Liu et al. 11 proposed that the catheter should stay in situ for at least 7 days postoperatively since they observed that PV is increased for the first week, most likely due to inflammatory changes induced by laser energy application. 11 Bertolo et al. 10 reported a high acute urinary retention rate (19.2%), which was mostly seen in patients with early catheter removal at 3–4 days, thus confirming the aforementioned hypothesis. 10 Rest complication rates were low with 0% need for transfusion, 1.9–2.3% hematuria, 3.7–36.3% dysuria, 0.6–9.1% urinary tract infections, and 0.6–4.8% prostatic abscess formation.
Functional outcomes were improved in the vast majority of studies. Importantly, as highlighted by de Rienzo et al., 16 there seems to be a progressive improvement becoming more evident after 3–6 months. Tafuri et al. 24 reported a pooled Qmax at baseline of 8.69 ml/s, which improved among all timepoints (3 months, 13.17 ml/s; 6 months, 14.55 ml/s; and 12 months, 17.12 ml/s). Similarly, PVR decreased from 91.94 ml at baseline, to 36 ml at 3 months, 27.57 ml at 6 months, and 22.27 ml at 12 months. 24 IPSS decreased from a mean of 20.96 at baseline to 9.8 at 3 months, 6.92 at 6 months, and 6.4 at 12 months, with a similar decrease at the QoL component. 24 Although these pooled outcomes are more easily comprehended, the strength of evidence is low since they are derived from single-arm observational studies. Nevertheless, more recent data confirmed TPLA effectiveness among all of these objective and subjective outcomes. Minafra et al. 25 recently published an updated 3-year follow-up of their pilot studies, with 20 men who underwent TPLA and found a sustainable effectiveness, with IPSS being reduced by 37.2%, Qmax increased by 45.8%, PV decreased by 20.4%, and questionnaire scores related to sexual function improved by 60%, compared to baseline.
Sexual function preservation undoubtedly drives a decision during patient counseling for choosing BPH treatment options, especially in younger men. A recently published RCT by Bertolo et al., 10 focused on the evaluation of sexual function after TPLA in comparison to TURP. At the 1-month follow-up visit, 96% of the TPLA group retained ejaculation in contrast to 28% of patients who were assigned to TURP. 10 Importantly, TPLA did not differ significantly in terms of IPSS/QoL improvement and PVR reduction but was inferior to TURP in terms of Qmax increase. 10 Bladder neck preservation, along with sparing the muscular tissue surrounding the verumontanum, is considered the major component for avoiding retrograde ejaculation, 26 while the reduction in PV potentially permits better sperm flow. 17
This systematic review suffers from specific limitations derived from the design of included studies, which in the majority were observational and with a small sample size, thus adding the inherent limitations of these types of studies such as selection bias. In addition, due to the lack of comparative studies using the same groups, no meta-analysis was performed and results are presented in a systematic way. Nevertheless, this is an updated systematic literature review for TPLA adding the results of the first reported RCT on this technique.
Conclusion
TPLA of the prostate is an innovative, minimally invasive technique for managing patients with BPH. Existing studies indicate an effective technique in reducing IPSS and QoL scores, PVR reduction, and increase in Qmax, albeit the measured improvements in terms of Qmax are not equal to TURP. Although sexual function, namely ejaculation and erectile function are maintained in contrast to more invasive techniques, the mean catheterization time is 7 days and some studies report a rate of acute retention close to 20% after catheter removal. This technique can be offered as an alternative to patients, but it is important to mention that no long-term data are available for most patients, in the existing studies.
Supplemental Material
Supplemental material, sj-docx-1-tau-10.1177_17562872231198634 for Transperineal laser ablation as a new minimally invasive surgical therapy for benign prostatic hyperplasia: a systematic review of existing literature by Lazaros Tzelves, Santhosh Nagasubramanian, Alexandros Pinitas, Patrick Juliebø-Jones, Sanjeev Madaan, Giampaolo Sienna and Bhaskar Somani in Therapeutic Advances in Urology
Acknowledgments
None.
Footnotes
ORCID iDs: Patrick Juliebø-Jones  https://orcid.org/0000-0003-4253-1283
https://orcid.org/0000-0003-4253-1283
Sanjeev Madaan  https://orcid.org/0000-0003-4220-5613
https://orcid.org/0000-0003-4220-5613
Bhaskar Somani  https://orcid.org/0000-0002-6248-6478
https://orcid.org/0000-0002-6248-6478
Supplemental material: Supplemental material for this article is available online.
