Abstract
Background:
The duration of oxaliplatin-based chemotherapy in high-risk stage II, low-risk stage III, and high-risk stage III colon cancer (CC) patients is controversial. To reduce the risk of adverse events (AEs) without compromising efficacy while improving chemotherapy compliance is crucial.
Methods:
The authors searched Cochrane, Embase, Pubmed, and Web of Science databases for articles from inception to August 8, 2023, the main outcomes were disease-free survival, overall survival, chemotherapy completion rates, and AE frequency.
Results:
Six randomized controlled trials (RCTs) involving 10 332 patients were included. Disease-free survival analysis revealed that only the high-risk stage III CC patients experienced better results with the 6-month FOLFOX regimen when compared with the 3-month regimen [Hazard ratio (HR): 1.32, 95% CI: 1.15–1.51, P<0.0001). Overall survival (OS) analysis revealed that extending the use of FOLFOX and CAPEOX regimens did not provide survival benefits for stage III CC patients (HR: 1.16, 95% CI: 0.9–1.49, and HR: 0.89, 95% CI: 0.67–1.18, P=0.40). The completion rate of the 3-month oxaliplatin-based adjuvant chemotherapy regimen was significantly higher than that of the 6-month regimen [Relative risk (RR): 1.16, 95% CI: 1.06–1.27, P=0.002]. Moreover, the 3-month regimen had significantly lower AE rates than the 6-month regimen (RR: 0.62, 95% CI: 0.57–0.68, P<0.00001), with differences mainly concentrated in grade 3/4 neutropenia (RR: 0.70, 95% CI: 0.59–0.85, P=0.0002), peripheral sensory neuropathy at ≥grade 2 (RR: 0.45, 95% CI: 0.38–0.53, P<0.00001), and hand–foot syndrome at ≥grade 2 (RR: 0.36, 95% CI: 0.17–0.77, P=0.009).
Conclusion:
The 6-month FOLFOX regimen should only be recommended for high-risk stage III CC, while the 3-month regimen can be recommended for other stages. A 3-month CAPEOX regimen can be recommended for stage II–III CC.
Keywords: adjuvant chemotherapy, colon cancer, CAPEOX, FOLOFX
Introduction
Highlights
Treating high-risk stage III patients with the 6-month FOLFOX regimen prolonged disease-free survival.
Prolonging the duration of FOLFOX did not increase disease-free survival in high-risk stage II and low-risk stage III patients.
Extending CAPEOX did not benefit patients with high-risk stage II, low-risk stage III, and high-risk stage III colon cancer.
The 3-month arm had significantly fewer adverse events and higher treatment compliance than the 6-month arm.
Prolonging chemotherapy duration does not necessarily benefit colon cancer patients.
Colon cancer (CC) is one of the most common gastrointestinal malignancies worldwide1,2 and it tends to affect younger populations2–4. Although surgery is the only curative treatment for locally resectable CC, even in the presence of high-risk factors, adjuvant postsurgery chemotherapy is required to prevent recurrence and prolong patient survival5. Despite advances in various treatments, including targeted therapy and immunotherapy, oxaliplatin-based chemotherapy remains the main adjuvant treatment for stage II–III CC6,7.
FOLFOX and CAPEOX, oxaliplatin chemotherapies based on three (fluorouracil, calcium folinate, and oxaliplatin) and two (capecitabine and oxaliplatin), respectively, are recommended as standard regimens for postoperative stage II–III CC treatment. The 2023 National Comprehensive Cancer Network (NCCN) recommended 3-month (3M) CAPEOX or 3–6-month (3–6M) FOLFOX as therapy for low-risk stage III CC and 3–6M CAPEOX or 6M FOLFOX for high-risk stage III CC.
However, there is controversy regarding the optimal duration of using oxaliplatin-based first-line adjuvant chemotherapy regimens8–10. For example, the 2023 NCCN guidelines have several controversies. The noninferiority of 6M FOLFOX in patients with low-risk stage III CC as well as that of 3M versus 6M CAPEOX in patients with high-risk stage III CC are unclear. The 2023 NCCN guidelines recommend 6M FOLFOX as adjuvant chemotherapy for high-risk stage II CC. However, a clinical study by the Hellenic Oncology Research Group11 and a phase III (ACHIEVE–2) trial in Japan12 showed that disease-free survival (DFS) did not differ significantly between 6M and 3M FOLFOX. At the same time, longer cycles of chemotherapy mean larger doses of chemotherapeutic agents as well as greater economic burden, which may be associated with higher rates of adverse events (AEs).
This study presents updated findings from the latest RCTs and assesses the effectiveness, compliance, and safety of 3M versus 6M oxaliplatin-based adjuvant chemotherapy in patients with stage II–III CC. Subgroup analyses were conducted on different chemotherapy regimens, pathological stages, and high-risk factors to assess the associations between 3M and 6M first-line oxaliplatin-based chemotherapy and CC prognosis. The study also aimed to address the controversial aspects of the 2023 NCCN guidelines and provides evidence-based medical evidence for individualized treatment of postoperative patients with stage II–III CC with different risk factors.
Methods
Registration and information
Ethical approval was not needed because the study analyzed publicly available data. This work has been reported in line with the PRISMA13 (Supplemental Digital Content 1, http://links.lww.com/JS9/B897, Supplemental Digital Content 2, http://links.lww.com/JS9/B898) (ID: CRD42023452245) and AMSTAR–2 Guidelines14 (Supplemental Digital Content 3, http://links.lww.com/JS9/B899).
Literature search
Literature was searched on Cochrane, Embase, PubMed, and Web of Science. Search queries were adjusted based on the respective database’s syntax. The searches included the period from database inception to 8 August 2023, without language restriction. The search terms included CC, colonic neoplasm, colon adenocarcinomas, postoperative, postsurgical, adjuvant, XELOX, CAPOX, FOLFOX, 5‐FU, fluorouracil, oxaliplatin, capecitabine, and leucovorin (Tables S1–S4, Supplemental Digital Content 4, http://links.lww.com/JS9/B900).
Study selection
The study included phase II–III RCTs with participants (>18 years) for stage II–III CC who received curative surgery. The study included high-risk stage II/III and low-risk III stage CC cases. High-risk stage II cases are characterized by T4, <12 detected lymph nodes, poor histological differentiation, lymphatic/vascular invasion, bowel obstruction, nerve invasion, or intestinal perforation. Low-risk stage III cases have T1–3 and N1. High-risk stage III patients had T4 and N1–2 or Tany and N212,14. The trial interventions were 3M FOLFOX (six cycles) or CAPEOX (four cycles) in the experimental group and 6M FOLFOX (12 cycles) or CAPEOX (eight cycles) in the control group. The outcomes should have reported one or more of the following clinical outcomes: OS, DFS, chemotherapy completion rate, and AEs. The study discussed AEs in grade 3 or higher, peripheral neurotoxicity (PSN) in grade 2 or higher, and hand–foot syndrome (HFS) in grade 2 or higher. Patients with multiple cancers, presurgery neoadjuvant therapy, postsurgery nonchemotherapy (e.g. radiotherapy, targeted therapy, immunotherapy, and alternative therapies), and duplicate studies or studies with incomplete data were excluded from the analysis.
Data extraction
Key information extracted from the studies retrieved by our search included first author name, publication year, demographic data, baseline data, interventions, and outcomes like DFS, OS, and AEs. Information was independently extracted by two authors (J.W. and K.L. sequentially) and verified by another author (Z.K.).
Risk of study bias
The risk of bias in the included studies was independently assessed by two authors. Using the Cochrane Handbook for Systematic Reviews and disagreements were resolved by another one. The risk of bias assessment included evaluation of the studies’ random sequence generation, allocation concealment, blinding (participants, personnel, and outcome assessors), incomplete outcome data, and selective reporting, and it was categorized as low, unclear, or high-risk.
Statistical analysis
Review Manager 5.4.1 was used to conduct a meta-analysis of the included studies. Basic information from the retrieved literature was used to analyze DFS, OS, chemotherapy completion rates, and AEs. Hazard ratio (HR) and 95% CI were used to examine survival data. Relative risk (RR) and 95% CI were used to analyze numerical data. The hypothesis test was two-sided, with P<0.05 indicating statistically significant differences. Review Manager 5.4.1 was used for data visualization. Where subgroup analysis results were reported separately, results from various subgroups were analyzed as separate studies.
The I 2 statistic was used to evaluate the heterogeneity of intergroup investigations, with I 2 scores of 0–25%, 25–50%, 50–75%, and 75–100% indicating no, mild, moderate, and extreme heterogeneity, respectively15. A fixed-effect model was used to analyze studies with low heterogeneity (I 2<50%). Subgroup analysis was used to determine heterogeneity sources in studies with significant heterogeneity (I 2>50%). A random-effects model was used if clinical heterogeneity was absent.
Results
Literature search and characteristics
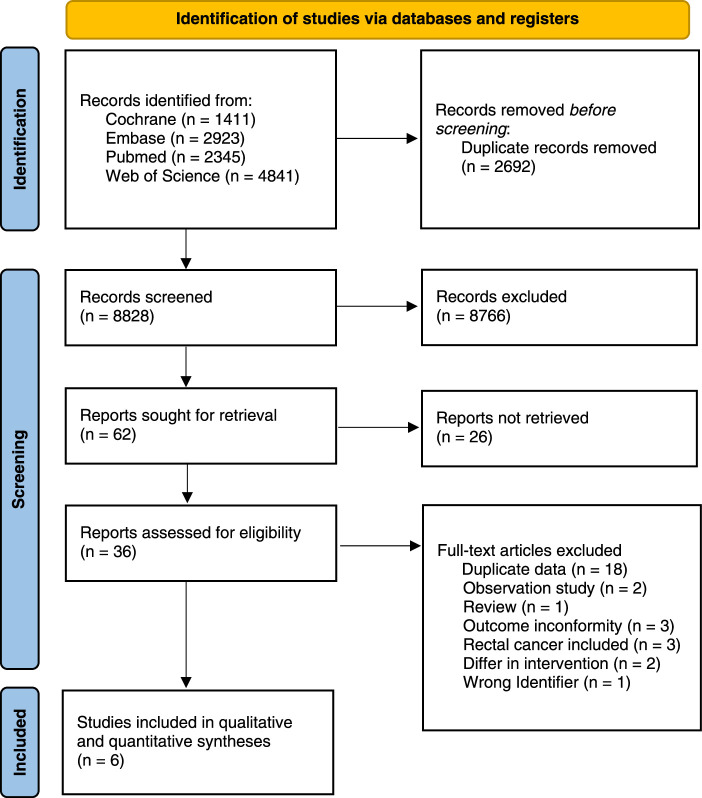
Literature searches retrieved 11 520 articles and 8828 remained after using Endnote 20 for plagiarism check. Of these 8766 were eliminated after title and abstract review. Of the remaining 62 articles, 26 had abstracts only or their original texts were unavailable. After reading the original text, 30 articles (18 duplicates, two observational studies, one review, three studies with cancer inconsistencies, two with intervention inconsistencies, and three with outcome inconsistencies) were excluded. A search for registries that met the inclusion criteria found that one study had a different registerium than this article, which was judged to impact reliability and reporting accuracy. The study was therefore excluded. Six studies11,12,16–19 (Fig. 1 and Table 1), including five clinical studies of The International Duration Evaluation of Adjuvant Chemotherapy (IDEA) collaboration (involving 10 332 patients) were included in the final analysis.
Figure 1.
Study selection flowchart.
Table 1.