Contributor Information
Lazaros Tzelves, Department of Urology, University College London Hospitals, London, UK.
Santhosh Nagasubramanian, Department of Urology, University College London Hospitals, London, UK.
Alexandros Pinitas, Department of Urology, General Hospital of Athens ‘Gennimatas’, Athens, Greece.
Patrick Juliebø-Jones, Department of Urology, Haukeland University Hospital, Bergen, Norway.
Sanjeev Madaan, Department of Urology, Darent Valley Hospital, Dartford and Gravesham NHS Trust, Dartford, UK.
Giampaolo Sienna, Department of Urology, Careggi Hospital, University of Florence, San Luca Nuovo, Florence, Italy.
Bhaskar Somani, University Hospital Southampton NHS Trust, Tremona Road, Southampton SO16 6YD, UK.
Declarations
Ethics approval and consent to participate: Not applicable.
Consent for publication: All authors have consented to their participation in the paper. As it is a review article, individual patient consent was not required.
Author contributions: Lazaros Tzelves: Formal analysis; Methodology; Writing – original draft.
Santosh Nagasubramanian: Data curation; Writing – review & editing.
Alexandros Pinitas: Data curation; Writing – review & editing.
Patrick Juliebø-Jones: Writing – review & editing.
Sanjeev Madaan: Writing – review & editing.
Giampaolo Siena: Writing – review & editing.
Bhaskar Somani: Conceptualization; Project administration; Supervision; Writing – review & editing.
Funding: The authors received no financial support for the research, authorship, and/or publication of this article.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials: Not applicable.
References
- 1. Gravas SCJ, Gacci M, Gratzke C, et al. EAU Guidelines on management of non-neurogenic male lower urinary tract symptoms (LUTS), incl. benign prostatic obstruction (BPO). In: Paper presented at the EAU annual congress, Amsterdam, 2022. Available at: https://d56bochluxqnz.cloudfront.net/documents/full-guideline/EAU-Guidelines-on-Non-Neurogenic-Male-LUTS-2023.pdf. accessed 1 May 2023. [Google Scholar]
- 2. Egan KB, Burnett AL, McVary KT, et al. The co-occurring syndrome-coexisting erectile dysfunction and benign prostatic hyperplasia and their clinical correlates in aging men: results from the national health and nutrition examination survey. Urology 2015; 86: 570–580. [DOI] [PubMed] [Google Scholar]
- 3. Huang S-W, Tsai C-Y, Tseng C-S, et al. Comparative efficacy and safety of new surgical treatments for benign prostatic hyperplasia: systematic review and network meta-analysis. BMJ 2019; 367: l5919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Herrmann TRW. Enucleation is enucleation is enucleation is enucleation. World J Urol 2016; 34: 1353–1355. [DOI] [PubMed] [Google Scholar]
- 5. Ahyai SA, Gilling P, Kaplan SA, et al. Meta-analysis of functional outcomes and complications following transurethral procedures for lower urinary tract symptoms resulting from benign prostatic enlargement. Eur Urol 2010; 58: 384–397. [DOI] [PubMed] [Google Scholar]
- 6. Moher D, Liberati A, Tetzlaff J, et al. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg 2010; 8: 336–341. [DOI] [PubMed] [Google Scholar]
- 7. Sterne JA, Hernán MA, Reeves BC, et al. ROBINS-I: a tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016; 355: i4919. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ 2019; 366: l4898. [DOI] [PubMed] [Google Scholar]
- 9. Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017; 358: j4008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Bertolo R, Iacovelli V, Cipriani C, et al. Ejaculatory function following transperineal laser ablation vs TURP for benign prostatic obstruction: a randomized trial. BJU Int 2023; 132: 100–108. [DOI] [PubMed] [Google Scholar]
- 11. Liu RQ, Liu JB, Duan SB, Cheng SG, et al. Dynamic prostatic and laser-ablated lesion volume change after transperineal laser ablation in canine: preliminary observation and its clinical significance. Int J Hyperthermia 2020; 37: 1260–1267. [DOI] [PubMed] [Google Scholar]