Characteristics of included studies.
| Source | Name of trials | Countries | No. of centers | Follow-up (range/median, month) | Start-stop time | Sample size | Position | Pathological Stage | Risk of metastasis | Age(range/median,y) | Male (%)/Female | Male% |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Andre et al. 201816 | IDEA France | France | 129 | 39.6–63.6/51.6 | 2009.5.12–2014.5.21 | 2010 | Left=1161/right=746/both=16/missing=87 | stage III for all | NA | 58–70.8/64.7 | 1144 (56.9)/866 | 56.9 |
| Kim et al. 202217 | KCSG C009-07 |
Korea | 21 | 78.7 | 2010.1–2016.1 | 1788 | Left=1208/right=566/both=14/missing=0 | II=392/III=1396 | High=856/low=932 | 52–65/58 | 1050 (58.7)/738 | 58.7 |
| Petrelli et al. 202018 | IDEA TOSCA | Italy | 130 | 84 | 2007.6–2013.3 | 3614 | Ascending=1482/descending=2007/both=108 | II=1249/III=2340 | High=804/low=2785 | 21–83.8/63.3 | 1600 (44.3)/2014 | 44.3 |
| Sougklakos et al. 201911 | IDEA HORG | Greece | 24 | 38.3–126/67 | 2009.4–2015.10 | 1115 | Left=621/right=494 | II=413/III=702 | High=290/low=825 | 22–82/65(6 months) 20–81/67 (3 months) |
624(56)/492 | 56 |
| Yamazaki et al. 202112 | IDEA ACHIEVE-2 | Japan | 102 | 36.1 | 2014.2–2017.1 | 514 | Left=306/right=205/unclassifiable=3 | Stage III for all | High-risk for all | 23-81/66 (6 months) 30–83/66 (3 months) |
292(56.8)/222 | 56.8 |
| Yoshino et al. 201919 | IDEA ACHIEVE | Japan | 244 | 39 | 2012.8–2014.1 | 1291 | NA | Stage III for all | NA | 34–82/67 (6 FOLFOX) 28–85/65 (6 XELOX) 31–85/69 (3 FOLFOX) 29–83/65 (6 XELOX) |
649(50.2)/642 | 50.2 |
Literature quality evaluation
Because the six included studies were randomized and they described their randomization methods in detail, they were all classified as low-risk in random sequence generation. Except for one study that was rated as having an unclear risk because it did not describe its allocation concealment design. Because the studies were not blinded, we reasoned that there was potential unblinding of the patients’ psychological state. Hence, they were judged to be blinding participants and personnel, and the blinding outcomes were assessed as high-risk. Low-risk in all studies because of complete outcome measures and no selective reporting, and of unknown risk in all studies because of the lack of assessment information on other bias aspects (Fig. 2).
Figure 2.
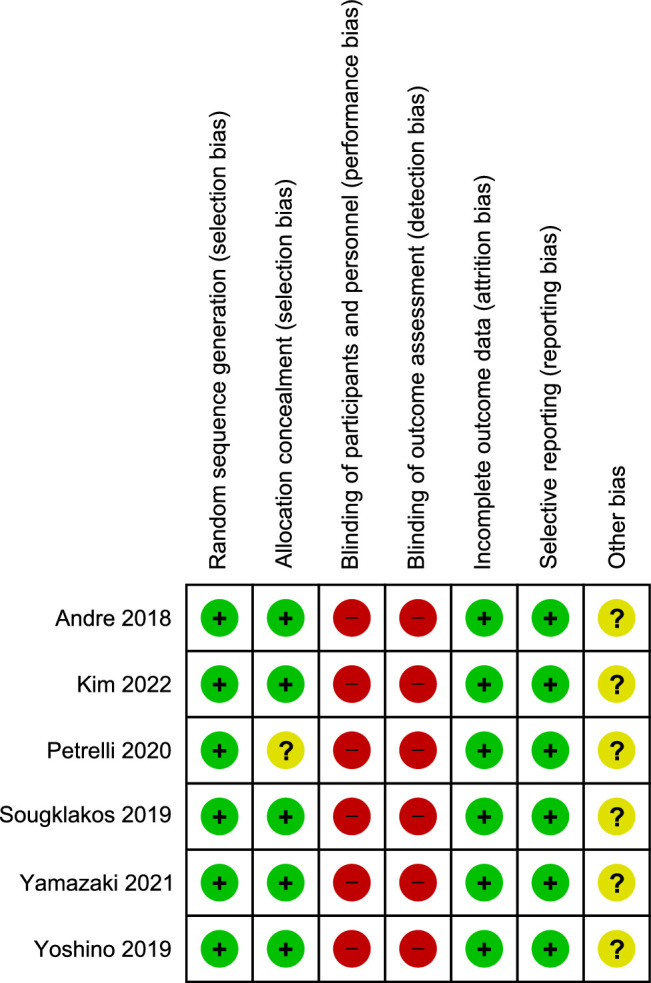
Risk of bias summary for all included studies.
DFS
Five studies11,12,16,17,19 reported DFS, Petrelli et al. 18 reported recurrence-free survival, and because Petrelli et al. defined recurrence-free survival as ‘assessed from the time of treatment allocation to relapse or death from any cause’, which is similar to DFS’s definition, this allowed for results pooling.
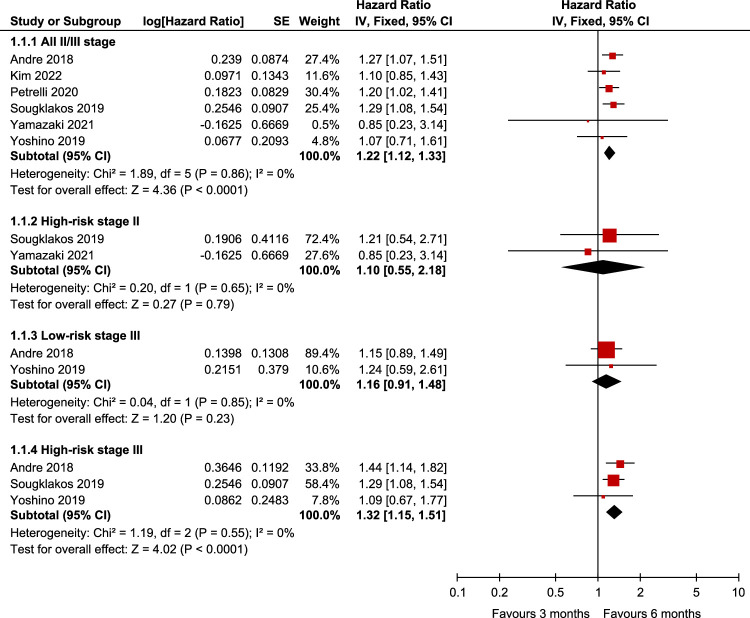
Six studies11,12,16–19, involving a total of 10 332 patients, showed that 3M FOLFOX had poorer DFS than 6M FOLFOX in patients with stage II–III CC (HR: 1.22, 95% CI: 1.12–1.33, P<0.0001), and no significant heterogeneity was found between studies (P=0.86, I 2=0%). We divided patients with stage II–III CC using FOLFOX into three subgroups for analysis: high-risk stage II, low-risk stage III, and high-risk stage III subgroups according to the previous settings. The inclusion of two RCTs11,12 in the high-risk II stage subgroup, which included a total of 1629 patients, showed no statistically significant difference in DFS between 3M FOLFOX and 6M FOLFOX in high-risk stage II (HR: 1.10, 95% CI: 0.55–2.18, P=0.79), and no significant heterogeneity was found between studies (P=0.65, I 2=0%). Two RCTs16,19 totaling 3301 patients were included in the low-risk phase III subgroup, in which there was no significant difference in DFS between 3M and 6M (HR: 1.16, 95% CI: 0.91–1.48, P=0.23), and no significant heterogeneity was found between studies (P=0.85, I 2=0%). The high-risk stage III subgroup included three RCTs11,16,19 including 4416 patients, and pooled data showed that in high-risk stage III CC, 6M FOLFOX was associated with longer DFS than 3M FOLFOX (HR: 1.32, 95% CI: 1.15–1.51, P<0.0001), and no significant heterogeneity was found between studies (P=0.55, I 2=0%) (Fig. 3).
Figure 3.
Assessment of disease-free survival (DFS) between 3M FOLFOX and 6M FOLFOX.
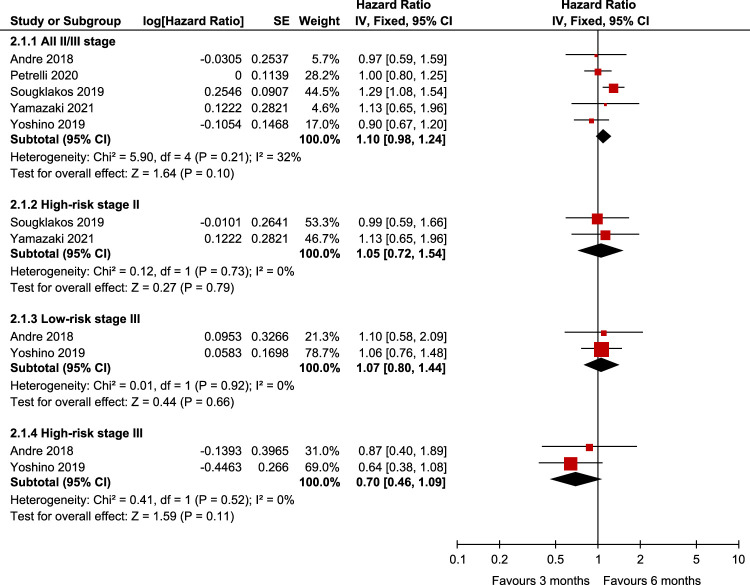
Five studies11,12,16,18,19 involving 8544 patients showed no significant difference in DFS between 3M CAPEOX and 6M CAPEOX in patients with stage II–III CC (HR: 1.10, 95% CI: 0.98–1.24, P=0.10), with mild heterogeneity between the included studies (P=0.21, I 2=32%). Following the same paradigm, we divided patients with stage II–III CC who used CAPEOX into the high-risk II stage subgroup, low-risk stage III subgroup, and high-risk stage III subgroup. The High-risk II stage subgroup included two studies11,12 with a total of 1629 patients and showed no significant difference in DFS between 3M and 6M chemotherapy (HR: 1.05, 95% CI: 0.72–1.54, P=0.79), with no significant heterogeneity between studies (P=0.73, I 2=0%). The low-risk stage III subgroup, which included two studies16,19 with a total of 3301 patients, showed no significant difference in DFS between 3M and 6M chemotherapy (HR: 1.07, 95% CI: 0.80–1.44, P=0.66), and no significant heterogeneity was found between studies (P=0.92, I 2=0%). High-risk stage III, which included two studies16,19 with a total of 3301 patients, also showed no significant difference in DFS between 3M and 6M chemotherapy (HR: 0.70, 95% CI: 0.46–1.09, P=0.11), and no significant heterogeneity was detected between studies (P=0.52, I 2=0%) (Fig. 4).
Figure 4.
Assessment of disease-free survival (DFS) between 3M CAPEOX and 6M CAPEOX.