- 12. Cai H, Zhu C, Fang J. Ultrasound-guided perineal laser ablation versus prostatic arterial embolization for benign prostatic hyperplasia: two similar short-term efficacies. Acta Radiol 2022: 64: 2841851221140214. [DOI] [PubMed] [Google Scholar]
- 13. Cai HJ, Fang JH, Kong FL, et al. Ultrasound-guided transperineal laser ablation for percutaneous treatment of benign prostatic hyperplasia: a new minimally invasive interventional therapy. Acta Radiol 2022; 63: 553–558. [DOI] [PubMed] [Google Scholar]
- 14. Laganà A, Di Lascio G, Di Blasi A, et al. Ultrasound-guided SoracteLite™ transperineal laser ablation (TPLA) of the prostate for the treatment of symptomatic benign prostatic hyperplasia (BPH): a prospective single-center experience. World J Urol 2023; 41: 1157–1162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Frego N, Saita A, Casale P, et al. Feasibility, safety, and efficacy of ultrasound-guided transperineal laser ablation for the treatment of benign prostatic hyperplasia: a single institutional experience. World J Urol 2021; 39: 3867–3873. [DOI] [PubMed] [Google Scholar]
- 16. de Rienzo G, Lorusso A, Minafra P, et al. Transperineal interstitial laser ablation of the prostate, a novel option for minimally invasive treatment of benign prostatic obstruction. Eur Urol 2021; 80: 95–103. [DOI] [PubMed] [Google Scholar]
- 17. Manenti G, Perretta T, Calcagni A, et al. 3-T MRI and clinical validation of ultrasound-guided transperineal laser ablation of benign prostatic hyperplasia. Eur Radiol Exp 2021; 5: 41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Pacella CM, Patelli G, Iapicca G, et al. Transperineal laser ablation for percutaneous treatment of benign prostatic hyperplasia: a feasibility study. Results at 6 and 12 months from a retrospective multi-centric study. Prostate Cancer Prostatic Dis 2020; 23: 356–363. [DOI] [PubMed] [Google Scholar]
- 19. Patelli G, Ranieri A, Paganelli A, et al. Transperineal laser ablation for percutaneous treatment of benign prostatic hyperplasia: a feasibility study. Cardiovasc Intervent Radiol 2017; 40: 1440–1446. [DOI] [PubMed] [Google Scholar]
- 20. Sessa F, Polverino P, Bisegna C, et al. Transperineal laser ablation of the prostate with EchoLaser™ system: perioperative and short-term functional and sexual outcomes. Front Urol 2022; 2. [Google Scholar]
- 21. Checcucci E, Veccia A, De Cillis S, et al. New Ultra-minimally invasive surgical treatment for benign prostatic hyperplasia: a systematic review and analysis of comparative outcomes. Eur Urol Open Sci 2021; 33: 28–41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Fiori C, De Cillis S, Volpi G, et al. iTIND for BPH: technique and procedural outcomes: a narrative review of current literature. Turk J Urol 2021; 47: 470–481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Sessa F, Polverino P, Siena G, et al. Transperineal laser ablation of the prostate (TPLA) for lower urinary tract symptoms due to benign prostatic obstruction. J Clin Med 2023; 12: 793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Tafuri A, Panunzio A, De Carlo F, et al. Transperineal laser ablation for benign prostatic enlargement: a systematic review and pooled analysis of pilot studies. J Clin Med 2023; 12: 1860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Minafra P, Rienzo GDE, Gerbasi S, et al. Three years outcomes of transperineal laser ablation of the prostate. Minerva Urol Nephrol 2023; 75: 471–478. [DOI] [PubMed] [Google Scholar]
- 26. Dorschner W, Stolzenburg JU, Dieterich F. A new theory of micturition and urinary continence based on histomorphological studies. 2. The musculus sphincter vesicae: continence or sexual function? Urol Int 1994; 52: 154–158. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplemental material, sj-docx-1-tau-10.1177_17562872231198634 for Transperineal laser ablation as a new minimally invasive surgical therapy for benign prostatic hyperplasia: a systematic review of existing literature by Lazaros Tzelves, Santhosh Nagasubramanian, Alexandros Pinitas, Patrick Juliebø-Jones, Sanjeev Madaan, Giampaolo Sienna and Bhaskar Somani in Therapeutic Advances in Urology