OS
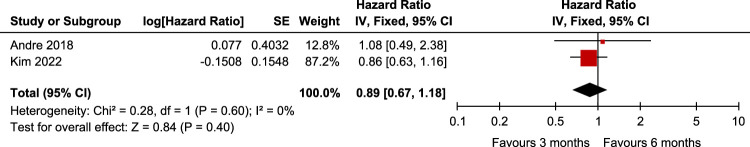
Two studies16,17 involving 3798 patients found no statistically significant difference in the OS of 3M versus 6M CAPEOX in patients with stage III CC (HR: 0.89, 95% CI: 0.67–1.18, P=0.40). No significant heterogeneity was detected between the studies (P=0.60, I 2=0%) (Fig. 5). Only one study16 found no statistically significant difference in the OS of 3M versus 6M FOLFOX in patients with stage III CC (HR: 1.16, 95% CI: 0.9–1.49). Due to the limited data, it is impossible to make subgroup analysis according to different risk factors.
Figure 5.
Assessment of overall Survival (OS) between 3M CAPEOX and 6M CAPEOX.
Completion of the chemotherapy regimen
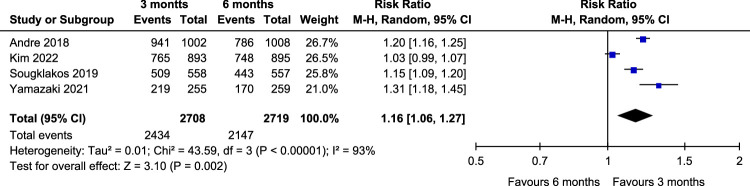
Evaluation of oxaliplatin-based chemotherapy regimen completion in the four studies11,12,16,17 that reported the number of patients who completed the 3M and 6M standard chemotherapy regimens (RR: 1.16, 95% CI: 1.06–1.27, P=0.002) found that completion was significantly higher in the 3M arm versus the 6M arm and that there was significant heterogeneity between studies (I 2=93%, P<0.00001, Fig. 6).
Figure 6.
Assessment of completion of 3M arm and 6M arm.
Safety evaluation
We assessed the safety of the included studies (mainly ≥grade 3 AEs, ≥grade 2 PSN, and ≥grade 2 HFS) and the combined results. Three studies11,12,16 reported on the number of people who ≥grade 3 AEs (3M arm: 470/1814, 6M arm: 763/1825), showed that the rate of ≥grade 3 AEs was significantly lower in the 3M arm than in the 6M arm (RR: 0.62, 95% CI: 0.57–0.68, P<0.00001), no significant heterogeneity was detected between the studies (P=0.60, I 2=0%). Subgroup analyses of the different types of AEs revealed that grade 3/4 neutropenia (RR: 0.70, 95% CI: 0.59–0.85, P=0.0002), ≥grade 2 PSN (RR: 0.44, 95% CI: 0.37–0.52, P<0.00001), and ≥grade 2 HFS (RR: 0.36, 95% CI: 0.17–0.77, P=0.009) were significantly lower in the 3M arm than the 6M arm. However, other types of ≥grade 3 AEs, such as leucopenia (RR: 0.41, 95% CI: 0.08–2.07, P=0.28), thrombocytopenia (RR: 0.94, 95% CI: 0.15–5.83, P=0.95), anaemia (RR: 0.11, 95% CI: 0.01–2.09, P=0.14), diarrhea (RR: 0.78, 95% CI: 0.55–1.09, P=0.14), nausea (RR: 0.83, 95% CI: 0.48–1.43, P=0.50), vomiting (RR: 1.27, 95% CI: 0.74–2.18, P=0.39), stomatitis (RR: 0.34, 95% CI: 0.04–3.23, P=0.35), and anorexia (RR: 0.65, 95% CI: 0.29–1.48, P=0.31) there were no significant difference between 3M and 6M (Fig. 7).
Figure 7.
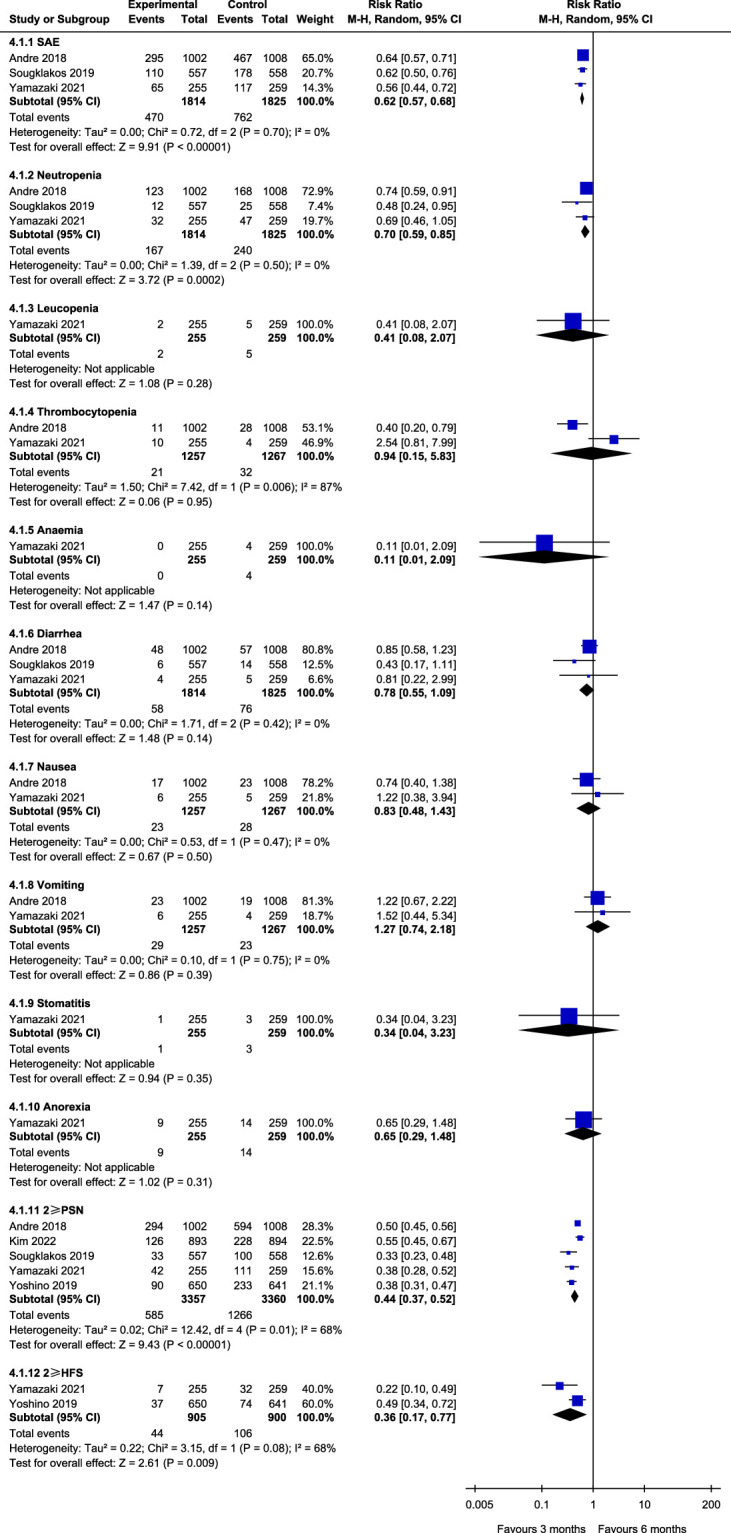
Assessment of safety.
We also used descriptive analyses to compare differences in the types of adverse events between FOLFOX and CAPEOX chemotherapy regimens. Andre et al. 16 showed that the incidence of ≥grade 3/4 neutropenia was higher in the FOLFOX (15%) group than in the CAPEOX group (8%) (P=0.0116). Patients treated with FOLFOX had less grade 3/4 diarrhea and vomiting than those treated with CAPEOX (diarrhea: 5 vs. 19%, P=0.0046; vomiting: 2 vs. 5%, P=0.0068). Souglakos et al. 11 reported a higher incidence of grade 3/4 neutropenia in patients treated with FOLFOX than in patients treated with CAPEOX (15 vs. 10%, P=0.0372). However, the incidence of grade 3/4 diarrhea was lower in patients treated with FOLFOX than in patients treated with CAPEOX (7 vs. 13%, P=0.0461). Yoshino et al. 19 showed the 3M arm (8 vs. 16%, P=0.04) and the 6M CAPEOX arm exhibited a significantly lower PSN incidence for 3 years when compared with FOLFOX (21 vs. 34%, P=0.02).
Discussion
Meta-analyses are considered to have higher quality than RCTs20. In this meta-analysis of six RCTs involving patients with stage II–III CC, we compared the effects of 3M and 6M oxaliplatin-based first-line chemotherapy on different CC stages and compared our findings with a 2019 meta-analysis21 to address the controversial aspects of the NCCN guidelines.
Postoperative adjuvant chemotherapy is the basic treatment of stage II–III CC. However, clinical efficacies of the two first-line adjuvant chemotherapy regimens (FOLFOX and CAPEOX) do not differ significantly22 and regimen choice is often based on clinician and patient preferences23. This is not only common in clinical practice but has also been observed in multiple IDEA clinical trials, such as the FOLFOX regimen that began in France24,25, in IDEA’s multicenter clinical study in France, FOLFOX was used more than CAPEOX, although it was likelier to cause PSN26, while clinicians in Asian countries like Japan often preferred CAPEOX, which was used to treat 432 out of 514 patients and 75% of the patients in two separate phase III ACHIEVE trials12,19. Hence, this meta-analysis focuses on the clinical efficacy of different duration of chemotherapy, rather than differences in efficacy between chemotherapy regimens.
Our meta-analysis revealed that the DFS of 6M versus 3M FOLFOX did not differ significantly in patients with high-risk stage II and low-risk stage III CC and that there was no significant difference in the DFS of 6M CAPEOX versus 3M CAPEOX. However, comparing 6M FOLFOX with 3M in patients with high-risk stage III CC revealed that 6M FOLFOX was associated with significantly longer DFS, and DFS did not differ significantly in 6M versus 3M CAPEOX. There were no significant survival differences between 3M versus 6M FOLFOX/CAPEOX in patients with stage III CC, probably because most studies lacked OS data.
The 3M arm was associated with significantly better compliance than the 6M arm, likely because fewer chemotherapy cycles mean shorter treatment time and a lower probability of AEs. This was confirmed by our results indicating that the 3M arm had significantly fewer ≥grade 3 AEs, which mainly manifested as grade 3/4 neutropenia, ≥grade 2 PSN, and ≥grade 2 HFS. PSN is one of the main dose-limiting oxaliplatin toxicities27,28 and it impacts CC prognosis because it can be long-term or permanent29,30. Hence, it is critical to reduce the duration and dose of oxaliplatin.
Because OS data were lacking, we mainly used DFS to assess clinical efficacy to answer three controversial aspects of the NCCN CC guidelines and to demonstrate the existing recommendations of the NCCN guidelines based on the meta-analysis results.
First, the noninferiority of 3M FOLFOX when compared with 6M FOLFOX, which is mentioned in the 2023 NCCN guidelines, has not been demonstrated in patients with low-risk stage III CC. Our meta-analysis shows that the efficacies of 3M and 6M against low-risk stage III CC are comparable and that the probability of AEs can be avoided by reducing the chemotherapy cycles, which would also improve compliance.
Second, the noninferiority of 3M CAPEOX when compared with 6M CAPEOX in patients with high-risk stage III CC has not been demonstrated. Our meta-analysis indicates that the efficacies of 3M and 6M CAPEOX are comparable regardless of CC stage or risk factors and that the probability of AEs is lower.
Finally, it is unclear if the 6M regimen is optimal when FOLFOX is used as adjuvant chemotherapy in high-risk stage II CC. Our meta-analysis indicates that the efficacies of 3M and 6M FOLFOX against high-risk stage II CC are comparable, highlighting 3M FOLFOX as an alternative for high-risk stage II CC treatment.
We also demonstrated the conclusion from the 2023 NCCN guidelines that if FOLFOX is chosen as an adjuvant for patients with high-risk stage III (T4 and/or N2) CC, the 6M regimen is required to minimize recurrence, and that if CAPEOX is selected as the adjuvant chemotherapy for low-risk stage III CC, the 3M regimen should be preferred because it has the similar efficacy while effectively reducing adverse reactions and increasing compliance.
This study has several limitations. A key limitation of this study is the small number of studies it included. Because this is a relatively novel field, few RCTs are available, which was also confirmed by exclusion and only two studies were included in multiple subgroups. Moreover, OS data are lacking. For instance, in the evaluation of 3M versus 6M FOLFOX, only one study evaluated OS in patients with stage III CC. Although DFS can indicate OS, it is not clear if DFS translates into a survival benefit. Therefore, the inclusion of more high-quality RCTs will help improve our understanding of the threshold effect of oxaliplatin-based first-line adjuvant chemotherapy. Another limitation results from methodological flaws in the included studies. Although the randomization scheme of the included studies is clear, because of different chemotherapy cycles, all included RCTs are open-label. Although it is difficult to use blinding in cancer clinical trials because remaining blinded after disease progression may adversely affect the subsequent treatment of the participants, the lack of blinding can cause bias. Finally, physicians’ personal habits, ethnic and regional differences are also important factors affecting the choice of postoperative adjuvant chemotherapy regimen for CC patients. Thus, studies are required to establish whether national, regional, and ethnic differences affect treatment, and to guide guideline formulation.
Conclusions
In this meta-analysis, we updated and analyzed several RCTs of IDEA, which are the largest prospective RCTs to date investigating the optimal duration of CC adjuvant chemotherapy with high confidence. Based on our meta-analysis results, we make several recommendations on how to improve the duration of postoperative adjuvant chemotherapy in real-world settings. Shortening the duration of chemotherapy can reduce AE incidence, lower economic burden, and increase patient compliance without affecting clinical efficacy. When compared with a meta-analysis published in 2019, we further refined the use of FOLFOX and CAPEOX in patients with various CC pathological stages and risk factors. We recommend using the 6M FOLFOX regimen only for high-risk patients with stage III CC and the 3M regimen for other stages. The 3M CAPEOX regimen is recommended regardless of pathologic stage and risk factors as a reference for personalized treatment of stage II-III stage CC.
Ethical approval
Not applicable.
Consent
Not applicable.
Sources of funding
This study was supported by the Innovation Team and Talents Culture Program of National Administration of Traditional Chinese Medicine; Beijing Science and Technology Planning Project (Grant No: Z221100003522021).
Author contribution
Z.K., J.W., K.L., J.W., J.L.: had full access to all of the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis; Z.K., J.W., and K.L.: are joint first authors; Z.K., J.W., and K.L.: acquisition, analysis, or interpretation of data; Z.K., J.W., K.L., J.W., and J.L.: drafting of the manuscript; Z.K., J.W., K.L., J.W., and J.L.: critical revision of the manuscript for important intellectual content; Z.K., J.W., and K.L.: statistical analysis; J.L.: obtained funding; Z.K., J.W., and K.L.: administrative, technical, or material support.
Conflicts of interest disclosure
The authors have no potential conflicts of interest to disclose.
Research registration unique identifying number (UIN)
Registry used: PROSPERO.
Unique identifying number or registration ID: CRD42023452245.
Hyperlink to your specific registration: https://www.crd.york.ac.uk/prospero/.
Guarantor
Jie Li.
Data availability statement
Ethical approval was not needed because the study analyzed publicly available data. All data were extracted separately by more than two authors.
Provenance and peer review
Not commissioned, externally peer-reviewed.
Assistance with the study
Not applicable.
Presentation
Not applicable.
Supplementary Material
Footnotes
Sponsorships or competing interests that may be relevant to content are disclosed at the end of this article.
Supplemental Digital Content is available for this article. Direct URL citations are provided in the HTML and PDF versions of this article on the journal’s website, www.lww.com/international-journal-of-surgery.
Published online 13 February 2024
Contributor Information
Ziyu Kuang, Email: 20220941390@bucm.edu.cn.
Jiaxi Wang, Email: 13661069062@163.com.
Kexin Liu, Email: bjlk2010@126.com.
Jingyuan Wu, Email: 20220941391@bucm.edu.cn.
Jie Li, Email: qfm2020jieli@yeah.net.
References
- 1.Wong MCS, Huang J, Lok V, et al. Differences in incidence and mortality trends of colorectal cancer worldwide based on sex, age, and anatomic location. Clin Gastroenterol Hepatol 2021;19:955–966.e61. [DOI] [PubMed] [Google Scholar]
- 2.Leopa N, Dumitru E, Dumitru A, et al. The clinicopathological differences of colon cancer in young adults versus older adults. J Adolesc Young Adult Oncol 2023;12:123–127. [DOI] [PubMed] [Google Scholar]
- 3.Murphy CC, Wallace K, Sandler RS, et al. Racial disparities in incidence of young-onset colorectal cancer and patient survival. Gastroenterology 2019;156:958–965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Altieri MS, Thompson H, Pryor A, et al. Incidence of colon resections is increasing in the younger populations: should an early initiation of colon cancer screening be implemented? Surg Endosc 2021;35:3636–3641. [DOI] [PubMed] [Google Scholar]
- 5.Klaver CEL, Kappen TM, Borstlap WAA, et al. Laparoscopic surgery for T4 colon cancer: a systematic review and meta-analysis. Surg Endosc 2017;31:4902–4912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Dey A, Mitra A, Pathak S, et al. Recent advancements, limitations, and future perspectives of the use of personalized medicine in treatment of colon cancer. Technol Cancer Res Treat 2023;22:15330338231178403. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kishore C, Bhadra P. Current advancements and future perspectives of immunotherapy in colorectal cancer research. Eur J Pharmacol 2021;893:173819. [DOI] [PubMed] [Google Scholar]
- 8.Collienne M, Arnold D. The optimal duration of adjuvant chemotherapy in colon cancer. Cancers (Basel) 2020;12:2509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Gelibter AJ, Caponnetto S, Urbano F, et al. Adjuvant chemotherapy in resected colon cancer: When, how and how long? Surg Oncol 2019;30:100–107. [DOI] [PubMed] [Google Scholar]
- 10.Tanis PJ. Refining the role of adjuvant chemotherapy in stage III colon cancer. Colorectal Dis 2019;21:135–136. [DOI] [PubMed] [Google Scholar]
- 11.Souglakos J, Boukovinas I, Kakolyris S, et al. Three- versus six-month adjuvant FOLFOX or CAPOX for high-risk stage II and stage III colon cancer patients: the efficacy results of Hellenic Oncology Research Group (HORG) participation to the International Duration Evaluation of Adjuvant Chemotherapy (IDEA) project. Ann Oncol 2019;30:1304–1310. [DOI] [PubMed] [Google Scholar]
- 12.Yamazaki K, Yamanaka T, Shiozawa M, et al. Oxaliplatin-based adjuvant chemotherapy duration (3 versus 6 months) for high-risk stage II colon cancer: the randomized phase III ACHIEVE-2 trial. Ann Oncol 2021;32:77–84. [DOI] [PubMed] [Google Scholar]
- 13.Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Int J Surg 2021;88:105906. [DOI] [PubMed] [Google Scholar]
- 14.Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews thatinclude randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017;358:j4008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Higgins JP, Thompson SG, Deeks JJ, et al. Measuring inconsistency in meta-analyses. BMJ 2003;327:557–560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.André T, Vernerey D, Mineur L, et al. Three versus 6 months of oxaliplatin-based adjuvant chemotherapy for patients with stage III colon cancer: disease-free survival results from a randomized, open-label, international duration evaluation of adjuvant (IDEA) France, Phase III Trial. J Clin Oncol 2018;36:1469–1477. [DOI] [PubMed] [Google Scholar]
- 17.Kim ST, Kim SY, Lee J, et al. Oxaliplatin (3 months v 6 months) With 6 Months of Fluoropyrimidine as Adjuvant Therapy in Patients With Stage II/III Colon Cancer: KCSG CO09-07. J Clin Oncol 2022;40:3868–3877. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Petrelli F, Rulli E, Labianca R, et al. Overall survival with 3 or 6 months of adjuvant chemotherapy in Italian TOSCA phase 3 randomised trial. Ann Oncol 2021;32:66–76. [DOI] [PubMed] [Google Scholar]
- 19.Yoshino T, Yamanaka T, Oki E, et al. Efficacy and long-term peripheral sensory neuropathy of 3 vs 6 months of oxaliplatin-based adjuvant chemotherapy for colon cancer: the ACHIEVE Phase 3 randomized clinical trial. JAMA Oncol 2019;5:1574–1581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zeng X, Zhang Y, Kwong JS, et al. The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: a systematic review. J Evid Based Med 2015;8:2–10. [DOI] [PubMed] [Google Scholar]
- 21.Boyne DJ, Cuthbert CA, O’Sullivan DE, et al. Association between adjuvant chemotherapy duration and survival among patients with stage II and III colon cancer: a systematic review and meta-analysis. JAMA Netw Open 2019;2:e194154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Conroy T, Hebbar M, Bennouna J, et al. Quality-of-life findings from a randomised phase-III study of XELOX vs FOLFOX-6 in metastatic colorectal cancer. Br J Cancer 2010;102:59–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Blinman P, Martin A, Jefford M, et al. Patients’ preferences for 3 months vs 6 months of adjuvant chemotherapy for colon cancer. JNCI Cancer Spectr 2020;5:pkaa107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.de Gramont A, Figer A, Seymour M, et al. Leucovorin and fluorouracil with or without oxaliplatin as first-line treatment in advanced colorectal cancer. J Clin Oncol 2000;18:2938–2947. [DOI] [PubMed] [Google Scholar]
- 25.André T, Bensmaine MA, Louvet C, et al. Multicenter phase II study of bimonthly high-dose leucovorin, fluorouracil infusion, and oxaliplatin for metastatic colorectal cancer resistant to the same leucovorin and fluorouracil regimen. J Clin Oncol 1999;17:3560–3568. [DOI] [PubMed] [Google Scholar]
- 26.Argyriou AA, Velasco R, Briani C, et al. Peripheral neurotoxicity of oxaliplatin in combination with 5-fluorouracil (FOLFOX) or capecitabine (XELOX): a prospective evaluation of 150 colorectal cancer patients. Ann Oncol 2012;23:3116–3122. [DOI] [PubMed] [Google Scholar]
- 27.Calls A, Carozzi V, Navarro X, et al. Pathogenesis of platinum-induced peripheral neurotoxicity: insights from preclinical studies. Exp Neurol 2020;325:113141. [DOI] [PubMed] [Google Scholar]
- 28.Sałat K. Chemotherapy-induced peripheral neuropathy-part 2: focus on the prevention of oxaliplatin-induced neurotoxicity. Pharmacol Rep 2020;72:508–527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cavaletti G, Marmiroli P. Management of oxaliplatin-induced peripheral sensory neuropathy. Cancers (Basel) 2020;12:1370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Peng S, Ying AF, Chan NJH, et al. Prevention of oxaliplatin-induced peripheral neuropathy: a systematic review and meta-analysis. Front Oncol 2022;12:731223. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Ethical approval was not needed because the study analyzed publicly available data. All data were extracted separately by more than two authors.